Abstract
Usual Interstitial Pneumonia (UIP) is a progressive fibrotic lung condition most commonly observed in older men, yet sex-specific clinical outcomes remain underexplored. This retrospective cohort study aimed to examine demographic, clinical, radiological, and mortality-related characteristics of patients diagnosed with a UIP pattern based on high-resolution computed tomography (HRCT), using real-world data from a tertiary chest clinic in Turkey between 2015 and 2025. A total of 100 patients were included, of whom 72% were male. The mean age was 68.4 years, and the average smoking burden was 32.5 pack-years. Radiological progression occurred in 48% of cases, and overall mortality was 26%. Malignancy was an independent predictor of mortality (adjusted odds ratio [OR] = 2.85, 95% confidence interval [CI]: [1.10, 7.43]; p = .032), alongside older age (per year: adjusted OR = 1.06, 95% CI = [1.01, 1.12]; p = .041). Despite the high smoking prevalence, no significant association was found between smoking load and mortality. These findings underscore the disproportionate burden of UIP among older men and highlight the importance of routine malignancy screening and individualized follow-up strategies in this population. The study supports the integration of interstitial lung disease surveillance into comprehensive men’s health care frameworks and calls for increased awareness of UIP’s clinical implications in at-risk male populations.
Keywords
Introduction
Interstitial lung diseases (ILDs) are a heterogeneous group of over 150 pulmonary disorders that share similar clinical, radiographic, and histopathological features but differ in etiology and prognosis (Harari & Caminati, 2010). These diseases primarily affect the pulmonary interstitium and may be associated with environmental exposures, autoimmune disorders, infections, or drug toxicity; however, many cases are idiopathic in nature (Travis et al., 2013). Among the idiopathic forms, idiopathic pulmonary fibrosis (IPF) is the most common and severe and is histologically and radiologically characterized by the UIP pattern (Raghu et al., 2022).
The UIP pattern is typified by subpleural fibrosis, temporal heterogeneity, and honeycombing, with high-resolution computed tomography (HRCT) serving as the cornerstone of diagnosis. In the appropriate clinical context, HRCT findings can obviate the need for lung biopsy, allowing a non-invasive diagnosis of IPF in patients exhibiting a “definite” or “probable” UIP pattern (Shih et al., 2021). However, diagnostic challenges remain due to radiologic-pathologic discordance and overlap with other fibrosing ILDs, including those secondary to connective tissue disease or occupational exposure (Gotarredona et al., 2022; Yagihashi et al., 2016).
Importantly, a disproportionate burden of UIP and IPF has been observed among older men. Epidemiologic studies consistently report a male predominance in UIP diagnoses, with estimates ranging from 60% to 80% male representation (Song et al., 2024). This sex-based disparity may reflect cumulative environmental exposures such as smoking and occupational inhalants but also points toward underlying biological mechanisms, including differences in sex hormones, genetic susceptibility, and immune response regulation (Molinari et al., 2024; Park et al., 2024). Moreover, male patients tend to exhibit more rapid functional decline and worse survival outcomes compared to their female counterparts, even when controlled for comorbidities (Solomon et al., 2015; Yang et al., 2023). In addition to environmental and occupational exposures, biological and behavioral factors may contribute to diagnostic delays in men, including lower symptom reporting, delayed health care-seeking, and sex-based disparities in lung disease awareness (Pinkerton et al., 2015).
Cigarette smoking remains a well-established risk factor for the development and progression of UIP. In male-dominant populations, smoking-related interstitial abnormalities such as respiratory bronchiolitis or combined pulmonary fibrosis and emphysema (CPFE) are frequently observed and may predispose to UIP transformation over time (Douglas et al., 2023). In patients with UIP, especially among older men, the structural alterations caused by chronic fibrosis are thought to contribute to a microenvironment conducive to tumor development (Dos Santos et al., 2022; Gayá García-Manso et al., 2024). This may explain the observed increase in lung cancer incidence within this population, warranting closer malignancy surveillance in clinical practice.
Despite global efforts to better characterize IPF, there remains a notable paucity of population-based epidemiological data specific to Turkey. National registry systems for ILDs are either fragmented or institution-based, limiting accurate prevalence estimates and masking regional disparities in diagnosis and outcomes (Aycicek et al., 2022). Sociocultural dynamics and systemic limitations often result in delayed diagnosis, particularly among male patients, who are frequently identified at more advanced stages of disease. Factors such as low health literacy, symptom overlap with more common conditions like chronic obstructive pulmonary disease (COPD), and insufficient recognition of ILDs in primary care settings may delay timely referral and accurate diagnosis. In this context, men may face additional risks stemming from occupational exposures common in Turkey’s industrial and agricultural sectors. Occupations such as construction work, mining, and metal processing are disproportionately male-dominated and often involve chronic exposure to silica, asbestos, welding fumes, and organic dusts—factors known to contribute to the development of pulmonary fibrosis (Trethewey & Walters, 2018; Walters, 2020). In Turkey, silicosis and other dust-related fibrotic lung diseases remain common among male workers in mining and construction sectors, often in poorly regulated environments (Akgün & Ergan, 2018). These locally relevant exposures are rarely captured in international IPF cohorts, underscoring the importance of regional studies that address sex-specific and occupational risk factors in disease development and progression.
Despite these trends, there is a paucity of real-world, sex-disaggregated data describing the clinical course, comorbidity burden, and mortality predictors in patients with UIP, especially in underrepresented populations such as those in Turkey. In particular, the prognostic relevance of malignancy, comorbid COPD, and HRCT-based radiologic progression in male-dominated cohorts remains unclear in routine clinical practice settings.
This retrospective cohort study aimed to evaluate the clinical, radiological, and mortality characteristics of patients diagnosed with a UIP pattern via HRCT at a tertiary chest clinic in Turkey over a 10-year period (2015–2025). With a predominantly male study population, the analysis focused on age, sex, smoking history, symptom burden, comorbidities, radiological progression, and malignancy as potential predictors of mortality. In addition, this study aimed to assess the utility of hospital information management systems (HIMS) as a platform for real-world data generation in men’s pulmonary health surveillance and decision-making.
Method
Study Design and Setting
This retrospective cohort study was conducted at Bolu Abant Izzet Baysal University Training and Research Hospital, a tertiary referral center in northwestern Turkey. Data were collected from patients who presented it to the Pulmonology Outpatient Clinic between January 2015 and January 2025. The follow-up period for all patients was concluded on January 31, 2025. Mortality status was verified using national databases as of this date, and patients who were alive were censored at their last recorded visit or imaging prior to this cut-off. The study was approved by the university’s Non-Interventional Clinical Research Ethics Committee (Protocol No.: 2025/142), and institutional permission was granted by the hospital’s chief physician.
Although no a priori power analysis was conducted due to the retrospective design of the study, the sample size (n = 100) was based on all eligible patients with a confirmed UIP pattern and complete clinical and mortality data available in the institutional database over a 10-year period. This pragmatic approach aimed to maximize data utility and minimize selection bias by including the entire accessible cohort within the defined time frame. Future prospective studies should incorporate formal power calculations to ensure adequate statistical power for subgroup analyses.
All HRCT evaluations were conducted independently by two board-certified pulmonologists with experience in ILDs. The reviewers were not blinded to the patients’ clinical characteristics due to the retrospective nature of the study and the integrated electronic record system. However, to minimize observer bias, a third senior pulmonologist reviewed all discordant cases, and consensus was achieved through joint discussion. Although formal interobserver agreement metrics (e.g., kappa statistics) were not calculated, this consensus-based approach provided methodological consistency. In future prospective studies, blinded and reproducible HRCT assessments are recommended to improve diagnostic objectivity and generalizability.
Study Population
Patients were eligible if they were aged 18 years or older, had a diagnosis of ILD (International Classification of Diseases [ICD]-10: J84.x) documented in the HIMS, had a chest HRCT report consistent with a “definite” or “probable” UIP pattern based on the 2022 ATS/ERS/JRS/ALAT guidelines (Raghu et al., 2022), and had initiated antifibrotic therapy (pirfenidone or nintedanib) under supervision of the study team.
Patients were excluded if they were under the age of 18 years, had previously been diagnosed and treated for ILD at another health care facility, had discontinued or switched antifibrotic therapy due to adverse effects, or had missing clinical or mortality data. Of the 138 patients initially screened, 22 were excluded due to HRCT findings not consistent with diagnostic criteria, and 16 were excluded due to incomplete data. These 16 patients had missing data in critical variables such as mortality status, antifibrotic therapy duration, or HRCT classification, which were essential for primary analyses. Including these cases would have compromised the integrity of the statistical results. Since most missing values were observed in dependent variables, multiple imputations were deemed inappropriate and therefore not applied. Thus, a total of 100 patients met the inclusion criteria and were included in the final analysis. The patient flow is summarized in Figure 1.

Participant Flow Diagram for the Retrospective Cohort Study.
Radiologic Assessment
HRCT images and reports were independently reviewed by two pulmonologists trained in ILD. Discrepancies were resolved by consensus in consultation with a third pulmonologist. “Definite” and “probable” UIP patterns were classified according to the 2022 ATS/ERS/JRS/ALAT criteria (Raghu et al., 2022). Radiological progression was defined as an increase in fibrotic changes, including honeycombing or reticulation, observed on follow-up imaging. Although formal interobserver agreement metrics (e.g., kappa statistics) were not initially planned, HRCT reports stored were re-reviewed by the same two pulmonologists independently for a subset of cases (n = 40), and interobserver agreement was calculated post hoc. This yielded Cohen’s kappa values of 0.81 for UIP pattern classification and 0.77 for radiological progression, indicating excellent and substantial agreement, respectively. These results have been incorporated to strengthen the methodological rigor of radiologic assessment.
Data Sources and Collection
Data were obtained retrospectively from both the HIMS and physical patient records. The sources included electronic patient files and demographic data, CT imaging reports, discharge summaries, and medication initiation and follow-up notes. All data were recorded using standardized forms in accordance with the RECORD guidelines (Benchimol et al., 2015). Variables collected included age, sex, smoking status and load (pack-years), symptom profiles, comorbidities, dyspnea severity (measured by the modified Medical Research Council [mMRC] scale), radiological findings, antifibrotic treatment duration, and mortality status. Smoking history and comorbidity information were obtained through self-report and verified by medical records. Data on malignancy type and diagnosis timing were extracted from patient records. Occupational categories were classified based on the available documentation in medical records. “Worker” referred to individuals engaged in manual labor such as construction, metalwork, or manufacturing and was therefore classified as a high-risk occupation. Similarly, “Farmer” was categorized as high-risk due to agricultural exposure. “Officer” (i.e., administrative or desk jobs) and “Housewife” were considered low-risk occupations. These definitions were used to derive a binary variable (high risk vs. low risk), which was incorporated into the multivariable logistic regression analysis. Mortality data were confirmed through national identification number verification with official health system databases. Detailed definitions of all variables and the corresponding coding algorithms are provided in the Supplementary Appendix.
Statistical Analysis
All analyses were conducted using SPSS version 20.0 (IBM Corp., Armonk, New York). Descriptive statistics were presented as mean ± standard deviation (SD) or median (min–max) for continuous variables and as frequencies and percentages for categorical variables. The Shapiro–Wilk test was used to evaluate the normality of data distribution. Group comparisons were performed using independent-samples t-tests for normally distributed variables and Mann–Whitney U tests for non-normally distributed variables. Pearson’s chi-square test or Fisher’s exact test was used to assess associations between categorical variables.
To evaluate the associations between binary outcomes (e.g., mortality, malignancy, radiological progression) and clinical predictors, logistic regression analysis was performed. Occupational exposure (high risk vs. low risk) was included as a covariate in an updated logistic regression model assessing mortality. The regression model included age, sex, smoking load, mMRC score, comorbidities, and duration of antifibrotic therapy. Model fit was assessed using the Hosmer–Lemeshow test. Correlations between continuous variables were analyzed using Spearman’s rank correlation. Comorbidities, including cardiovascular disease, diabetes mellitus, and chronic kidney disease, were included in the multivariate models based on their clinical significance and their imbalance between sexes. To account for time-to-event data and potential risk overestimation with odds ratios (ORs), we conducted a supplementary Cox proportional hazards regression analysis. The time variable was defined as the duration between the HRCT-based diagnosis and the date of death or censoring (January 31, 2025). Independent variables included in the model were age, sex, mMRC score ≥2, presence of malignancy, radiological progression, and occupational exposure. Sex was retained in both models (logistic and Cox) to assess adjusted associations with mortality, controlling for potential confounders such as age and comorbidities. A p-value < .05 was considered statistically significant.
All data were anonymized prior to analysis, and personal identifiers were removed. Data handling and extraction were conducted solely by the research team (S.K. and E.O.) in accordance with ethical and institutional approvals.
Results
A total of 100 patients diagnosed with a UIP pattern were included in the final analysis. The mean age was 68.4 ± 9.7 years, with no significant sex-based difference (p = .249). Male patients had a significantly higher smoking load compared to females (34.7 ± 13.9 vs. 27.3 ± 12.4 pack-years, p = .012), and heavy smoking (≥30 pack-years) was markedly more common among men (61.1% vs. 14.3%, p < .001). Occupational distribution also differed between sexes, with male patients predominantly engaged in manual labor (54.2% vs. 21.4%, p = .003), while most female patients were housewives (32.1% vs. 5.6%, p = .001). Other baseline clinical features, including dyspnea, cough, sputum, chest pain, mMRC scores, and auscultation findings, did not differ significantly between sexes (all p > .05).
Among the 100 patients, 19 had a history of malignancy, of which 7 (7%) were lung and 12 (12%) were non-lung malignancies. Female patients had a slightly higher proportion of non-lung malignancies compared to males (17.9% vs. 9.7%). Dyspnea (82%) and dry cough (77%) were the most reported symptoms, and 61% of all patients had an mMRC dyspnea score ≥2. On auscultation, Velcro-like inspiratory crackles were present in 88% of patients, predominantly among male participants. Baseline clinical characteristics are presented in Table 1.
Baseline Demographic and Clinical Characteristics by Sex.

Note. Values are presented as mean ± standard deviation (SD) for continuous variables and as number and percentage (n, %) for categorical variables. Percentages are calculated based on subgroup totals. †Independent-samples t-test. ‡Pearson’s chi-square test. §Fisher’s exact test. Malignancy timing is defined relative to the HRCT-based UIP diagnosis. Bolded p-values indicate statistical significance at p < .05.
Radiological progression was observed in 48% of patients, with a mean time to progression of 14.3 ± 6.2 months. Radiological progression did not show a statistically significant association with any of the assessed clinical variables, including mMRC score, cumulative smoking exposure, and the duration of antifibrotic treatment (all p > .05) as shown in Table 2.
HRCT Features and Correlation With Clinical Predictors.
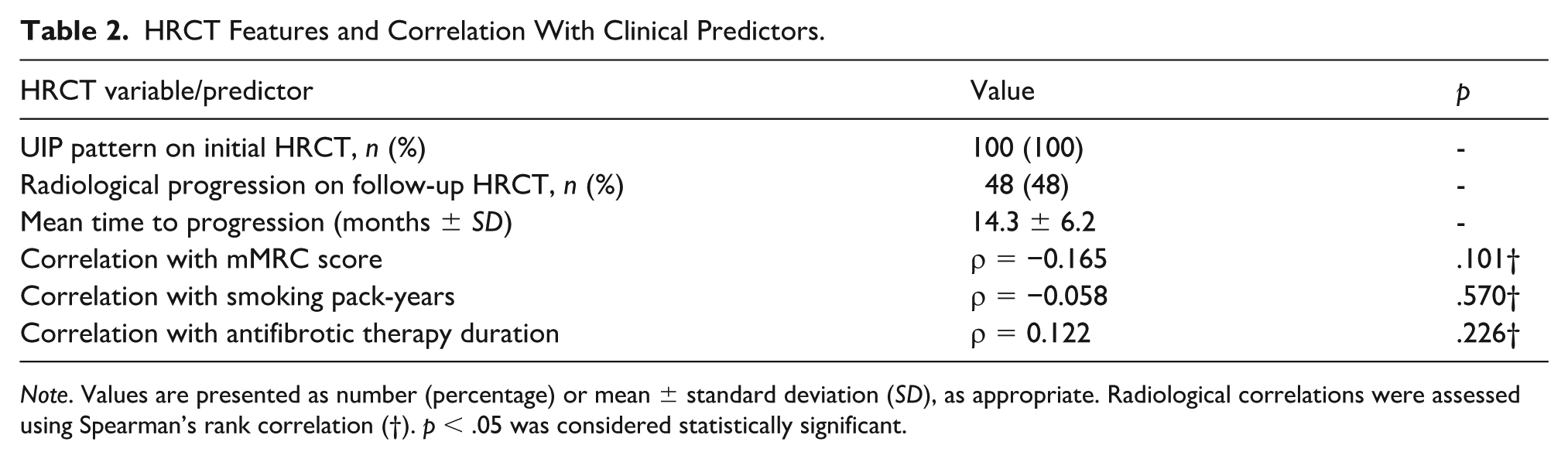
Note. Values are presented as number (percentage) or mean ± standard deviation (SD), as appropriate. Radiological correlations were assessed using Spearman’s rank correlation (†). p < .05 was considered statistically significant.
Comorbidities were highly prevalent, particularly among male patients. The most frequent were hypertension (47%), COPD (39%), and diabetes mellitus (22%). Malignancy was diagnosed with 19% of patients during follow-up, and 9% of all deaths were attributed to cancer. The overall mortality rate in the cohort was 26%.
Notably, mortality was significantly higher among patients with malignancy (47.4%) compared to those without (21.0%), corresponding to an OR of 3.39 (95% confidence interval [CI] = [1.19, 9.66]; p = .038). COPD was also significantly associated with mortality (χ2 = 4.15, p = .042), whereas cardiovascular disease, hypertension, and diabetes did not show significant associations as detailed in Table 3. Although men comprised most of the cohort (72%), the proportional mortality rate was unexpectedly higher among female patients (42.9%) compared to males (19.4%). However, further exploratory analysis revealed that female patients, despite being slightly younger on average (66.5 vs. 69.1 years), had a higher prevalence of cardiovascular disease (50% vs. 27.8%). These differences may partially explain the elevated mortality rate observed in women. Nonetheless, due to the limited sample size of female patients (n = 28), no statistically significant sex-based mortality difference was identified (p = .066). Furthermore, after adjusting for age and comorbidities, sex was not an independent predictor of mortality in either logistic or survival models, indicating that the observed crude difference may be attributable to confounding rather than true sex effect. These findings highlight the importance of adjusting for age and comorbidity burden in future sex-disaggregated analyses. It should also be noted that this subgroup comparison may have been underpowered due to the small sample size, increasing the likelihood of a Type II error. In univariate analysis, malignancy was associated with a higher risk of mortality (crude OR = 3.39, 95% CI = [1.19, 9.66]; p = .038). This association persisted in the multivariate logistic regression model, where malignancy remained an independent predictor of mortality (adjusted OR = 2.85, 95% CI = [1.10, 7.43]; p = .032).
Univariate Associations of Comorbidities and Malignancy With Mortality.

Mann–Whitney U-test. ‡Pearson’s chi-square test. §Fisher’s exact test. ¶Regression. Bolded p-values indicate the statistical significance at p < .05.
Multivariate logistic regression analysis identified older age ([OR] = 1.06 per year increase, 95% CI = [1.01, 1.12]; p = .041) and the presence of malignancy (OR = 2.85, 95% CI = [1.10, 7.43]; p = .032) as independent predictors of mortality. Occupation was not significantly associated with mortality in the revised regression model (p = .399), although its inclusion allowed evaluation of stated exposure-related hypotheses. In the revised multivariate model including comorbidities, malignancy (OR = 2.85, p = .032) and age (OR = 1.06, p = .041) remained significant predictors of mortality, while cardiovascular disease was not statistically significant (OR = 1.48, p = .218). While both mMRC score ≥2 and radiological progression showed a trend toward increased mortality risk, these associations did not reach statistical significance as shown in Table 4.
Multivariate Logistic Regression Analysis of Mortality Predictors.

Note. Binary logistic regression (¶) was used to identify independent predictors of mortality. The model included age, sex, mMRC score, radiological progression, presence of malignancy, occupational exposure, and comorbidities (cardiovascular disease, diabetes mellitus, and chronic kidney disease). Occupational exposure was coded as high risk versus low risk. Sex was coded as female = 0, male = 1 (reference group). Comorbidities were included due to clinical relevance and observed sex imbalance. Model fit was assessed using the Hosmer–Lemeshow test. Bolded p-values indicate the statistical significance at p < .05.
In the supplementary Cox proportional hazards analysis, malignancy was independently associated with higher mortality risk (hazard ratio [HR] = 2.91; 95% CI = [1.11, 7.65]; p = .029) along with older age (HR = 1.05 per year; 95% CI = [1.01, 1.09]; p = .034; see Supplementary Table 1 for full details). Other variables, including sex, mMRC score, radiologic progression, and occupational exposure, were not statistically significant predictors. Similarly, in the Cox regression model, the inclusion of these comorbidities did not materially alter the results. Malignancy (HR = 2.74, p = .037) and age (HR = 1.05, p = .033) remained significant predictors. These results further support malignancy and age as independent mortality predictors in UIP patients. Notably, although crude mortality was higher in female patients, sex was not significantly associated with mortality in either model after adjustment (Cox model: HR = 1.38, 95% CI = [0.75, 2.54], p = .296; Logistic model: OR = 1.43, 95% CI = [0.58, 3.56], p = .428).
Among male patients specifically (n = 72), 14 deaths (19.4%) occurred, compared to 12 deaths (42.9%) among female patients. Despite the higher absolute number of deaths in men, the proportional mortality was greater in women. However, when adjusted for age, smoking, and comorbidities, mortality risk remained more strongly associated with malignancy status than sex alone.
Discussion
This retrospective cohort study investigated clinical, demographic, and radiological features of 100 patients diagnosed with a UIP pattern via HRCT, with a focus on predictors of mortality. The analysis, based on real-world data collected from 2015 to 2025, aimed to inform clinical practice by identifying factors associated with outcomes in a population composed of older adults and predominantly male patients.
The cohort had a mean age of 68.4 years, with 72% of participants being male. This demographic pattern aligns with national and international findings, suggesting that UIP and IPF disproportionately affect older men (Song et al., 2024). The observed sex disparity may be attributed to cumulative environmental exposures (e.g., smoking, occupational dust), genetic predispositions, and sex hormone–related immune modulation (Molinari et al., 2024; Park et al., 2024). Our findings reinforce the importance of incorporating sex-specific factors into risk stratification and surveillance strategies for patients with UIP.
Smoking history was notable, with an average burden of 32.5 pack-years. While smoking is a recognized risk factor for the development of fibrotic ILD, we found no significant association between smoking load and mortality. This suggests that smoking may contribute more to disease onset than progression. Contrastingly, Raghu et al. (2022) reports conflicting findings, due to differences in smoking definitions, duration of follow-up, or presence of overlapping conditions like CPFE.
Comorbid conditions were prevalent (89%), particularly COPD (39%) and hypertension (47%). While several comorbidities were evaluated, only malignancy showed a statistically significant association with mortality (p = .038), confirming its role as a key prognostic factor. The proportion of malignancy-related deaths (9%) supports previous reports suggesting that fibrotic lung tissue may foster a microenvironment conducive to carcinogenesis (Dos Santos et al., 2022; Gayá García-Manso et al., 2024).
The observation that over one-third of malignancy cases were primary lung cancers reinforces the importance of early lung cancer screening in patients with fibrotic ILDs, especially those diagnosed with UIP. This finding is consistent with prior studies reporting an elevated lung cancer risk in patients with IPF or UIP-like patterns, due to shared risk factors such as smoking, age, and chronic epithelial injury (Zhang et al., 2025). Furthermore, large-scale population-based cohort analyses have confirmed the increased incidence of lung cancer in UIP/IPF patients, highlighting the relevance of early surveillance strategies (Lee et al., 2021). Recent reviews have further emphasized common pathogenic pathways and molecular mechanisms underlying the coexistence of fibrosis and neoplasia (Jo et al., 2024).
Further exploration of the subgroup of patients who developed malignancy in our cohort suggested trends toward older age and higher smoking exposure, although precise mean values were not calculated due to limited sample size. In addition, a greater proportion had pre-existing COPD and a history of occupational exposure, particularly in male-dominated sectors such as construction. Although these trends did not reach statistical significance, they align with previously reported risk factors for lung cancer in patients with fibrotic ILD (JafariNezhad & YektaKooshali, 2018; Vancheri et al., 2010). A recent meta-analysis reported a significantly elevated risk of lung cancer in IPF patients, with prevalence estimates ranging from 11% to 48%, particularly among older male smokers with emphysematous changes (Grant-Orser et al., 2023). Taken together, these findings underscore the importance of heightened vigilance and personalized surveillance strategies for malignancy in fibrotic ILD patients, particularly among high-risk subgroups such as older male smokers with coexisting emphysema or occupational exposure.
Radiological progression occurred in 48% of patients, with a mean progression time of 14 months. Although not statistically linked to mortality in our cohort, this high rate of progression aligns with the known natural history of UIP, which is often marked by functional decline and structural deterioration (Raghu et al., 2022). Despite a clear trend toward higher mortality in patients with elevated mMRC scores and radiological progression, these associations did not achieve statistical significance. This may be partially explained by limited statistical power or variability in the timing and frequency of follow-up HRCT scans. Furthermore, the mMRC score, while widely used, is inherently subjective and may not accurately reflect disease severity in all patients. Previous studies have shown that radiological progression often precedes functional decline (Sun et al., 2022; Taha et al., 2020), suggesting that a composite index incorporating both imaging and physiological metrics may be more predictive. The absence of standardized follow-up intervals and potential interobserver variability in HRCT interpretation may also have diluted the observed effect. The variability in progression among patients highlights the heterogeneity of UIP and the need for individualized follow-up schedules incorporating repeat HRCT imaging. The unexpectedly higher proportional mortality in women, despite lower absolute numbers, may reflect confounding factors such as advanced age or comorbidity profiles. Given the underrepresentation of female patients in UIP cohorts, this observation warrants further investigation in larger, sex-balanced studies.
In line with the 2022 ATS/ERS/JRS/ALAT guidelines, HRCT-based diagnosis remains the cornerstone for identifying UIP, particularly in appropriate clinical contexts (Raghu et al., 2022). Our study confirms that both “definite” and “probable” UIP patterns are clinically meaningful and may guide treatment decisions without the need for histological confirmation. Regular imaging follow-up should be emphasized in practice, particularly for patients showing early signs of radiologic progression.
From a data perspective, the integration of HIMS and national mortality databases enabled accurate mortality tracking and real-world analysis. Such digital platforms hold promises for building decision-support systems in respiratory medicine, especially in resource-constrained settings. However, reliance on retrospective data introduces limitations, including potential coding errors, variability in clinical documentation, and missing data. In our case, exclusion of 16 patients due to incomplete records may have introduced selection bias.
Additional limitations include the use of self-reported smoking and comorbidity data, single-center HRCT interpretation, and unmeasured confounders such as physical activity level, symptom onset time, and treatment adherence. Initially, comorbidities were not included in the multivariate models. However, revised analyses incorporating cardiovascular disease, diabetes, and chronic kidney disease demonstrated that these variables did not significantly impact the overall findings or alter the significance of the main predictors. Furthermore, subgroup analyses, particularly those involving female patients and malignancy cases, were underpowered due to small group sizes. The potential for Type II errors should be considered when interpreting non-significant results. These factors limited the statistical power to detect certain associations. Our inability to model radiological progression or mortality in relation to these unmeasured variables underscores the need for standardized data collection in future prospective studies.
Clinically, our findings emphasize several actionable priorities. First, routine screening for malignancy should be an integral part of UIP management, particularly in older male patients. Second, the high prevalence of radiological progression supports incorporating serial imaging into follow-up protocols. Third, a multidisciplinary care model involving pulmonologists, radiologists, oncologists, and rehabilitation specialists may enhance patient outcomes by addressing the multifactorial nature of disease progression. Finally, reinforcing long-term adherence to antifibrotic therapy remains essential, particularly in real-world settings where dropout rates may be higher than in clinical trials.
Given the disproportionate burden of UIP among men and the strong association between occupational exposure, smoking, and malignancy, sex-specific prevention strategies should be prioritized. These may include periodic low-dose chest CT screening for high-risk male patients with significant smoking history or occupational exposure, particularly in sectors such as construction, metalworking, and agriculture. Preventive strategies in occupational settings should prioritize engineering controls and exposure mitigation, developed in coordination with occupational medicine specialists and regulatory institutions. Such measures are particularly vital in industries with high dust and fume exposure, which disproportionately affect male workers in sectors like construction, mining, and manufacturing. In parallel, public health campaigns targeting middle-aged and older men could improve early symptom recognition and encourage timely specialist referral. Integrating men’s health education into primary care protocols may also enhance disease awareness and screening uptake, reducing diagnostic delays.
In conclusion, this study adds to the growing body of evidence supporting sex-specific risk profiles, comorbidity surveillance, and real-world data integration in UIP care. Malignancy emerged as the strongest independent predictor of mortality, underscoring the importance of oncology-informed follow-up strategies. This association was further confirmed in a time-to-event Cox regression model, which reinforced malignancy and older age as significant mortality predictors, independent of other covariates. Although mMRC dyspnea scores and radiological progression were not statistically significant predictors, their trends suggest clinical relevance. Prospective studies with standardized methodologies are needed to validate these findings and improve long-term outcomes in this vulnerable patient group.
Conclusions
This retrospective cohort study identified malignancy and older age as independent predictors of mortality in patients with a usual interstitial pneumonia (UIP) pattern confirmed by HRCT. While mMRC dyspnea scores and radiological progression showed trends toward increased mortality, these associations were not statistically significant. Notably, malignancy was associated with more than a threefold increase in the risk of death, highlighting its prognostic significance.
Given the male predominance of UIP and the high burden of smoking and occupational exposures in this group, our findings support the implementation of sex-specific follow-up strategies. These may include periodic low-dose CT screening for high-risk men, targeted education for symptom recognition, and improved referral pathways from primary care. Integrating occupational history and sex-based risk profiling into national ILD guidelines could help identify vulnerable patients earlier in the disease course. A multidisciplinary care model—bringing together pulmonologists, radiologists, oncologists, and occupational health specialists—will be essential to address the complex needs of this population and improve long-term outcomes.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883251397902 – Supplemental material for Sex-Specific Clinical Characteristics and Mortality Predictors in Patients With Usual Interstitial Pneumonia Pattern: A Retrospective Cohort Study Highlighting Male Health Risks
Supplemental material, sj-docx-1-jmh-10.1177_15579883251397902 for Sex-Specific Clinical Characteristics and Mortality Predictors in Patients With Usual Interstitial Pneumonia Pattern: A Retrospective Cohort Study Highlighting Male Health Risks by Suat Konuk, Alp Ozel, Emine Ozsari and Sena Melis Sert in American Journal of Men's Health
Footnotes
Acknowledgements
The authors have reviewed and edited all content and take full responsibility for its accuracy and integrity.
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by the Non-Interventional Clinical Research Ethics Committee of Bolu Abant Izzet Baysal University (protocol code 2025/142 and April 8, 2025 date of approval).
Informed Consent
Patient consent was waived due to the retrospective nature of the study and the use of anonymized data obtained from electronic health records. No identifiable personal information was used, and ethical approval was obtained from the relevant institutional ethics committee.
Author Contributions
Conceptualization: S.K., E.O., and A.O.; methodology: S.K. and E.O.; validation: A.O. and S.M.S.; formal analysis: A.O.; investigation: S.K., E.O., S.M.S., and A.O.; resources: S.K. and E.O.; data curation: A.O.; writing—original draft preparation: S.K., E.O., A.O., and S.M.S.; writing—review and editing: A.O.; and supervision: S.K. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used in this study were obtained from electronic health record systems affiliated with the Ministry of Health of the Republic of Turkey. Due to patient confidentiality and ethical considerations, these data are not publicly available. Access to the data set was restricted to researchers who had obtained the necessary approvals from the relevant ethics committee and institutional authorities. The analytical codes and variable definitions used in this study may be shared by the corresponding author upon reasonable request and with appropriate justification.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
