Abstract
Paget disease (PD) of the breast is a rare malignancy of the nipple–areola region that is almost always associated with an underlying in situ or invasive carcinoma. It can affect both women and men. It is more commonly seen in postmenopausal women. Clinical features may be the only sign of cancer during the initial presentation, making a diagnosis challenging. Early changes in PD include itching, scaling, and erythema of the nipple that can mimic other benign conditions, such as eczema. Histologically, it is characterized by an intraepidermal proliferation of malignant glandular epithelial cells in the nipple–areolar region. Imaging plays an important role in evaluating the extent of involvement and in deciding patient management. This article will provide an overview of the clinical features, histopathology, associated imaging findings, and management of PD.
Introduction
Paget disease (PD) of the breast is a rare malignancy of the nipple–areola region that accounts for approximately 1%–4% of all primary breast malignancies. 1 It is characterized by an intraepidermal proliferation of malignant glandular epithelial cells in the nipple–areolar complex (NAC). PD has been reported in patients aged 24–90 years1–3; however, it is more common in postmenopausal women in the sixth decade of life.1–4 The median age reported at diagnosis is 61 (Ma). Although uncommon, PD has been reported to be bilaterally, 5 and it is rare in men.1,6,7 PD risk factors, outside those recognized for breast cancer, have not been identified.1,8
In 1874, Sir James Paget published his findings that an eczematous lesion of the nipple was associated with an underlying cancer. The “disease began as an eruption on the nipple and areolar, and cancer of the mammary gland followed within two years”. 9 Clinically, PD can present with an itchy, scaly, erythematous nipple with extension to the areola and surrounding skin of the breast. Because the clinical findings can appear innocuous and similar to nipple eczema, a delay in diagnosis can occur. 10 A diagnosis of PD disease can be made from a wedge biopsy, a superficial “shave” biopsy of the epidermis, or punch biopsy of the nipple–areolar region. Since more than 90% of PD cases are associated with an underlying malignancy, ductal carcinoma in situ (DCIS) or invasive breast cancer-no special type (IBC-NST),1–3,11,12 imaging should be performed to detect the underlying carcinoma. Patients with a palpable mass or suspicious mammographic findings have a higher probability of an underlying invasive cancer. 13 Even with a normal mammogram and no palpable mass, an underlying malignancy is likely present. 12 Breast magnetic resonance imaging (MR) is an effective diagnostic tool for evaluating the extent of involvement in PD and in deciding patient management. This article will provide an overview of the clinical features, histopathology, associated imaging findings, and management of PD.
Clinical features
PD of the breast develops slowly and insidiously over months to years as it extends from the nipple to the areola in a centrifugal growth pattern.14–16 Most PD cases are unilateral.1,2,17 At an early stage, the affected nipple may be pruritic and initially the nipple may appear normal.1,2,10,12,14,18 As the disease progresses, scaling and erythema of the nipple and/or areola occur and can be misdiagnosed as eczema or other benign inflammatory conditions, such as dermatitis, psoriasis, or nipple adenoma.1,12,19–21 Advanced cases of PD can be accompanied by nipple erosion or ulceration, nipple retraction, and bloody discharge.1,12,22,23 In a study consisting of 223 women with PD of the nipple, the authors reported that 98% (219/223) of the patients presented with eczema or ulceration of the nipple and 10% (23/223) had bloody nipple discharge. 18
Topical corticosteroids may temporarily improve the inflammatory component of PD.2,10,12,14,16 Thus, masking the underlying cancer and potentially causing a delay in diagnosis. Malignancies to consider in the differential diagnosis of PD include Bowen’s disease (squamous cell carcinoma in situ), basal cell carcinoma, cutaneous extension of breast carcinoma, and melanoma.1,12 Clinically, pigmented PD (PPD) can mimic melanoma since it presents as a dark macule without other skin changes of the nipple–areolar region. 1 In addition, the presentation of PD is different from inflammatory breast cancer in which there is diffuse erythema of the breast with secondary involvement of the nipple and skin. 19
Although PD can develop alone without underlying cancer, DCIS or IBC-NST, the majority of cases of PD coexist with an underlying breast cancer.1,2,5,7,12 Thus, in addition to visual inspection of the breast, a physical examination of the breast should be performed, since approximately 50% of patients with PD will have an associated palpable mass. 12 It has been reported that greater than 90% of patients with PD that have a palpable mass will have associated invasive cancer. 12 In contrast, patients with PD who present without a palpable mass are more likely to have DCIS. 23 The underlying carcinoma, which may or may not be palpable, tends to be located close to the areola or centrally. 23 However, the underlying carcinoma may also be located peripherally in the breast. In a study consisting of 35 patients with PD, the authors reported that 45% (5/11) of the palpable invasive carcinomas were located in the upper, outer quadrant of the breast. 24
The diagnosis of PD can be made from a superficial “shave” biopsy of the epidermis, a wedge biopsy, or a punch biopsy of the affected nipple and/or the areolar region. However, a shave biopsy may not provide a sufficient number of Paget cells, especially when the nipple is ulcerated. On the contrary, a wedge biopsy adequately samples the epidermis and most likely includes part of a lactiferous duct that may be involved with an underlying carcinoma of the breast. Although a punch biopsy includes underlying tissue and possibly part of a duct, it can yield limited epidermis to be examined. 19
Pathogenesis
The pathogenesis of mammary PD is unknown. Nevertheless, two theories have been proposed. The first and most widely accepted theory is the epidermotropic theory. It suggests that Paget cells originate from an underlying breast carcinoma and migrate along the basement membrane of the ducts to the nipple. 25 The basement membrane of the ducts and lobules is in continuity with the basement membrane of the skin; therefore, DCIS can be present in both the epidermis and the breast ductal system in the absence of stromal invasion. 25 The migration of tumor cells is facilitated by a motility factor called heregulin-alpha, which is synthesized by keratinocytes and interacts with the human epidermal growth factor receptor (HER2). 26 This theory is supported by the high incidence of PD with underlying DCIS.1,26 IBC-NST may also be present. 1 Further evidence to support this theory is the high level of HER2 overexpression in Paget cells, which reflects the HER2 overexpression of the underlying carcinoma. 16 The results of immunohistochemical analysis demonstrate similar staining for Paget cells and ductal epithelial cells, but not for keratinocytes, which is direct evidence of the epidermotropic theory. 7
The transformation theory is based on a change of the epidermal keratinocytes in the absence of an underlying carcinoma.11,27 Paget cells are transformed in situ and derive from cells in the terminal lactiferous duct at its junction with the epidermis. 7 Toker cells have also been hypothesized to undergo malignant transformation into Paget cells.1,27 This theory is supported by the morphological similarity between Paget cells and Toker cells, which are non-malignant, cytoplasm-rich epithelial cells of sebaceous glands.7,28 However, unlike Paget cells, which overexpress HER2 and Ki-67, Toker cells are HER2 negative and express low levels of Ki-67. 28 This theory may explain the low incidence (13%) in which PD is not associated with an underlying carcinoma or the carcinoma is distant from the nipple–areolar region.1,7,26.
Histopathology
PD is characterized by an intraepidermal proliferation of malignant glandular epithelial cells in the nipple–areolar region.
1
The malignant epithelial cells, known as Paget cells, are large in size and contain abundant pale cytoplasm with pleomorphic nuclei containing prominent nucleoli that have variable mitotic activity
1
(Figure 1). Paget cells are usually identified at the dermoepidermal junction and can be seen as single cells or clusters that rarely form glandular structures.
1
Up to 40% of Paget cells contain mucin and may phagocytose melanin, mimicking melanocytes.
1
When Paget cells are associated with a marked increase of non-neoplastic melanocytes it is termed pigmented PD.
1
Dermal invasion by Paget cells has been reported in 4%–8% of PD cases.
1
Dermal invasion is diagnosed when Paget cells invade through the basement membrane and there is a separation of the Paget cells from an underlying breast cancer. An associated lymphocytic infiltrate can also be seen in the underlying dermis.
1
Paget disease of the breast. (A) An intraepidermal malignant proliferation of epithelial (Paget) cells (arrow) is identified in a thickened epidermis of the nipple. There is also ulceration of the epidermis (arrowhead), H&E (×20). (B) Paget cells (arrows) are large in size and contain abundant pale cytoplasm with pleomorphic nuclei, H&E (×200). (C) Paget cells stain positive for the immunostain cytokeratin-7 (open arrow) (×200) and for (D) the epithelial membrane antigen (arrow) (×200). H&E: hematoxylin and eosin.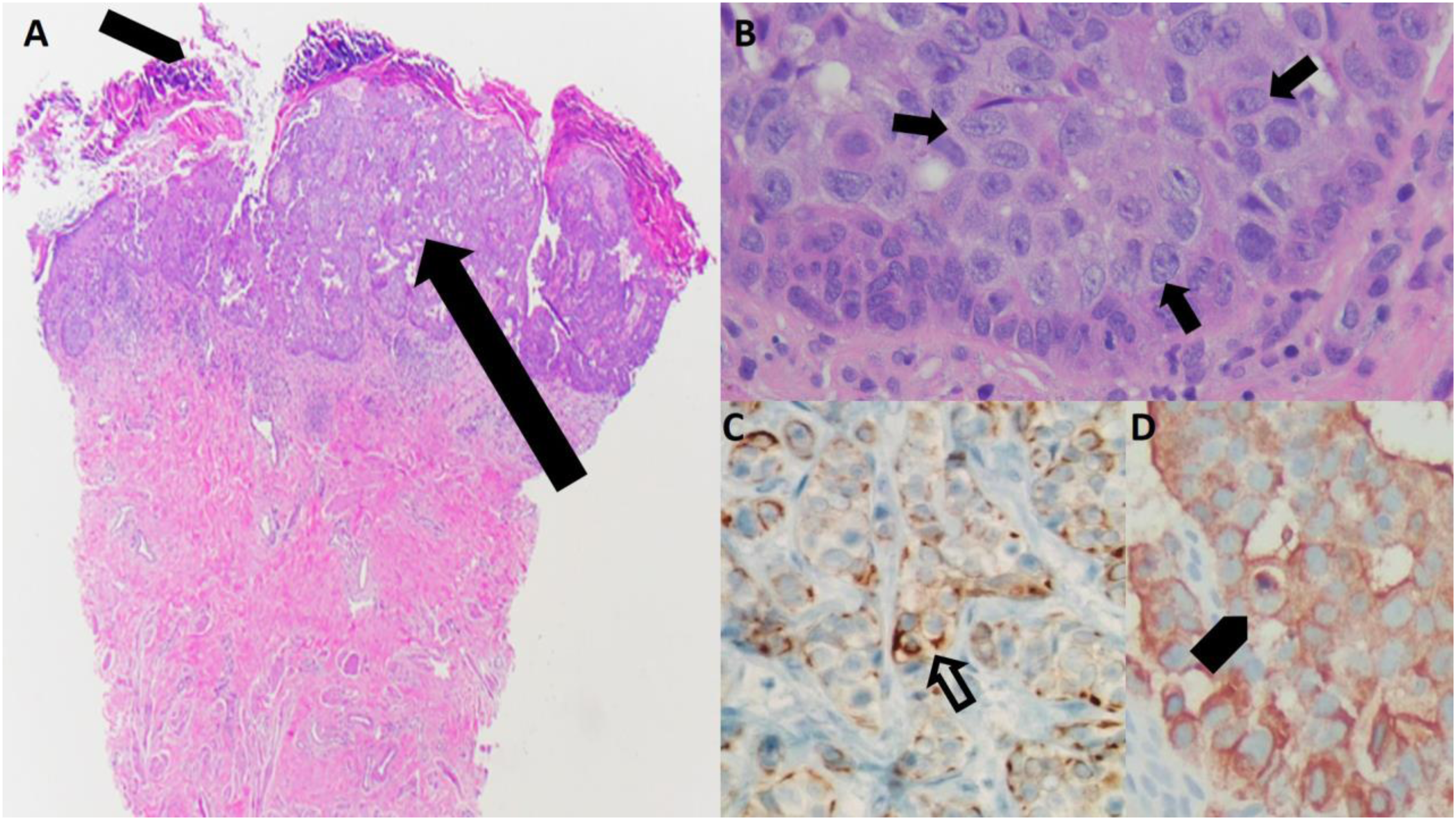
Paget cells are typically immunohistochemically positive for low-molecular weight cytokeratins, such as CK7, and for epithelial membrane antigen (Figure 1). 1 HER2 overexpression is present in 80%–90% of cases.8,23 Whereas the estrogen receptor (ER) and the progesterone receptor (PR) are positive in 40% and 30% of cases, respectively. 1 Paget cells usually have the same immunoprofile as the underlying carcinoma. 1 Toker cells may resemble Paget cells, but lack atypical or malignant features and are HER2-negative and ER-positive.1,8
Histologically, PD can mimic malignant melanoma, especially if Paget cells have incorporated melanin from the adjacent epidermis. Using special stains such as mucicarmine, which highlight cytoplasmic mucin vacuoles in Paget cells, it is possible to differentiate PD from melanoma. 8
The prevalence of an associated underlying carcinoma in PD ranges from 67% to 100%, and most studies report the presence of a coexisting malignancy in more than 90% of patients with PD.2,7,12,26,29 DICS accounts for 24%–43% 1 of the cases; while most commonly, high-grade IBC-NST accounts for 53%–64% of the cases.1,7,14 In a Surveillance, Epidemiology, and End Results Program (SEER) analysis of 5398 patients with PD, Yao et al. reported that 1725 (32.0%) had PD-DCIS, 3047 (56.4%) had PD-IDC, and that 88.4% of patients with PD presented with an underlying carcinoma. 7 The reported incidence of axillary nodal involvement in patients with PD-only, PD-DCIS, and PD-IBC is 20.2%, 3.3% and 33.2%, respectively. 7
The underlying cancer in patents with PD is usually located centrally, but it can also be located elsewhere in the breast in up to 41% of cases, and be multifocal or multicentric in distribution, consistent with the ability of the cancer cells to traverse the ducts.4,5 The prevalence of multifocality and multicentricity of the underlying carcinoma in PD has been reported to be up to 63% and 34%, respectively. 23 Both DCIS and IBC-NST associated with PD are more often of a higher histological grade, hormone receptor negative and HER2 positive (80% of cases) than cancers that are not associated with PD.1,7,14,23
PD without an underlying carcinoma is less common with a reported incidence of 0%–13%.1,7 Furthermore, PD has been incidentally identified on histology in 15% of mastectomy specimens for invasive carcinoma that were subclinical.1,27
Diagnosis
The diagnosis of PD can be made from a superficial “shave” biopsy of the epidermis, a wedge biopsy, or a punch biopsy of the affected nipple and/or the areolar region. However, a shave biopsy may not provide a sufficient number of Paget cells, especially when the nipple is ulcerated. On the contrary, a wedge biopsy adequately samples the epidermis and most likely includes part of a lactiferous duct that may be involved with an underlying carcinoma of the breast. Although a punch biopsy includes underlying tissue and possibly part of a duct, it often yields limited epidermis to examine. 19
Imaging features
After a diagnosis of PD is confirmed, breast imaging should be performed due to the high likelihood of an underlying DCIS and/or IBC.12,25 Initial workup should include diagnostic mammography and ultrasound. The mammogram may be negative in up to 50% of cases. 12 Breast magnetic resonance imaging (MR) can help detect clinically and mammographically occult cancer in patients with PD.30–32 MR can also be utilized to establish the extent of the disease in patients with PD for appropriate surgical management.
Mammography
Although PD is a clinical diagnosis, the majority of patients have an underlying carcinoma.12,23,25 Thus, the initial evaluation should include diagnostic mammography and ultrasound to further characterize the clinical findings and determine the extent of disease. Magnification views of the NAC can also be obtained as part of the diagnostic evaluation to increase detection of calcifications or other subtle findings just deep to the NAC. 5
Mammographic findings that can be seen with PD include thickening, retraction, or flattening of the nipple and periareolar skin of the affected side (Figures 2 and 3).5,11 Other findings include a suspicious mass or masses, malignant calcifications, asymmetry, or architectural distortion in the retroareolar region or elsewhere in the breast (Figures 3 and 4).11,12,33 In a study with 52 patients with PD, the most common imaging findings in 17 (33%) patients with clinically evident PD were microcalcifications in 3 (18%), mass with microcalcifications in 5 (29%), a mass in 2 (12%) and negative in 7 (41%) cases.
4
In the same study, there were 35 (67%) patients without clinically evident PD, and the mammographic findings for this cohort were calcifications in 15 (43%), mass with calcifications in 12 (34%), a mass in 7 (20%) and negative in 1 (3%) case.
4
In a study by Ikeda et al. with 58 patients with PD, the mammographic findings in 34 (59%) patients with clinically evident PD were nipple thickening in 1 (3%), calcifications involving the nipple and breast in 4 (11.7%), breast calcifications only in 1 (3%), a mass with or without calcifications in 11 (32.3%) and negative in 17 (50%) cases.
33
In the same study, there were 24 (41%) patients without clinically evident PD, the mammographic findings for this cohort were calcifications involving the nipple and breast in 1 (4%), breast calcifications only in 9 (38%), a mass with or without calcifications in 12 (50%), architectural distortion in 1 (4%) and negative in 1 (4%) case.
33
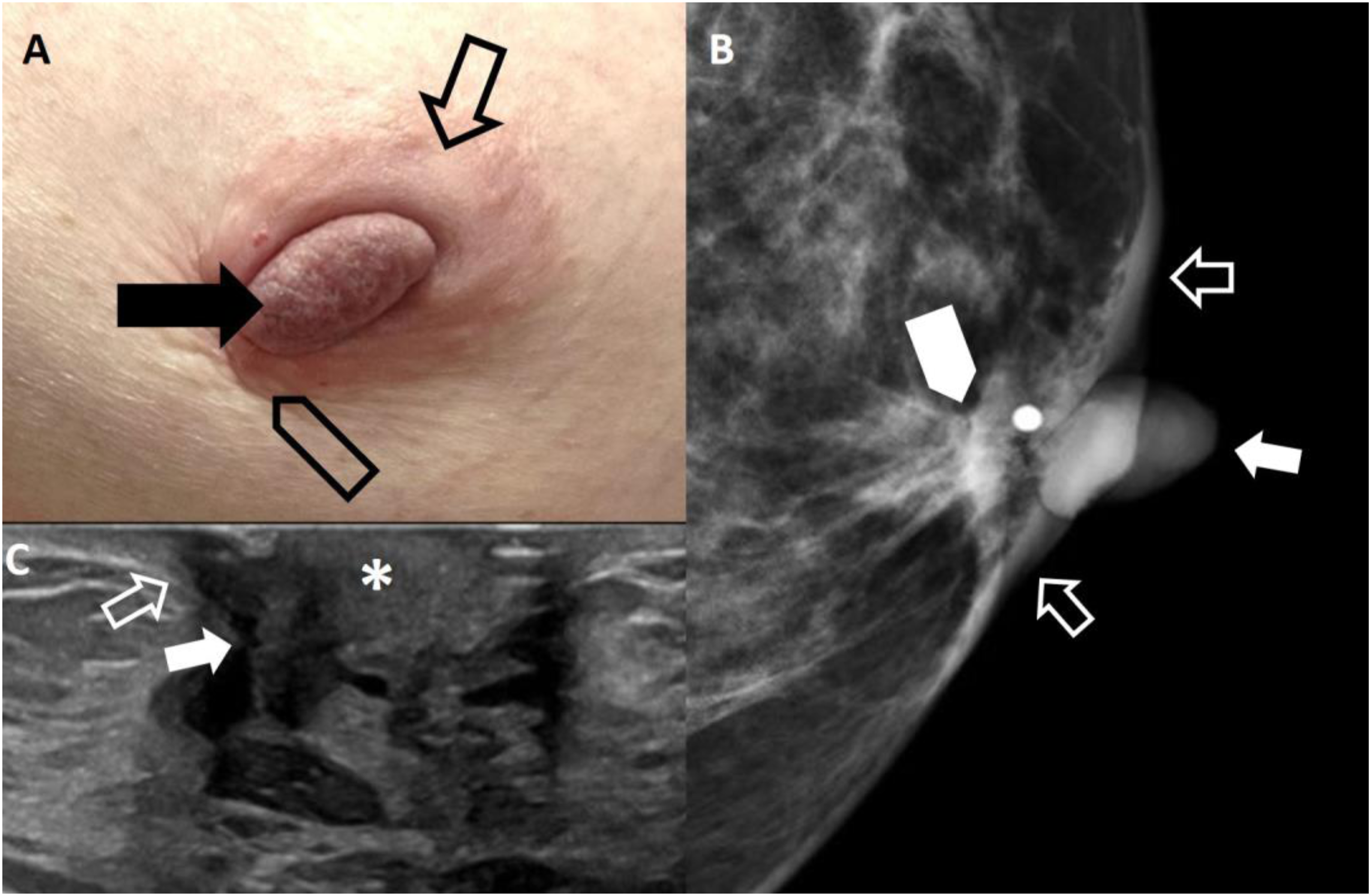
A 52-year-old, female with a history of breast implants presented with itching and “eczema-like” changes of the left nipple that did not respond to topical steroid cream. (A) Photograph of the left nipple demonstrates excoriation and erythema (arrow). (B) Enlarged mammographic mediolateral oblique view of the right and left nipple–areolar complex demonstrates subtle flattening and thickening of the left nipple (arrow). The rest of the mammogram (not shown) was normal. A left breast US was performed and was negative. The patient underwent a left nipple punch biopsy and confirmed the diagnosis of Paget disease. A MR was performed to evaluate for an underlying lesion(s). (C) Pre-contract, T1-weighted, axial image demonstrates thickening and flattening of the left nipple (arrow). (D) Post-contrast, T1-weighted, subtracted, axial image shows asymmetric enhancement of the left nipple (arrow) is seen. There was no underlying lesion in the breast. The patient has bilateral subpectoral silicone implants (*). The patient opted for a left mastectomy. On final pathology, only-Paget disease of the nipple was present. A 62-year-old, female with a history of left nipple itching and tenderness for 6 months presented with a palpable retroareolar mass. (A) Photograph of the left nipple shows thickening of the nipple with some scaling (arrow). In addition, there is thickening of the areolar region (open arrow) and a palpable mass with skin retraction (open arrowhead). (B) Left breast mediolateral oblique spot compression magnification mammogram demonstrates thickening of the nipple (arrow) and periareolar region (open arrows) with an underlying 1 cm, irregular mass (arrowhead) in the subareolar region. (C) Ultrasound shows a 1 cm, irregular, hypoechoic mass (arrow) with nipple (*) and skin involvement skin involvement (open arrow). The patient underwent a left nipple punch biopsy that confirmed the diagnosis of Paget disease. In addition, a needle core biopsy of the retroareolar mass was performed under ultrasound guidance that revealed an invasive ductal carcinoma, grade 2, ER+PR+HER2-. The patient underwent a left breast central lumpectomy with a sentinel lymph node dissection. Final pathology demonstrated a 1.1 cm, invasive ductal carcinoma, grade 2, ER+PR+HER2- with both dermal and nipple involvement. One of two sentinel lymph nodes was positive for nodal involvement. A 42-year-old, female presented with a history of right nipple tenderness for 1 year. (A) Right breast mediolateral oblique view and craniocaudal mammogram images show extensive linear and branching calcifications (arrows) in a segmental distribution extending from the upper outer quadrant to the nipple–areolar complex. The patient underwent a right nipple punch biopsy that confirmed a diagnosis of Paget disease. In addition, a needle core biopsy of the calcifications was performed and revealed ductal carcinoma in situ, grade 3, solid subtype with necrosis that was ER+PR+HER2-. A MRI was performed to evaluate for extent of disease and to evaluate the contralateral breast. (B) Post-contrast, T1-weighted, subtracted, sagittal image of the right breast demonstrates clumped, nonmass enhancement in a segmental distribution (arrows) with extension to and involvement of the nipple–areola complex (arrowheads). The left breast (not shown) was negative. The patient underwent a right simple mastectomy with a sentinel lymph node dissection. Final pathology revealed 14 cm of DCIS with involvement of the nipple and no nodal involvement in two sentinel lymph nodes.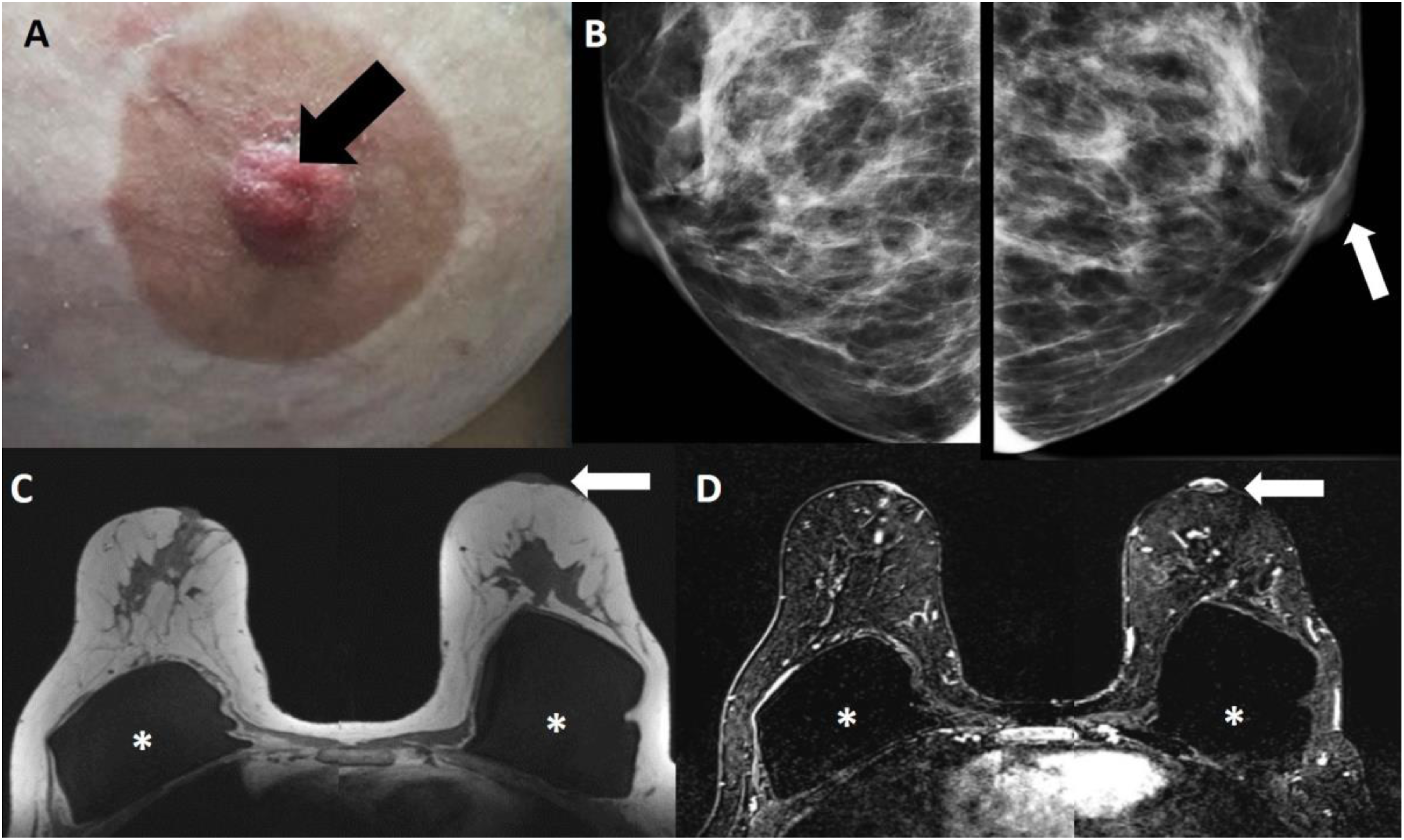
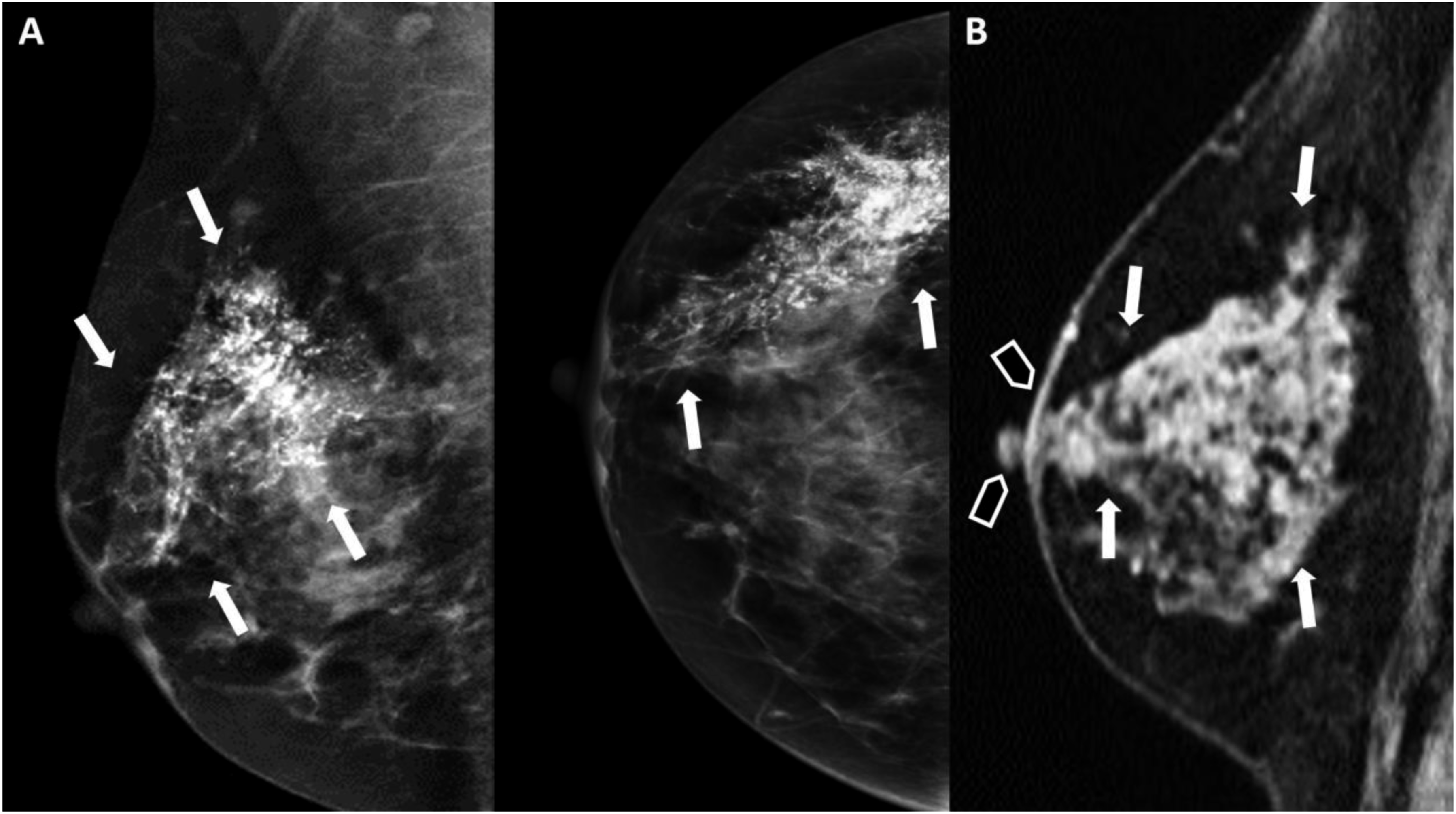
In patients with PD and the presence of a palpable mass, the sensitivity of mammography to detect an underlying carcinoma is more than 90%.22,25 However, the sensitivity of mammography in detecting an underlying carcinoma decreases to 50% in patients with PD when there is no palpable mass even with typical clinical features of PD.2,12,22,33 Due to the limitations of mammography, further evaluation with US and/or MR imaging should be considered to detect an underlying carcinoma and disease extent.
Ultrasound
Ultrasound (US) examination should be included as part of the initial imaging evaluation of PD even when there are no mammographic findings or palpable mass. Although there are no characteristic US features of PD, findings can include a mass, ductal ectasia, calcifications, parenchymal heterogeneousness, or flattening and/or thickening of the NAC (Figure 3).4,5,11,25,33 In a study of 52 patients with PD, US detected 43 (83%) underlying masses. Thirty-five (81%) of the masses were described as lobulated or irregular in shape, and 41 (95%) were without posterior acoustic shadowing. Overall, 15% of the cases were mammography occult and 13% were occult by both mammography and US. 4
Color Doppler may be useful in the evaluation of the nipple in patients with PD. In a study involving 12 patients, 7 (58.3%) with PD and 5 (42%) with dermatitis, color Doppler in the affected nipple of patients with PD was marked compared to those in the unaffected nipple. In addition, the authors reported that no color Doppler effects were observed in the affected and unaffected nipples of patients with simple dermatitis. 34
Magnetic resonance imaging
MR has a high sensitivity for the detection of breast cancer. Because mammography and US have limitations in the evaluation of patients with PD, especially in the setting of clinically and mammographically occult disease, additional evaluation with contrast-enhanced MR can aid in detecting underlying carcinoma, both IBC and DCIS.2,4,27,35 As the underlying carcinoma may be multifocal and/or multicentric, MR is recommended to evaluate the extent of disease and therefore aid in the treatment planning of these patients.31,32,35 It can also allow for the evaluation of axillary nodes, which may detect suspicion of nodal involvement, and evaluation of the contralateral breast.
MRI findings that can be seen with PD include an asymmetric shape, thickening, flattening, or retraction of the NAC, as well as an asymmetric and abnormal enhancement of the nipple (Figures 2 and 4). Normal NAC enhancement patterns are variable, including absent, mild, or intense enhancement. 12 However, asymmetric enhancement is observed in the affected nipple with PD (Figure 2). 36 Nipple involvement can also be seen even when there is no clinical suspicion of PD. 36 In a study by Moon et al., they reported that in their 10 patients with PD who underwent MR imaging, all (100%) cases showed abnormal enhancement of the NAC, 7 (70%) with complete and 3 (30%) with partial enhancement. 37 On MR, inversion or retraction of the nipple was observed in 6 cases, periareolar skin thickening was present in 5 cases, and a continuous association with a subareolar mass was identified in 5 cases. 37 However, asymmetric abnormal enhancement of the NAC is not pathognomonic for PD, since this finding can be seen with a nipple adenoma or extension of breast neoplasms toward the nipple. 38
Because patients with PD may also have an underlying IBC or DCIS, other MR findings that can be seen include an associated suspicious enhancing mass or non-mass enhancement in the retroareolar location or elsewhere in the breast, or in combination (Figure 4).2,5,8,12,27 These findings may be present even when PD is not clinically suspected.2,5,27,37 In the study by Moon et al., 3 of 5 patients with PD and DCIS had enhancement of the NAC with linear or combined clumped nonmass enhancement. 37
In a study consisting of 32 patients with PD, 7 (22%) had associated invasive ductal carcinoma and 25 (78%) had DCIS. 35 Nineteen (59%) of the cancers were unifocal and located in the central quadrant of the breast. Mammography detected 11 cancers and accurately demonstrated the extent of disease in 9 (81.8%) of those patients. MR detected 7 cancers and accurately demonstrated the extent of disease in 6 (85.7%) of those patients. Twenty-three (71.2%) patients had a negative mammogram and MR detected occult disease in 4 of 8 patients. 35 MR also accurately demonstrated the extent of disease in 4 of 4 patients and ruled out underlying cancer in 1 patient. There were 3 (9.4%) patients (2 unifocal DCIS and 1 invasive cancer) whose cancer was occult on MR. 35 Although MR imaging may detect occult cancer in patients with PD, a negative MR study does not reliably exclude underlying occult cancer. The study showed that 94% of patients with PD as the only physical finding had an underlying cancer. 35 Thus, MR imaging in patients with PD can facilitate treatment planning, especially in those who desire breast conservation surgery.
Management
If no underlying carcinoma is present, PD of the breast is staged as Tis (Paget). 39 If DCIS is present with PD, it is staged as Tis (DCIS). 39 If invasive cancer is present with PD, then staging is the based on the underlying invasive carcinoma.
Mastectomy, with or without axillary dissection, has been considered the conventional surgical treatment in patients with PD. More recently, breast conservation therapy (BCT), which involves complete resection of the NAC, followed by adjuvant radiation therapy (RT) in a select group of patients, has been shown to be a viable alternative to mastectomy when the underlying cancer is limited to the central quadrant/subareolar region of the breast.10,11 A SEER analysis of 1739 cases of PD reported that patients who underwent a central lumpectomy or mastectomy had similar outcomes. 29
The National Comprehensive Cancer Network (NCCN) guidelines for patients with PD and DCIS follow the treatment guides for patients diagnosed with DCIS-only. These include BCS without lymph node sampling unless the excision is in an anatomic location compromising a future sentinel lymph node (SLN) procedure followed by potential radiation therapy (RT). In patients undergoing a total mastectomy, SLN sampling may be performed or omitted. The guidelines also include the potential addition of endocrine therapy for all patients with hormone receptor positive DCIS. 40 The guidelines also recommend that patients with PD-only, may undergo a central lumpectomy (including the NAC) or a total mastectomy, and SLN sampling may be performed or omitted. In PD-only patients with BCT that do not undergo SLN sampling, the guidelines recommend adjuvant RT. In patients with IBC and PD, the NCCN advises following their IBC treatment guidelines. 40 Adjuvant RT and chemotherapy are based on the underlying invasive pathology and nodal involvement.
Alternatives to surgery for PD have been proposed. Cryosurgery is a local treatment that uses extreme cold produced by liquid nitrogen or argon gas to destroy cancer cells. It freezes the tissue and causes cell death in the treated. 41 Another alternative is an investigational procedure called photodynamic therapy (PDT) that involves the use of a drug known as a photosensitizer. A topical or intravenous drug is administered to the patient and is absorbed by the affected cells. The drug is then activated by a specific wavelength of light, which destroys the affected cells.8,42 Initial results have demonstrated that PDT is safe and well tolerated. However, the data is limited and more research is needed to determine the safety and effectiveness of the procedure.
Prognosis
The prognosis of PD depends on the presence or absence of an underlying carcinoma and the stage of the disease. The 5-year overall survival rates range from 94% to 98% and from 73% to 93% when PD is associated with invasive carcinoma.2,22,25,43 Although PD in men is clinically the same as PD in women, the 5-year survival rate is lower, with an estimated 5-year survival rate of 20%–30%.27,43 Invasion of the dermis by PD is not associated with a poor prognosis. 1 However, the distinction from skin involvement by locally advanced breast cancer is significant, as such involvement is associated with a poor prognosis compared to dermal invasion in PD. 1
Conclusions
It is important to know the clinical manifestations of PD of the breast because they may be the only signs of underlying cancer. PD should be considered when eczematous involvement of the nipple is not responding to treatment with topical steroids. Mammography may depict changes of the NAC, calcifications, or a mass in the breast, or may be normal. Thus, magnetic resonance imaging can be helpful in the detection of an underlying malignancy and in determining the appropriate patients for breast conservation treatment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
