Abstract
Eco-friendly dyeing by using supercritical carbon dioxide as a medium has already been investigated worldwide due to the advantages of dyeing without water and recyclability of dyes and carbon dioxide. In this article, dyeing mechanism of poly(m-phenylene isophthalamide) was investigated in supercritical carbon dioxide. The obtained results showed that the dye uptake of Disperse Red 60 increased moderately with the temperature raising at constant pressure and achieved dyeing equilibrium after 70 min. By adding the carrier, diffusion coefficients of Disperse Red 60 in the polymer increased significantly in supercritical carbon dioxide. The activation energy for diffusion of Disperse Red 60 with and without carrier was 1165.91 and 1050.66 kJ mol−1, respectively. Moreover, the distribution coefficient, the standard affinity, the standard enthalpy, and the standard entropy of dyeing were also determined in supercritical carbon dioxide. These fundamental data are of vital importance on the green dyeing production of poly(m-phenylene isophthalamide).
Introduction
Poly(m-phenylene isophthalamide) (PMIA) is widely used for thermal protective materials in the defense field because of its outstanding flame retardance and tenacity.1–5 However, the hydrophobic surface of PMIA displays poor dyeing performance due to its high degree of orientation and crystallization.6–9 Various attempts have been performed to dye PMIA, of which aqueous dyeing with carriers is one of the basic approaches.10,11
Generally, most carriers used are aromatic compounds, such as butyl benzoate, methylnaphthalene, dichlorobenzene, diphenyl, and acetophenone, 2 , 6 which can emulsify in dyebath and increase the dyeing rate. 12 Deep shades for PMIA can be achieved by adding carriers since the hydrogen bonding and van der Waals force between PMIA and dye molecules are formed. 11 Nevertheless, the carriers currently used always present high toxicity and bad smell, bringing in great inconvenience to the commercial production. 13 Furthermore, a huge amount of wastewater generated from the water dyeing procedure exacerbates concerns as high concentrations of unused dyes, surfactants, and other chemicals are contained. 14
Recently, supercritical carbon dioxide dyeing and finishing has already been investigated worldwide due to the distinct advantages of no water and recyclability of dyes, auxiliaries, and carbon dioxide.15–18 With the implementation of more strict environmental laws and regulations around the world, the development of supercritical carbon dioxide dyeing on polyethylene terephthalate (PET) is accelerating.19–21 There are also some trials in applying this approach to dye aramid. Choi and Kim found that Basic Yellow 21, Basic Blue 41, and Basic Red 46 displayed comparatively higher exhaustion yield to PMIA fiber in comparison with other dyes. 22 In our previous work, the influence of supercritical carbon dioxide on the physicochemical properties of PMIA were investigated, and the obvious changes in the wettability and the surface morphology of PMIA were observed. 23 Kinetics and thermodynamics of PMIA dyeing, however, have been rarely documented although they are important since supercritical carbon dioxide is an entirely different dyebath compared with water. Therefore, a systematic study on this subject is desirable.
In this work, adsorption kinetics and thermodynamics of PMIA fiber on Disperse Red 60 were investigated with CINDYE DNK as carrier. The kinetic parameters were determined by diffusion coefficient and diffusion active energy. The thermodynamic parameters in supercritical carbon dioxide, including distribution coefficient, dyeing affinity, enthalpy, and entropy, that governing the dyeing were also studied.
Experimental
Materials
PMIA fabrics with diameters of 133 µm were provided by Teijin Aramid Asia Co. Ltd. Crude Disperse Red 60 (C20H13NO4, CAS No. 17418-58-5, >98%; Figure 1) was purchased from Zhejiang Longsheng Group Co., Ltd. (China). CINDYE DNK (oral rabbit LD50: >5000 mg kg−1), a kind of non-toxicity aromatic amide compounds showing a similar structure with PMIA, 13 was received from Shenzhen Kangyi Health Products Co., Ltd (China). Analytical reagent grade sodium hydroxide and N,N-dimethylformamide were supplied from Tianjin Kemiou Chemical Reagent Co., Ltd. (China).

Chemical structure of Disperse Red 60.
Dyeing procedure
The dyeing of PMIA was conducted in the same apparatus as previously reported. 9 As depicted in Figure 2, scoured PMIA and 4.5% o.m.f. solid dyes were put into a dyeing vessel and a dye vessel. CINDYE DNK was pumped from a co-solvent vessel into the apparatus through a liquid pump with a speed of 3 g min−1 and mixed with high-pressure carbon dioxide in a mixer. Subsequently, supercritical carbon dioxide with carriers flowed into the dye vessel to dissolve dyes and then injected into the dyeing vessel. PMIA fabrics were then dyed at 30 MPa for 10–90 min from 80°C to 140°C according to our previous work. 9 The dyed PMIA samples were finally obtained after the carbon dioxide and dyes were separated in a separator.

Schematic diagram of the dyeing apparatus.
Curve of the dyeing rate
The quantity of the dyes adsorbed on a dyed PMIA sample was evaluated by extraction of the polymer with N,N-dimethylformamide. About 0.25 g PMIA was added to 25 mL N,N-dimethylformamide to achieve the complete dye extraction. The absorption intensity of the extract liquor was then tested by the ultraviolet-visible spectrophotometer.
Standard absorbance-concentration curve of the dye solution
A quantitative estimation of Disperse Red 60 was obtained by spectroscopic determination using an ultraviolet-visible spectrophotometer at the maximum absorbance peak. A series of standard dye solutions at different concentrations were freshly prepared in N,N-dimethylformamide solution. Standard curve of the dye was generated, where Y and X refer to the absorbance of Disperse Red 60 solutions and the concentration (g L−1). As depicted in Figure 3, the standard absorbance-concentration curve conforms with Bouguer–Lambert–Beer law
where A is the absorbance of the dyeing solutions, I0 refers to the intensity of incident light and I is the intensity of transmission light, and T is the transmittance.

Standard absorbance-concentration curve of Disperse Red 60.
Results and discussion
Dyeing kinetics
Influence of dyeing temperature and time
Dyeing rates present a more practical importance in comparison with the dyeing equilibrium since few equilibrium can be reached in the actual dyeing production. 24 Unfortunately, the rates of dyeing are usually affected by some factors, such as the liquor ratio, the temperature, as well as pH. 12 The relationship between dye uptake and time is shown in Figures 4 and 5.

The effect of time and temperature on the dye uptake by PMIA fiber.
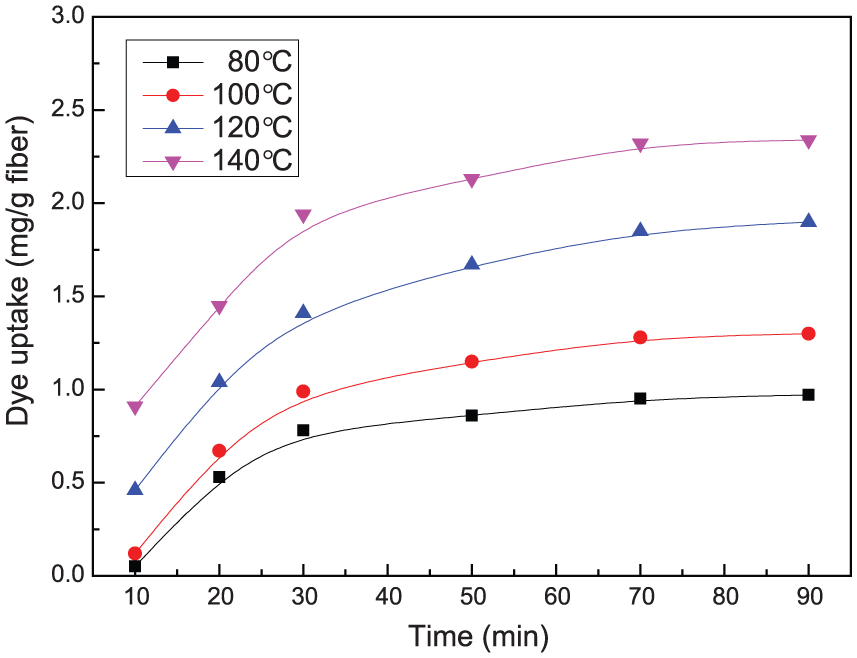
The effect of time and temperature on the dye uptake by PMIA fiber in the presence of carrier.
As seen from Figures 4 and 5, the dye uptake of Disperse Red 60 on PMIA increased initially with the time prolonging. The increase rate of dye uptake slowed after 30 min, and the equilibrium was reached after 70 min. Moreover, the dye uptake increased gradually with higher temperature due to the extremely high glass-transition temperature and high crystallinity of PMIA. The maximum dye uptake appeared at 140°C. These revealed that the higher temperature for carbon dioxide, the greater dye molecules absorbed into PMIA fiber. Furthermore, it is observed that the dye uptake was improved significantly when carrier was added into supercritical carbon dioxide. Higher carbon dioxide temperature can increase the flexibility of polymer chains, leading to greater permeability and diffusivity of dye molecule. Simultaneously, carrier is instrumental to supercritical carbon dioxide dyeing procedure. For one thing, CINDYE DNK combines with PMIA molecules with hydrogen bonds and van der Waals force, forming the fiber-to-carrier bonds. 9 During this process, the bonding force between the PMIA fibers reduced while the occurrence probability of the holes in polymer increased, thereby improving the dyes’ diffusion rate. For another, CINDYE DNK also has good plasticizing capacity for PMIA fiber. The activity ability of polymer chains enhances, resulting in the increase in the free volume. However, the highest dye uptake of Disperse Red 60 on PMIA with carrier presented less than 2.5 mg g−1 fiber, which is one order of magnitude lower than polyester due to the high degree of orientation and crystallization. 25 In addition, previous studies showed that the solubility of disperse dyes and the swelling of polymers could be improved with the increasing pressure, leading to the rising of dye uptake. 9 Thus, 30 MPa was chosen as the experimental pressure.
Diffusion coefficients
In the dyeing procedure, dye’s diffusion within fibers is usually supposed to be a rate-controlling process. Moreover, the diffusion of dye into polymer is more difficult than in dye bath due to the interactions between dyes and fibers and the existing mechanical obstruction among fiber molecules. 26
Dyes added into the dye vessel were always overdose for the dyeing of PMIA fibers. This is in fact true that the concentration of Disperse Red 60 is fixed at a constant temperature and pressure. Therefore, it is assumed that the carbon dioxide bath is the infinite dyeing bath. The relationship of the dye concentration in fiber (qA) at time t, the dye concentration in fiber at equilibrium (q∞), and the diffusion coefficient (D) can be described using Fick’s second law
where m is the positive constant and l is the sample thickness.
For a short dyeing time, equation (2) can be simplified to equation (3) 27
where df is the fiber diameter.
As shown in Figures 4 and 5, the dye uptake of Disperse Red 60 increased slightly when the PMIA was dyed for 70 min at 140°C. Thus, the dye concentration at 140°C and 70 min was selected as dye concentration in polymer at equilibrium. For equation (3), it shows better adaptability only for short dyeing time. Hence, the dyeing data within 30 min were chosen. The plots of qA/q∞ versus t1/2 are depicted in Figures 6 and 7, and the diffusion coefficients of Disperse Red 60 were calculated from the slope of linear plots (as shown in Table 1).

Dye uptake (qA/q∞) as a function of t1/2.

Dye uptake (qA/q∞) as a function of t1/2 in the presence of carrier.
Diffusion coefficients of Disperse Red 60 in PMIA fiber.

PMIA: poly(m-phenylene isophthalamide).
As listed in Table 1, the diffusion coefficient of Disperse Red 60 increased with the dyeing temperature elevation, and the maximum value was reached at 140°C. By adding the carrier, diffusion coefficients of Disperse Red 60 increased prominently. This is mainly because the increase in dye’s diffusivity led to promoting the diffusion of dye molecules into fiber. However, the Disperse Red 60 concentration on the surface of the fiber decreased gradually after the dye molecules diffused into the PMIA. Afterwards, more dyes were absorbed onto fiber surface and gradually migrated into fiber, increasing the dye uptake. But the diffusion coefficients of Disperse Red 60 in PMIA fiber are lower than Eggers’s report for PET in supercritical carbon dioxide. 27
During dyeing, the decentralized state of dye and the thickness of viscous retardation layer on the fiber surface are major factors influencing the diffusion coefficient. Carrier could reduce the granularity of disperse dyes, making the dye aggregation disperse into single dye molecules. In addition, the swelling of the polymer could be improved with carrier, which resulted in the increase of the fiber free volume and the kinetic energy of dye molecules. All of these factors led to the diffusion coefficients of Disperse Red 60 in PMIA fiber increasing finally in supercritical carbon dioxide.
Activation energy
Rate of dyeing improves with the increase in temperature while the equilibrium exhaustion usually decreases. Thus, for a dye that quickly absorbs and rapidly reaches equilibrium, dyeing at lower temperature will result in the best color yield. For a dye which absorbs slowly, the best color yield occurs under higher temperatures as dye adsorption is faster and equilibrium is not approached. 12 An optimum dyeing temperature for dye is usually determined by using a temperature range test. In this, the deepest shade can be obtained at an optimum dyeing temperature and a convenient dyeing time under the given conditions.
Activation energy (E) represents the minimum energy required for a chemical reaction. In the dyeing process, the changes in diffusion coefficient are calculated from Arrhenius equation 28 , 29

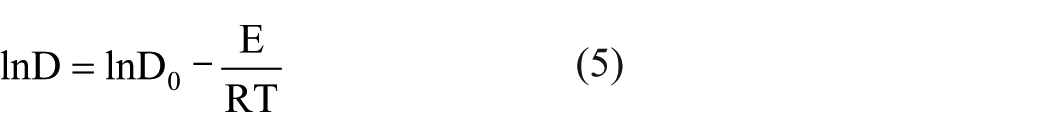
where T is the absolute temperature, D0 refers to a constant, and R refers to universal gas constant.
It can be seen in Figures 8 and 9 that the activation energy values for Disperse Red 60 with and without carrier were 1165.91 and 1050.66 kJ mol−1, respectively. Moreover, it is found that R2 was 0.99699 and 0.9587, which are close to 1. Theoretically, the activation energy can be considered as a measuring means for the barrier preventing the motion of dye molecules. The decrease in the activation energy proved that the movement barrier of Disperse Red 60 within PMIA fiber was lower by adding the carrier, thereby improving the permeability and diffusion of the dye.

ln D as a function of 1/T with Disperse Red 60.

ln D as a function of 1/T with Disperse Red 60 in the presence of carrier.
Dyeing thermodynamics
Distribution coefficients
Distribution coefficient is the ratio of dye concentration in fiber (q∞f ) and dye concentration in dye bath (q∞s ) at equilibrium. 30 , 31 Distribution coefficient can be calculated according to equation (6)

where K refers to the distribution coefficient.
The dye concentration for 70 min dyeing was regarded as equilibrium concentration in PMIA fiber because the dyeing equilibrium was approximately achieved. Accordingly, dye solubility was considered as the dye concentration at equilibrium in supercritical carbon dioxide. Shim reported that the density-based model Chrastil equation fits the experimental solubility data of Disperse Red 60 well. 32 Therefore, Chrastil equation was used to calculate the solubility of Disperse Red 60 under different temperatures.
Distribution coefficients of Disperse Red 60 are displayed in Table 2. The results revealed that the distribution coefficients reduced with the increase in the dyeing temperature. This means that at supercritical dyeing equilibrium, the increase in dye concentration in PMIA is less than the increase in dye solubility. On the contrary, by adding the carrier, the distribution coefficients increased moderately with the rising of temperature. It suggests that the increase in dye concentration in PMIA is higher than the increase in Disperse Red 60 solubility when the dyeing equilibrium was achieved.
Distribution coefficients of Disperse Red 60.

Standard affinity
In the dyeing process, dye is absorbed continually into PMIA fiber. Simultaneously, dye desorption is also occurred constantly. It is easy for dye molecule to absorb on the fiber surface with a large adsorption rate in the initial stage due to the higher chemical potential of the dye in supercritical carbon dioxide. Along with the proceeding of dyeing, the chemical potential of the dye in PMIA increased while the dye chemical potential in supercritical carbon dioxide decreased, accelerating the desorption rate. The adsorption rate will be equal to the desorption rate when dyeing equilibrium is obtained since the equal values of dye’s chemical potential in supercritical carbon dioxide and its chemical potential in PMIA were reached. The dye’s chemical potentials in medium (us) and in fiber (uf) are given by equations (7) and (8) 33


where as and af refer to the activities and the effective concentrations, respectively. (μs0) and (μf0) denote the standard chemical potentials of Disperse Red 60 in supercritical carbon dioxide and in PMIA, respectively.
At dyeing equilibrium of PMIA, us = uf. Hence, equation (9) is obtained

where −Δu0 is the standard affinity.
Theoretically, it can be assumed that the dye dissolved in the amorphous area of the fiber. The dye and the fiber were regarded as solute and solvent, respectively. The standard affinity can be calculated directly with the assumption that the term is a correct approximation for the activity quotient af/as. Thus, the standard affinity is acquired by equation (10)

As shown in Table 3, with the enhancing of temperature, the standard affinity of Disperse Red 60 decreased, which is similar to the conventional aqueous dyeing. Herein, the negative values of standard affinity of Disperse Red 60 demonstrated the spontaneous dye adsorption into PMIA occurred. 34 However, by adding carrier, the standard affinity values became bigger, which certified an increase in the driving force.
Thermodynamic affinities of Disperse Red 60.

Standard dyeing enthalpy and standard dyeing entropy
The enthalpy of dyeing and the entropy of dyeing can be derived from the temperature dependence of standard affinity by using free energy equation 35

where −Δu0 is the standard affinity, ΔH0 is the standard enthalpy, and ΔS0 is the standard entropy.
According to equation (11), the standard affinity is mainly depending on ΔH0 and ΔS0. ΔH0 plays a leading role at low temperature. When system reaches absolute zero, −Δu0 is determined by ΔH0. The contribution of ΔS0 increased moderately with the dyeing temperature raising. Under high-temperature condition, the influence of ΔS0 on affinity becomes more important. Therefore, ΔH0 and ΔS0 all play significant roles in the actual dyeing process.
By substituting equation (10) into equation (11), equation (12) is given as follows

As shown in Figures 10 and 11, ΔH° and ΔS° can be figured up according to the slope and intercept of the plots. The value of enthalpy −86.59 kJ mol−1 and the value of entropy −0.57 J/(mol °C) were given in the dyeing without the carrier. Furthermore, the enthalpy and the entropy with carrier in supercritical carbon dioxide were 807.85 kJ mol−1 and 18.17 J/(mol °C), respectively.

Distribution coefficients of Disperse Red 60.
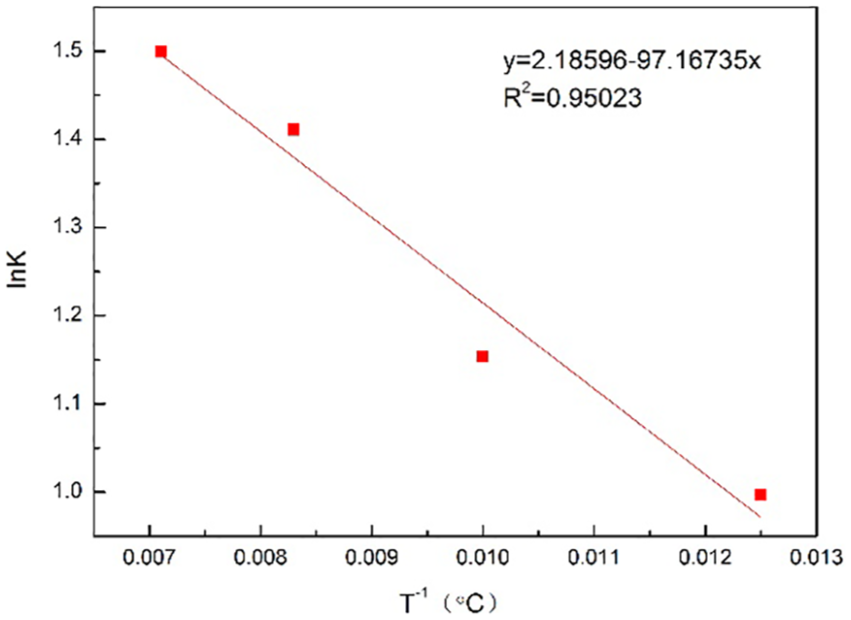
Distribution coefficients of Disperse Red 60 with carrier.
The negative enthalpy proved that there was an exothermic adsorption process existing, which presents a same trend as Banchero’s report. 25 After the carrier was introduced into supercritical carbon dioxide, the positive system enthalpy was observed, indicating that the dye uptake will be increased continuously with the dyeing temperature elevation. In addition, the value of entropy without the carrier presented smaller negative values, which represented system disorders reduced. After the carrier was introduced into the dyeing system, it found that the value of enthalpy was positive, which means that the immobilization of the dye in the PMIA fiber, relative to its freedom of movement in supercritical carbon dioxide, would give a substantial increase.
Conclusion
The dyeing kinetics and thermodynamics of PMIA were investigated with Disperse Red 60 in supercritical carbon dioxide using CINDYE DNK as a carrier. When the dyeing was conducted for 70 min at 30 MPa and 140°C, dye-uptake equilibrium was reached. The values of diffusion activation energy for Disperse Red 60 in PMIA with and without carrier were 1165.91 and 1050.66 kJ mol−1, respectively. This means that the movement barrier of the dye molecule within PMIA fiber was decreased in the presence of carrier. The standard affinity of Disperse Red 60 reduced with the rising of dyeing temperature, while the values with the carrier enhanced. In addition, the enthalpy and the entropy with and without carrier were −86.59 kJ mol−1 and −0.57 J (mol °C) −1 and 807.85 kJ mol−1 and 18.17 J (mol °C) −1, respectively.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (No. 21908015), China Postdoctoral Science Foundation (No. 2017M611420), Guidance Program of Liaoning Natural Science Foundation (2019-ZD-0285), and the Open Project Program of Key Laboratory of Eco-textiles, Ministry of Education, Jiangnan University (No. KLET1713).



