Abstract
There is a growing public interest in utilizing biomass and biomaterials to obtain products with high sustainability and less harm to the environment. This study reports on using electrospinning technique to produce nanofiber membranes based on homogeneous polymeric blends of gum Arabic, polyvinyl alcohol, and silver nanoparticles. The produced interconnected membranes were cross-linked via heat and plasma treatments, and the membranes were characterized for their chemical and physical characteristics. Fourier transform infrared spectroscopy shows a cross-linking of gum Arabic and polyvinyl alcohol through esterification during the heat treatment, and through graft polymerization with methyl groups after methane plasma treatment. The mechanical performance of the membranes showed an increase in the modulus of elasticity in the longitudinal direction (parallel to electrospun nanofibers) from 85 ± 4 MPa to 148 ± 5 MPa compared with the transverse direction. Also, well-dispersed nanoparticles in the spinning solution tend to increase the elasticity from 41 ± 3 MPa to 148 ± 5 MPa, while the agglomeration of these nanoparticles decreases the mechanical properties of the nanofibers. Results of the biodegradation tests confirmed the significant biodegradable nature of the produced nanofibers, where 99.09% of the material was degraded within 28 days. Moreover, samples showed significant bactericidal activity against Micrococcus luteus with significantly less-observed bacteria in the measured plate, while the inhibition zone for Escherichia coli was 1 cm. The produced biodegradable electrospun membranes have multiple potential applications in many fields; especially for medical, antibacterial, and food packaging. This work reports the results for moisture and oxygen transfer of the membranes as a proposed application in food wrapping.
Keywords
Introduction
Electrospinning is a technique that allows fabricating superfine nanofibers with diameters ranging from 10 µm down to 5 nm. 1 Due to its cost-efficiency and simplicity in fabricating very long fibers from different polymers, electrospinning received a lot of attention among other techniques for producing fine fibers. Up to date, a wide range of synthetic and natural polymers have been electrospun either separately or as polymer blends. 2 Synesthetic polymers are manmade from petroleum (a non-renewable fossil fuel), and cannot be broken down naturally by micro-organisms. This poses a major environmental challenge in terms of disposing these polymeric materials. 3 On the other hand, green nanotechnology encourages the use of nontoxic and renewable chemicals to eliminate the production of hazardous substances.4,5 Green nanotechnology uses natural, nontoxic, renewable, and biodegradable materials to produce nanofibers. 6
Natural polymers have unique biodegradability, biocompatibility, vast availability, cost-efficiency, and nontoxicity. 7 Therefore, they offer a reasonable substitute to synthetic materials in many applications; including gene therapy, drug delivery, regenerative medicine, scaffolds, filtration, and food packaging.3,6–14 Natural polymers are derived from plants, animals, and microbial sources, 15 where a wide range of polysaccharides and proteins can be described as hydrocolloids that form gel colloids when mixed in water. Hydrocolloids are derived from different origins that include plants (e.g. starch, pectin, and cellulose), trees gum exudate (e.g. gum Arabic (GA), gum tragacanth, gum kondagogu, gum karaya, and gum ghatti), seeds (e.g. guar gum, locus bean gum, and tara gum), red seaweeds (e.g. agar and carrageenan), brown seaweeds (e.g. alginate), microbial fermentation (e.g. xanthan gum, dextran, gellan gum, and cellulose), and animals (e.g. chitosan and soy protein). 16 Tree extrudates have been used in multiple food and non-food applications, and they have different colors that range from white to pale amber for GA, white to dark brown for tragacanth, and pale gray to dark brown for karaya gums. 16
GA has many advantages as it is abundant in nature, relatively cheap, biocompatible, biodegradable, and water soluble. 4 GA mainly consists of D-galactose, L-arabinose, low proportion of proteins, 17 and a complex mixture of salts; for example, calcium Ca2+, magnesium Mg2+, and potassium K+. There are other constituents of GA such as cyanogenic glyeosides, tannins, oxidases, pectinases, and peroxidases. 18 GA has antioxidant properties and a role in lipid metabolism, with promising results in treating some degenerative diseases such as kidney failure and gastrointestinal problems. Also, GA exhibits antimicrobial properties as reported in multiple publications.17,19–22 Bacteria form biofilms when they attach to a surface, and it becomes very difficult to remove these biofilms. 23 Biofilm inhibition can be achieved through two main processes: killing the bacteria before adhesion by antibiotics, and modifying the surface properties to prevent adhesion at the first place. Due to the multi-drug–resistant (MDR) developed by bacteria, the first mechanism for preventing biofilms via antibiotics becomes less effective. Researchers intensively investigated this issue to overcome the MDR, and the mechanism for antimicrobial activity of naturally occurring biopolymers was discovered. 24 In this study, we will show results for the antimicrobial activity of GA.
GA was utilized for centuries as an oral heath substance in multiple regions such as the Middle East and North African countries. 25 Nowadays, GA is widely developed and explored in many fields, for instance, it was used in food industry as thickener, stabilizer, and emulsifier agent. 26 GA is also used intensively in textiles, ceramics, cosmetics, and lithography. 26 In pharmaceutical industry, GA has been used as a drug carrier as it is considered as physiological harmless material. 2 However, nanofibers based on GA are not fully explored in applications such as the potential use as a food wrapping material.
Nanoparticles introduce a wide range of antimicrobial activities; for instance, silver was known for long time as an antimicrobial agent, but its use was suspended after the introduction of penicillin in the 1940s, then it emerges again after the bacterial resistance to antibiotics. Silver nanoparticles (Ag NPs) are among the most effective nanoparticles to treat infections caused by bacteria, viruses, as well as eukaryotic organisms. 27
Controlling surface properties of metals, ceramics, and polymers is a key for innovating new functional materials, because many special functions are operating at the surfaces and interfaces. Surface properties of polymers depend on many parameters including the chemical reactions that could take place at the surface, the chemical composition of the surface, and the surface morphology. 28 Surface modification can be performed through chemical reactions that involve the implementation of special elements (e.g. oxygen, nitrogen, and fluorine atoms), moieties (e.g. methyl, carbonyl, and carboxylate), or the grafting of special monomers. 29 Surface modification should be limited only to a monolayer depth and should not proceed to the bulk material. Hence, the ultraviolet (UV) light and the plasma are proper energy sources that can initiate the chemical reactions in shallow layers from the surface. Plasma exceeds the UV light as an energy source for surface modification because UV can only be transmitted through the bright parts of polymers, while plasma can be transmitted in materials with wide optical characteristics. 30 To generate plasma, three essential items must be provided: an energy source that starts the ionization, vacuum system to maintain the plasma, and a reaction chamber.31–34 In this study, nanofiber membranes are treated with methane plasma to enhance the hydrophobicity of the surface.
Mechanical properties of nanofibers are very important, as they determine the validity of use in certain applications. For example, in bone tissue engineering, electrospun scaffolds should have reasonable mechanical properties to sustain tension or compression stresses.24,25 Among the parameters that affect the mechanical properties of electrospun nanofibers are: the nanofibers orientation whether longitudinal (parallel to electrospun nanofibers) or transverse, and the dispersion of nanoparticles or nanofillers, if exist.
In this work, we will produce nanofibrous membranes based on the electrospinning of natural polymers (gum extrudates), and stabilize the membranes in water by cross-linking the polymer chains and inducing hydrophobic characteristics using plasma treatments. The antimicrobial properties for the produced membranes are investigated for pristine natural polymers as well as for polymers with added Ag-NPs. To allow the produced membranes for food packaging applications, we studied their mechanical and transmission (oxygen and water vapor) performances.
Experimental section
Materials
Poly(vinyl alcohol) (PVA) with a molecular weight (Mw) of 88,000 g/mol, GA from acacia trees (G9752) with a number average molecular mass (Mn) of 2.5 × 105 g/mol, silver nitrate (AgNO3), and sodium hydroxyl (NaOH) were purchased from Sigma-Aldrich.
Preparation of Ag NPs
The Ag NPs were prepared by adding 10 mL of 0.1 wt% AgNO3 to 2 mL of 0.5 wt% of a GA solution and a 3 mL of distilled water. The pH of the mixture was maintained at 8.0 using a 0.1% solution of NaOH. The solution was stirred at 70 °C until a yellowish color appeared.
Electrospinning and cross-linking nanofibers
Electrospinning solution was prepared by mixing 20 wt% of GA with 16 wt% of PVA in a balanced mixing ratio of 1:1. An amount of 50 µL from the prepared Ag NPs was dispersed in a total volume of 100 mL from the polymer solution, and the pH was adjusted at 7.0. The viscosity of 20 wt% GA solution was recorded using a rotational viscometer (Brookfield Engineering Laboratories, USA) before and after adding the 16 wt% PVA. Also, the conductivity was measured using Toledo FG3 electric conductivity meter (Mettler, USA), and the surface tension, was recorded using a tensiometer (KRUSS GmbH, Germany). All measurements were performed three times, and the values are reported here with their mean ± standard deviation (SD). At 20 °C, 20 wt% GA had a viscosity of 361 ± 11 cP, whereas its viscosity became 2630 ± 22 cP after blending with 16 wt% and Ag NPs. The conductivity and the surface tension of the polymer solution were 4.3 ± 0.04 mS/cm and 53.5 ± mN/m, respectively.
The electrospinning conditions were adjusted as follow: effective nanofiber width is 200–500 mm, spinning electrode width is 500 mm; spinning distance is 190–300 mm, substrate speed is 0.016–2.03 m/min, voltage is 32–37 KV, and process air flow is 80–120 m3/h. Membranes were cross-linked via heat treatment at 120 °C for 10 min, then were treated with methane plasma in a radio frequency reactor with a continuous flow that keeps the pressure at 40 Pa. The plasma was adjusted for 10 min with a power of 23 W and a voltage of 270 V.
Characterization
The synthesized Ag NPs were characterized using a 300 kV transmission electron microscopy (TEM; JEM-3010, JEOL, Japan), and the Attenuated Total Reflectance Fourier Transform Infrared Spectroscope (ATR-FTIR; NICLET IZ10 module, Thermo Scientific, USA). The ATR-FTIR was used in the range of 400–4000 cm−1 to investigate the functional groups of GA/PVA/Ag NPs in the nanofibrous membranes before and after the plasma treatment. To characterize the surface topology and estimate the average diameter of the nanofibers, samples were tested using the Ultra High Resolution Field Emission Scanning Electron Microscope Zeiss Ultra Plus (UHR FE-SEM Carl Zeiss ULTRA Plus) equipped with a microanalytic system (EDS + WDS + EBSD) that can reach a resolution of 1 nm at 15 kV. The hydrophilic/hydrophobic characteristics of the membranes surface were determined via measuring the water contact angle using the sessile drop method (Surface Energy Evaluation Systems, Czech Republic).
The mechanical properties of at least five specimens from the nanofibrous membranes were tested using an Instron testing machine according to the Standard method ASTM D-638. Also, biodegradation was tested by a continuous automatic Micro-Oxymax Respirometer with O2, CO2, and CH4 sensors. The inoculum characterization used sludge dry matter with the concentration of 4.3 g/L from a wastewater treatment plant in Liberec, Czech Republic. Bacterial colonies were brought from Czech Collection of Microorganisms (CCM), Czech Republic, and with a dilution coefficient of 105, the culture has about 1.2 × 108 colony-forming unit (CFU) per milliliter. Antibacterial activity was assessed using Kirby–Bauer method for both gram-positive and gram-negative, where Micrococcus luteus (ML) and Escherichia coli (EC) were used. Duplicates were used for each bacterium: twice the same sample for EC (CCM7395) and twice the same sample for ML (CCM144). The samples were prepared immediately after their production. Sterile scissors and tweezers (sterilized at 120 °C, 20 min) were used to cut the samples. The work was carried out in a controlled microbiological laboratory environment, and the used dishes were sterile from the manufacturer (VWR). Incubation was carried out in an Incucell thermostat (MMM Medcenter Einrichtungen GmbH, Germany) at 37 °C. Zones of inhibition were measured after 24 h of incubation at 37 °C. Water vapor permeability was measured using MOCON PERMATRAN-W 3/33 (MoCon Inc, USA) according to the CSN 770332 and the DIN 53 122 Standards. Also, the oxygen transmission rate (OTR) was measured for the produced membranes using OxTran 2/20 MH measuring system (MoCon Inc, USA).
Results and discussion
The GA was used as a reducing agent during the preparation of the Ag NPs. The NPs were characterized using TEM that is shown in Figure 1(a), with an average particle size of 4.5 ± 0.5 nm as shown in Figure 1(b).

(a) TEM image for the synthesized Ag NPs; (b) the particle size distribution for the Ag NPs indicating a mean value of 4 nm and a standard deviation (SD) of 0.5 nm.
The Ag NPs samples were found to lack homogeneous dispersion at the beginning of experiments, then a better dispersion was achieved via probe sonicating the solution (Fisherbrand™ Q500; Fisher Scientific, UK) for 10 min before the electrospinning process. SEM image (Figure 2(a)) shows an agglomeration of Ag NPs across the nanofibers. On the other hand, Figure 2(b) shows a better dispersion of the Ag NPs across the nanofibers after sonication.

SEM images of electrospun nanofibers with agglomerated Ag NPs (a), and without agglomeration (b).
The quality of NPs dispersion in polymer matrix is responsible for its properties, where the modulus of elasticity increased from 41 ± 3 for nanofibrous membranes with agglomerated particles to 148 ± 5 MPa for a well dispersed Ag NPs, as can be seen in Figure 3. This observation is in agreement with results of other studies.35,36 Also, an increase in the modulus of elasticity from 85 ± 4 MPa to 148 ± 5 MPa is observed in the longitudinal direction (parallel to electrospun nanofibers) compared to the transverse direction (Figure 4).

Modulus of elasticity of both a well-dispersed and agglomerated NPs.

Modulus of elasticity of both longitudinal and transverse directions.
FTIR spectra for GA, PVA, as well as the GA/PVA/Ag NPs (before and after plasma treatment) are shown in Figure 5. GA displayed a typical spectrum of polysaccharides. The –C = C stretch alkanes is predominant at 960 cm−1, while –C ≡ C– stretch alkynes are observed at 2165 cm−1. The stretch of O – H groups of alcohol/phenol are observed at 3400 cm−1, and the band at 1035 cm−1 can be attributed to the C – N stretch of aliphatic amines. The band at 1618 cm−1 is obtained due to the symmetric and the asymmetric stretches of –COO– groups, while the band at 1427 cm−1 can be attributed to the –OH bend of acid groups. The observed changes in the position and shape of the peaks at 3400 cm−1, and 1618 cm−1 after using GA as a reducing and stabilizing agent to Ag NPs confirms the reduction and stabilization processes. The higher intensity observed at 1427 cm−1 indicates the participation of carboxylate groups in the process of Ag NPs stabilization, which agreed with another study. 37

FTIR spectra of GA, PVA, GA/PVA/Ag NPs before and after plasma treatment.
The shift in frequencies from 1716 to 1760 cm−1 after methane plasma treatment can be attributed to the esterification reaction. The additional functionality observed in plasma-treated membranes may be a result of grafting polymerization of hydroxyl and carbonyl and carboxylate groups during the treatment, which agrees with the results of another study. 38 The peak at 1800 cm−1, which appeared after CH3 plasma treatment can be attributed to the C – H methyl rocking, which demonstrates the possible grafting polymerization.
An increase in water contact angle from 46° to 121° was observed after plasma treatment. Therefore, plasma treatment led to improving the hydrophobicity of the surface of nanofibers by the esterification reactions between carboxylic groups of the gum and the hydroxyl groups of the PVA, which was confirmed by the FTIR and agrees with the results of a previous study. 39
Evaluation of biodegradation is computed by comparing the weight of the initial sample (before test) with the weight of the sample after the test, as given in equation (1). The sample weight (after test) is determined by filtering the entire medium content and weighing the solid components. Duplicate readings for each specimen were taken
The initial mass of one sample was 0.0220 g and after biodegradation testing for 28 days, the sample weighed 0.0002 g. Therefore, the degradation ratio for the sample can be calculated as 0.0091, and the degradation percentage after these 28 days is D28 = 100—(mass ratio × 100) = 99.09%. That means, 99.09% of the nanofibrous membrane was degraded after 28 days, which confirms the biodegradable nature of the produced nanofibers. The results for duplicated samples were found statistically significant.
Antibacterial activity was assessed using Kirby–Bauer method for ML and EC to represent gram-positive and gram-negative bacteria, respectively. Duplicate was used for each bacterium and the results of the duplicates were fully consistent. The results were significant for ML as the whole plate was found with significantly less bacteria after 24 h of incubation at 37 °C as shown in Figure 6(a), while the inhibition zone for EC was about 1 cm (Figure 6(b)). These results may be due to the synergic effect of both GA and Ag Nps. The antimicrobial activity of GA can be attributed to the presence of individual constituents such as cyanogenic glycosides, tannins, oxidases, pectinases, and peroxidases.22,25 It was also reported that GA provides inhibition against dental plaque and the biofilms of bacteria that grow on the surface of the teeth. 40

Kirby–Bauer disks indicate inhibition zone for both Micrococcus luteus (a), and Escherichia coli (b).
The antibacterial activity of Ag NPs has been illustrated by numbers of scenarios; among them is the high affinity of Ag NPs to sulfur and phosphorous. The cell membranes of bacteria contain high amounts of sulfur, which promote the interaction of Ag NPs with the bacteria cell membranes, thus an increase in membrane permeability leading to the cell death. 41 Also, the DNA is highly abundant with phosphorous moieties, which interact with the silver ions released from Ag NPs leading to an inhibition of DNA replication. These ions also lead to inactivation of sulfur-containing enzymes through interacting with the sulfur in these enzymes. Matsumura et al. 42 reported the size and shape of NPs and their impact on its antimicrobial effect, where small Ag NPs have higher antibacterial activity compared to large Ag particles due to the high surface-to-volume ratio. In our study, the Ag NPs has a small size of nearly 5 nm, which significantly contributes to the antibacterial activity results; especially to ML as the whole plate was found with significantly reduced bacteria after 24 h. These results suggest the proper use of these membranes in applications that require antibacterial activity such as food packaging applications.
Food wrapping is among the proposed applications for the produced nanofibers membranes, which offers a sustainable alternative to the plastics that are commonly used in this sector. Therefore, the nanofibers mats should exhibit some barrier properties against gases and moisture. Table 1 presents the values of water vapor transmission rate (WVTR) measured in g m−2 day−1, and OTR in mL m−2 day−1 atm−1 for some commercial plastics commonly used in food packaging.
OTR and WVTR of some common packing materials.
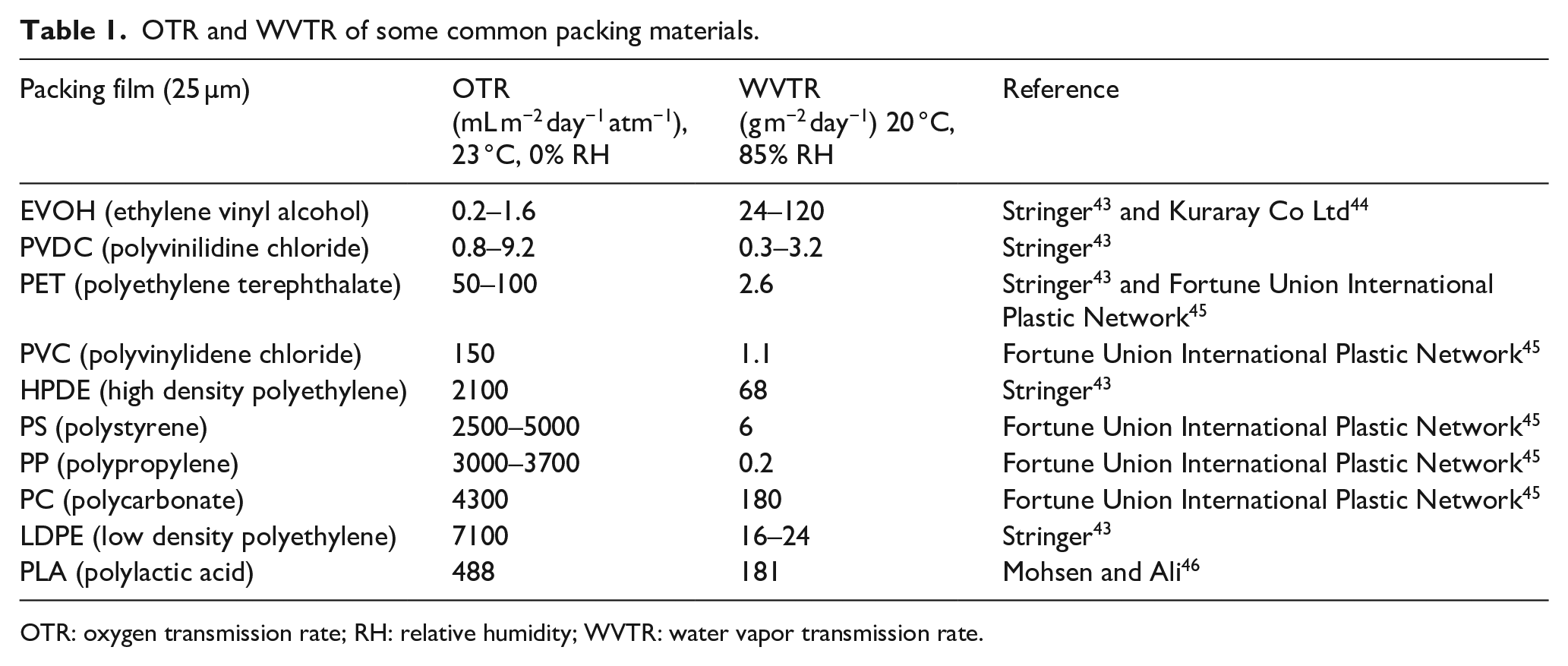
OTR: oxygen transmission rate; RH: relative humidity; WVTR: water vapor transmission rate.
On the other hand, the packaging barrier level in terms of OTR and WVTR may determine the suitable products to be packed in these materials. Table 2 shows ranges for both WVTR measured in g m−2 day−1, and OTR in mL m−2 day−1 atm−1 for some food products.
Barrier values for selected foods according to GAP Italy. 47

WVTR: water vapor transmission rate; OTR: oxygen transmission rate; VAP: vacuum skin packaging.
MAP (modified atmosphere packaging) is the replacement of air in packing material with a single or mixtures of gases.
WVTR was determined gravimetrically at 23 °C and 85% relative humidity (RH) using the MOCON PERMATRAN-W 3/33 according to CSN 770332 and DIN 53 122 standards. Five samples were tested in parallel. OTR was determined using the OxTran 2/20 MH measuring system at 23° C and RH of 0%. Thickness of tested films was determined by the micrometer (L&W type 051, AB Lorentzen & Wettre, Sweden). Water vapor permeability for the produced nanofibers with a well-dispersed Ag NPs was 174 g m−2 day−1. Also, OTR was measured as 10 mL m−2 day−1 atm−1. For better understanding of these values, barrier performance can be classified based on the OTR and the WVTR values, and they can be categorized from poor to high barriers as shown in Table 3.
Classification of packing materials according to OTR and WVTR and their ranges. 48

OTR: oxygen transmission rate; RH: relative humidity; WVTR: water vapor transmission rate.
Depending on the values in Table 3, our produced membranes can be considered as high barriers to oxygen, while low barrier to moisture. Moreover, the barrier properties of our produced nanofibrous membranes suggest (according to Table 2) their suitable use as warping materials for bakery products. In order to improve the moisture barrier of the membranes, further work should be done such as embedding nanowires/nanorods (e.g. nanoclay, cellulose nanowhiskers). This will allow the diffusing substances to navigate around the embedded nanowires and through the interfacial zones and increase their travel via tortious paths with longer diffusing time. Thus, enhancing the materials barrier performance during their shelf life.
Conclusion
Nanofibrous membranes based on tree extrudate (e.g. GA) were produced using electrospinning technology. The polymers helped the reduction and stabilization of Ag NPs, which were used during the production to increase the antimicrobial activities of the produced membranes. It is necessary to homogenize the polymer solution properly (e.g. via sonication) to obtain well-dispersed Ag NPs within the polymer matrix. Good dispersion of the Ag NPs positively affected the mechanical properties of the produced nanofibrous membranes with a significant increase in the modulus of elasticity from 41 ± 3 MPa (for a well-dispersed NPs) to 148 ± 5 MPa (for samples with agglomerated particles). Moreover, the nanofiber orientations in the membrane determine to a high degree the mechanical performance of the fibrous mat with an increase in the modulus of elasticity from 85 ± 4 MPa for samples analyzed in the longitudinal direction (parallel to electrospun nanofibers) compared with 148 ± 5 MPa for measurements at the transverse direction. Results of this study indicate the biodegradability and the antimicrobial activity of the produced nanofibers mats, which are prerequisite characteristics in many applications including food packaging. More research should be carried out to improve the WVTR of these nanofiber membranes via inclusion of different nanofillers (such as the nanoclays).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Youth and Sports in the Czech Republic under the “Inter Excellence – Action programme” within the framework of project “Micro-struCtural imaging as a Tool for modelinG fibrOus materiALS (μ-CT GOALS)” (registration number LTAUSA18135). Also, this work was supported by the Ministry of Education, Youth and Sports of the Czech Republic and the European Union - European Structural and Investment Funds in the frames of Operational Programme Research, Development and Education - project Hybrid Materials for Hierarchical Structures (HyHi, Reg. No. CZ.02.1.01/0.0/0.0/16_019/0000843).

