Abstract
In this work, a phase-change energy storage nonwoven fabric was made of polyurethane phase-change material (PUPCM) by a non-woven melt-blown machine. Polyethylene glycol 2000 was used as the phase transition unit and diphenyl-methane-diisocyanate as the hard segment to prepare PUPCM. Thermal stability of the PUPCM was evaluated through thermal stability analysis. The performance of pristine PUPCM was determined by Fourier transform infrared spectroscopy and differential scanning calorimetry to analyze the spinning technology of spinning temperature and the stretching process. Phase-change energy storage nonwoven fabric (413.22 g/m2) was prepared, and the morphology, solid–solid exothermic phase transition, mechanical properties, and the structures were characterized. The enthalpy of solid–solid exothermic phase transition reached 60.17 mJ/mg (peaked at 23.14°C). The enthalpy of solid–solid endothermic phase transition reached 67.09 mJ/mg (peaked at 34.34°C). The strength and elongation of phase-change energy storage nonwoven fabric were found suitable for garments and tent fabrics.
Introduction
Phase-change material (PCM) refers to a material that absorbs or releases large latent heat by phase transition between different phases of the material itself (solid–solid phase or solid–liquid phase) at certain temperatures.1–3 PCMs have high heat storage densities and melting enthalpies, which enable them to store relatively dense amounts of energy under the condition of almost constant temperature.4,5 PCMs are widely used in sustainable thermal energy storage and management,6,7 thermal energy, 8 waste heat recovery, 9 building energy conservation,10,11 solar energy utilization,12,13 thermal control of electronic devices, and other fields.14,15 Telkes and Raymond 16 first carried out research on PCM in 1949. PCMs were first used by the aerospace industry: Hale, Hoover, and O ′Neill 17 designed the thermal insulation system for the space vehicle. The earliest PCMs were inorganic salt hydrates 18 which store heat and release it through hydration and dehydration of such substances. Organic PCMs have a wide range of phase-change temperature (−5∼190°C) and can be adjusted by changing the phase-change functional group of organic matter. Known organic PCMs mainly consist of paraffin, polyethylene glycol, fatty acids, and their composite materials. These organic PCMs mainly store and release energy through solid–liquid phase transition. However, the before and after state changes of PCMs are often accompanied by liquid leakage, thus severely limiting their application. Organic PCMs (polyols, polyethylenes, polyurethanes, etc.) which store and release latent heat during solid–solid phase change have attracted more and more attention due to their special properties. 19
Intelligent temperature-regulating textile is a new development direction of the textile industry. With the development of science and technology, outdoor and extreme sports are favored by more and more people with the improvement of people’s living standard, and the demand for intelligent temperature-regulating textiles is also increasing. Intelligent temperature-regulating textiles can regulate the ambient temperature around the human body, isolate the human body from the surrounding atmospheric environment, and maintain a suitable environment for the skin on the human body even under the extremely harsh surrounding environment. 20
Pause 21 put forward the concept of the dynamic thermal insulation in 1995. After this, the active insulation textile material is called dynamic heat storage textile material or thermal regulation functional textile material. The suitable temperature range for the human body is 18°C–35°C, so the phase-change temperature of temperature-adjusted textiles should also be within this range. 22 N-tetradecane, n-hexadecane, n-eicosane, and their mixtures have been successfully applied in the field of PCMs for clothing. 23 At present, temperature-adjusted textiles are mainly prepared by encapsulating PCMs into microcapsules and adding it to textiles.24,25
Since Bryant and Colvin 26 developed phase-change energy storage fibers using microencapsulated PCMs, they have investigated a variety of methods to continuously increase the content of microencapsulated PCMs in the fibers in order to improve the thermal properties without affecting the mechanical properties. After that, researchers began to apply microcapsule PCMs to wet spinning of viscose fiber, acrylic fiber, and other fibers and then extended it to melt spinning to prepare phase-change energy storage polyethylene fiber and polypropylene fiber.27–30 The structure of the microcapsule determines that the addition amount of the microcapsule should not be too large (< 10%), and the processing technology needs to be precisely controlled, otherwise the microcapsule will rupture and affect the performance. 31 Based on them, microcapsule PCMs have been widely used in textiles, such as diving suits, heat insulation pads, temperature-controlled leather, etc.32,33
The addition of microencapsulated PCMs have some influence on the mechanical properties of the fiber more or less. In order to reduce the influence of the addition of PCMs on the fiber, researchers focused on the direct fibrillation or chemical grafting reaction of PCMs (to polymerize PCMs with fibrillable matrix materials). As a kind of PCMs with adjustable phase-change temperature, high enthalpy, and no toxicity, polyethylene glycol (PEG) has been widely studied in direct blending of phase-change fibers. However, due to the good water solubility of PEG, PCM fibers prepared by PEG have poor water resistance. To improve this problem, researchers shifted their focus from pure blends to graft polymerization of PEG with fiberable substrates, and the performance of PCM fibers prepared by graft polymerization depends on factors such as the molecular weight of PEG and the type of fiberable substrates. 34 In general, the addition of PEG through graft reaction can improve the thermal properties, hygroscopicity, and moisture regain of fibers.
In 2006, Hu 35 grafted copolymerized PEG with polyethylene terephthalate (PET), prepared PEG–PET fibers by melt spinning, and the phase-transformation properties of PEG–PET fibers were studied. The phase-transformation properties of the PCM fibers can be adjusted by controlling the molecular weight of PEG. The enthalpy of phase transformation reached 26.9 kJ kg−1, and the phase transformation temperature ranges from 19.6°C to 49.3°C, when the content of PEG in the fiber reached 45%.
In 2008, Meng and Hu
36
used bulk polymerization and PEG as a phase-change group to synthesize polyurethane phase-change material (PUPCM). PUPCM intelligent fiber was prepared by the melt-spinning method, and the performances were tested. The results showed that the prepared 100-dtex fiber only had a tenacity of 0.7 cN/dtex, too weak to suitable for garments. But the PUPCM intelligent fiber had large latent heat-storage capacity of about 100 J/g with a crystallizing temperature of 20.9°C and a melting temperature of 44.7°C. PUPCM is a block copolymer composed of flexible chain segments with large molecular weight and relatively small rigid chain segments with relatively small molecular weight. PUPCM was similar to ordinary PU in structure. Polyurethane (PU) was first discovered in 1937 by Professor O. Ayer.
37
It is a group of soft–hard alternate heterochain polymers, characteristic with repeat -NH-COO- groups.
38
The molecular structure formula of PU is shown in Figure 1, which is generated by the polymerization reaction between polyols and polyisocyanates. Polyol was the soft segment, and isocyanate and chain extender were the hard segment. It consists of a chain extender and isocyanate to form a hard segment, thus forming a hard segment microregion and serving as the backbone of PUPCM. The soft segment was composed of macromolecular polyols with phase-transition function, and the soft segment was used as the functional carrier of PUPCM. PUPCM is a kind of solid–solid phase-change energy storage material with high enthalpy, good thermal stability, adjustable phase-change temperature, and simple production process. Structure of polyurethane molecular chain and preparation schematics of PUPCM.
This work is mainly focused on the preparation of a phase-change energy storage nonwoven fabric (PCESNF). PUPCM was prepared with the aid of a chain extender 1,4 butanediol (BDO) and neutralizer triethylamine; polyethylene glycol was used as the phase-transition unit, diphenyl-methane-diisocyanate as the hard segment, and dibutyltin dilaurate as catalyst. Thermal stability of the PUPCM was evaluated through thermal gravimetric analysis (TGA). The performance of pristine PUPCM was determined by Fourier transform infrared (FTIR) and differential scanning calorimetry (DSC), and the spinning technology of spinning temperature and the stretching process were analyzed. Phase-change energy storage nonwoven fabric was prepared by a nonwoven melt-blown machine. And the morphology, mechanical properties, and structure were characterized. The enthalpy of solid–solid exothermic phase transition of PCESNF was evaluated through DSC.
Methods and materials
Preparation of polyurethane phase change material
The preparation schematics of PUPCM are shown in Figure 1. PEG (from Tianjin Guangfu Fine Chemical Research Institute, P.R. China) and diphenyl-methane-diisocyanate (MDI, Yantai Wanhua PU Co. Ltd, P.R. China) were dissolved in ethyl acetate (EA) (from Tianjin Guangfu Fine Chemical Research Institute, P.R. China) and heated to 60 C. Dibutyltin dilaurate (from Tianjin Guangfu Fine Chemical Research Institute, P.R. China) was added as a catalyst to prepare the prepolymer of PUPCM. Then BDO (from Tianjin Guangfu Fine Chemical Research Institute, P.R. China) was added to increase the PUPCM molecular chain, then cooled to 25–30°C, and ethylenediamine (from Tianjin Guangfu Fine Chemical Research Institute, P.R. China) was added to deplete the -NCO group. Finally, the EA was removed by vacuum distillation to produce PUPCM.
Before the reaction, the PEG must be vacuum-dried and dehydrated to remove as many water molecules as possible.
Preparation of phase-change energy storage nonwoven fabric
The spinning temperature is one of the most important parameters to determine the formation and the quality of the fibers in the spinning process. The softening temperature (ST 91.1°C) and flow temperature (FT 123.9°C) of PUPCM were measured by rheometer. The melt flow rate was 12.72 g/10 min (210°C/5 Kg), according to ASTMD1238. Production technology of the PUPCM melt-blown nonwoven web was based on a melt-blown machine (Shanxi Jingwei Chemical Fiber Machinery Co., Ltd). The nozzle diameter is 0.3 mm, and the number of holes is 360. The nozzle is designed with a 60-degree angle, and the die-to-collector distance (DCD) is 25 cm. The spinning process is shown in Figure 2. Spinning process of PCESNF.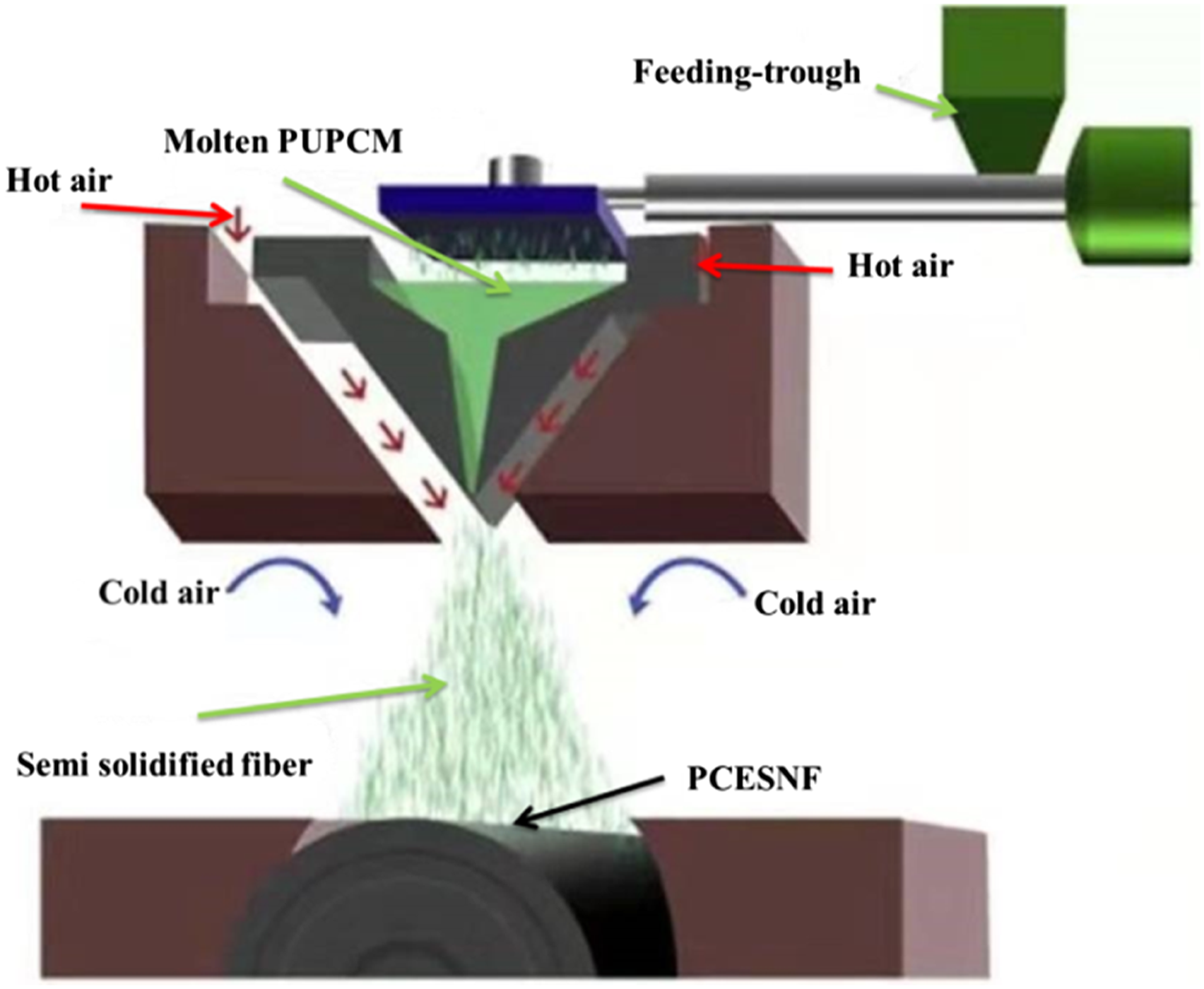
Process parameters of preparation of phase-change energy storage nonwoven fabric.

In a certain temperature range, with the increase of temperature, the mechanical properties of nonwoven fabrics are enhanced. In a certain range of hot air pressure, with the increase of hot air pressure, the mechanical properties of nonwoven fabrics are enhanced. In a certain flow range, with the increase of the melt flow rate, the mechanical properties of nonwoven fabric decrease. PUPCM has high-viscous flow-activation energy; therefore, the viscosity of the melt can be reduced and its fluidity can be improved by increasing the melt temperature, and the melt can be fully drawn by high-speed hot air flow. The bonding points between fibers increased, which enhanced the mechanical properties. However, the melt temperature should not be too high, high temperature makes the activity of macromolecular chain more intense and the probability of molecular chain fracture increases. On the other hand, when the viscosity reaches a critical low value, the friction between the melt and the spinneret is not enough to hold the filament, and the melt achieves a bad drafting; the fiber shrinks and appears “shot” phenomenon before cooled. Both of the two conditions will cause the mechanical properties of melt-blown nonwoven fabric to decrease.
The high spinning temperature can intensify the thermal decomposition of polymers and accelerate the movement of macromolecules in the fiber. The macromolecules are less prone to orientation due to the low-melt viscosity at high temperatures. All the spinning-zone temperatures of the extruder with stable spinning production and quenched air blow rate are presented in Table 1. The melt flow presented a uniform and continuous state and stable melt pressure. Therefore, it can be concluded that the PUPCM can be applied to spin nonwoven fabric with a reasonable range of temperature and gain good spinability.
Properties measurement
Fourier transform infrared spectra were obtained using an E55 FTIR spectrometer (Bruker, Germany). The physical properties, that is, shore hardness of PUPCM were measured as per ASTM D2240. The tensile strength and tear strength were measured as per ASTM D412 and ASTM D624, respectively. Differential scanning calorimetry and TGA of the samples were carried out using an STA449 C thermal analyzer (NETZSCH, Germany) synchronously at a heating rate of 10°C/min from room temperature to 600°C in the nitrogen. The thickness of nonwoven fabric was measured by an HD 10 thickness meter, and the average value is taken three times in different positions. Air permeability was carried out using a YG461E digital fabric air permeability meter (Ningbo textile instrument factory, China). Mechanical properties of the samples were carried out using an XQ 1 fiber tensile tester (Shanghai new fiber instrument Co., Ltd, China) with pneumatic clamp-operating and data processing function. The sample size was 100 mm × 20 mm, the Collet spacing was 50 mm, and the drawing rate was 250 mm/min.
Results and discussion
In preparation of raw materials for PUPCM, the water may exist in the PEG after contact with - NCO groups and water molecules will generate unstable carbamate, first, which then decomposes quickly and gives off carbon dioxide gas and generated from the amine. And the amine can further react with -NCO and generate urea; urea instead may react with -NCO to generate further biuret, water molecules, and isocyanate, and reactions as shown in Figure 3. These reactions will consume the -NCO group greatly, and the products produced by the substitution of urea and biuret quickly improve the viscosity of the system and reduce the liquidity of the system, and the carbon dioxide produced by the viscosity rise sharply after cannot escape from the system. Water in the system is easy to form aerogel, making normal polymerization is unable to continue. Therefore, before starting the reaction, the PEG must be vacuum-dried and dehydrated to remove as many water molecules as possible. Reaction of isocyanate with water.
Fourier transform infrared spectra analysis
The FTIR spectra of MDI, PEG2000, and PUPCM are shown in Figure 4. According to the mechanism of the reaction, the main change in the molecular structure is the transformation of the -N=C=O bonds to -N-C(=O)-O bonds. As shown in Figure 4(b), the IR absorption of the bending absorption peak of C-H appeared at 1470 cm−1, 1351 cm−1, 950 cm−1, and 839 cm−1, the stretching vibration peak for C-H appeared at 2880 cm−1, and the stretching vibration peak for C-O-C appeared at 1100 cm−1. The asymmetric stretching peak for -N=C=O appeared at 2270 cm−1 in Figure 4(a) MDI, and it almost disappeared in Figure 4(c) PUPCM, indicating that all -NCO bonds participated in the reaction with the PEG. The absorption peak of –OH at 3600 cm−1 appeared in both B and C, indicating the -OH in the system is excessive. As shown in Figure 4(c), the N-H deformation vibration peak of amides is at 1510 cm−1, the characteristic peak of C=O is at 1709 cm−1, and the hydrogen-bonded N-H stretching vibration peak is at 3400 cm−1. The above analysis shows that we have successfully synthesized PUPCM. Fourier transform infrared of PEG, MDI, and PUPCM (a) MDI, (b) PEG2000, and (c): PUPCM.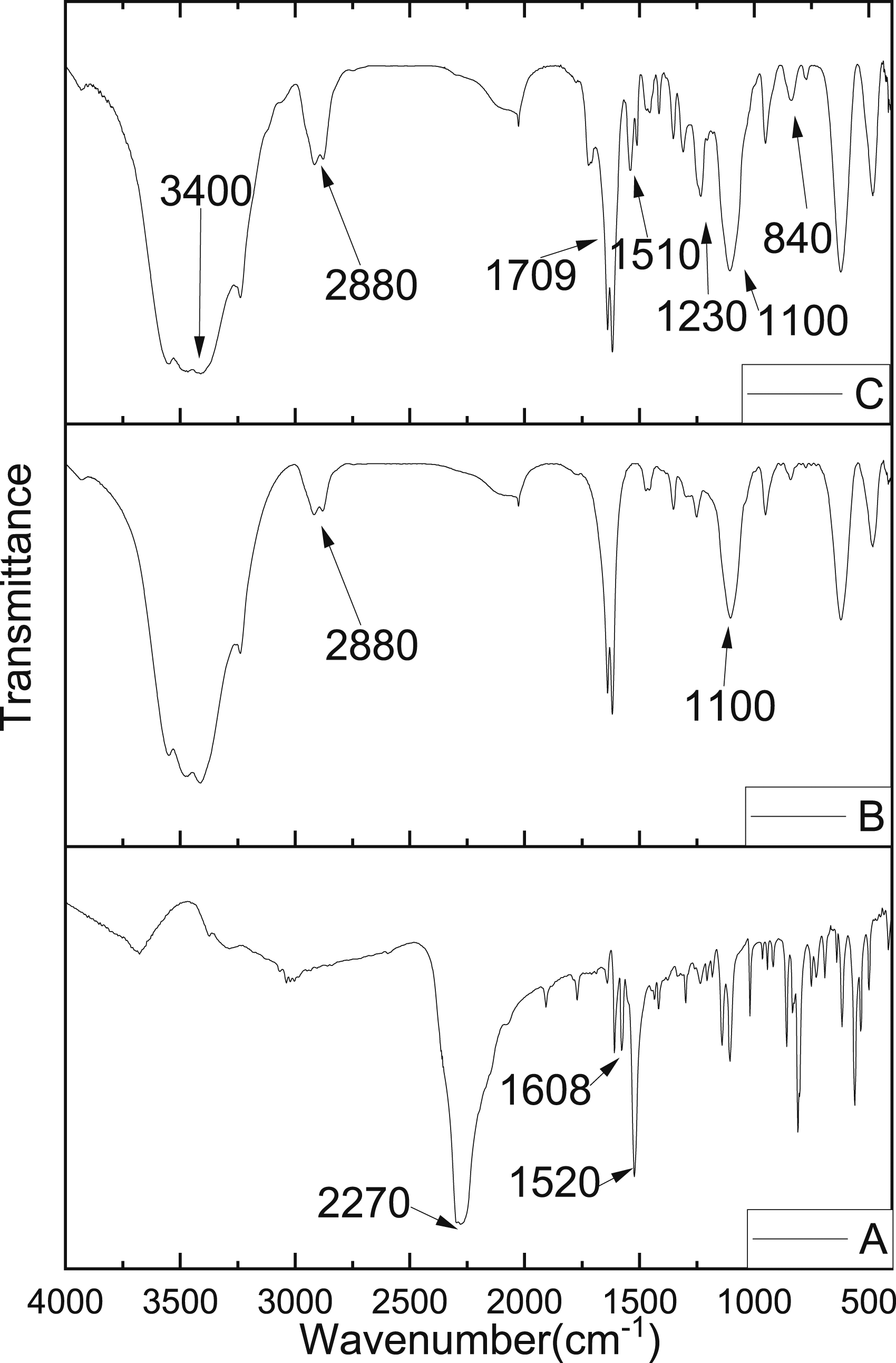
Thermal stability analysis
At normal temperature, PUPCM is a pale gray colloidal solid and has a good liquidity after heating. The results of the thermal stability analysis are shown in Figure 5. The initial thermal decomposition temperature of PEG2000 is 370°C, the temperature corresponding to the maximum thermogravimetric rate is 417.5°C, and the mass change is −98.92%. The initial thermal decomposition temperature of PUPCM is 305.4°C, the temperature corresponding to the maximum thermogravimetric rate is 411.3°C, and the mass change is −89.3%. The weight loss before 305.4°C of PUPCM was mainly caused by a volatile solvent. Although the decomposition temperature of PUPCM is lower than that of PEG, it is still within the melt-spinning temperature of itself. Therefore, the temperature of each zone of the screw extruder should be lower than the temperature of thermal decomposition of PUPCM. Thermal gravimetric analysis curves of PEG and PUPCM (a) PEG and (b) PUPCM.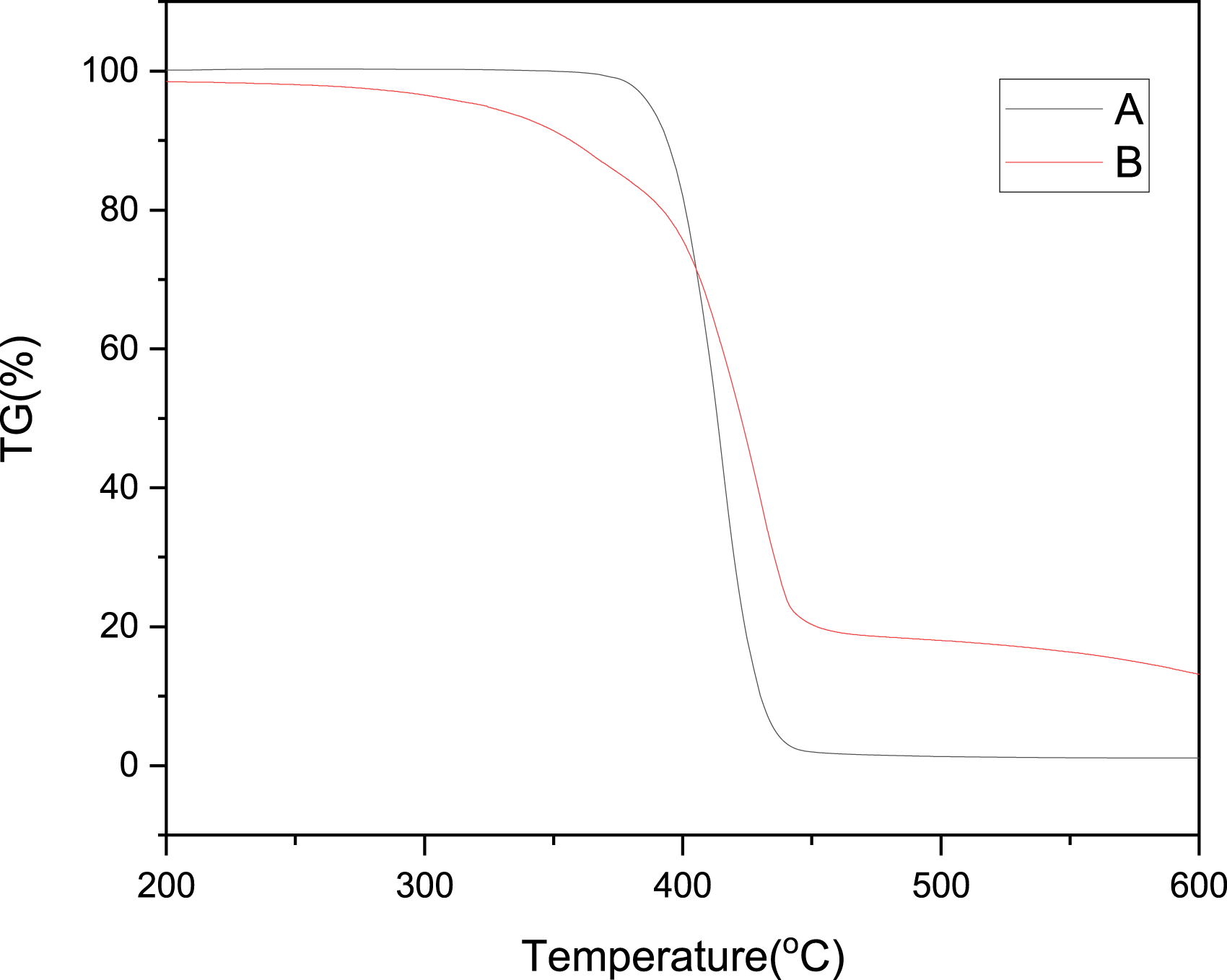
Microstructure of the nonwoven fabric sample
As shown in Figure 6(a), PCESNF has a uniform and stable structure. The internal structure of the fabric was porous structures, these porous structures ensure the air permeability of the nonwoven fabric. During the solidification, the fibers solidify upon cooling from the melt liquid, drafting and forming interlaced webs under the action of pressure and side blowing. In the process, the melt liquid is suspended in an air stream and then blown or forced onto a continuously moving belt where the web is formed. PUPCM has a large solid–solid phase transition enthalpy at a low temperature. Thus, the melt formed an outer-layer structure when it reached the moving belt, while the inner part may remain liquid. The PUPCM droplets are interwoven with each other under the action of hot wind, the fibers are naturally crimped and interwoven as shown in Figures 6(b) and (c). The cooling speed should be appropriately accelerated by an enlarged quench air blow rate, which is conducive to improve the strength and evenness of the fabric. Images of PCESNF: (a) Non-magnified, (b) magnified 1000 times, and (c) magnified 5000 times.
Differential scanning calorimetry of the nonwoven fabric sample
It is necessary to analyze the phase transition property which is basic to set the application environment of PCESNF. The DSC curve of PCESNF is shown in Figure 7, the endothermic direction downward. The peak at 110.91 C is the melting peak of solid–liquid phase transitioned, and the peak at 117.94 C is the solidification peak of liquid–solid phase transformation. Compared with the enthalpy of phase transition at low temperature, the melting enthalpy of PUPCM is much lower. The peak at 51.42 C appears only during the first heating process: it could be because the initial temperature is room temperature. The enthalpy of solid–solid exothermic phase transition reached 60.17 mJ/mg (peaked at 23.14 C). The enthalpy of solid–solid endothermic phase transition reached 67.09 mJ/mg (peaked at 34.34 C). The phase-transition property of PCESNF is reversible; PCESNF has a solid–solid phase change in the process of heating (absorbs heat) and cooling (releases heat). Because when isocyanate made the solid–liquid phase transition of PEG to a solid–solid phase transition of PCESNF, it increased the rigidity of the molecular chain and reduced the crystal size. Because PCESNF is 100% phase-change energy storage material, the phase-change enthalpy of PCESNF is much larger than the traditional microencapsulated phase-change energy storage textile materials. Differential scanning calorimetry curves of PCESNF.
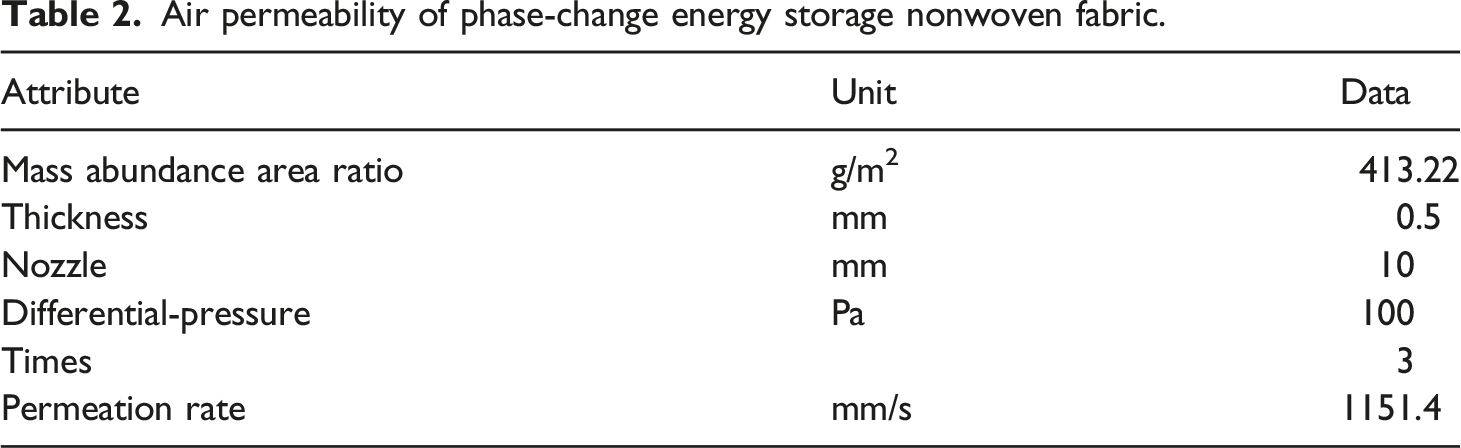
Air permeability
Air permeability of phase-change energy storage nonwoven fabric.
Mechanical properties of the nonwoven fabric sample
Physical properties of phase-change energy storage nonwoven fabric.

Conclusions
We have successfully synthesized PUPCM with polyethylene glycol and diphenyl-methane-diisocyanate. According to the TGA results, the initial thermal decomposition temperature of PUPCM is 305.4°C. The phase-change energy storage nonwoven fabric was made of PUPCM by a nonwoven melt-blown method based on a melt-blown machine (Shanxi Jingwei Chemical Fiber Machinery Co., Ltd). After heating, melting, and homogenization in a screw extruder, the melt is drawn by high-speed hot air flow to form extremely fine fibers, which are condensed on the roller and self-adhesive to form nonwoven fabric. According to the microscope photograph, PCESNF has uniform and stable structure. The air permeability of PCESNF (413.22 g/m2, 0.5 mm thickness) was 1151.4 mm/s; this is because the internal structure of the PCESNF has porous structures, and these porous structures ensure the air permeability of the nonwoven fabric. The enthalpy of solid–solid exothermic phase transition reached 60.17 mJ/mg (peaked at 23.14 C). The enthalpy of solid–solid endothermic phase transition reached 67.09 mJ/mg (peaked at 34.34 C). The phase-change enthalpy of PCESNF is much larger than the traditional microencapsulated phase-change energy storage textile materials (about 26 mJ/mg). The strength and elongation of PCESNF are suitable for garment and tent fabric.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the financial support for the research, authorship, and/or publication of this article: This work was supported by Directive Project of China Textile Industry Federation grant number 2018061, Key Research Projects of Shaanxi Province in 2020 grant number 2020GY-267 and Xi‘an Polytechnic University Start-up Research Fund.
