Abstract
The silica leached from coal fly ash using alkali, via the hydrothermal method, can be used as the raw material for the synthesis of xonotlite fibers through the hydrothermal synthesis method. This investigation was made to examine how the fly ash desilicated liquid influences the crystal growth and microstructure of xonotlite fibers. The obtained samples were characterized by X-ray diffraction and scanning electron microscope techniques to investigate their mineralogical composition and morphological characteristics. The results indicated that the pure desilication liquid leached from coal fly ash could be used to prepare xonotlite fibers. Xonotlite fibers with single crystal characteristics and large aspect ratio of 100–400 were successfully fabricated from fly ash desilication liquid, which is used as the silica material, at 240°C for 6 h.
Introduction
Xonotlite is an excellent thermal insulation and refractory material, with distinct physical and chemical properties, such as thermal stability, low heat conductivity, low density, fibrous form, and environmental friendliness. The products are widely used in energy-saving, fire-resistant, freeze-resistant, and other fields. 1 The xonotlite material can generally be formed via dynamic hydrothermal synthesis method using calcareous materials and different kinds of high purity silicas as the raw materials, and the molar ratio of CaO/SiO2 1.0 as the initial concentration. However, the study of synthesis conditions on xonotlite, such as the material source, impurities, crystal growth, and micromorphology, were little explained.
China is the largest among countries run by coal-fired power, and it is developing its power industry rapidly. This development has resulted in a sharp increase in emissions of fly ash produced by coal-fired power plants. 2 Currently, about 720 million tons of coal fly ash has been generated in China3,4 By 2020, fly ash emissions will reach approximately 800 million tons, which is expected to cause tremendous pressure on China’s national economy and environmental protection efforts. Meanwhile, the utilization rate of coal fly ash is low in China, it is generally used for construction industries, such as road construction, cement, and others. The silica and alumina coexist in fly ash produced by some power plants, and the total content can be as high as 80%. For this reason, using coal fly ash as raw material in silica and alumina production can alleviate the strain on diminishing bauxite resources.5–7 In recent years, pre-desilication soda lime sintering method has become the main developing trend for extracting silica and alumina from coal fly ash. The main process involves using sodium hydroxide solution mixed with coal fly ash, and dissolving silica under certain conditions to obtain fly ash desilicated liquid, and finally after lime causticization reaction, the active calcium silicate byproduct is obtained (Figure 1). In this process, utilizing the characteristics of fly ash containing amorphous SiO2, the fly ash was subjected to a desilication reaction with sodium hydroxide solution, providing a sodium silicate solution containing SiO2. So, the amorphous SiO2 can be chemically extracted from fly ash and converted into fly ash desilicated liquid, the remaining solid residue can be used as a base raw material to extract alumina by lime sintered method.8–10

Coal fly ash pre desilication soda lime sintering process.
In this experiment, the fly ash desilicated liquid developed from fly ash was used as raw material for synthesis of xonotlite fibers. It aimed at finding an effective way to deal with silicon products obtained after fly ash desilication and seeking a cheap and fine raw material for synthesis of xonotlite fibers. Thus, the production cost of xonotlite fibers could be reduced tremendously because of the low cost of silica of fly ash. At the same time, the xonotlite fibers quality will be improved because of the high activity in fly ash desilicated liquid. As a result, it is expected to be environmentally and economically benign.
Experiment
Raw materials
The fly ash desilicated liquid used in this study was prepared from coal fly ash by extraction with alkali solution. The coal fly ash was collected with bag house procedure from Toketo thermal power plant. The chemical composition of the coal fly ash was analyzed by X-ray fluorescence and the result is listed in Table 1, “Others” in the Table 1 includes unburned carbon, magnesium oxide, sodium oxide and loss on ignition, and other trace substances. The desilication of coal fly ash was started by mixing 1 kg of raw materials with 1.2 kg of water inside a thermo jacket vessel. The mixture was heated to the boiling point temperature. As soon as the mixture was boiled, 0.7 kg of sodium hydroxide solid was added for each kilogram of ash. After the temperature farther reached to 125°C, the mixture was kept at that temperature for 45–60 min to complete the desilication process. When the desilication process completed, the mixture was subjected to filtration without cooling to get the desilicated liquid.
Chemical composition of the raw coal fly ash used in the experiments.

Because of the existing impurities from fly ash, such as iron and titanium, it is difficult to get a highly pure xonotlite fibers product. Thus, we developed a pretreated method to purify the fly ash desilicated liquid using carbon dioxide. When the concentration of carbon dioxide was controlled at about 48% and the pH value of the solution was controlled in the range of 11–11.2, impurities such as ferric oxide and titanium were precipitated out from the solution. The sodium silicate liquor was then purified after filtration. Through X-ray diffraction (XRD) analysis of solid content in purified liquids, it was found that the main component was pure sodium silicate (Figure 2). The desiliconization liquid obtained in this experiment was analyzed and the concentrations of various components are shown in Table 2.

XRD patterns of the solid content from pure fly ash desilicated liquid.
Chemical compositions of fly ash desilicated liquid before and after the purification process.

Total Na was represented as Na2O.
Preparation of xonotlite fibers
The fly ash desilicated liquid was mixed with lime milk as well as silicon oxide and calcium oxide in the ratios of CaO/SiO2 = 1.0, Na2O/SiO2 = 0.6, and water–solid ratio was 40.0. Then, the mixture was put in autoclave miscellaneous high-pressure reactor and heated to 240°C for 6 h. During the crystal formation process, continuous stirring is maintained and the stirring speed is kept at 400 r/min. The crystal was then separated from the mixture by filtration. The crystal collected then was mashed with water at pH less than 8 and dried in an electric oven at 105°C for 12 h. After that, the final product obtained was the xonotlite fibers. The equation for the synthesis reaction is as follows
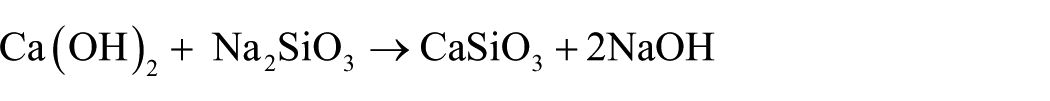

Characterization of materials
The morphology of the products was examined by scanning electron microscope (SEM, Model Hitachi S-3400N, operating at 20 and 15 kV, Japan). The mineralogical compositions of the synthesized samples and residues of fly ash after desilication were determined by XRD (Model PANalytical TD-3500, Holland) using Cu Kα radiation at 35 kV and 40 mA. The thermal conductivity and strength characteristics of the sample were also tested.
Results and discussion
Effect of purification of silica material on the crystalline phases of xonotlite
Figure 3(a) shows the XRD patterns of sample synthesized using purified fly ash desilicated liquid as silicon source while Figure 3(b) shows the XRD patterns of an anhydrite synthesized using an unpurified desiliconization solution as slicon source. The XRD patterns of the sample shows that the purification of silicon raw materials used in preparation of coal fly desilication liquid have little influence on the crystalline phases of the products. Clearly, the main crystalline phases prepared by hydrothermal synthesis method with fly ash desilicated liquid are xonotlite fibers, corresponding to 6CaO·6SiO2·H2O.

XRD patterns of the products hydrothermally synthesized using (a) purified fly ash desilicated liquid and (b) unpurified desilicated liquid as silicon source.
Effect of purification of silica material on the microscopic crystal structure of xonotlite
Figure 4(a) and (b) shows the SEM image of xonotlite products synthesized by pure and non-pure coal fly desilication liquid, respectively. It showed that the microstructure of the products obtained using pure coal fly desilication liquid as silica raw material, mainly consists of whisker fibers. It is obvious that the product xonotlite fiber has a good crystal growth, and its aspect ratio is approximately 100–400. However, the microstructure of the contrast control set shows dysplasia of whisker fibers and intertwining of xonotlite crystals, the fiber structure is incompletely formed and curved. Its aspect ratio is less than 50. Through contrast and analysis of two kinds of synthetic methods, if high whisker length and aspect ratio are expected, high concentrations of Fe3+, Ti2+, and Al3+in sodium silicate solution should be avoided, because the tilt angle of the bridge-erecting part of tetrahedral framework with higher degree of freedom can be changed by these metal ions, the C-axis of lattice is elongated and the lattice volume is increased, so that the stability of tobermorite with lamellar structure is enhanced, the lattice of anhydrite shrinks and it restrains the growth of whisker fibers.

SEM images of the different silica material products hydrothermally synthesized. (a) SEM images of products synthesized by pure coal fly desilication liquid. (b) SEM images of products synthesized by non-pure coal fly desilication liquid.
Conclusion
In this study, the fly ash desilicated liquid is used as silicon raw material for synthesis of xonotlite fibers. The desilicated liquid is obtained after extracting silica from coal fly ash via a hydrothermal process with alkali and impurities are removed by carbon dioxide separation method. Our study reveals that xonotlite produced from industrial waste has a promising potential in the future. The findings are summarized below:
The optimal dynamic hydrothermal synthesis of xonotlite fibers reaction conditions are: temperature of 260°C, holding time was 6 h, CaO/SiO2 ratio of 1.0, Na2O/SiO2 = 0.6, and water–solid ratio was 40.0.
Pure and non-pure fly ash desilicated liquid have little influence on the crystalline phases of xonotlite fibers but pose a great impact on their morphologies. Purified fly ash desilicated liquid synthesized well-developed crystal microstructure of xonotlite fibers, the aspect ratio can reach 100–400.
In the reaction process, a certain amount of impurities such as Fe2+, Al3+, Ti2+ can replace Si4+ or Ca2+. It will affect the formation of intermediate products of tobermorite. Therefore, the effective removal of Fe2+, Al3+, and Mg2+ impurities in the silica raw materials is particularly important for the synthesis of high-quality xonotlite fibers. The raw materials should be effectively purified and removed before the reaction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by the Scientific Research Projects of Inner Mongolia University of Technology (ZZ201816) and Scientific research project of Inner Mongolia University (NJZY20065).
