Abstract
The dissolution and recovery of cellulose from pine wood bits with two types of ionic liquids and co-solvents were explored. The results revealed that 1-ethyl-3-methylimidazolium diethylphosphonate ionic liquid/dimethylsulfoxide (mass ratio 1:0.5) could dissolve pine cellulose within 8 min at 105°C. Moreover, the degree of polymerization of the regenerated cellulose reached 244. The dissolved cellulose could be regenerated from the component solution when water was added. Then, the regenerated cellulose was characterized by X-ray powder diffraction, Fourier-transform infrared spectroscopy, and thermal gravimetric analysis measurements. The results revealed that there was no derivatization of the reaction in the process of dissolution and that dissolving the pine cellulose was a direct process. The crystalline structure of the cellulose was converted from cellulose I to cellulose II after regeneration. The regenerated cellulose was also found to have good thermal stability.
Introduction
In recent years, with the development of society, reserves of non-renewable resources have decreased, and serious environmental pollution has increased such that people have begun to focus on the applications of natural cellulose. 1 , 2 Cellulose is the most abundant biodegradable resource in the world, the conversion of cellulose into useful chemicals has attracted significant attention in green and sustainable chemistry.3–5 Cellulose is the most abundant non-food biomass and renewable carbon source on earth and is considered a long-term renewable fossil fuel alternative. 6 As a main component of plant cell walls, cellulose is a macromolecular polysaccharide that consists of D-glucose with β-1, 4 glycosidic bonds. 7 , 8 Due to the large number of extremely strong hydrogen bonds (HBs) and high crystallinity, the crystal structure of cellulose is so firm that cellulose is difficult to dissolve in common solvents. 9 , 10 An efficient pretreatment is decisive for processes based on lignocellulose, and the recalcitrance of lignocellulosic biomass poses a major challenge for its sustainable and cost-effective utilization. 11 Therefore, finding an efficient solvent to dissolve cellulose is very important. Currently, cellulose solvent systems can be divided into traditional and new solvent systems. Traditional solvent systems mainly comprise copper ammonia solution systems and carbon disulfide/sodium hydroxide (CS2/NaOH) systems. These traditional solvents have many shortcomings, such as complex technological processes, serious pollution, difficulty in recycling, and high cost. A large number of new solvent systems, such as lithium chloride (LiCl)/dimethylacetamide (DMAC), 12 N-methylmorpholine-N-oxide (NMMO), 13 , 14 and NaOH/urea, 15 , 16 have been found to be efficient in the dissolution of cellulose. However, limitations remain, such as the demand of a high processing temperature, which occasionally involves additional environmental, toxicity or other problems. 17 Ionic liquids (ILs) are a widespread focus in the chemical industry due to their unique and beneficial solvent resistance, strong polarity, lack of volatility, lack of oxidation, stability and other excellent qualities. As room-temperature molten salts, ILs have emerged as promising environmental solvents in recent years. 18 , 19 Swatloski et al. 20 examined an IL as a cellulose solvent for the regeneration and chemical modification of polysaccharides. In this study, 1-butyl-3-methylimidazolium ([BMIM]+) ILs were found to dissolve cellulose when the anion was Cl-, Br-, or SCN-. When the anion was BF4- or PF6-, the IL cannot dissolve cellulose. Therefore, the structures of the cations and anions of ILs affect cellulose dissolution. Based on the research about the cationic side chain contained a double bond, and the hydroxyl group of the IL has a strong ability to dissolve cellulose, ILs with different functions were designed to dissolve cellulose. 21 However, the viscosity of [BMIM]+ ILs is too high. N-allylpyridinium chloride IL ([APy]Cl) and 1-ethyl-3-methylimidazolium diethylphosphonate IL ([EMIM]DEP) were also used as the solvent for the dissolution of cellulose, which have low viscosity. 22 , 23 However, there are still some limitations to the application of ILs, such as their relatively high viscosity, which slows down the dissolution of cellulose. 24 To improve the disadvantages of ILs, some solvents have been added to ILs as co-solvents. The addition of a solvation co-solvent can reduce the viscosities of ILs and exhibit a certain swelling and even dissolving effect on the IL. Lignocellulose mainly consists of cellulose, hemicellulose, and lignin. The effective separation of lignocellulose and the utilization of the whole component have attracted significant attention in the world in recent years. At present, some study and exploration has been made on the separation of lignocellulose using IL, and some research results have been obtained. Xie and colleagues 25 , 26 systematically studied the dissolution of wood in ILs. The results showed that both hardwoods and softwoods are readily soluble in various imidazolium-based ILs under gentle conditions. And 1-butyl-3-methylimidazolium chloride IL and 1-allyl-3-methylimidazolium chloride IL have good solvating power for Norway spruce sawdust and Norway spruce and Southern pine thermomechanical pulp (TMP) fibers. After dissolution, wood can be regenerated as an amorphous mixture of its original components. The cellulose of the regenerated wood can be efficiently digested to glucose by a cellulase enzymatic hydrolysis treatment. They also investigated homogeneous acylation and carbanilation reactions of wood-based lignocellulosic materials in ILs. They found that highly substituted lignocellulosic esters can be obtained under mild conditions (2 h, 70°C) by reacting wood dissolved in ILs with acetyl chloride, benzoyl chloride, and acetic anhydride in the presence of pyridine. A number of reactions were also carried out in completely recycled ILs, verifying their utility for potential applications beyond the laboratory bench. Fort et al. 27 presented a simple and novel alternative approach for the processing of lignocellulosic materials that relies on their solubility in solvent systems based on the IL 1-n-butyl-3-methylimidazolium chloride ([C4mim]Cl). Cellulose could be readily reconstituted from the IL-based wood liquors in fair yields by the addition of a variety of precipitating solvents.
In this article, a systematic study of the dissolution and regeneration of cellulose in different types of ILs and different types of co-solvents was performed. The morphologies of the treated pine wood bits and regenerated cellulose were observed using a polarized optical microscope. The degree of polymerization (DP) was evaluated using Ubbelohde viscometer.
Experimental
Materials
Pine wood bits were obtained from Nature Pine Furniture Factory in Shijiazhuang, China. The chemical reagents, including sodium hydroxide, sodium hypochlorite, hydrogen peroxide, nitric acid and hydrochloric acid were acquired from Tianjin Yongda Chemical Reagent Co., Ltd. Ethylene diamine, dimethylsulfoxide (DMSO) were obtained from Beijing Chemical Reagents Co., China. DMAC and N, N-dimethylformamide (DMF) were acquired from Tianjin Kemiou Chemical Reagent Co., Ltd.
Pine wood bits were passed through a 40–60 mesh sieve (0.45–0.65 mm) and washed, dried, and boiled using a 15% sodium hydroxide solution for 2 h. After cooling, a sodium hypochlorite: hydrogen peroxide (3:4) mixed liquid bleaching was performed for 30 min at 45°C. Sawdust was acidized with diluted hydrochloric acid for 10 min.
Preparation of [APy]Cl and [EMIM]DEP
[APy]Cl and [EMIM]DEP were synthesized as described in previous work. 22 , 23 [APy]Cl was synthesized by the following process. Pyridine and chloropropylene were mixed in a three-mouth flask at a molar ratio of 1:1.1. Anhydrous ether was added as the solvent and N2 was filled as protective atmosphere. The mixture was refluxed and reacted for 12 h under 60°C. After the reaction, the solvent was removed by vacuum suction filter to get crude product. The crude product was washed with ethyl acetate for 3 times and freeze-dried for 24 h. The target compound was in the form of light yellow-brown powder. 1H-NMR (CDCl3, 500 MHz,δ): 9.72 (d, 2H, HC—N=CH), 8.59 (t, 1H, C—H on pyridine ring), 8.19 (t, 2H, C—H on pyridine ring), 6.16∼6.24 (m, 1H, CH=CH2), 5.78 (d, 2H, N—CH2), 5.51∼5.70 (q, 2H, CH=CH2).
[EMIM]DEP was synthesized by the following process. 0.5 mol of an amine was weighed into a dry three-mouth flask and 0.5 mol of triethylphosphate was added dropwise to ensure isothermal reaction conditions. The reaction mixture was stirred for 3 days at 120°C under a N2 atmosphere. 1H-NMR (CDCl3, 500 MHz), δ × 10–6: 10.617(1H, s, NCHN), 7.378 (2H, m, NCHCHN), 4.279 (2H, q, NCH2CH3), 3.982 (3H, s, NCH3), 3.857 (4H, m, P(OCH2CH3)2), 1.480 (3H, t, NCH2CH3), and 1.159 (6H, t, P(OCH2CH3)2).
Dissolution and regeneration of cellulose
Organic solvent such as DMSO, DMAC, DMF or pyridine was added to the ILs to form a component solution via mechanical stirring at a desired temperature. Then, the cellulose was added to observe its partial dissolution in the component solution using a polarizing microscope (XP-203, Shanghai, China). When the cellulose was completely dissolved, the solution was filtered while hot. The filtrate was poured into the watch glass, and distilled water was added to regenerate the cellulose. The regenerated cellulose was soaked for 24 h and repeatedly washed with distilled water and then dried in a vacuum oven at 70°C for 2 h.
Measurements of the DPs
Several drops of concentrated nitric acid were used to remove the oxide film on the surfaces of copper grains (20–25) in a beaker. The copper grains were soaked with ethylene diamine solution and repeatedly washed with distilled water. Regenerated cellulose (0.02 g; cut into pieces) was added to a beaker filled with 5 mL copper ethylene diamine solution and 5 mL distilled water. The regenerated cellulose was dissolved with magnetic stirring for 1 h. Then, the DP of the regenerated cellulose was measured using an Ubbelohde viscometer.
Results and discussion
Dissolution and regeneration of pine cellulose
ILs perform better in dissolving cellulose. 28 To investigate the effect of the IL, [EMIM]DEP and [APy]Cl were examined in terms of their ability to dissolve the cellulose of sawdust. As presented in Table 1, after adding DMF and pine cellulose, [EMIM]DEP/DMF required a lower temperature and a shorter time to dissolve the pine cellulose. More importantly, the cellulose regenerated with [EMIM]DEP/DMF was membranous, which indicated that it had better mechanical properties, but the other cellulose that was regenerated with [APy]Cl/DMF was flocculent. In addition, the high dissolution temperature caused the regenerated cellulose to exhibit serious thermal degradation. Taken together, these results led to the use of [EMIM]DEP in the further experiments.
Performance comparison of two ionic liquids for dissolving sawdust cellulose.

DMF: N, N-dimethylformamide; [APy]Cl: N-allylpyridinium chloride; [EMIM]DEP: 1-ethyl-3-methylimidazolium diethylphosphonate; IL: ionic liquid.
Co-solvents have solvation effects and can make [APy]Cl and [EMIM]DEP exhibit liquid forms below their melting points. In addition, co-solvents can reduce the viscosity of the solvent system. Therefore, co-solvents are good for swelling and even dissolving cellulose. To investigate the dissolution time of the co-solvents, DMF, DMAC, DMSO and pyridine were measured and compared. Equal quantities of the solvent and pine cellulose (3% IL) were added to [EMIM]DEP at 100°C, and the DPs of the regenerated cellulose were detected (Table 2).
Performance comparison of co-solvents for dissolving sawdust cellulose.

DMF: N, N-dimethylformamide; DMSO: dimethylsulfoxide; DP: degree of polymerization; DMAC: dimethylacetamide.
As presented in Table 2, DMSO/[EMIM]DEP required the shortest time to completely dissolve the pine cellulose. Although the DP of the regenerated cellulose was slightly lower than that of the pyridine component solvent system with the IL, DMSO had the absolute advantage of the dissolution time compared with the other co-solvents. Therefore, DMSO was used to optimize the experimental conditions.
Table 3 presents the effects of the reaction temperature and the mass ratio of [EMIM]DEP and DMSO. At the same temperature, as the mass fraction of DMSO increased, the dissolution time of [EMIM]DEP/DMSO greatly increased, and the DP of the regenerated cellulose decreased. For instance, the dissolution time increased from 75 min to 120 min and the DP decreased from 320 to 200 with the mass fraction of DMSO increased from 1:0.5 to 1:1.2 at 90℃. When the DMSO mass fraction was 1:0.5 and dissolution temperature increased from 90℃ to 105℃, dissolution time decreased from 75 to 8 min and the DP decreased from 320 to 244. It suggested that in the condition of a constant mass fraction of DMSO, as the dissolution temperature increased, the time required for cellulose dissolution and the DP of the regenerated cellulose decreased. This might have occurred because of the molecular motion increasing, and the cellulose molecular chain was breaking as the temperature increased. Therefore, the IL was more easily changed into the cellulose molecule and formed new HBs with cellulose molecules to dissolve the cellulose, which greatly shortened the dissolution time. The newly developed membrane with regenerated cellulose seemed to show no difference with the changes of DMSO mass ratios. However, when the thermal property of these regenerated membrane was tested by thermal gravimetric analysis (TGA), the results showed that the thermal stability of these regenerated membrane decreased with the increase of DMSO mass ratio (Supplemental Figure S1). The reason may be that with the increase of DMSO mass ratio, the regenerated cellulose DP decreased which caused the decrease of thermal stability. Although the degradation of the cellulose itself occurred, the DP might have declined. In addition, with the increase in the DMSO mass fraction, the cost will rise. At 105°C and with a mass ratio of [EMIM]DEP: DMSO of 1:0.5, the complete dissolution time of the pine cellulose was 8 min, and the DP of the regenerated cellulose was up to 244, which indicated that the cellulose dissolving reaction was active at this temperature and mass fraction of DMSO. It needs further investigation to find out which is having greater influence.
Performance comparison of sawdust cellulose dissolution with different mass ratios ([EMIM]DEP: DMSO) and temperatures.

[EMIM]DEP: 1-ethyl-3-methylimidazolium diethylphosphonate; DMSO: dimethylsulfoxide; DP: degree of polymerization.
The dissolution process of pine cellulose in [EMIM]DEP/DMSO under a polarizing microscope is presented in Figure 1. At the beginning of dissolution, it can be seen that there were some bright cellulose fibrils. As time progressed, the cellulose became dispersed and dissolved. The pulp was invisible in the polarizing microscope after only 24 min in [EMIM]DEP/DMSO. After dissolution, the cellulose was regenerated. Figure 2 depicts the regenerated membrane.

Optical microscopy photos (×40) of cellulose dissolution at different times: (a) 0 min, (b) 5 min, (c) 15 min, and (d) 24 min. Dissolution conditions: 100°C, pine cellulose was dissolved in (EMIM)DEP/DMSO (the mass ratio of (EMIM) with DMSO was 1:0.75).

Photograph of the regenerated membrane.
Structural characterization of the cellulose before and after regeneration
To determine any chemical changes in the cellulose before and after regeneration, the Fourier-transform infrared spectroscopy (FTIR) spectra of the treated and regenerated cellulose were compared (Figure 3). The characteristic peaks of cellulose at 1054 cm–1, 3439 cm–1 and 2917 cm–1 represented the asymmetric stretching vibration peak of C-O-C, stretching vibration peak of O-H, and stretching vibration peak of C-H. The stretching vibration peak of O-H at 3439 cm–1 moved to 3429 cm–1 after the regeneration of the cellulose, which indicated that the association between the hydroxyl groups was lessened and that the force of the hydrogen bonding was weakened. The prominent peak at 1722 cm–1 in the treated wood was attributed to a C=O stretching vibration in the acetyl groups of the hemicelluloses, 29 and it shifted to 1718 cm–1 after regeneration, which indicated that some of the hemicellulose was removed in the recovery step. 30 , 31 The peak emerged at 1054 cm–1 can be assigned to C-O of secondary alcohols and ethers in cellulose. It became stronger after regeneration. The vibration at 898 cm–1 can to beta-glycosidic links, which shifted to 887 cm–1 after regeneration. These changes also demonstrated that some of the hemicellulose was removed in the recovery step. 32 The peak wave numbers of the regenerated cellulose and the treated sawdust were very similar, which indicated that the process of dissolving pine cellulose was a direct process. Moreover, there was no derivatization of the reaction in the process of dissolution.

IR spectra of cellulose before and after regeneration. A: treated pine cellulose; B: regenerated cellulose. Dissolution conditions: 100°C, the pine cellulose was dissolved in [EMIM]DEP/DMSO.
Figure 4 illustrates the wide-angle X-ray diffraction curves of the treated pine cellulose (A) and the regenerated cellulose (B). The diffraction curve of the native cellulose was typical of a cellulose I structure and exhibited major intensity peaks that were to the crystalline structure at 2θ values of approximately 14.36°, 16.42° and 21.76°, which corresponded to the (1 1 0), (

XRD patterns of the (A) treated pine cellulose and (B) the regenerated cellulose. Dissolution conditions: 100°C, the pine cellulose was dissolved in [EMIM]DEP/DMSO.
Investigation of the thermal properties of natural fibers is important to gauge their applicability for biocomposite processing. 29 Figure 5 displays the TGA curves of the regenerated cellulose film from [EMIM]DEP/DMSO solution (A) and the treated pine cellulose (B). The thermal decomposition process of the treated pine cellulose and regenerated cellulose had three stages. A small initial drop occurred near 100°C in all cases and was due to the evaporation of the retained moisture. As presented in Figure 5, the decomposing temperature of the regenerated cellulose (200°C) was lower than that of the treated pine cellulose (250°C), which illustrated that the thermal stability of regenerated cellulose was less than that of the treated pine cellulose. However, the residual of the regenerated cellulose was higher than that of the treated pine cellulose. This result was probably because the crystallinity of the cellulose decreased, and the number of intermolecular HBs was reduced, thus weakening the intermolecular force and allowing the molecular chains to be more freely active. This change led to decrease in the thermal decomposition temperature and a reduction in the temperature range.
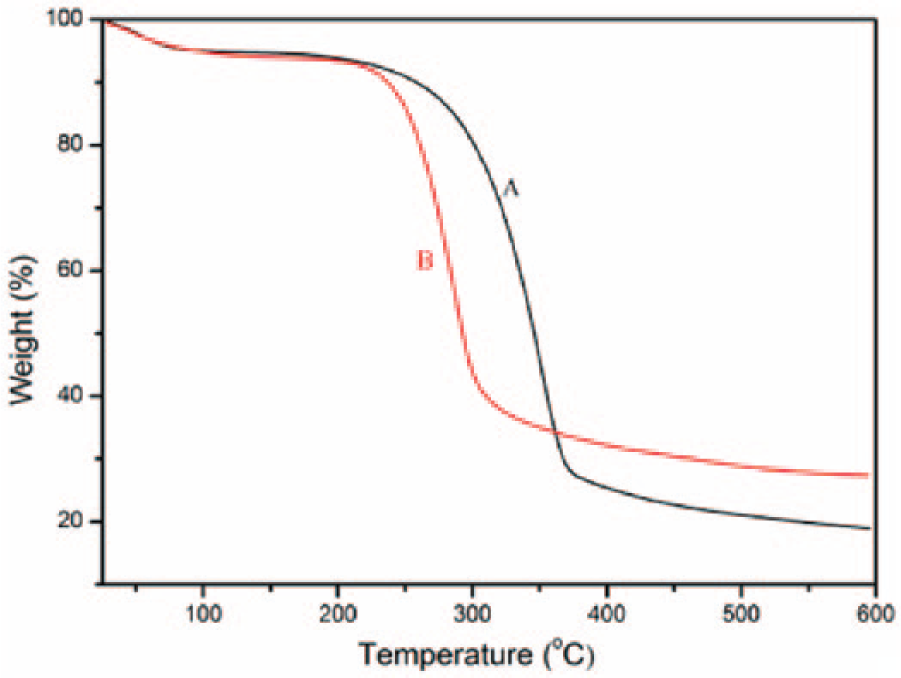
TGA curves of the (A) treated pine cellulose and (B) regenerated cellulose film from [EMIM]DEP/DMSO.
Conclusion
The dissolution properties of pine cellulose in ILs and co-solvents were measured. DMSO was better than several other solvents under the same conditions for dissolving pine cellulose. When the [EMIM]DEP:DMSO ratio was 1:0.5, 3% of the cellulose could be dissolved at 105°C within 8 min, and the DP of the regenerated cellulose reached 244. The crystalline structure of the cellulose was converted from cellulose I to cellulose II after regeneration. There was no derivatization of the reaction in the process of the dissolution, as evidenced from IR spectroscopy. The regenerated cellulose exhibited good thermal stability according to TGA analysis. Therefore, [EMIM]DEP/DMSO is a promising solvent for recovering cellulose from wood off cut.
Supplemental Material
Supporting_Information_(JEFF-18-0009)1.26 – Supplemental material for Dissolution and recovery of cellulose from pine wood bits in ionic liquids and a co-solvent component mixed system
Supplemental material, Supporting_Information_(JEFF-18-0009)1.26 for Dissolution and recovery of cellulose from pine wood bits in ionic liquids and a co-solvent component mixed system by Ran Liu, Juan Zhang, Shuang Sun, Yuhang Bian and Yanhui Hu in Journal of Engineered Fibers and Fabrics
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for the support of the National Nature Science Foundation of China (Grant No. 21106032), Education department of Hebei province (Grant No. ZD201606, Grant No. QN2017067), and Scientific treatment of haze and air pollution prevention and control key research projects of Hebei University of Science and Technology (Grant No. 2017020).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
