Abstract
A kind of magnetic poly (lactic acid) (PLA) melt blown nonwoven fabric (MB) was fabricated by the introduction of ferroferric oxide (Fe3O4) nanoparticles to improve its air filtration performances. In view of the poor compatibility of two components, the poly (L-lactic acid) (PLLA) molecular chains was firstly grafted onto the Fe3O4 nanoparticles surface via the ring opening polymerization (ROP). Then, PLA/Fe3O4-g-PLLA composite masterbatches with different mass ratios were prepared by melt-blending method and processed into the corresponding composite MB. The structures and performances of PLA/Fe3O4-g-PLLA composite masterbatches and their MB were investigated. The results showed that the addition of Fe3O4-g-PLLA nanohybrids hardly influenced the glass transition, cold crystallization and melting behaviors of the composite masterbatches. Though the melt fluidity of the composite masterbatches reduced with the Fe3O4-g-PLLA content increasing, the composite masterbatches still could present the appropriate processability in the range of 210°C to 230°C. Fe3O4-g-PLLA could be uniformly dispersed in PLA matrix and had a good interfacial compatibility with the matrix. Compared with pure PLA MB, the fiber surface of the composite MB became slightly rough, the pore size and distribution of the fiber web increased. The addition of Fe3O4-g-PLLA endowed PLA MB with magnetism. With the increasing of Fe3O4-g-PLLA content, the air permeability of the composite MB was improved and their filtration resistance obviously reduced. When the content of Fe3O4-g-PLLA was 0.5 wt%, the filtration efficiency of the composite MB reached the maximum. Moreover, the composite MB have higher longitudinal tensile strength and elongation at break than those of pure PLA MB.
Introduction
With the rapid development of industry and acceleration of urbanization, air pollution has been a serious problem endangering the survival of human being. A large amount of particles matter (PM) and volatile organic compounds (VOCs) exist in the air and enter human’s body during the breath process, which causes the irreversible harm to human health, living quality and ecosystems.1–7 In order to deal with PM pollution in the air, fabric filters have been widely used for air filtration due to relatively energy-efficiency, low-cost, and ease of scalable fabrication.8–10 Melt blown nonwoven fabric (MB), a kind of innovative fabric obtained by the one-step melt-blowing process, has drawn people’s interest in being used as the high-efficiency air filtration filters or N95-certified mask to treat the serious event of air pollution according to its unique performances.2,3,11–14 Compared with traditional woven fabrics, MB has the simpler manufacturing process, lower prices and better air permeability, meanwhile, still remains the high surface area, small pore size, good barrier and mechanical properties. Thus, MB is regarded as a desired energy saving and consumption reducing replacement for traditional fabric filters. However, most of the used raw materials for the MB are petroleum-based polymers. Once these MB products are abandoned after having been used, it is difficult for them to be recycled and degraded, which causes again the environmental pollution and energy crisis. Fortunately, many biodegradable polymers have appeared, and their application can relieve the press from environment and energy.15–23 Some of them have appropriate spinnability and are used for the fabrication of MB.24–26
Poly (lactic acid) (PLA), mainly derived from starch, corn, sugar and some other renewable plant resources, is a typical eco-friendly bio-based polymer and has exhibited the vast commercial market prospects in the fibers, film and biomedical materials.24–27 Since being used for melt-blowing process (trade name Ingeo 6252D and 6201D), PLA displays the available process ability and service properties.25,26,28 Especially, as the air filter, PLA MB not only process excellent filtering capability, but reduce the waste disposal and meet the requirements of sustainable development. However, the fast development of modern industry and agriculture make the pollutant compositions in the air more and more complex, which raises the higher demands for the air filtration products. Thus, PLA MB air filter need to further enhance its air filtration performances and mechanical properties to face new challenges.
Happily, there have been existed some approaches to the improvement of the filtration performances of MB air filters. For example, some reports show that the filtration efficiency of MB can be enhanced by reducing the pore size among fibers and fiber diameter in MB web.29–31 These changes in geometric structures of MB web are involved in the adjustment of production process or realized by the introduction of some inorganic particles, such as graphite oxide (GO), reduced-GO (rGO) and inorganic nanotubes.32–38 However, small pore size and fine fiber diameter increase filtration resistance of MB, which will cut the service life of air filters. 29 The other popular way to improve the filter efficiency of MB is the electret treatment.39–43 The electret treatment enhances the electrostatic attraction of the MB surface for PM in the air when the PM is close to MB, which is helpful to the adsorption, collection and interception of MB filters for PM. Unfortunately, the electret treatment effect should gradually weaken until vanish as time goes on because the electrostatic charge dissipates as more and more PM are deposited on the filter. Besides, the increase in ambient humidity can also accelerate the charge dissipation. Thus, the electret MB air filters cannot remain the long-term high filtration efficiency. 44
At present, it has been found that once the air filter is endowed with magnetism, its filtration performances should be enhanced because the presence of magnetism increases the attraction and collection capacities of the filter to the dust.44,45 Ferroferric oxide (Fe3O4) is a kind of magnetic inorganic nanoparticle and has aroused many scholars’ interest on account of its advantages, such as superior magnetic responsiveness, nontoxic and inexpensive. 46 Blending PLA with Fe3O4 nanoparticles is a feasible way to make PLA MB obtain the magnetism. However, the existing studies on the magnetic PLA materials are mostly focused on medicine carrier, microwave or absorbing materials.47–49 Few of reports are about the fabrication of magnetic PLA MB and its application on air filtration. Additionally, the natural incompatibility between the hydrophilic Fe3O4 nanoparticles and hydrophobic PLA matrix need to be considered for the fabrication of magnetic PLA MB with outstanding filtration performances. 50 Many strategies have been used to increase the interfacial interaction between inorganic nanoparticles and PLA matrix, including the physical surface pretreatment, the addition of the coupling agent or the surfactant, and the interfacial chemical modification.51–54 Among them, grafting PLA molecular chains onto the surface of inorganic nanoparticles via ROP strategy has been proven to have a satisfying effect on the improvement of the interfacial compatibility because the grafted molecular chains on the surface of inorganic nanoparticles can form the strong interaction with PLA matrix.55–57
The aim of this paper is to provide some basic and original data to the development of a magnetic degradable PLA-based MB filter. In order to improve the interfacial compatibility between PLA matrix and Fe3O4 nanoparticles, Fe3O4-g-poly (L-lactic acid) (PLLA) nanohybrids was firstly synthesized by grafting PLLA molecular chains onto the Fe3O4 surface via ROP and characterized. Then PLA/Fe3O4-g-PLLA composite masterbatches with different mass ratios were prepared by melt-blending and processed into MB by use of a micro-experiment melt-blowing machine. The influence of Fe3O4-g-PLLA nanohybrids on the thermal and rheological behaviors and morphologies of PLA/Fe3O4-g-PLLA composite masterbatches were analyzed, and the magnetism, geometric structures and air filtration performances of PLA/Fe3O4-g-PLLA composite MB were investigated.
Experimental
Materials
A commercial PLA (6252D) with a D-isomer lactide content of about 6.86% were supplied by NatureWorks LLC (USA). Its density was 1.24 g·cm−3, and the number average molecular weight (Mn) was about 105 g·mol−1. L-Lactide (with the purity in excess of 99.5%) was provided by the Shenzhen Bright China Industrial Co., Ltd (China). Fe3O4 nanoparticles with the mean particle diameter of about 20 nm and Tin (II) 2-ethylhexanoate (Sn(Oct)2) were purchased from the Shanghai Macklin Biochemical Co., Ltd, respectively. 3-(Trimethoxysilyl) propylmethacrylate (KH570) was supplied by Hangzhou Feidian chemical company. Ethanoic acid, xylene, dichloromethane, absolute ethanol and glacial acetic acid (HAc) were provided by the Hangzhou Gaojing Fine Chemical Industry Co., Ltd, respectively. All of these chemicals used in this work were the analytical grade. Additionally, both xylene and dichloromethane were dried by calcium hydride before being used.
Synthesis of Fe3O4-g-PLLA nanohybrids
Prior to ROP, the surface of Fe3O4 nanoparticles were firstly modified by KH570. The modified procedure could be briefly summed up as follow: 1 mL KH570 was injected into a 500 mL flask containing the mixed solution of 300 mL absolute ethanol and deionized water (the volume ratio = 3:1), and the pH value of the mixed solution was adjusted to 3.5–4.0 by ethanoic acid. Then, the obtained solution was stirred at 50°C for 10 min for the purpose of pre-hydrolysis of KH570. Subsequently, 3 g Fe3O4 powder was put into the above solution and dispersed by using an ultrasonic homogenizer (JY 98-N, Ningbo Scientz Biotechnology Co., Ltd, China) with a power of 800 W for 10 min. After that, the suspension was transferred into a three-necked flask and stirred in oil bath at 70°C for 5 h in the nitrogen atmosphere. The precipitation was filtered out from the suspension by centrifugation, washed with excess absolute ethanol for three times, and dried in the vacuum at 50°C for 12 h. The modified Fe3O4 nanoparticles by KH570 were gained.
Next, Fe3O4-g-PLLA nanohybrids were synthesized via ROP. 10 g L-lactide and 1 g modified Fe3O4 by KH570 were put into 40 mL dried xylene in a four-neck round-bottom flask, respectively. At the same time, 0.05 g stannous iso-caprylate (Sn(Oct)2) was also added into the dried xylene as catalyst. The suspension was stirred for 12 h at 125°C in the nitrogen atmosphere. PLLA was grafted onto the Fe3O4 surface by reacting with the hydroxyl groups provided by modified Fe3O4 by KH570. After the polymerization had been finished, the resulting suspension was cooled to the room temperature, dissolved by dichloromethane and precipitated by the excess absolute ethanol. The dissolution and precipitation were repeated three times. Then some black precipitate could be obtained and filtered out by Millipore polytetrafluoroethylene membrane with a mean pore size of 0.22 μm. After that, the black precipitate was washed with the chloroform. The washing, centrifugalizing and filtering processes were repeated at least four times to remove the unreacted L-lactide and free PLLA that was not grafted onto the Fe3O4 surface. The final solid product, Fe3O4-g-PLLA nanohybrids, was dried in the vacuum oven at 60°C for 24 h. The above modified and synthetic routes were described in Figure 1.

The synthetic route of Fe3O4-g-PLLA nanohybrids.
Preparation of PLA/Fe3O4-g-PLLA composite masterbatches and their MB
PLA/Fe3O4-g-PLLA composite masterbatches were obtained by three steps. Prior to processing, pure PLA resins and Fe3O4-g-PLLA nanohybrids were dried in the vacuum drying oven at 80°C for 12 h. Then, PLA were mixed with Fe3O4-g-PLLA nanoparticles in different masse ratios by means of a highspeed mixer (Nanjing GAINT Machinery Co., Ltd, Nanjing, China). After that, the PLA/Fe3O4-g-PLLA composite masterbatches with different mass ratios were prepared by the melt-blending method with a twin-screw extruder (TSE-30A, Nanjing Ruiya Extrusion System Co., Ltd, China) at 180°C and 300 rpm. The melt-blending method has the environmentally friendly advantage. This method can also be used in preparation of composites with negative electromagnetic parameters, which has potential application in electronic device.58,59 The screw diameter (D) of the extruder was 25 mm and the length-diameter ratio (L/D) was 40. Pure PLA was also processed as a control sample following the same process for the normalized evaluation. Finally, pure PLA and PLA/Fe3O4-g-PLLA MB were fabricated by a micro-experiment melted spray machine (Hangzhou Dian-Zi Univeristy, Hangzhou, Zhejiang, China) with a single hole. The die diameter was 0.5 mm, and the die temperature was near 220°C. The temperature of hot air was approximately 260°C with a drawing air flow press of 0.2 MPa, and the collector distance was 20 cm. In this paper, the PLA/Fe3O4-g-PLLA composites masterbatches with 0, 0.5, 1 and 2 wt% Fe3O4-g-PLLA content were signed as C0, C0.5, C1 and C2, respectively, and the corresponding MB were marked as MB0, MB0.5, MB1 and MB2, respectively. The preparation process of PLA/Fe3O4-g-PLLA composite masterbatches and MB was shown in Figure 2.

The preparation process of PLA/Fe3O4-g-PLLA composite masterbatches and their MB.
Methods
Fourier transform infrared spectroscopy (FTIR)
FTIR spectra were performed using a Nicolet 5700 FTIR spectrometer from 400 to 4000 cm−1 with a resolution of 4 cm−1 and an accumulation of 32 scans. Before the measurement, pure Fe3O4 nanoparticles, modified Fe3O4 and Fe3O4-g-PLLA nanohybrids were mixed with potassium bromide (KBr) powder and pressed into the thin disk specimens, respectively.
Wide-angle X-ray diffraction (XRD)
XRD patterns were carried out by a D8 Discovery X-ray diffractometer, using Cu Kα (λ=1.5406 Å) radiation source in the diffraction angle (2θ) range of 5° to 70° by the steps of 5 deg·min−1 at 40 kv and 40 mA.
Hydrogen nuclear magnetic resonance (1HNMR)
1HNMR spectra were executed with a Bruker Ascend400 nuclear magnetic resonance spectrometer at a frequency of 400 MHz, Deuterated chloroform (CDCl3) and tetramethylsilane (TMS) were used as the solvent and the internal calibration agent, respectively.
Differential scanning calorimeter (DSC)
The thermal behaviors of pure PLA and PLA/Fe3O4-g-PLLA composite masterbatches were investigated by the Perkin-Elmer DSC8000. All of the samples were heated from 25 to 200°C at 100°C·min−1 and stayed at 200°C for 3 min to erase the previous thermal history, then cooled down to 25°C at 10°C·min−1. After that, the samples were reheated from 25°C to 200°C at 10°C·min−1, and the second heating curves were recorded. All of the measurements were performed under the nitrogen atmosphere. The cold crystallization enthalpy (△Hcc) and the melting enthalpy (△Hm) of the samples could be obtained from their second heating curves. The relative degree of crystallinity (Xc) was calculated using the following equation (1). 60
where △H0 was the fusion enthalpy of the completely crystalline PLA, the value was taken as 93 J·g−1 from the literatur, 61 C was the weight fraction of PLA in the composite masterbatches.
Rheological behaviors
The melt flow index (MFR) of pure PLA and the composite masterbatches was evaluated by a P/90L-Z1B1 melt flow indexer. The testing temperature was 180°C to 230°C under the loading of 2.16 kg, the inner diameter of the die was about 2 mm, and the preheating time of the samples was 2 min. The dynamic rheological behaviors were determined by Anton Paar Physica MCR 301 rheometer at 180°C and 190°C in the frequency scan range of 0.1 to 500 rad·s−1, respectively. The measurements were performed in the linear viscoelastic region with dynamic oscillatory mode. The diameter of the samples was 25 mm, and the gap of the parallel cone-plate was about 0.4 mm.
Scanning electron microscopy
Scanning electron microscopy (SEM) was performed by use of a Carl Zeiss AG ULTRA55 instrument. The morphologies of the fracture surface of pure PLA and the composite masterbatches and the surface of the corresponding MB were taken at an acceleration voltage of 3 KV. Prior to the measurement, the samples of pure PLA and composite masterbatches were prepared by snapping in a liquid nitrogen environment. All of the samples were sputtered with gold to provide enhanced conductivity.
Magnetic performance
The VSM 7407 micro-vibration sample magnetometer was used to measure the magnetic hysteresis loops of PLA/Fe3O4-g-PLLA composite MB in the solid state with an external 2 T magnetic field at room temperature.
The geometric structures
The pore size and distribution of pure PLA and the composite MB was characterized via a PSM 165/H pore size meter. All of the samples were cut as a circle with the diameter of 1 cm, then placed in the instrument and tested after immersion with Topas standard solution.
The filtration performances
The air permeability and filtration resistance of pure PLA and the composite MB were analyzed by the YG461E-III automatic permeability meter at the temperature of 25°C and the relative humidity of 60%. Additionally, the WPS-1000xp electron-laser particle spectrometer was used to measure the filtration efficiency of MB. According to GB/T 6165-2008, sodium chloride (NaCl) with in the particle size range of 0.3 to 0.5 μm and the concentration of 10,000 grains·m−3 was used as the testing aerosol at 5.3 cm·s−1 filtration velocity in the air. Five replicates were performed on each sample and the average values were recorded.
The mechanical properties
Tensile properties of pure PLA and the composite MB were determined according to GB/T 24218.3-2010. All of the samples were conditioned for 48 h at 23°C and 50% relative humidity in a constant temperature and humidity chamber before the measurements. The tensile data were obtained using an Instron 3369. The samples were cut into rectangular shapes of 20 mm wide and 900 mm long. The length of the samples was parallel to the longitudinal direction of MB. The initial grip separation and the crosshead speed were set at 50 mm and 10 mm·min−1, respectively. The reported longitudinal tensile strength and elongation at break were the average values of at least five samples.
Results and discussion
Characterization of Fe3O4-g-PLLA nanohybrid
Figure 3(a) presented FTIR spectra of Fe3O4, modified Fe3O4 by KH570 and Fe3O4-g-PLLA nanohybrids. In Fe3O4 spectrum, there was an intense peak at around 594.7 cm−1, corresponded to the stretching vibration peak of Fe-O. The obvious peaks at around 1632.8 and 3429 cm−1 reflected the stretching and bending vibration peak of hydroxyl groups, respectively, which owes to the adsorption effect of Fe3O4 surface on water. Compared with Fe3O4, the spectrum of modified Fe3O4 exhibited two new weak absorption peaks at around 2922.9 cm−1 and 2848.5 cm−1, which reflects the stretching vibration peaks of methyl and methylene of KH570. Moreover, there appeared some new stretching peaks in 1091–1184 cm−1, which belongs to the stretching vibration peak of Si-O-C. These phenomena indicated that the surface of Fe3O4 nanoparticles were modified by KH570. For Fe3O4-g-PLLA nanohybrids, besides the characteristic peaks of modified Fe3O4, the strong peak at around 1754.2 cm−1 appeared, attributing to the carbonyl (C=O) stretching vibration of the ester group of PLLA. The peaks at around 1182.8 cm−1, 1130 cm−1 and 1088 cm−1 belonged to the symmetric O-C-O stretching vibrations of the ester group of PLLA. Thus, it could be believed that PLLA molecular chains were successfully grafted onto the modified Fe3O4 surface.

FTIR spectra (a). WAXD patterns (b) and 1H-NMR spectra (c) of Fe3O4, modified Fe3O4 and Fe3O4-g-PLLA nanohybrid.
This suggestion on the grafting reaction was also supported by the WXAD patterns observed in Figure 3(b). WAXD pattern of Fe3O4 exhibited several characteristic diffraction peaks at about 18.3°, 30.1°, 35.5°, 43.2°, 57.4° and 62.6°, corresponding to the (111), (220), (311), (400), (511) and (440) crystal planes of Fe3O4, respectively. 62 In WXRD pattern of modified Fe3O4 by KH570, all of the characteristic diffraction peaks belonging to Fe3O4 appeared, showing that the modification treatment did not destroy the crystalline structure of Fe3O4. For the Fe3O4-g-PLLA nanohybrids, there appeared two noticeable new peaks at about 16.6° and 19.0°, which is the characteristic diffraction peaks of PLLA. Thus, the results from WAXD analysis could also confirm that Fe3O4-g-PLLA nanohybrids were synthesized.
In order to ascertain the molecular weight of the grafted PLLA onto the modified Fe3O4 surface, the 1HNMR spectra of the Fe3O4-g-PLLA nanohybrids was tested and shown in Figure 3(c). The chemical shift peak marked by “A” at around 5.16 ppm belonged to methyl and methine protons in the structure unit of PLLA. The peak marked by “a” at around 4.36 ppm was ascribed to the methine protons connected to the ester groups of PLLA within the molecular chain backbone and the hydroxyl groups at the chain end. The Mn value of the grafted PLLA molecular chains on the Fe3O4 surface was calculated by the equation (2). 50
where AA and Aa represented the integrated areas of the two chemical shift peaks “A” and “a”, respectively. According to the calculated result, it was confirmed that the Mn value of PLLA grafted onto the Fe3O4 surface was about 1.47×104 g·mol−1. The long chain molecules instead of monomers grafted on the Fe3O4 surface was more conducive to the formation of the strong interfacial interaction of Fe3O4 and PLA matrix.
The thermal behaviors of the composite masterbatches
DSC second heating scans of pure PLA and the composite masterbatches were plotted in Figure 4 and analyzed in order to reveal the effect of Fe3O4-g-PLLA nanohybrids on thermal behaviors of the composite masterbatches. Some thermal parameters were summarized in Table 1. It could be seen that the glass transition peak temperature (Tg) of pure PLA were around 59.95°C. Compared with pure PLA, the Tg values of PLA/Fe3O4-g-PLLA composite masterbatches were almost constant, indicating that the addition of Fe3O4-g-PLLA did not affect the motion of PLA molecule chains. With the temperature increasing, every DSC curve showed an exothermic peak corresponding to the cold crystallization process of PLA because of the mobility and the rearrangement of PLA molecule chains. The cold crystalline peak temperature (Tcc) of pure PLA was about 122.63°C. When the content of Fe3O4-g-PLLA were 0.5 wt% and 1 wt%, the Tcc values of composite masterbatches slightly increased. However, when the content of Fe3O4-g-PLLA reached 2 wt%, the Tcc value of composite masterbatches was close to that of pure PLA again. Moreover, the Xc value of pure PLA was 2.91%. After Fe3O4-g-PLLA were added, the Xc values of composite masterbatches declined from 1.84% to 1.05%. There were two aspects might be taken into account to explain this phenomenon. On the one hand, the addition of Fe3O4-g-PLLA disrupted the regularity of the PLA crystalline structure and caused the formation unperfect crystalline structure. On the other hand, unlikely other inorganic nanoparticles with a crystalline nucleation capacity, Fe3O4-g-PLLA might act as the blocking sites to hinder the growth of PLA crystals.60,63 When the heating temperature continued to rise, two distinct melting peaks were observed for all of the samples. The dual melting behavior could be explained by the presence of the multiple crystal structure and melt-recrystallization during the second heating process. 64 The lower melting peak temperature (Tm1) reflected the melting process of some small crystals with unperfect structure, while the higher melting peak temperature (Tm2) was regarded as the main melting behaviors of the samples. For pure PLA and the composite masterbatches, their Tm2 values were 164.5°C or so, indicating that the incorporation of Fe3O4-g-PLLA did not change the melting behaviors of the composite masterbatches.

DSC second heating scan curves of pure PLA and PLA/Fe3O4-g-PLLA composite masterbatches.
Thermal behaviors parameters of pure PLA and PLA/Fe3O4-g-PLLA composite masterbatches.

The rheological behaviors of the composite masterbatches
MFR might roughly reflect the melt fluidity of polymer materials. Figure 5(a) illustrated MFR of pure PLA and the composite masterbatches in the considerable processed temperature range. MFR of pure PLA increased from 78.15 g·10 min−1 to 370.90 g·10 min−1 when the testing temperature rose from 180°C to 220°C. Compared with pure PLA, MFR of the composite masterbatches decreased, meaning that the melt fluidity of PLA molecular chains slowed down due to the addition of Fe3O4-g-PLLA. It was suggested that Fe3O4-g-PLLA took up part of free volume and shrank the motion space of PLA chains. Besides, MFR of the composite masterbatches increased with the testing temperature increasing because the higher temperature provided the more energy for the motion of molecular chains. MFR could intuitively evaluate the spinnability of the polymer resins. Generally, the required MFR for PLA MB should be higher than 150 g·10 min−1. It was seen in Figure 5(a), when the testing temperature reached 210°C or above, MFR of both pure PLA and the composite masterbatches were more than 150 g·10 min−1. Thus, though the addition of Fe3O4-g-PLLA decreased MFR of the composite masterbatches, the appropriate processing temperature of our MB still could be selected in the range of 210°C to 230°C.
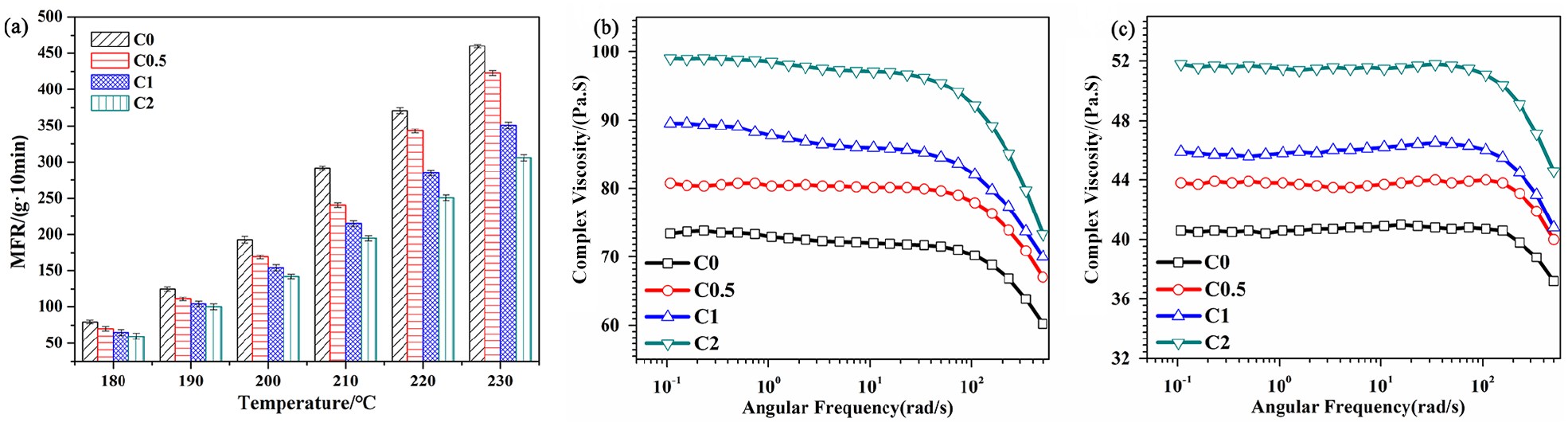
The MFR bar chart (a). The complex viscosity versus angular frequency curves of pure PLA and PLA/Fe3O4-g-PLLA composite masterbatches at 180°C (b) and190°C (c).
The processing dynamics and web structure of MB are influenced by the rheological properties of masterbatches. 62 The rheological behaviors of pure PLA and the composite masterbatches at 180°C and 190°C were shown in Figure 5(b) and (c), respectively. The complex viscosity of the samples kept constant at low angular frequency, which exhibits the characteristic of Newtonian plateau. However, their complex viscosity decreased when the angular frequency was more than 100 rad·s−1, which reveals a frequency dependent behavior of “shear thinning”. When the testing temperature rose from 180°C to 190°C, the complex viscosity of the samples reduced. Moreover, compared with pure PLA, the complex viscosity of the composite masterbatches increased with the Fe3O4-g-PLLA content increasing at two studied temperatures, which confirms the conclusion from MFR measurements that the addition of Fe3O4-g-PLLA could reduce the flow orientation of PLA chains.
Morphologies of the composite masterbatches and MB
The fracture morphologies of pure PLA and the composite masterbatches were observed through SEM and shown in Figure 6. Seen from Figure 6(a), the fracture surface of pure PLA was flat and homogeneous structure. In Figure 6(b–d), when Fe3O4-g-PLLA was added, the morphologies of the composite masterbatches were typical “sea-island” structure. Fe3O4-g-PLLA, marked by red circles in Figure 6(b–d), could uniformly disperse in PLA matrix with the diameter of about 0.1 to 0.4 μm even if its content reached 2 wt%. In our previous research, 65 when Fe3O4 was directly added in PLA matrix, some small cracks were seen at the interface of these two compositions. And some holes were able to be observed on the fracture surface of PLA/Fe3O4 blends because it was easy for Fe3O4 to fall off from the PLA matrix during the brittle fracture process. These phenomena indicated the poor interfacial compatibility between Fe3O4 nanoparticles and PLA matrix. Herein, there were no obvious cracks or holes on fracture surface of the PLA/Fe3O4-g-PLLA composite masterbatches, which means the good interfacial interaction between Fe3O4-g-PLLA and PLA matrix. The reason is that once PLLA molecular chains are grafted on the surface of Fe3O4, the strong interfacial interaction force of Fe3O4-g-PLLA and PLA was formed due to the physical entanglement of the molecular chains of PLLA and PLA matrix.

SEM images of fracture surfaces of pure PLA and PLA/Fe3O4-g-PLLA composite masterbatches. (a) C0. (b) C0.5. (c) C1. (d) C2.
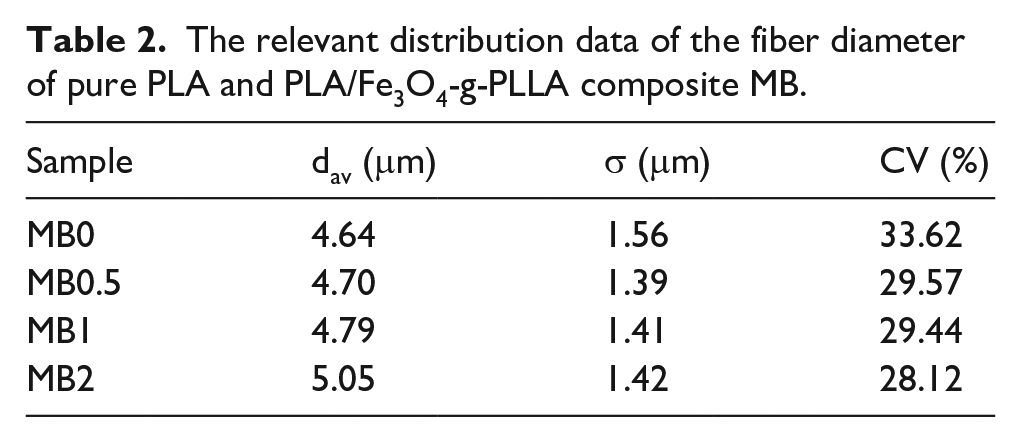
SEM images of the surface of pure PLA and PLA/Fe3O4-g-PLLA composite MB were exhibited in Figure 7(a–d). Their fiber diameter distribution was analyzed by a measurement tool named Image J Pro according to Ellison’s work 66 and shown in Figure 7(a′–d′). The mean fiber diameter (dav) of the samples were presented in Table 2, and the variance (σ) and the coefficient of variation (CV), obtained from the curve fitting of Figure 7(a′–d′), were also listed in Table 2. In Figure 7(a), the fiber surface of pure PLA MB was smooth. For the composite MB, with the increasing of Fe3O4-g-PLLA content, some small protrusions, marked by the red circles in the Figure 7(b–d), appeared on the fiber surface, which makes the fiber surface slightly rough. In Table 2, the mean fiber diameter (dav) of pure PLA MB was about 4.64 μm. The dav values of the composite MB had a negligible increase. Generally, the rough fiber surface in MB web should lead to an increase in the fiber diameter. The larger fiber diameter, the lower filtration efficiency of MB. However, for our composite MB, the rough surface did not sharply change the dav values, which was similar to the conclusion of Tan’s paper. 67
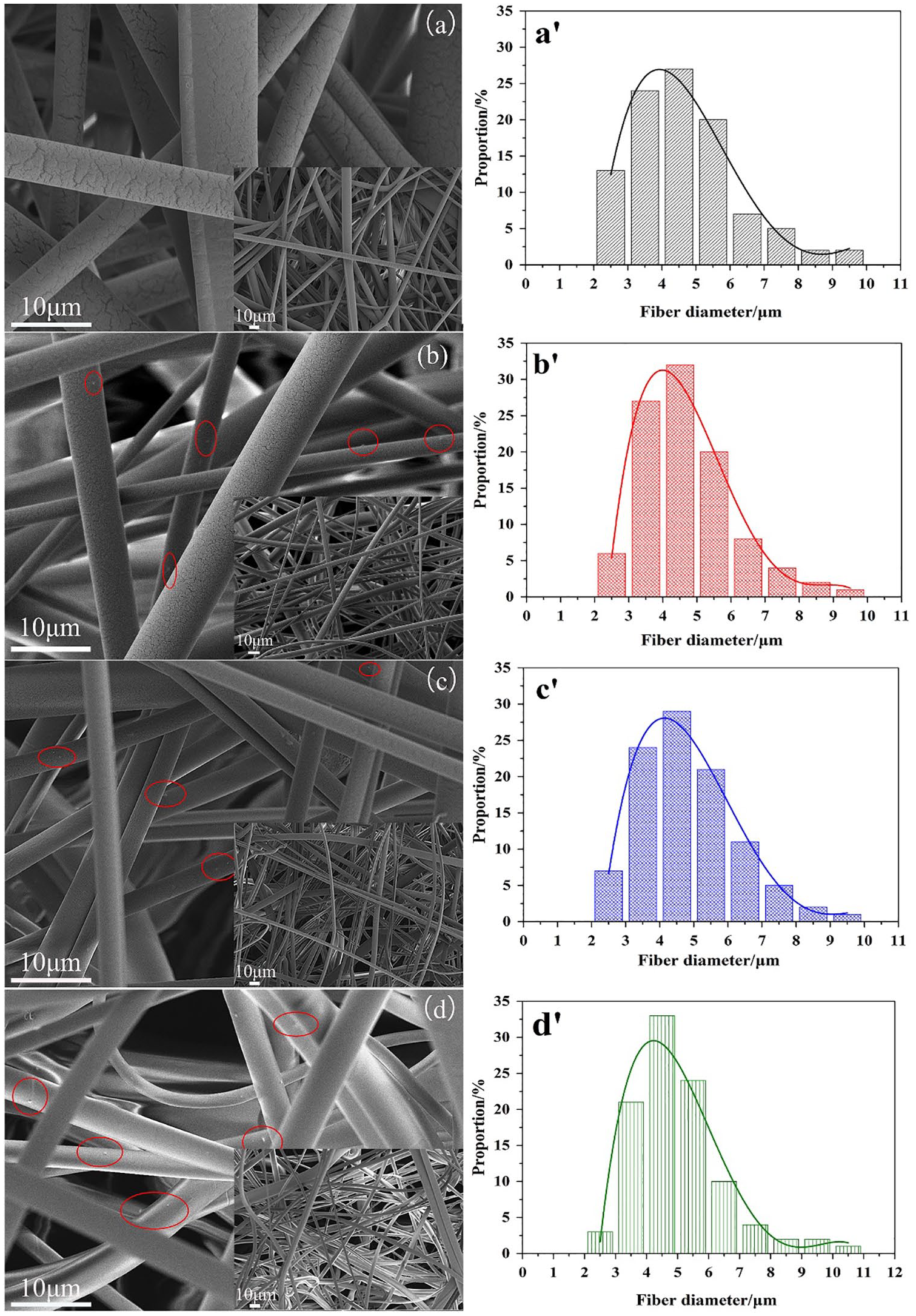
SEM images of the surfaces of pure PLA and PLA/Fe3O4-g-PLLA composite MB. (a) MB0. (b) MB0.5. (c) MB1. (d) MB2. The fiber diameter distribution of the MB. (a′) MB0. (b′) MB0.5. (c′) MB1. (d′) MB2.
The relevant distribution data of the fiber diameter of pure PLA and PLA/Fe3O4-g-PLLA composite MB.
Magnetic performance of the composite MB
The magnetization hysteresis loops of PLA/Fe3O4-g-PLLA composite MB were shown in Figure 8. As we all known, pure PLA MB had nonmagnetic nature and no magnetic response. However, the composite MB presented magnetic characteristic and achieved their saturation magnetization values (Ms), which is the maximum magnetization intensity corresponding to the value of the atom magnetic moment completely displayed in the same direction. With the content of Fe3O4-g-PLLA increasing from 0.5 wt% to 2 wt%, the Ms values of the composite MB increased from 0.044 to 0.101 emu·g−1. Of course, the magnified Figure 8 showed that the hysteresis curves of the composite MB had the certain remanence (Mr), which should cause loss of magnetism to some extent. But, the Mr values of the composite MB were no more than 0.01 emu·g−1, which does not obviously impair the paramagnetism of MB. Thus, the composite MB still owned the certain magnetic response after the melt-blowing processing.

The magnetization hysteresis loops of PLA/Fe3O4-g-PLLA composite MB.
The geometric structures and filtration performances of the composite MB
The geometric structures of MB, including the pores size and distribution of the fiber web, have an important effect on the filtration performances of MB. The pore size and distribution of pure PLA and PLA/Fe3O4-g-PLLA composite MB were plotted in Figure 9(a). As seen from Figure 9(a), the mean pore size of pure PLA MB was about 34.91 μm, and its pore size distribution was in the range of 35 to 40 μm. When the Fe3O4-g-PLLA content changed from the 0.5 wt% to 2 wt%, the mean pore size of the composite MB increased from the 40.8 μm to 42.6 μm, and their pore size distribution broadened. The increasing pore size and distribution were attributed to the decrease of the self-adhesive points among the fibers in the MB web caused by the rougher fiber surface of the composite MB.

The mean pore size and distribution (a). The filtration performances (b) of pure PLA and PLA/Fe3O4-g-PLLA composite MB.
The air permeability, filtration efficiency and resistance of pure PLA and PLA/Fe3O4-g-PLLA composite MB were illustrated in Figure 9(b). The air permeability of pure PLA MB was about 321.6 mm·s−1. With the increasing of the Fe3O4-g-PLLA content, the air permeability of the composite MB increased, while their filtration resistance decreased. The reason was that as a result of the input of Fe3O4-g-PLLA, the larger mean pore size and the wider pore size distribution made PM accompanied with air easier to pass through the MB web. The increase in air permeability and the decrease in filtration resistance could relieve the pressure on the MB web during air filtration process, which is favorable to expend the service life of the filtration materials. Furthermore, the filtration efficiency of pure PLA MB was about 32.16%. When Fe3O4-g-PLLA with 0.5 wt% content was added, the filtration efficiency of the composite MB increased to 36.44%. Several well-known mechanisms have been accepted to explain the filtration process of the nonwoven fabrics: inertial impaction, interception, diffusion, electrostatic attraction, and so on. 40 The researches have shown that the instantaneous high pressure would generate when the PM in the air was close to the magnetic air filter, which enhances the electrostatic adsorption and traction of fabric to PM and results in the higher filtration efficiency.44,45 Besides, the magnetic air filters can effectively filter metallic oxide dust in the air, which also could improve the filtration efficiency. 44 However, when the Fe3O4-g-PLLA content continued to increase, the filtration efficiency of the composite MB started to decline and even slightly lower than that of pure PLA MB when Fe3O4-g-PLLA content was 2 wt%. Compared with the previously reported PLA MB air filter, 40 the filtration efficiency of our composite MB did not markedly increase. One of the reasons was that the increasing pore size and distribution of the MB web brought about the decreasing interception rate of the composite MB for PM. Additionally, the magnetic response intensity of the composite MB was not high enough due to the low adding content of Fe3O4-g-PLLA. The filtration efficiency of our composite MB will be tired to enhance by increasing the adding content of Fe3O4-g-PLLA and optimizing fabrication technique to in the following study.
Mechanical properties of the composite MB
The mechanical properties of pure PLA and PLA/Fe3O4-g-PLLA composite MB were tested by tensile experiments, and their longitudinal tensile strength and elongation at break were listed in Table 3. Tensile strength and the elongation at break of pure PLA MB were 0.54 ± 0.10 N and 29.26 ± 2.06%, respectively. Compared to pure PLA MB, tensile strength of the composite MB increased depending on the content of Fe3O4-g-PLLA at different levels and reached to the maximum when the content of Fe3O4-g-PLLA was 1 wt%. The increase in tensile strength was suggested by the reinforcement effect of Fe3O4-g-PLLA as a kind of inorganic rigid particles and good compatibility between Fe3O4-g-PLLA and PLA matrix. However, tensile strength of the composite MB dropped to 0.75 ± 0.08 N when the content of Fe3O4-g-PLLA was up to 2 wt%, which is due to the reduce in the self-adhesive points among the fibers in the MB web. On the other hand, the elongation at break of the composite MB was higher than that of pure PLA MB and had the highest value when Fe3O4-g-PLLA with 1 wt% content was added, which may be considered as a result of the larger pore size of the composite MB. Moreover, the inhibition of Fe3O4-g-PLLA to the crystallization of PLA also contributed to the increase in the elongation at break.
The mechanical properties parameters of pure PLA and PLA/Fe3O4-g-PLLA composite MB.

Conclusion
In this paper, PLA was expected to endow the magnetism by means of being mixed with Fe3O4 nanoparticles in order to obtain a new functional PLA-based MB for air filtration. In consideration of the poor interfacial compatibility between PLA matrix and Fe3O4, the Fe3O4 surface was grafted by PLLA chains via ROP. Then, PLLA/Fe3O4-g-PLLA composite masterbatches with different mass ratios were prepared by the melt-blending and made into the corresponding MB by a micro-experiment melt-blowing machine. The results illustrated that Fe3O4-g-PLLA were successfully synthesized. The addition of Fe3O4-g-PLLA did not change the Tg, Tcc and Tm2 of PLA/Fe3O4-g-PLLA composite masterbatches. With the Fe3O4-g-PLLA content increasing, the melt fluidity of the composite masterbatches reduced. However, they still could achieve the appropriate MFR for the good processibility in the range of 210°C to 230°C. Through the observation of the morphologies, it was revealed that the Fe3O4-g-PLLA were uniformly dispersed in PLA matrix and formed good compatibility with the matrix. On account of the addition of Fe3O4-g-PLLA, PLA/Fe3O4-g-PLLA composite MB got magnetism, the fiber surface became slightly rough, the pore size among fibers got bigger, and pore size distribution broadened. With the increasing of Fe3O4-g-PLLA content, the air permeability of the composite MB increased, while their filtration resistance decreased. The filtration efficiency of the composite MB reached the maximum when the adding content of Fe3O4-g-PLLA was 0.5 wt%. The input of Fe3O4-g-PLLA increased the longitudinal tensile strength and elongation at break of the composite MB.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Zhejiang Provincial Natural Science Foundation of China (Grant No. LY19E030011).


