Abstract
Background
Trastuzumab emtansine (T-DM1) is indicated for HER2-positive early breast cancer with residual invasive disease after neoadjuvant chemotherapy (NAC). However, the extent of benefit T-DM1 will provide over trastuzumab plus pertuzumab (HP) in patients with microscopic residual invasive disease after NAC remains unclear.
Methods
Data of patients with HER2-positive breast cancer who were treated with HER2-targeted therapy as NAC and completed HP or T-DM1 as adjuvant therapy from October 2018 to October 2022 were retrospectively extracted from the institutional database. Invasive disease-free survival (IDFS), overall survival (OS), and safety were analyzed.
Results
Of the 110 patients, 61 achieved pCR (55.4%), and 49 had residual invasive disease. Of the 49 patients, 29 received HP, and 20 received T-DM1 as adjuvant therapy. The proportion of ypT1aN0 was 31.0% (n = 9/29) in the HP group and 45.0% (n = 9/20) in the T-DM1 group. The 3-year IDFS and OS were 96.6% and 95.0%, and 96.6% and 100% in the HP and T-DM1 groups, respectively. The 3-year IDFS and OS of patients with ypT1aN0 were 100% in both groups. The most common adverse events of any grade in the T-DM1 group were aspartate aminotransferase increased (90%) and alanine aminotransferase increased (85%) aminotransferase and platelet count decreased (70%).
Conclusion
The 3-year IDFS and OS of patients with ypT1aN0 disease who received HP or T-DM1 as adjuvant therapy were comparable.
Keywords
Introduction
Patients with HER2-positive early breast cancer with residual invasive disease at surgery following neoadjuvant chemotherapy (NAC) have a worse prognosis than those with a pathologic complete response (pCR); thus, the development of a response-guided approach is remarkably advanced. 1 Until 2018, the only HER2-targeted therapy available for HER2-positive early breast cancer was trastuzumab, a humanized monoclonal antibody directed against domain IV of the extracellular domain of HER2. 2 Pertuzumab was approved in October 2018 for the perioperative treatment of HER2-positive breast cancer.3–7 Pertuzumab is a humanized monoclonal antibody that binds to the dimerization domain of HER2 and inhibits the heterodimerization of HER2 with other HER receptors. 8 Meanwhile, trastuzumab emtansine (T-DM1) was approved in Japan in August 2020 for patients with non-pCR after NAC based on the results of the KATHERINE trial. 9 T-DM1 is an antibody–drug conjugate (ADC) consisting of trastuzumab and emtansine (DM1), a maytansine derivative and microtubule inhibitor. 9 The definition of pCR in the KATHERINE trial was ypT0/isN0; therefore, ypT1aN0 is an indication for T-DM1 post-surgery. However, the extent of benefit T-DM1 that will provide over trastuzumab plus pertuzumab (HP) in patients with microscopic residual invasive disease after NAC remains unclear. In the subgroup analysis of the KATHERINE trial, the invasive disease-free survival (IDFS) at 7 years in patients with ypT1mic-T1b and ypN0 disease was 85.7% in the T-DM1 group and 76.7% in the trastuzumab group (hazard ratio 0.62; 95% CI, 0.37 to 1.03). 10 However, IDFS data on patients with ypT1aN0 were not available in the KATHERINE trial. The KATHERINE trial compared T-DM1 with trastuzumab, and pertuzumab was used as NAC in only 18% of patients; therefore, T-DM1 was not compared with HP. To explore the prognosis of patients with ypT1aN0 disease after NAC who received HP for adjuvant therapy, we conducted a retrospective analysis in a single institution to determine the survival of patients with HER2-positive breast cancer who had microscopic residual invasive disease after NAC.
Patients and methods
We retrospectively extracted data and medical record of patients with HER2-positive breast cancer who were treated with taxanes plus HP as NAC and completed HP or T-DM1 as adjuvant therapy from October 2018 to October 2022 from the institutional database. Patients who did not receive HER2-targeted therapy as NAC, with occult cancer due to lack of pathological evaluation of the breast, and who received an investigational drug were excluded. IDFS, overall survival (OS), and safety were analyzed. IDFS and OS curves were generated using the Kaplan–Meier method and compared using the log-rank test. Probability values between groups were evaluated by Fisher’s exact test. For all tests, P < 0.05 was considered statistically significant. All statistical analyses were performed with EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). 11 More precisely, it is a modified version of R commander (version 1.60) designed to add statistical functions frequently used in biostatistics.
Results
We screened 126 patients with HER2-positive breast cancer who completed HP or T-DM1 as adjuvant chemotherapy after taxanes plus HP with or without anthracycline as NAC from October 2018 to October 2022. We excluded 15 patients due to the following: eight did not receive pertuzumab as NAC; four received only trastuzumab as adjuvant therapy; one discontinued NAC due to adverse events; one did not receive any HER2-targeted drug as NAC; one participated in a clinical trial; and one had occult cancer (Figure 1). Overall, 55.4% (n = 61/110) of the patients achieved pCR (ypT0/isN0) with NAC, and 49 patients were analyzed (Figure 1); 29 were treated with HP, and 20 were treated with T-DM1 as adjuvant therapy. The median duration of follow-up was 49 months in the HP group and 36 months in the T-DM1 group. Patient flow diagram.
Patient characteristics.

The 3-year IDFS and OS were 96.6% and 95.0%, and 96.6% and 100% in the HP and T-DM1 groups, respectively; they were not statistically different between both groups (Figure 2(a) and (b)). The 3-year IDFS and OS of patients with ypT1aN0 disease were 100% in both groups (Figure 2(c) and (d)). There was no difference in the background characteristics, for example, lymphovascular invasion of patients with ypT1aN0 disease between the HP and T-DM1 groups. (a) 3-year invasive disease-free survival of all patients; (b) 3-year overall survival of all patients; (c) 3-year invasive disease-free survival of patients with ypT1aN0; (d) 3-year overall survival of patients with ypT1aN0.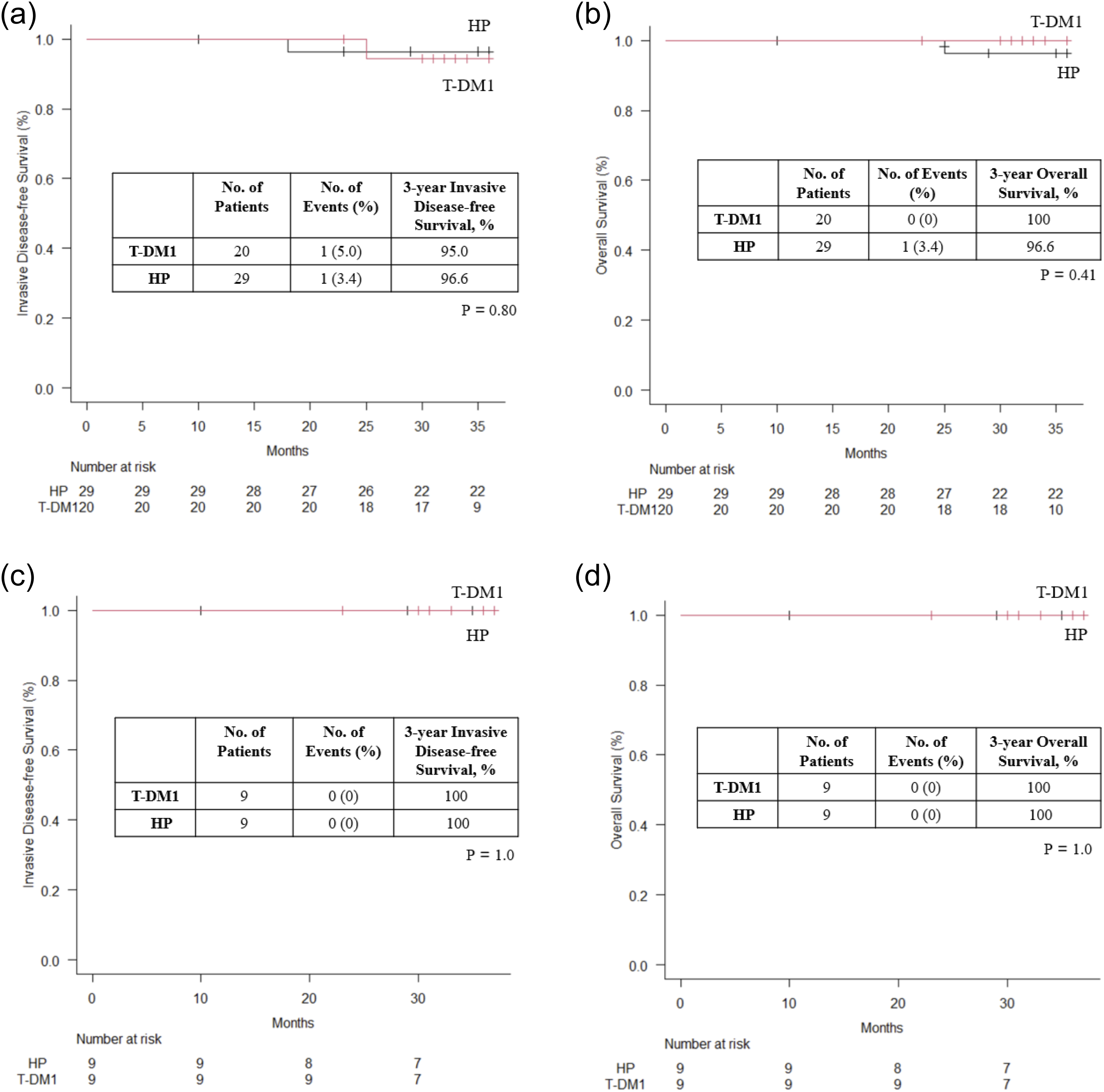
Adverse events.

Discussion
In this study, patients with ypT1aN0 disease who received HP as adjuvant chemotherapy after surgery following NAC had no recurrence at the 3-year follow-up and had a very good prognosis. In the subgroup analysis of the KATHERINE trial, the IDFS at 7 years in patients with ypT1mic-T1b and ypN0 disease was worse in the trastuzumab group (76.7%) than in the T-DM1 group (85.7%) (hazard ratio 0.62; 95% CI, 0.37 to 1.03). The proportions of patients who received pertuzumab and anthracycline in this study were 100% and 96.6% in the HP group, 100% and 100% in the T-DM1 group, while those in the KATHERINE trial were 18.7% and 75.9% in the trastuzumab group, 17.9% and 77.9% in the T-DM1 group. 9 The differences in the usage rate of pertuzumab as NAC may have caused the difference in IDFS. Furthermore, the fact that slightly more patients in the KATHERINE study had worse pathological stage than in this study may have been related to the difference in IDFS. The BERENICE trial, which continued HP as adjuvant chemotherapy after surgery following NAC with HP plus taxane and anthracycline, reported a 4-year IDFS rate and 5-year OS rate of 92.6% and 96.1%, respectively. 12 The results of this trial suggest that receiving HP as NAC and continuing postoperative HP have a favorable prognosis. However, no analysis focusing on patients with ypT1aN0 disease has been conducted. To the best of our knowledge, this is the first report showing the good prognosis of patients with ypT1aN0 disease who received HP instead of T-DM1 after NAC, suggesting that HP is a viable option in this setting.
The incidences of any grade of aminotransferase (AST) increased (90%) and alanine aminotransferase (ALT) increased (85%) and platelet count decreased (70%) in the T-DM1 group were higher than those reported in the KATHERINE trial (28.4%, 23.1%, and 28.5%, respectively). 9 A meta-analysis of randomized clinical trials comparing T-DM1 with a control treatment showed that the relative risk for all-grade AST and ALT elevations was 3.24 (95% CI 2.16–4.86; p < 0.00001) and 2.90 (95% CI 1.98–4.23; p < 0.00001), respectively. 13 T-DM1-induced hepatotoxicity sometimes caused treatment interruption and dose adjustment. T-DM1 also induces nodular regenerative hyperplasia (NRH), and with 19 cases of NRH having been attributed to T-DM1 according to the World Health Organization adverse drug reaction (ADR) database. 14 The incidence of any grade of platelet count decreased (69.8%) among Japanese patients with advanced breast cancer was higher than that in the KATHERINE trial (28.5%). 15 Nausea of any grade is also common in T-DM1, occurring in 41.5% of patients in the KATHERINE trial. 9 In comparison, the NeoSphere study showed a nausea rate of only 14% with HP. 6 Adverse events leading to discontinuation of the drug occurred in 10% of patients in the T-DM1 group, similar to that in the KATHERINE trial (18%). 9 In the BERENICE trial, only 5.0% of patients discontinued HP due to adverse events. 12 In our study, only 1 patient (3.4%) discontinued HP due to a grade 2 decrease in ejection fraction, although the discontinuation rate was similar to previous studies, despite the limitation of the small patient numbers. There is no survival difference between T-DM1 and HP, and from a toxicity point of view, HP may be a viable option as adjuvant therapy after NAC for patients with microscopic residual invasive disease such as ypT1aN0.
One patient in the HP group whose tumor had low HER2 expression after NAC died. The KATHERINE trial noted that 8.3% of patients who initially had HER2-positive tumors changed to HER2-negative tumors post-NAC. 16 Among these patients, the 3-year IDFS rate was 100% in the T-DM1 group (n = 28) and 70.1% in the trastuzumab group (n = 42). 16 Other studies have also reported that a change from HER2-positive to HER2-negative status after NAC, particularly with the use of HER2-targeted therapies, is associated with a poorer prognosis.17,18 T-DM1 as adjuvant therapy will reduce the risk of disease recurrence in patients in whom the residual disease HER2 status changes from positive to negative.
The pCR rate after NAC consisting of taxane plus HP with anthracycline in this study was 55.4%, which was slightly lower than that reported in a previous study on HER2-positive breast cancer.7,19 However, our study showed that the T-DM1 group exhibited good 3-year IDFS or OS despite non-pCR. An analysis of 11 randomized trials reported that pCR cannot be used as a trial-level surrogate for event-free survival or OS following NAC for HER2-positive breast cancer. 20 In HER2-positive early breast cancer, pCR after NAC may not be used as a surrogate for survival with the development of HER2-targeted drugs. The HER2DX genomic test, which predicts survival and treatment response by analyzing 27 genes for HER2-positive breast cancer, has recently been developed. 21 In line with this, the ATEMPT trial revealed that the HER2DX risk score was associated with IDFS in patients with stage I HER2-positive breast cancer. 22 The APT trial also showed that the HER2DX risk score was associated with IDFS and recurrence-free interval. 23 In the future, it will be important to evaluate the HER2DX risk score in addition to the clinical risk and to consider whether to escalate or de-escalate the drug comprehensively.
Our study has several limitations. The number of patients was limited due to this being a single institutional study. Randomized controlled trials are ideally needed; however, due to the scarcity of the target population, they may be difficult to conduct from the viewpoint of feasibility. Hence, retrospective data, such as those used in our study, or a large real-world database is crucial. The follow-up period in this study was short, and long-term follow-up is needed. This was a retrospective study; therefore, there may be selection bias for HP or T-DM1.
In conclusion, the 3-year IDFS and OS of patients with ypT1aN0 disease who received HP or T-DM1 as adjuvant therapy were comparable. Long-term follow-up and accumulation of cases are needed to validate our results.
Footnotes
Acknowledgments
We thank all the participants in this study, including patients, caregivers, physicians, and medical workers.
ORCID iDs
Ethical considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee.
Author contributions
Conception: Meiko Nishimura and Takayuki Kobayashi; data curation: Meiko Nishimura and Tomo Osako; analysis of data: Meiko Nishimura; preparation of the manuscript: Meiko Nishimura, Yosuke Aoyama, Saori Kawai, Jun Masuda, Yukinori Ozaki, Mari Hosonaga, Ippei Fukada, Tetsuyo Maeda, Nami Yamashita, and Toshimi Takano; revision for important intellectual content: Takayuki Kobayashi and Takayuki Ueno; and supervision: Takayuki Kobayashi and Takayuki Ueno.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Meiko Nishimura has received honoraria for lectures from Pfizer, Kyowa Kirin, Chugai, Daiichi Sankyo and Eizai. Yosuke Aoyama has received honoraria for lectures from TAIHO, MSD, and Pfizer. Yukinori Ozaki has received honoraria from Pfizer, Kyowa Kirin, Eli Lilly, and Daiichi Sankyo. Tomo Osako has received honoraria from Roche Diagnostic, Daiichi Sankyo, and Exact Sciences. Toshimi Takano has received honoraria for lectures from Chugai, Daiichi Sankyo, and Eli Lilly. Takayuki Ueno has received payments to his institution from Eli Lilly and payments from Chugai, Eisai, AstraZeneca, and Novartis. The other authors did not report any conflicts of interest.
