Abstract
This study aimed to improve the strength and hydrophobic properties of polyvinyl alcohol/microfibrillated-cellulose composite films and thereby solve problems such as the poor water resistance of polyvinyl alcohol films and defects in the packaging. Polyvinyl alcohol/microfibrillated-cellulose composite films were prepared with the silane coupling agent KH151. The mechanical, optical, crystalline, and other properties of the composite films were tested. After the modification of the polyvinyl alcohol/microfibrillated-cellulose films, their strength and hydrophobic and barrier property were greatly improved. Moreover, their oxygen transmittance decreased by 85.9%, and the water contact angle of the film surface increased by 44%. The internal structure of a polyvinyl alcohol/microfibrillated-cellulose film is formed by KH151, which improves the strength and barrier properties of the film, forms an alkane-based layer on the film surface, and improves the hydrophobic property of the film. Thermogravimetric analysis shows that the thermal stability of composite materials has been greatly improved.
Introduction
Microfibrillated cellulose (MFC) is prepared by using cellulose as a renewable material and is then added to different polymer materials to increase their strength. 1 Cellulose is treated in several ways, and TEMPO (2, 2, 6, 6-tetramethylpiperidine oxynitride) oxidation via the TEMPO/NaBr/NaClO system is currently being used. This oxidation method uses the high selective oxidation of TEMPO to convert the primary hydroxyl group at the C6 position on the surface of the cellulose fiber into a carboxyl group, thereby improving the electrostatic repulsion of the fiber in the aqueous phase system. The method also aids in ultrasonic treatment by separating MFCs from one another. The MFC prepared by the TEMPO oxidation method has a diameter of 2–4 nm, a length of several micrometers, and a high aspect ratio, all of which can improve the strength of composite materials. 1
Polyvinyl alcohol (PVA) is a water-soluble polyhydroxy polymer with good film-forming properties, flexibility, and solvent resistance. It is often used to prepare membrane materials, which have high flexibility and plasticity but poor mechanical rigidity and strength. PVA and MFC are highly hydrophobic, and they complement each other in terms of mechanical strength. After adding MFC, the mechanical properties and thermal stability of PVA/MFC composites are improved. 2
Many silane coupling agents are used to modify the hydrophobic properties of PVA and cellulose. The chemical group reacts to support the alkyl group on the surface of the material, and the water contact angle varies greatly. The types of silane coupling agents currently used are KH560, KH570, KH602, KH171, dodecyltriethoxysilane, octadecyltrichlorosilane, trimethylchlorosilane, and so on.3–11 The main reaction principle is that the silane coupling agent is hydrolyzed under acidic conditions and cross-linked with a hydroxyl group on PVA or cellulose. The PVA and cellulose can be modified into a hydrophobic substance. PVA can even be modified into a water-repellent coating, which is used on gypsum to improve its waterproof performance.
In the use of vinyl trimethoxysilane (VTMS) to improve the thermal stability and barrier properties of PVA film, oxygen permeability decreased from 6.12 to 0.17, and the glass transition temperature and decomposition temperature of the composite film were improved to some extent. This indicates that after the cross-linking occurs, the structure of the PVA is made tighter, and the mechanical properties of the film are improved. The preparation of water-repellent all-cellulose nanocomposites (ACNC) with vinyl triethoxysilane greatly reduces the hydrophobic properties of cellulose, and the cellulose changes from hydrophobic to hydrophobic. These indicate that the silane coupling agent can not only react with PVA but also form a bond with cellulose, and can enhance the strength of the film.
The bonding of PVA and cellulose by hydrogen bonding is deficient, and thus, different cross-linking agents are continuously selected to enhance the performance of PVA/MFC films. PVA and MFC have the strongest mechanical properties in the microstructure, but the combination of PVA and MFC is still bonded by hydrogen bonding, and the strength of the composite film can be further improved. Some researchers have used NFC/glycerol/PVA composite membranes with very high transparency, and the use of glycerin to improve the toughness of the composite membrane, and increased the composite membrane to block UV in food packaging, but did not increase hydrophobicity and compounding. The water resistance of the film is such that the composite film is left in the air for a long time, so that the composite film absorbs water for a long time, and the strength is continuously lowered. Some researchers have combined PVA and MFC with UV photocrosslinking technology to prepare photocrosslinked PVA/NFC and PVA/MFC with good water resistance, good strength, and functionality: Cellulose (PVA/(M/NFC)/poly-HEMA) composite.12–15
In this article, the silane coupling agent KH151 is used as a cross-linking agent, which does not only increase the film strength but also can modify the film into a hydrophobic film.
Materials and methods
Preparation of composite membrane
Materials and equipment
Materials: glacial acetic acid (Tianjin Damao chemical reagent factory), vinyl triethoxysilane (Nanjing Chuangshi chemical additive Co., Ltd.), PVA (Source Leaf Biological Co., Ltd.), glycerol (Tianjin Fuqi chemical Co., Ltd.), MFC (self-provided by the laboratory).
Equipment: electronic analytical balance (AR2130, Ohaus instrument Co., Ltd.), electric thermostat blast drying oven (DHG-9203A, Shanghai Yiheng scientific instrument Co., Ltd.), constant speed agitator (Shanghai Shensheng biotechnology Co., Ltd.), thickness gauge (GY-12.7*0.001, Dongguan Guangyi instrument Co., Ltd.), electronic universal material testing machine (Instron3369, American instron), X-ray diffractometer (XRD) (D/max-2500 type, Japanese science corporation), Fourier-transform infrared spectrometer (FT-IR) (iS5, American Nicolet), breathable tester (GDP-C, Bruggar, Germany), scanning electron microscope (SU1510, Hitachi High-Tech Co., Ltd.) thermogravimetric analyzer (TGA-Q50, TA company, USA), differential scanning calorimeter (8000, Elmer, Platinum, USA).
PVA/MFC/KH151 hybrid film
In a ratio of MFC:PVA of 5:95, 2% glycerol and 1%–5% KH151 were added to the mixed solution to achieve a good configuration. Then, 2% acetic acid was added dropwise, and the pH of the mixture was maintained between 3.0 and 4.5. The mixture was stirred under mechanical conditions for 6 h until the mixture was homogeneous. It was then sonicated for 2 h to remove any air bubbles. The mixture was made colorless and transparent. The prepared mixture was uniformly poured onto a glass plate and evenly spread. The glass plate was placed in an oven at 50℃ to dry enough to form a film.
Reaction mechanism
As shown in Figure 1, the silane coupling agent KH151 continued to hydrolyze at a pH of 3.0 to 4.5 and reacted with hydroxyl groups. The by-product was ethanol, which is non-toxic and harmless. The silane coupling agent KH151 increased the strength of the PVA/MFC film. As shown in Figure 2, inside the film, KH151 cross-linked with the hydroxyl groups on the PVA and MFC. The reaction improved the mechanical properties of the film. The hydrophobicity of the film surface also increased. Given the molecular arrangement of the film surface, KH151 continuously condensed, and the vinyl group was arranged on the outside of the film to form a dense hydrophobic structure (see Figure 3).

Hydrolysis of KH151.

Diagram of KH151 cross-linking PVA and microfibrillated fibers.

Molecular diagram of hydrophobic surface of KH151 modified PVA/MFC film.
Methods
Mechanical property test
According to “GB/T 1040.3-2006 Determination of tensile properties of plastics” 16 Part 3: Film and sheet test conditions, the film samples were cut into rectangular strips of 150 × 10 mm and then measured by a universal material testing machine in terms of tensile strength and elongation at break. Each set of samples was averaged 10 times and the stretching speed was 20 mm/s.
Scanning electron microscopy
Scanning electron microscopy (SEM) was used to analyze the surface morphology of the film samples. Prior to analysis, gold was sprayed on the fractured surface of the films. The magnification was 800×.
Fourier-transform infrared spectroscopy
The prepared film was tested at ambient temperature using a Vector 22 FT-IR with a wavenumber in the range of 4000–500 cm−1. Each film was placed in the scanning position at a scan rate of 4 cm−1 and then scanned 16 times.
X-ray diffractometer
The test was carried out using a D/max-2500 X-ray manufactured with a tube voltage of 40 kV, a current of 100 mA, a CuKα ray, in the range of 1° to 50°, and a scan rate of 4°/min.
Water contact test
The water contact angle of the film surface was measured using a VCA optima goniometer. A film of 1 × 10 cm was taken, and a droplet with a diameter of 1–2 mm was dropped on the surface thereof. The water contact angle of the film was then measured by a seat drop method, and the data were recorded. Ten measurements were taken for each sample, and the final results were averaged.
Optical performance test
The spectral transmittance of the modified film was measured by a UV-2700 spectrometer to characterize the transparency of the film. The spectral wavelength range was set to 200–800 nm.
Oxygen permeability test
Samples were prepared according to GB/T 1038-2000, and the oxygen permeability of the film samples was measured using a GDP-C permeability tester. Ten parallel samples were selected for each sample during the test. The calculation formula of the permeability coefficient

where,
Differential calorimetric scanning test
The thermal stability of the PVA/MFC/KH151 film was measured by a DSC (differential calorimetric scanning) 8000 differential calorimeter. The thermal stability of the PVA/MFC/KH151 film was heated from 30 to 250℃ at a heating rate of 10℃/min under N2 protection conditions, eliminating the heat history, and then cooling the temperature to 10℃/min to at 30℃, the temperature was finally raised to 250℃ at a rate of 10℃/min.
Thermogravimetric test
The thermal stability of the PVA/MFC/KH151 film was measured with a TGA Q500 thermogravimetric analyzer and heated from 25 to 800℃ at a heating rate of 10℃/min under N2 protection conditions.
Results and discussion
Mechanical properties results
The mechanical properties of the PVA/MFC composite membrane are shown in Figure 4. As the hydrogen bonding between the PVA molecule and MFC is weak and the molecular flexibility is strong, the tensile strength is 33.1 MPa, but the elongation at break can reach 197%. However, as the amount of KH151 increases, the PVA and MFC molecules continuously combine the two substances through a coupling agent. Given the small amount of the coupling agent in the beginning, the elongation at break of the material increases continuously. When the amount of coupling agent is increased, the elongation at break begins to decrease. When the amount of coupling agent KH151 is 3%, the elongation at break is 305.35%, but the composite layer is combined with KH151. The elongation at break is rapidly reduced. However, the strength is still increasing at this time. When the amount of the coupling agent reaches 4%, the tensile strength peaks up 42.87 MPa.

Effects of KH151 on tensile properties.
SEM analysis results
As shown in Figure 5, the compatibility of the PVA and MFC composite film is continuously improved with the increasing use of KH151. The cross-section scanning shows that the film’s cross section is relatively flat. When the amount of KH151 reaches 3% (Figure 5(c)), the phase is at the best capacity. As the KH151 dosage continues to increase, a gap begins to form, and delamination of the composite film occurs. As the more the amount of KH151 increases further, the gap and delamination become greatly obvious. This result is due to the low amount of KH151 that can be evenly distributed on the film substrate and act as a cross-linking agent. When the amount of KH151 is small, the composite film cannot be made uniform, and phase separation occurs.

SEM images of KH151 hybrid film: (a) KH151 1% 800×, (b) KH151 2% 800×, (c) KH151 3% 800×, (d) KH151 4% 800×, and (e) KH151 5% 800×.
With the increase of the KH151 dosage, the phase separation phenomenon is gradually reduced. When the dosage is 3%, the internal distribution of the composite membrane becomes uniform. However, as the dosage continues to increase, KH151 undergoes a self-condensation reaction, and the vinyl group becomes an alkyl group. The group, without combining with any materials, then shows stratification. The occurrence of defects such as voids and delamination causes a sharp drop in the elongation at break of the composite film.
The void phenomenon that occurs at 4% (Figure 5(d)) KH151 dosage is due to the short molecular layer formed by the condensation of KH151 itself. Under this condition, bending easily occurs, and voids appear. When the amount of KH151 reaches 5% (Figure 5(e)), the molecular layer formed by the condensation of KH151 becomes long, which leads to delamination inside the film. The occurrence of this delamination causes the destruction of the continuous phase inside the film, which is then pulled. The tensile strength and the elongation at break increase sharply.
When the addition amount of the silane coupling agent is 4%, a certain agglomeration phenomenon will occur inside the composite material at this time. At this time, it can be seen from the electron microscope scanning chart that the agglomeration effect is obvious, which is due to silane coupling. The addition of the agent reaches a certain amount, so that the silane coupling agent appears self-crosslinking, forming a closed structure, and agglomeration occurs in the electron microscope.
FT-IR analysis results
Figure 6 shows the characteristic peak of the hydroxyl group at 3280 cm−1. The area of the peak decreases as the amount of coupling agent increases. At 2900 cm−1, the absorption peak of C-H shows stretching vibration, but it decreases under the action of KH151. The coupling agent is supported on the hydroxyl group, so that the peak intensity is reduced. A characteristic peak of C = C double bond is observed near 1600 cm−1, which indicates the reaction of KH151 with the hydroxyl groups of PVA and MFC. The double bond appears inside the composite membrane. The characteristic peak of Si-O-C is 970 cm−1, indicating that KH151 and PVA and MFC form a cross-linked network structure. The characteristic peak of Si-O-Si is at 1010 cm−1, and KH151 itself undergoes a condensation reaction. As KH151 increases, the peak value increases continuously.

Infrared spectrum of PVA and MFC composite membrane.
XRD analysis results
As shown the XRD pattern in Figure 7, a new peak appears at a position of about 2θ = 10°, and as the amount of KH151 increases, the peak intensity increases, indicating that a new peak at 9.6 nm appears inside the film. The ordered structure is the ordered arrangement of KH151’s continuous cross-linking reaction. 2θ = 20° is the intensity of the PVA (101) reflection peak as it drops. This condition indicates that the number of hydroxyl groups on the PVA is continuously decreasing and reacts with KH151 to form a new cross-linked network structure. The barrier and strength properties of the composite film are greatly improved.

Effects of KH151 on the crystallization of the composite membrane.
Oxygen permeability test results
As shown in Figure 8, the oxygen permeability coefficient decreases from 8.332 × 10−6 cm3·cm/(cm2 s Pa) to 1.174 × 10−6 cm3 cm/(cm2 s Pa) as the amount of KH151 increases. This condition indicates that the formed network structure is continuously increasing the barrier property of the film. The oxygen permeability coefficient decreases by 85.9%, and the oxygen barrier property increases because a new network structure is formed to produce a dense crystal structure. The path and difficulty for oxygen to permeate the film are increased, and the barrier property of the film strengthens.

Effects of ethylene triethoxysilane on oxygen permeability.
Contact angle test results
As shown in Figure 9, the contact angle increases by 44% from 71.5° to 103°. As the amount of KH151 increases, the water contact angle increases as well, indicating that the hydrophobicity of the composite membrane also increases with the increase of KH151. In this way, we can infer that the hydroxyl group formed by the hydrolysis of KH151, and the hydroxyl groups of PVA and MFC are consumed by the self-condensation reaction, forming a structure shown in Figure 3. Consequently, the surface of the composite film becomes hydrophobic as the amount of KH151 increases. The denser the hydrophobic structure, the lesser the hydrophilic groups on the film surface, and the greater the more hydrophobic effect.
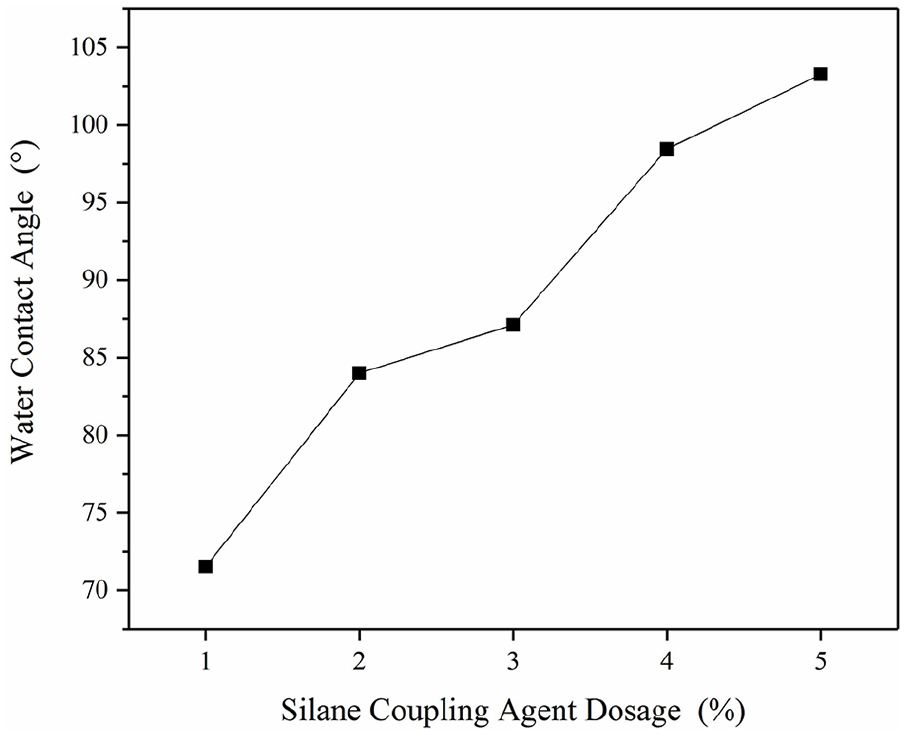
Effects of KH151 on water contact on composite film surface.
Optical properties results
As shown in Figure 10, as the amount of KH151 increases, the transparency of the film decreases, and the light transmission spectrum in the visible light region (200–800 nm) is slightly affected by KH151. As the amount of KH151 increases, the film transparency decreases to a certain extent, indicating that after the condensation reaction occurs, the cross-linking reaction forms crystal particles. When the diameter of the crystal particles is close to the wavelength corresponding to visible light, light scattering occurs in visible light, resulting in a decrease in UV-visible light transmittance. When the content of the silane coupling agent is 1%, the transparency of the film is best.

Effects of KH151 on the transparency of composite films.
DSC and T g results
The DSC curve of pure PVA presents an endothermic peak at 201℃ corresponding to the melting of the PVA crystallites (see Figure 11). This endothermic peak was shifted to lower temperature and became smaller as the KH151 content increased. With the increase of silane connection, the cross-linking effect is more obvious, and the absorption peak gradually disappears. In the last figure, 5% has no absorption peak at 201℃.

DSC thermograms of the PVA/MFC KH151 hybrid films.
The thermal decomposition of the hybrid films shifts toward a higher temperature range than that of PVA, indicating an enhancement of thermal stability in the PVA/MFC/KH151 hybrid films. This enhancement in the thermal stability and change in the decomposition pattern were ascribed to the high heat resistance and structural change exerted by the KH151, which originated both from the condensation of KH151 with hydroxyl groups of PVA and strong interfacial interactions between KH151 and PVA. The temperature at the last inflection point of the

TGA thermograms of the PVA/MFC/KH151 hybrid films.
Conclusion
The PVA/MFC composite membrane was prepared by cross-linking the hydroxyl groups of PVA and MFC by KH151. A composite membrane is formed by the cross-linking between the hydroxyl group of PVA/MFC and the silanol group of the hydrolyzed KH151, in which case a condensation reaction occurs. When the amount of KH151 used reaches 3%, the tensile strength of the composite film is 38.8 MPa, the elongation at break is 300.6%, and the water contact angle of the film is 87°. These properties basically improve the hydrophobic ability of the film. Moreover, the oxygen permeability coefficient reaches 3.322 × 10−6 cm3 cm/(cm2 s Pa), and good performance, particularly in terms of transparency, is achieved. The silane coupling agent can improve the thermal stability of the composite film, and the maximum decomposition temperature of the film is raised from 417 to 442℃.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
