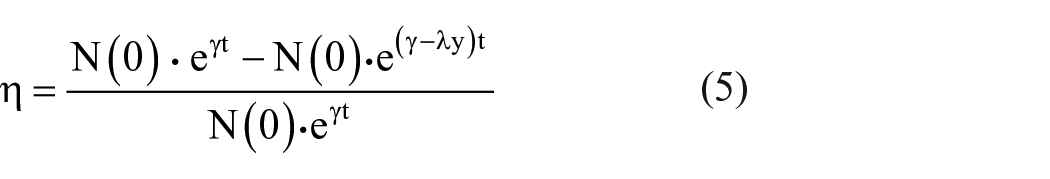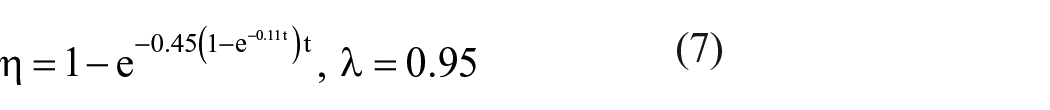Abstract
A food packing system requires an antimicrobial environment with high air permeability to guarantee absolutely both food safety and long and reliable durability. A hollow fiber membrane containing silver ions is the best candidate for this purpose. However, a safe and controllable release process has become a pressing issue in the practical application. Here, we use the predator–prey model to predict the antimicrobial activity of silver ions released from a hollow fiber, and Staphylococcus aureus was applied to the antibacterial experiment. Both theoretical and experimental results show that there are an optimal concentration of the silver ions and an optimal release time. This article sheds a bright light on the design of a new kind of food packing systems using the hollow fiber membrane.
Introduction
A food packing system should guarantee absolutely both food safety and long and reliable durability. To this end, the system should have, first, high air permeability to ensure a fresh air in the system; second, it should prevent microbial resources from entering into the system; third, the material of system should be environmentally friendly and harmless to humans. Hollow fibers with silver ions have good antibacterial property and high air permeability, and the natural fibers have also excellent antimicrobial property, and their geometrical properties contribute to their bio-functional properties.1–6 It is interesting to note that the fractal dimensions of many nature fibers are closed to the golden meaning (1.618), implying an optimal geometric structure. Polar bear hairs are hollow fibers which have an excellent thermal insulation property,1–3 cocoons with hierarchical structures have excellent air permeability, 4 fractal-like wool fibers have high heat release rate from its body, 5 and geckos enable smart adhesion property to their foot hairs. 6 It can be concluded that natural hollow fibers containing silver ions are the best candidate for the food packing system, a packing system with hollow fiber membranes has high air permeability and good vibration resistance.7,8 This article is to study the bio-inspired hollow fibers and to study the silver ion release from the artificial fibers. However, a safe and controllable release process has become a pressing issue in practical applications.
Silver ion release from hollow fibers has been caught much attention,9–13 and its antibacterial ratio plays an important role in practical applications, especially in food packing systems. Recently, an intelligent packaging system was designed successfully by cellulose–polypyrrole–ZnO films, 14 which sheds a new light on new packing systems by advanced nanotechnology.
Polyethylene terephthalate (PET) hollow fiber membranes containing silver ions have a significant antimicrobial activity and high air permeability,4,15,16,17 making them the best candidate for food packings. Nanoscale hollow fibers can now be produced by the bubble electrospinning.18–25 Figure 1 shows the cross section of the Ag/PET hollow fiber. The silver particles are attached to the inner wall of the fibers and distributed evenly, when the hollow fibers’ free ends are immersed into water or meet water moisture; the released ions have a good antibacterial property, because silver ions can cause a fast diffusion of proteins in live bacteria; 26 and additionally silver nanoparticles have high surface energy (geometric potential)27–29 and also have good antibacterial activity.30–32 TiO2-Ag nanoparticles and zinc oxide nanoparticles can be also used for this purpose.33,34

Inner surface of the silver hollow fiber. The scale bar is 10 mm.
Zanetti et al. 35 gave a systematical review on the active packaging using nanotechnology; Kurek et al. 36 proposed a mathematical model for the antimicrobial agent release from a bio-based film; and Esmaeili et al. 37 studied numerically ellipsoidal particles deposition; however, the mathematical models for ion release were rare and very preliminary 11 ; in this article, we will use the well-known predator–prey model 11 to study the antibacterial ratio, so that the pressing issue of a safe and controllable release process can be solved.
An antimicrobial model of silver ions
The Lotka–Volterra model or named by the predator–prey model is used to describe the relationship between predators and prey in an ecosystem.38,39 It is similar to the silver-bacterium process. In the process, silver ions are viewed as predators that feed on bacteria, and bacteria are referred as prey. According to our previous results, 11 the antimicrobial model can be expressed as
where N(t) is the biomass density of the prey-bacteria, y is the mass density of the predator-silver ions, γ is the growth rate of the prey, b is the death rate of the predator, λ is the rate of change in the prey, µ is the rate of change in the predator.
We assume that the releasing process of silver ions is not affected by the number of bacteria in the antibacterial process. The nonlinear system is given in equations (1) and (2) by Taylor method, 40 the variational method,41,42 the variational iteration method,43,44 and others.45,46 The approximate solution reads
where
The antibacterial rate (η) can be calculated as
Experimental verification47–49
The PET-based hollow fiber with silver particles (Ag/PET hollow fiber) as an antibacterial material was bought from Shenzhen Yang Qian Material Application Technology Co, Ltd, China. Silver nitrate and nitric acid were bought from Sinopharm Group Chemical Reagent Co, Ltd, China. Nutrient agar medium, nutrient broth medium, 0.9% saline, disodium hydrogen phosphate, phosphoric acid II hydrogen potassium, and deionized water were bought from Ningbo Aobo Scientific Instrument Co, Ltd, China. Staphylococcus aureus (No. ATCC 6538) was bought from Guangdong Huankai Microbial Technology Co, Ltd, China.
The inductively coupled plasma atomic emission spectrometer (ICP-OES) was used to determine the amount of silver ions. The scanning electron microscope (Hitachi 3-4800) was used to observe the cross section of the Ag/PET hollow fiber.
S. aureus was used as a sample bacteria. A single colony was taken on a petri dish, and then a nutritious broth medium was covered on it for overnight culturing. The concentration of the bacteria was diluted to 108 CFU (colony-forming unit)/mL. Polyethylene terephthalate (PBS) solution of 200 mL was added into 1 mL bacteria solution, and the resultant solution was termed as a control group. PBS solution of 1 mL with 0.1 g of Ag/PET hollow fiber was termed as a test group. Bacterial suspension of 1 mL from the control group and the test group at various periods (0, 1, 2, 4, 4, 6, 8, 8, 12, and 16 h) was taken to count the bacteria number and to make the curve of the bacterial growth. Thus, we can calculate the antibacterial rate and draw the antibacterial curve based on the control group and test group. The fixed bacterial samples were sputter-coated with a gold layer and observed under a scanning electron microscope.
We investigated the morphology of S. aureus bacteria after incubation with Ag/PET hollow fiber by scanning electron microscopy (SEM). As shown in Figure 2(a), images exhibited no change in the control test, and the average size of S. aureus cell is 1–2 µm. Conversely, there was a remarkable alteration when treated with Ag/PET hollow fiber (Figure 2(b)). The bacterial cell’s membrane was expanded and became rough, which indicated that the Ag/PET hollow fiber had the ability to detect the presence of bacterial in the text sample.

SEM images of S. aureus on a food packing system (a) without and (b) with silver ions. The scale bar is 1 mm.
The bacteria will grow at the natural rate of growth, and the growth curve of the bacteria is shown in Figure 3. The fitting equation of the above test data results in
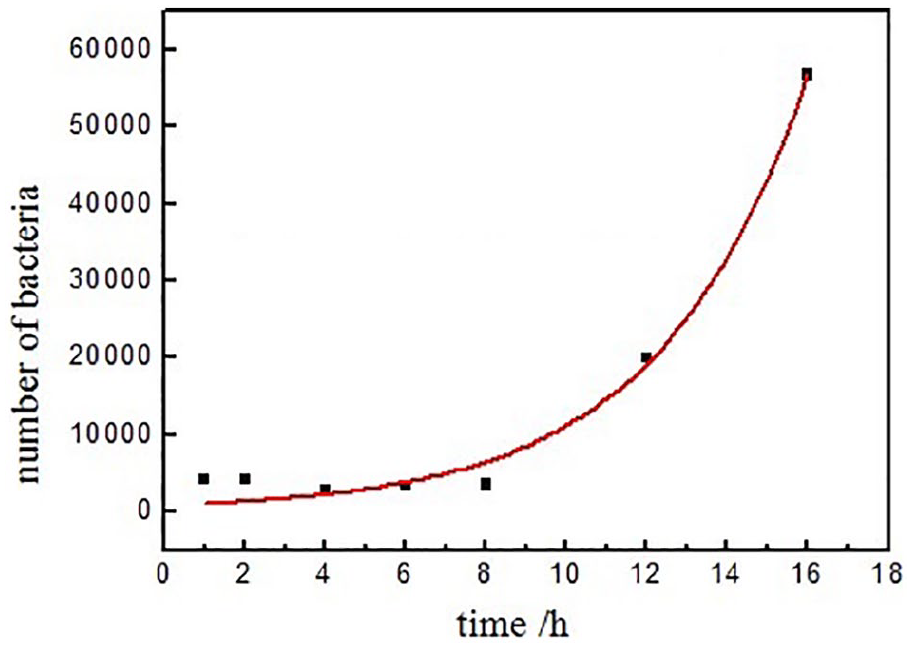
The growth of S. aureus unloaded Ag/PET hollow fibers.
The growth of the bacteria is inhibited by the silver ions, when the Ag/PET hollow fiber is added. At this time, the relationship of the antibacterial rate of the Ag/PET hollow fibers and the contact time is shown in Figure 4. The fitting equation of the above test data is

The antibacterial rate curve of S. aureus contacted with Ag/PET hollow fiber.
When y is equal to the ratio of γ to λ, the number of S. aureus does not increase over times, and the corresponding amount of silver ions reaches the threshold concentration. The antibacterial properties of Ag/PET hollow fiber depend on the silver ions released from the fiber. The threshold time for antibacterial silver ions is named as T. The threshold concentration of the silver ions is calculated as follows
The time required to the threshold concentration of the silver ions is calculated as
Discussion
This article proposed a self-contained mathematical model for ion release process and antimicrobial activity of silver ions simultaneously, and the theoretical analysis is of critical importance for the design of a packing system. The present food packing system might not be harmful to the human physiological system; however, an experimental verification is needed to validate our prediction.
This model can predict the optimal ion concentration and the optimal release time, and the present experiment can be used a paradigm for real applications. Although the mathematical model seems to be rigorous, the assumptions can be made to fix the real conditions, and a fractal modification might be very much needed. Recently, a fractal model was suggested12,13,39 to study the release property of ions from a hollow fiber, and the fractal calculus and fractional calculus50–53 can predict more accurately the antibacterial activity.
Conclusion
This article shows that the silver ions released from hollow fibers have effective antibacterial activity. We use the well-known predator–prey model to predict the antibacterial activity, and the experimental results show the theoretical prediction is reliable and can be used for optimization of hollow fibers.
In this article, S. aureus was applied to the antibacterial experiments for the model, and the results showed that the optimal concentration of the silver ions is 0.28 mg/L and the optimal release time is 8.2 h for our experiment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.