Abstract
The present study reports on the preparation of novel nanofibre membranes from the thermoplastic polymer polyvinylidene fluoride coated with chitosan to enhance membrane properties such as hydrophilicity, mechanical properties, water flux and salt rejection. Initially, a supporting layer was produced from polyvinylidene fluoride using phase inversion methods, followed by being coated with chitosan using either electrospinning or immersion methods. Two types of fabricated membranes with different coating methods were characterized and tested for physical and chemical performance using field-emission scanning electron microscopy, tensile tests, permeation tests (water flux and salt rejection) and contact angle measurements. It was found that the support membrane (polyvinylidene fluoride) produced by the phase inversion method that was coated with chitosan using electrospinning showed better performance, with a salt rejection up to 70% for MgSO4, a decreased the contact angle (52°) and improved the elongation at the breaking point (~82%).
Keywords
Introduction
Recently, the scarcity of fresh water has become a troublesome concern around the world. The United Nations global environmental report released in 2000 reveals that along with global warming issues, shortages of fresh water have also become a significant concern. Fresh water shortages are becoming more concerning due to the rapid growth of the human population and a lack of fresh water resources. 1 A recent report on ocean water depicts that oceans contain 97% of the water on earth, which is too salty for drinking. 2 However, a report on areas of North Africa, the Middle East and parts of Europe reveals that these areas might face droughts and numerous fresh water shortages in the next 20 years. 3 Therefore, developing substitute water resources could be a vital solution to mitigate fresh water shortages. In this scenario, many techniques are being used to attain fresh water from salt water bodies, and one of the most important and commonly used techniques is water desalination. This technique is classified into thermal desalination and membrane desalination approaches. The salt from water is separated by evaporation and condensation in thermal desalination, whereas in the membrane separation technique, pressure and semi-permeable forces are used.
Nanofibre membranes (NFMs) are known to be very advantageous due to their high surface-area-to-volume ratio, high retention of multivalent organic molecules and ions, high flux and operation pressure and low maintenance cost.4–6 In recent years, NFMs have been widely used predominantly for drinking and brackish water filtration, especially for the removal of turbidity, removal of dissolved organics, water softening and colour removal. Non-woven NFMs are produced by one of the most prominently used techniques called electrospinning,7–10 and the resulting NFMs possess splendid characteristics such as large surface-area-to-volume ratios, high gas permeability, minute interfibrous pore sizes, 11 diameter control 12 and high porosity. Moreover, NFMs produced using electrospinning also have excellent mechanical properties and the ability to efficiently eradicate harmful/hazardous materials, which allows electrospun NFMs to be used in various applications, such as tissue engineering, drug delivery, filtration, composite reinforcement and so on.13–15
Polyvinylidene fluoride (PVDF) is one of the most prevalent thermoplastic polymers used in various membrane filtration processes due to its thermal stability, exceptional chemical resistivity and mechanical properties.16–18 However, PVDF is intrinsically hydrophobic, which in turn intensifies membrane fouling. Therefore, by increasing the hydrophilic nature of the membrane surface, membrane fouling can be reduced.19–21 Several methods have been proposed to enhance the hydrophilicity of membranes, such as coating, blending 22 and surface modification.23,24 In another study, composite of PVDF/PVDF-g-PVP (poly(vinylpyrrolidinone)) porous membranes were prepared using an immersion precipitation phase inversion process, and the results revealed that the surface modification of the PVDF increased the hydrophilic nature along with protein absorption resistance. However, increasing the amount of PVDF-g-PVP from 0 to 50 wt% increased the flux recovery from 37% to 77%, approximately. 25 Boributh et al. 26 modified the hydrophobic surfaces of a PVDF membrane with chitosan and enhanced the hydrophilicity and reduced protein fouling. Furthermore, Li et al. 27 prepared PVDF/fluorinated acrylate copolymer tree-like NFMs for membrane distillation and reported a substantial increase in mechanical strength, high liquid entry pressure, a superior rejection rate (above 99.5%) and high permeate flux. Shaulsky et al. 28 generated PVDF-co-hexafluoropropylene and reported that these nanofibres possessed high permeate flux. Shen et al. 29 constructed a hydroxyethyl acrylate/PVDF membrane via a radiation grafting process and found that the water contact angle was reduced by 10°, indicating a hydrophilic natured membrane. Mun et al. 30 modified the surface of PVDF membranes by cross-linking with a PVA solution and obtained enhanced water flux and fouling resistance. Zuo et al. 31 suggested that simple surface modification of PVDF membranes can enhance membrane stability and fouling resistance. Therefore, the mechanisms underlying the surface modification of PVDF membranes deserve rigorous study.
This work deals with the preparation of NFMs from a thermoplastic polymer (PVDF) coated by chitosan to improve membrane properties such as hydrophilicity, surface roughness, water flux and salt rejection. To achieve these properties, a supporting layer was produced from thermoplastic PVDF polymer by using phase inversion methods, which was then coated with chitosan using either electrospinning or immersion methods. Two types of fabricated membranes with different methods for coating have been characterized for their physical and chemical performances using tensile tests, water contact angle, water flux and salt rejection tests.
Experimental work
Materials
The reagents used in this research study were PVDF (Solef®6010; medium viscosity), supplied by the Solvay chemical co. Belgium; magnesium sulphate (MgSO4; analytical grade, 1000 ppm of concentration) purchased from J.T. Baker chemical co. USA; and PVP with an average molecular weight of 10,000, chitosan from shrimp shells that were ⩾75% (deacetylated), tetrahydrofuran (THF),
Polymeric dope and supporting layer preparation
For the preparation of the polymeric dopes, PVDF (15 wt%) was dissolved in a mixture of two different solvents, that is, DMF and THF, along with 2.5% PVP added as a pore former. The solution was mixed homogenously using magnetic stirring for approximately 24 h. Moreover, for the preparation of the PVDF supporting layer, a required amount of PVDF casting solution was taken and mixed in different ratios (10/0, 8/2, 7/3 and 6/4) with DMF/TFA solvents. The solution mixtures with different ratios were then applied between a silicon wafer and a casting knife, which were placed at a fixed distance of 250 μm, and then casted at a speed of 70 mm/s. The temperature was constantly maintained at 25°C with an environmental humidity of 30%.
Chitosan coating onto the supporting layer
Two separate chitosan 2 wt% polymeric solutions were prepared by dissolving chitosan in acetic acid and TFA solvents for immersion and electrospinning, respectively. A total of six samples were prepared via the two methods, of which four of the PVDF supporting layer samples were coated with chitosan by the immersion method, that is, by immersing the prepared layer in a specific chitosan concentration for a controlled amount of time (samples 1–4). The remaining two samples of PVDF supporting layer were coated with chitosan using the electrospinning method (samples 5 and 6), using an NF-500 electrospinning machine supplied by MECC CO., LTD, Japan. Approximately 20 mL of chitosan solution was electrospun by adjusting instrument parameters such as a 22-kV electrical potential, a distance of 10 cm between the needle tip and rotating disc and a 0.4-mL/h flow rate. During the electrospinning process, the humidity and temperature were observed to be 10% and 25°, respectively. Polymeric nanofibres were collected on the collector for approximately 7 h, resulting in a thick layer of nanofibres (Scheme 1). The specifications of the sample preparation are given in Table 1.
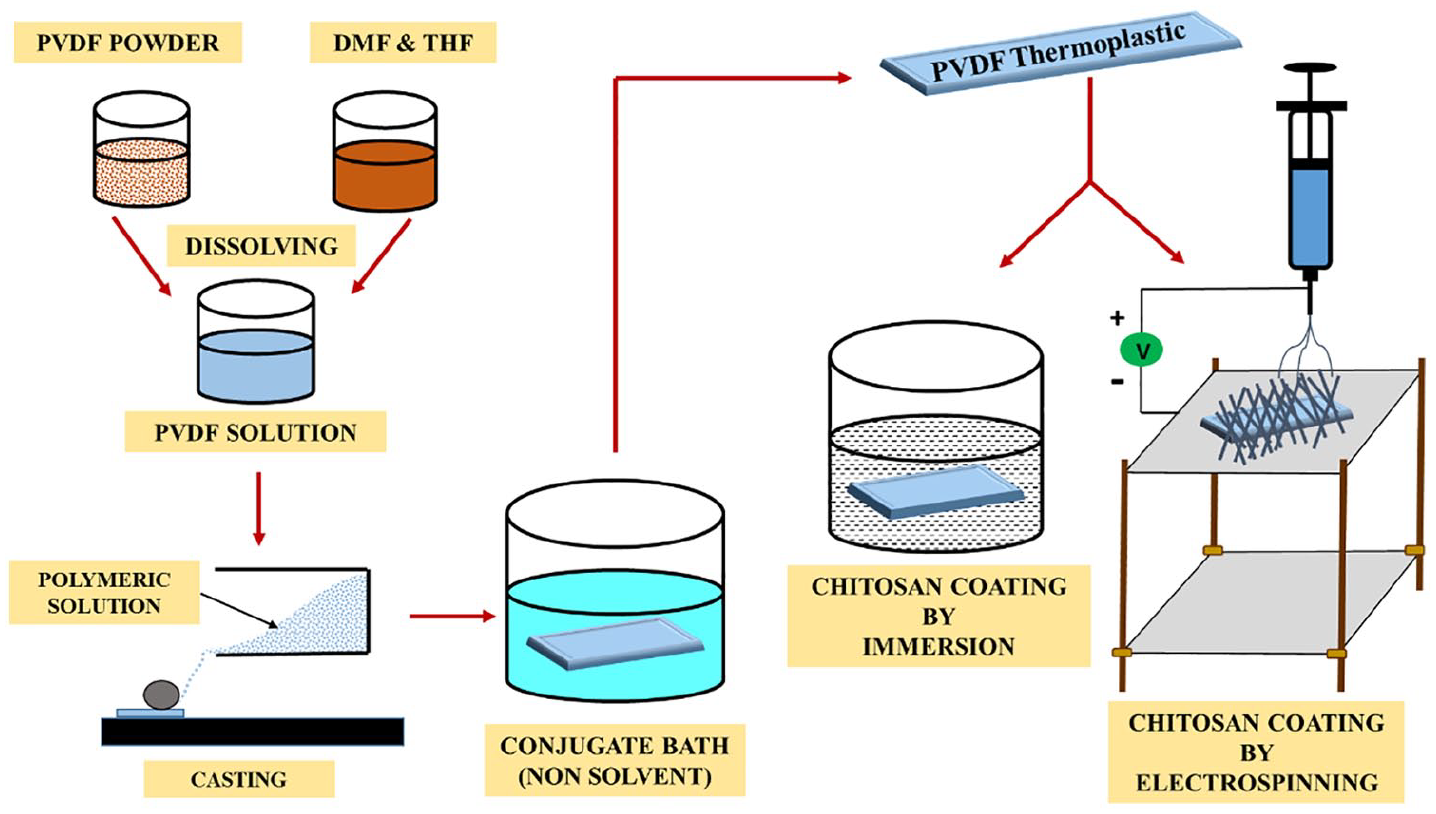
Schematic diagram outlining the experimental process.
Specifications of sample preparation.
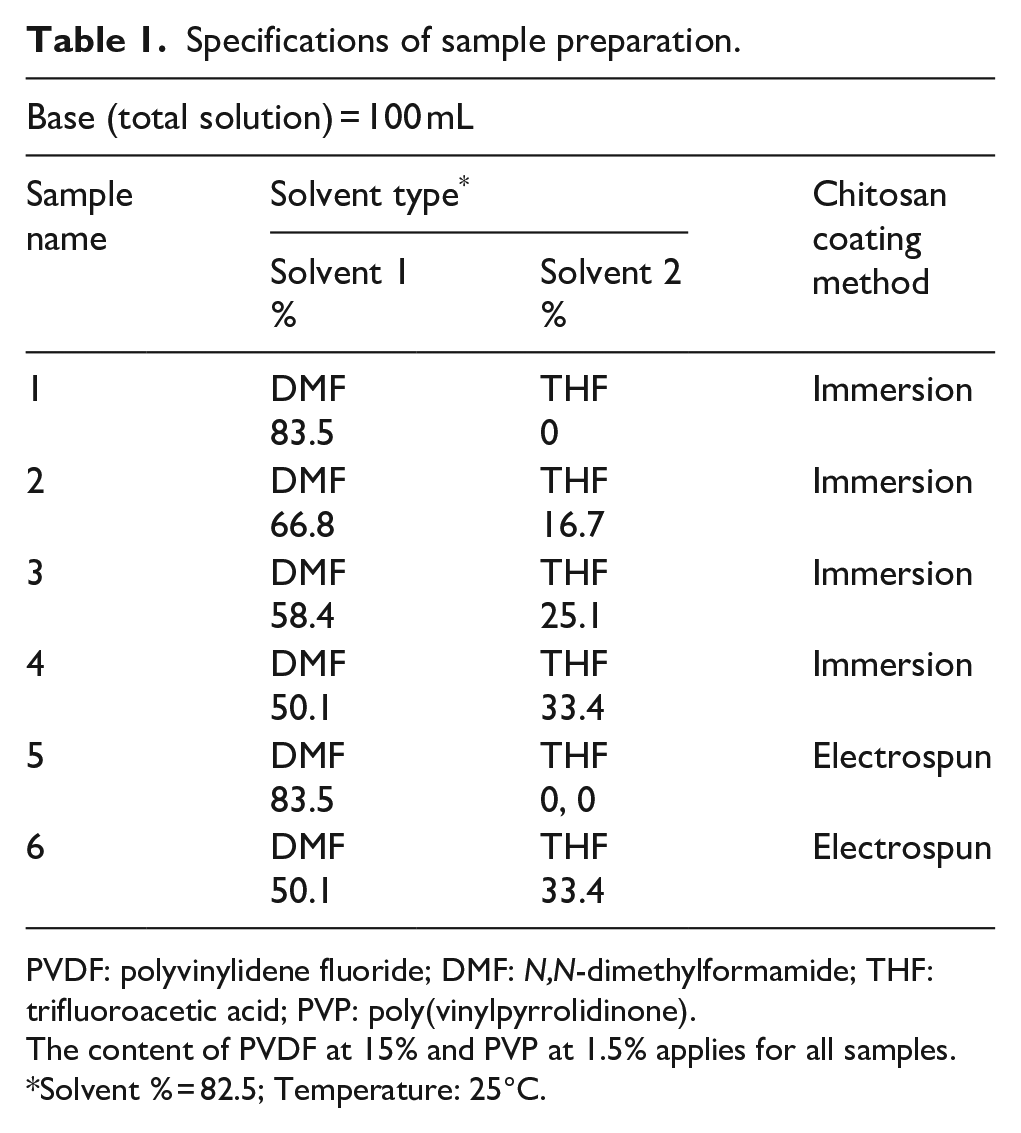
PVDF: polyvinylidene fluoride; DMF:
The content of PVDF at 15% and PVP at 1.5% applies for all samples.
Solvent % = 82.5; Temperature: 25°C.
Membrane characterizations
Morphological observation
Morphological observation of the NFMs was done by means of a field-emission scanning electron microscope (FE-SEM) provided by JEOL Ltd, Japan (Model: JSM-7600). The characterized samples were electron sputtered (platinum coated) using a sputter coater and viewed at an acceleration voltage ranging between 8 and 15 kV.
Contact angle measurements
Contact angle measurements were done on fabricated sheet membranes using a Rame-Hart contact angle goniometer by the sessile drop method. Images of the contact angles made by the droplets on the membranes were acquired using a built-in image system and were analysed using a linked computer.
Mechanical property measurement
Uniaxial tensile tests were performed on the pristine and coated membranes to investigate the mechanical properties using a micro tensile machine (MTD - 500 PLUS). Five samples each of the pristine and coated membranes were tested by cutting the samples into dog-bone shape following the type-5 ASTM D638-10 standard. The cross-head speed, thickness and initial length were 10 mm/min, 1 mm and 40 mm, respectively. Measurements (force and elongation) were recorded until sample failure.
Salt rejection and water flux measurements
MgSO4 rejection was done using a stainless steel stirred dead-end cell HP4750, supplied by Sterlitech Corporation, USA. To accomplish this, a 1000 ppm concentrated solution of MgSO4 was prepared by dissolving 4 g of MgSO4 in 1000 mL of H2O. The prepared solution was mounted on to the membranes with an active area of 14.6 cm2, and a 69-bar pressure handling capacity was placed on the cell connected with a nitrogen supply to control the pressure. The measurements were at different feed and pressure rates. The permeate concentrations collected were tested for the total dissolved salts using a Myron supplied Ultrameter cell.
The percent

The water flux was directly determined by measuring the permeate flow in terms of litres per square metre per hour (L/m2*h).
Result and discussions
Effect of solvent type and concentration on the performance and morphology of the membranes
Choosing the appropriate solvent is an important criterion for preparing a good polymeric solution as it plays a prominent role in dissolving the polymers completely. The PVDF solution used for the preparation of the membranes is obtained by dissolving the PVDF in DMF, dimethyl sulfoxide (DMSO), THF and so on. The use of THF alone might not be effective as it does not fully dissolve PVDF. Therefore, in this study, membranes were prepared using a mixture of DMF and a maximum of 40% THF. The membrane morphologies are presented in Figure 1. However, during FE-SEM analysis, it was seen that the membranes in Figure 1 (a)–(d) produced using the immersion technique possessed sponge-like structures, with finger-shaped cavities with uniform thicknesses. Moreover, it was detected that a dense layer of chitosan was covering the electrospun PVDF membranes coated with chitosan as shown in Figure 1 (e) and (f)

FE-SEM images of cross-sectioned PVDF/chitosan composite membranes for (a) sample 1 (DMF–THF–PVP: 83.5%–0%–1.5%), (b) sample 2 (DMF–THF–PVP: 66.8%–16.7%–1.5%), (c) sample 3 (DMF–THF–PVP: 58.4%–25.1%–1.5%) and (d) sample 4 (DMF–THF–PVP: 50.1%–33.4%–1.5%) and top-surface of the coated chitosan layer for (e) sample 5 (DMF–THF–PVP: 83.5%–0%–1.5%) and (f) sample 6 (DMF–THF–PVP: 50.1%–33.4%–1.5%).
Water flux and salt rejection measurements
Salt rejection and water flux measurement were conducted using a stainless steel stirred dead-end cell HP4750. Figures 2 and 3 show the salt and water rejection measurements. An increase in salt rejection and a decrease in water flux were observed for membranes that were produced with increasing THF solvent concentration during solution preparation. In addition, membranes modified using the immersion method had the lowest salt rejection and highest water flux when the THF concentration was reduced. Interestingly, the electrospun membranes coated with chitosan both showed higher salt rejection and water flux (shown in Tables 2 and 3).
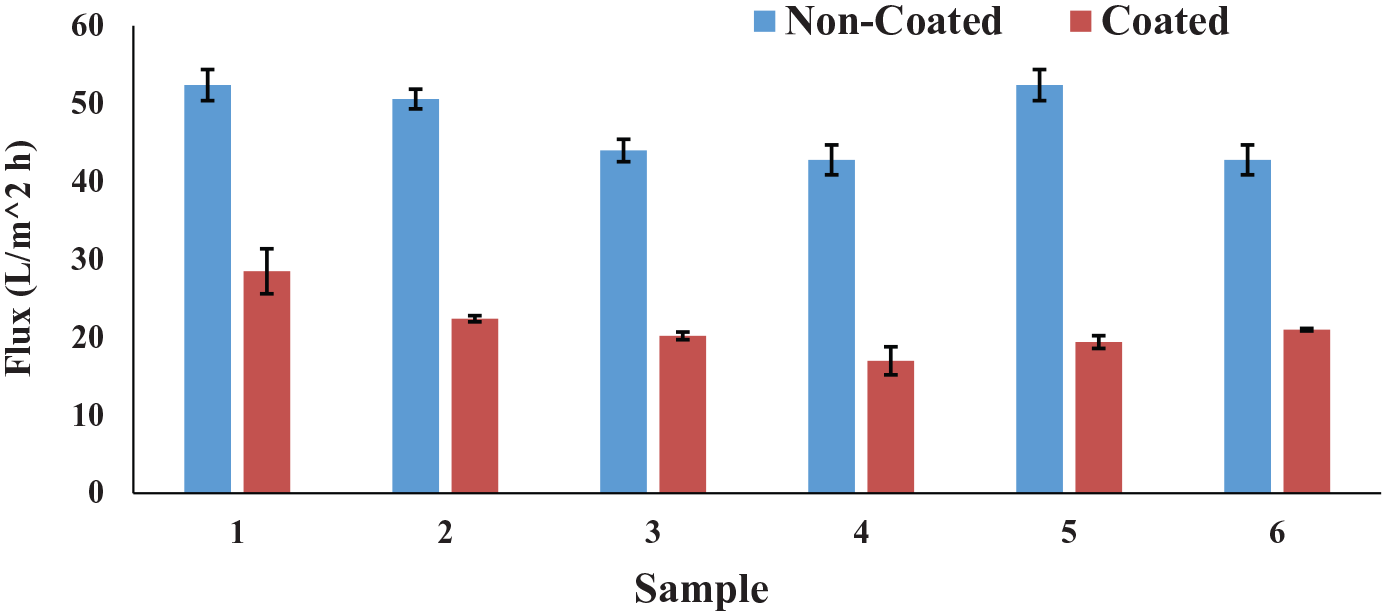
Flux of the PVDF/Chi composite membrane.

Salt rejection of the PVDF/Chi composite membrane.
Flux of the PVDF/Chi composite membrane.

PVDF: polyvinylidene fluoride; Chi: chitosan.
Rejection of the PVDF/Chi composite membrane.

PVDF: polyvinylidene fluoride; Chi: chitosan.
Tables 4 and 5 show comparisons between samples for total dissolved solid (TDS; ppm) removal with varied pressure (bar) for the supporting PVDF membranes (Table 4) and modified PVDF membranes coated with chitosan (Tables 5) by each method, respectively. Here, the electrospun membranes coated with chitosan show higher TDS removal rates at lower pressures and lower TDS removal rates at higher applied pressures (Table 5).
TDS of the PVDF supporting membrane with different pressures.

PVDF: polyvinylidene fluoride; TDS: total dissolved solid.
TDS of the PVDF/Chi membranes with different pressures.

PVDF: polyvinylidene fluoride; TDS: total dissolved solids; Chi: chitosan.
Contact angle tests
The surface wettability or hydrophilicity of membranes is generally measured using contact angle measurement tests. The hydrophilicity of a membrane is said to be higher when the contact angle made by the water with the membrane is smaller. In this study, we measured the contact angles of the prepared membranes; to check the repeatability and accuracy, the measurement was done three times for each sample, and the average results are given in Table 6. The PVDF membranes are hydrophobic in nature, but due to the addition of 2.5% PVP in the PVDF solution, a decrease in the hydrophobicity was observed. In addition, it was observed that membranes coated with chitosan using both the immersion and electrospinning techniques had increased hydrophilicity compared to the non-coated membranes. Interestingly, it was observed that the support membranes that were surface modified by the addition of the chitosan layer using the electrospinning technique (samples 5 and 6) had higher hydrophilicity than the immersion-modified membranes (samples 1–4). Moreover, it was also observed that membranes generated using lower proportions of DMF and higher proportions of THF with a maximum of 40% had decreased contact angles.
Contact angle measurements of the PVDF/Chi composite membranes.

PVDF: polyvinylidene fluoride; Chi: chitosan.
Mechanical properties of the fabricated membranes
The mechanical properties of the membranes are one of the most important parameters as the membranes endure stress during operation. Because of this, the mechanical responses, that is, the breaking strength and percentage of elongation of the fabricated membranes (sample 1–6), were tested until failure, with at least four repeated tests. The assessed tensile test values of the pure PVDF and PVDF support with coated chitosan are shown in Figure 4. The results indicate that the amount of elongation at the breaking point was almost the same for the membranes coated with different concentrations of chitosan using the immersion method. The membranes coated with a chitosan layer using electrospinning (samples 5 and 6) showed a dramatic increase in the tensile stress and elongation at the breaking point. Therefore, from the above mechanical responses, it can be concluded that these responses strongly depend on the interactions between the support (PVDF) and the coated layer (chitosan).
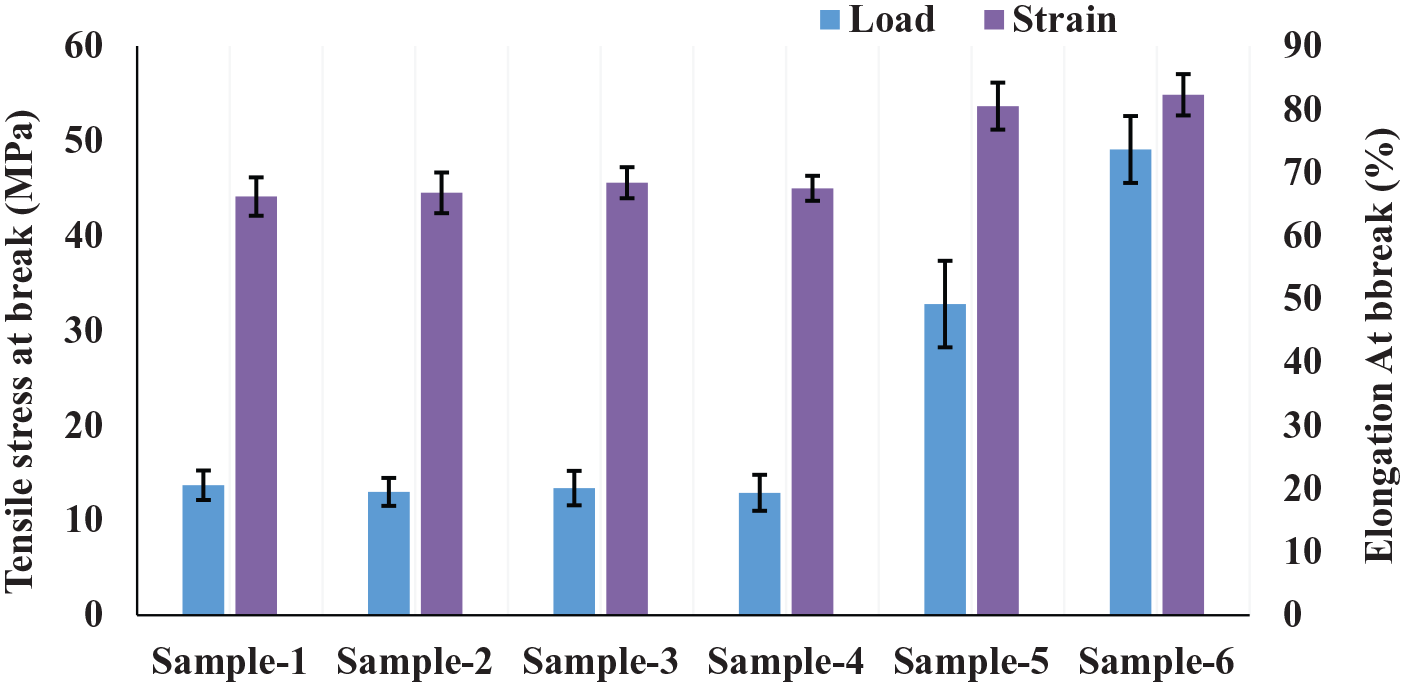
Mechanical property measurement of the PVDF/chitosan composite membrane.
Conclusion
In this study, we analysed and compared membranes of PVDF (support) coated with chitosan (layer) using immersion and electrospinning methods. A large improvement in the hydrophilicity, water flux, salt rejection and mechanical properties was attained when electrospinning was used for coating chitosan onto the PVDF supporting layer. The conclusions from this study are as follows: (a) THF is a weak solvent for PVDF (~40% maximum percentage of THF solvent could be used), and it was impossible to be used alone for dissolving PVDF. The use of a strong solvent such as DMF was necessary. (b) The modification of the hydrophobic PVDF membrane with chitosan was performed to increase the hydrophilicity. (c) The hydrophilicity of the modified membrane increased (water contact angle decreased) with increasing concentrations of THF. (d) Using electrospinning for coating chitosan onto the PVDF membrane resulted in better performance compared to the immersion method on the basis of water flux, salt rejection and mechanical properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to extend their sincere appreciation to the Deanship of Scientific Research at King Saud University for funding this work through research group (RGP-1439-042).
