Abstract
The combination of Syto16/7-AAD allows the detection of apoptosis at an earlier stage than that of annexin V/7-AAD. The effects of the earlier apoptosis of spermatozoa on fertilization rate and post-embryonic development have not been investigated systematically. The objective of this study is to assess the relationship between the earlier apoptosis of spermatozoa in IVF patients and seminal parameters (concentration and morphology), to assess the effect of the earlier apoptosis of spermatozoa on ART outcomes, especially with regard to clinical pregnancy. The earlier apoptosis of spermatozoa was detected by syto16/7AAD using flow cytometry (FCM) in 98 couples (98 IVF) before and after density gradient centrifugation (DGC), and its relationships with the fertilization rate, the normal fertilization rate, the cleavage rate, the good embryo rate and the cumulative clinical pregnancy rate were assessed. Meanwhile, the correlation with seminal parameters (concentration, motility and morphology) was also analyzed. The rate of the earlier apoptosis was significantly negatively correlated to sperm concentration(r = −.302, p = .003) and membrane integrity rate(r = −.862; p < .01) after DGC. There was also a positive correlation between the rate of the earlier apoptosis and sperm mortality rate (r = .225, p = .027) after DGC. A highly significantly negative correlation was found between the rate of the earlier apoptosis and outcomes in IVF patients: the fertilization rate and the normal fertilization rate decreased as the rate of the earlier apoptosis increased (r = −.271, −.288; p = .008,.004). The cumulative clinical pregnancy rate also significantly differed between patients with the earlier apoptotic rate ≥15% and <15% (57.89% vs. 81.69%, p < .01) after DGC. The earlier apoptosis of spermatozoa seems to affect fertilization, however, it does not affect the early embryo development proficiency in IVF procedures. Because of the decreased fertilization rate, the earlier apoptosis of spermatozoa can compromise the number of available embryos and good embryos, resulting in cumulative clinical pregnancy loss.
Keywords
Background
Over the last decade, we have seen the increasing use of assisted reproductive technology (ART) to overcome the problem of couple infertility. Currently, ART is routinely proposed in the cases of couples with a severely sub-fertile male partner who wishes to have a child. However, the pregnancy rate is still relatively low and somewhat unpredictable, given the number of variables possibly involved in the multistep process of fertilization, embryo cleavage, implantation and pregnancy. Several studies have stressed the importance of conventional seminal parameters (sperm concentration, motility and morphology) as predictors of ART outcome. However, data demonstrate that traditional seminal analysis is of limited value in the determination of the couples’ fertility status and the prediction of fertility and embryonic development potential in ART (Del Giudice et al., 2022; Sharbatoghli et al., 2012; Yetunde & Vasiliki, 2013).
To improve the prediction of fertility and embryonic development potential in clinical practice, the use of sperm function biomarkers, that is, of the markers of apoptosis, has been proposed. Apoptosis is a mode of programmed cellular death that leads the cell to suicide without eliciting an inflammatory response. Apoptosis occurs throughout spermatogenesis, maintaining sperm count and semen quality, specifically, it is increased in spermatozoa of infertile men affected by cryptorchidism, infection and varicocele. There is an increased focus on the correlation between sperm apoptosis and outcomes in ART(Sharbatoghli et al., 2012; Simon et al., 2017; Zhao et al., 2021). DNA fragmentation, potentially related to late apoptosis, has been the most thoroughly investigated factor to date (Marinaro & Schlegel, 2023; Newman et al., 2022; Peel et al., 2023; Simon et al., 2019) Most studies focus on the effect of DNA fragmentation on embryo quality and/or development in IVF/ICSI technology. The existing data suggest that the influence of sperm DNA damage on embryo quality may be less significant in IVF compared to ICSI cycles based on the natural selection that occurs during IVF, the natural selection barriers are bypassed during ICSI entirely and fertilization with highly DNA fragmented sperm may happen (Andrabi et al., 2024). Further research indicates that the influence of sperm DNA damage on the embryo appears to be related to embryo development more than embryo quality (Mateo-Otero et al., 2022; Sakkas et al., 1998). Thompson-Cree et al. (2003) found that DNA fragmentation in spermatozoa that did not achieve a pregnancy was substantially higher than that in spermatozoa that resulted in pregnancy. Similarly, Spano et al. (2005) and Evenson et al.(2008) showed that patients with high proportions of spermatozoa with abnormal DNA in their ejaculate were less likely to father a child. The notion of the correlation between sperm DNA integrity and fertilization is still controversial. Several studies suggest that spermatozoa with DNA damage are still capable of fertilization and defects may not become evident until the embryo has divided (Zheng et al., 2018). Other studies show a significant inverse relationship between fertilization and the presence of DNA fragmentation(Al-Saleh et al., 2021).
However, DNA fragmentation is considered the terminal stage of the apoptotic pathway, and more spermatozoa are still in the phase of early apoptosis, whose pathological changes are too subtle and slight to be easily discerned. If indeed spermatozoa in the early stages of apoptosis adversely affect ART outcomes, then removal of the early apoptotic spermatozoa would be desirable and more effective than removal of spermatozoa with DNA fragmentation to improve ART outcomes, because removal of early apoptotic spermatozoa can potentially improve ART outcomes by both eliminating early apoptotic spermatozoa and reducing the production of spermatozoa with DNA fragmentation. To date, there is little research on the effects of the earlier apoptosis of spermatozoa on fertilization and embryo development. Some evidence indicated that the use of annexin V coupled with magnetic-activated cell sorting (MACS) improved sperm quality on cryopreserved spermatozoa and ICSI outcome (Grunewald et al., 2009; Grunewald & Paasch, 2013; Salehi Novin et al., 2023; Sheikhi et al., 2013). In contrast, another study found that the removal of annexin V-positive sperm for intra-cytoplasmic sperm injection in ovum donation cycles did not improve reproductive outcomes (Romany et al., 2014). Therefore, the effects of early apoptosis in spermatozoa on fertilization rate and post-embryonic development are still controversial.
In general, the dye Syto16, which, besides functioning as a Pgp probe, stains DNA and RNA structure (Sparrow et al., 2006), and the combination of Syto16/7-AAD allows the detection of apoptosis at an earlier stage than that of annexin V/7-AAD. The effects of the earlier apoptosis of spermatozoa on fertilization rate and post-embryonic development have not been investigated systematically and adequately. Therefore, this study aims to assess the impacts of the earlier apoptosis of spermatozoa as detected by syto16/7AAD on the fertilization rate, cleavage rate and cumulative clinical pregnancy rate.
Methods
Collection and Classification of Clinical Examples
Semen samples obtained from 98 patients undergoing IVF treatments were collected at the Assisted Reproduction Center of the Guangdong Women and Children Hospital from June 1, 2024 to June 30, 2024. The mean age of women included in the study was 30.93 ± 0.36 years, and the mean BMI was 21.28 ± 1.26 kg/m2. The mean age of men was 33.15 ± 0.55 years, and the mean BMI was 24.36 ± 3.01 kg/m2. All subjects were the partners of women who failed to conceive after 24 months of unprotected intercourse. Semen samples were collected by masturbation into sterile containers after 2 days of sexual abstinence. Routine analysis was performed according to World Health Organization standard guidelines (World Health Organization, 2010).
Semen Preparation
Sperm samples were collected by masturbation after 2 days of sexual abstinence. After half an hour, the spermatozoa were assessed according to the WHO guidelines as the raw sperm parameters and purified free of seminal plasma by using a two-step (90%/45%) discontinuous Pure Sperm gradient (Nicadon, Gothenburg, Sweden). After density gradient centrifugation (DGC) at 350g for 20 min, the pellet was suspended in a volume of 5 ml of Spermrinse medium, processed by centrifugation at 350g for 5 min, and resuspended in 0.5 ml of IVF30 medium for IVF (PureSperm wash, Nidacon, Gothenburg, Sweden). Before and after DGC, part of the seminal sample was analyzed for both sperm concentration and morphology following WHO procedures (WHO, 2011), as well as the expression of syto16/7ADD and CD46 by the multiparameter FC analysis.
Multiparameter FC Analysis
FC analysis was performed to assess simultaneously sperm viability, apoptosis and acrosomal integrity by using a FACS Calibur four-colour (Becton Dickinson, San Jose`, CA, USA). Briefly, 100 µl of whole semen was stained for 20 min in the dark at room temperature (RT) using 2 ml of a 10 mM solution of Syto 16 Green-Fluorescent nucleic acid stain from Molecular Probes (Eugene, OR, USA) (final concentration 200 nM), 7-AAD (Via-Probe, BD Pharmingen, San Diego, CA, USA) and phycoerythrin (PE)-conjugated monoclonal anti-human CD46-PE antibody (CD46-PE; Ancell, Bayport, MN, USA) that recognizes an antigen on the inner acrosomal membrane. A Flow-Count TM fluorospheres vial (Beckmann-Coulter, Fullerton, CA, USA), at a concentration of 1 × 106 beads/ml, was gently mixed for 10 to 12 s, immediately, prior to analysis, and 100 µl of fluorospheres were added accurately to each tube by precision reverse pipetting with wet tip. After the 20-min incubation period, 1ml of cold phosphate-buffered saline (PBS) was added to each tube, and the samples were analyzed by FCM. Unique region was set to include both Syto 16low and Syto 16high sperm, and such a gated population was then analyzed in another cytogram, Syto 16 versus 7-AAD, where the Syto 16 population displayed different expressions of 7-AAD. By using this gating strategy, it was possible to distinguish between viable (membrane integrity), apoptotic and dead sperm. Syto 16high/7-AADneg sperm were defined as sperm with membrane integrity (viable spermatozoa), Syto 16low/7-AADneg sperm as apoptotic and Syto 16low/7-AADpos sperm as dead. The combination of Syto 16/7-AAD and CD46 staining was sensitive not only for discriminating viable (sperm with membrane integrity) from apoptotic spermatozoa but also for identifying the acrosomal integrity and the spontaneous acrosomal reaction of sperm in different conditions. The apoptotic rate, mortality rate and the rate of membrane integrity rate were the number of apoptotic sperm, dead sperm and sperm with membrane integrity divided by the number of the entire sperm population detected by FCM, respectively. All analyses were performed on each sample before and after DGC.
Ovarian Stimulation and Embryo Development
After 3 weeks of desensitizing with gonadotropin-releasing hormone (GnRH) analog (Decapeptyl; Ipsen Biotech, Paris, France), ovarian stimulation was obtained by administering recombinant follicle-stimulating hormone (FSH) (Gonal-F, Serono France SA, Boulogne, France; or Puregon, Organon, Oss, The Netherlands), with monitoring by plasma estradiol levels and vaginal ultrasound. When ovarian follicles had reached or more than a diameter of 18 mm, ovulation was triggered with 10,000 IU of human chorionic gonadotropin (hCG; Organon). Thirty-six hours after their release, oocytes were retrieved by vaginal ultrasound-guided puncture. Sixteen to eighteen hours after insemination, the oocytes were assessed for the two pronuclei (2PN) stage, then the normal fertilization rate was calculated as the ratio of number of the oocytes in the 2PN stage to the number of punctured oocytes, and then the fertilization rate was calculated as the ratio of number of the fertilized oocytes to the number of punctured oocytes. At 70 hr after fertilization, the embryos normally reached the eight-cell stage and were graded according to their morphological criteria. Embryo grading criteria were as follows: according to morphology, grade 1 is defined as embryos in which all blastomeres have an equal size with <10% of the volume of the embryo with anucleate fragments. Grade 2 is defined as embryos in which all blastomeres have an slightly equal size with 10% to 20% of the volume of the embryo with anucleate fragments. Grade 3 has anucleate fragments comprising 21% and 50% of the embryo volume with unequal-sized cells. Grade 4 has anucleate fragments of more than 50% of the embryo volume with unequal-sized cells. According to the distribution of fragmentation: Type 1: Limited fragment location. Type 2: Scattered fragments with similar volumes. Type 3: Large and scattered fragments, accompanied by uneven size. For example, 8C/2.1: at the eight-cell stage, 10%-20% fragmentation with slightly equal-sized cells, limited fragment location; 8C/2.2: at the eight-cell stage, 10% to 20% fragmentation with slightly equal-sized cells, scattered fragments with similar volumes; 6C/2.1: at the six-cell stage, 10% to 20% fragmentation with slightly equal-sized cells, limited fragment location; 6C/2.2: at the six-cell stage, 10%-20% fragmentation with slightly equal-sized cells, scattered fragments with similar volumes, and so on. Embryo graded 6C/2.1, 6C/2.2, 7C/2.1, 7C/2.2, 8C/2.1, 8C/2.2……12C/2.1, 12C/2.2 was denoted as “good embryo,” and two to three “good embryos,” were used to transfer at 72 hr after fertilization. After transfer, the remaining embryos exhibiting good morphology were cryopreserved. The normal cleavage rate was defined as the ratio of the number of embryos to the number of fertilized oocytes in the 2PN stage. The good embryo rate was the number of “good embryos” divided by the number of fertilized oocytes in the 2PN stage. The cumulative clinical pregnancy rate was defined as the number of patients with the first fetal heartbeat following the use of all fresh and frozen embryos derived from a single ovarian stimulation cycle divided by the total number of ovarian stimulation cycles for all patients, and biochemical pregnancy by the presence of only one positive β-hCG measurement.
Statistical Analysis
The statistical tests were performed using SPSS version 20.0 software (SPSS, Chicago, IL, USA), and all data were expressed as mean ± SD. All data were analyzed from September 1, 2024 to September 15, 2024. The Kolmogorov–Smirnov test was used to determine whether the data were random samples from a normal distribution. The chi-square test was used to compare clinical pregnancy rates in different groups. The independent Student’s t-test was used to determine the differences in the various parameters (conventional seminal parameters, apoptosis rate, acrosomal integrity rate and ART outcome) between two groups. Pearson’s correlation, valid for normally distributed data, was applied to determine the correlation between the earlier apoptotic rate and various parameters (conventional semen parameters, acrosomal integrity rate, fertilization rate, normal fertilization rate, cleavage rate and good embryo rate). All tests were two-tailed, and significance was indicated by p < .05 and highly significant by p < .01.
Results
Description of the Population
A total of 98 couples who underwent IVF (corresponding to 98 cycles, one cycle per couple) since January 1, 2024 were investigated. The average age of men involved in the study was 33.15 years (±0.55 years). The etiology of infertility was teratozoospermia in 43 cases (43.87%), asthenozoospermia in 3 cases (3.06%),
Clinical Characteristics of 98 Infertile Couples Included in the Study.

DGC = density gradient centrifugation.
The Correlation of the Earlier Apoptotic Rate and Conventional Sperm Characteristics
Flow cytometry analysis of a representative semen sample showed the earlier apoptosis of sperm stained with syto 16/7-AAD (Figure 1), and the percentages of the earlier apoptotic spermatozoa shown in the gated area were 64.36%, 25.77%, and 6.21%, respectively. Flow cytometry analysis also exhibited a reduction in the percentage of the earlier apoptotic sperm after DGC, and the percentage of the earlier apoptotic sperm was 23.09% versus 3.28% before and after DGC (Figure 2). Furthermore, Table 2 summarizes the correlation analysis between the earlier apoptotic rate and conventional semen characteristics before and after DGC. The earlier apoptotic rate was significantly negatively correlated to the rate of membrane integrity and sperm concentration after DGC (r = −.862; p = .000; r =

Flow Cytometry Analysis of the Earlier Apoptosis of Spermatozoa Staining with Syto16/7-AAD.

The Comparison of the Percentage of Sperm Apoptosis Before DGC with That After DGC.
Correlation Analysis Between Sperm Parameters and the Early Apoptotic Rate (syto16-/7AAD-), Before and After DGC.
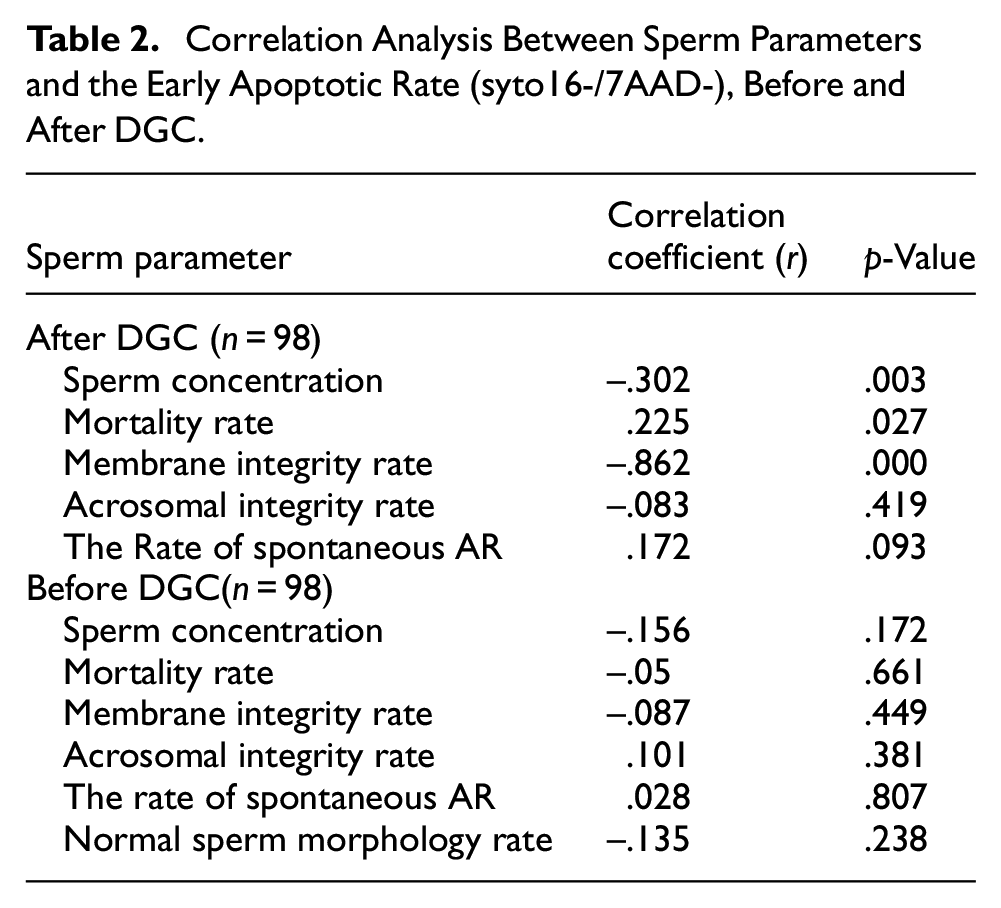
The Correlation of the Earlier Apoptotic Rate (Before and After DGC) and ART Outcome
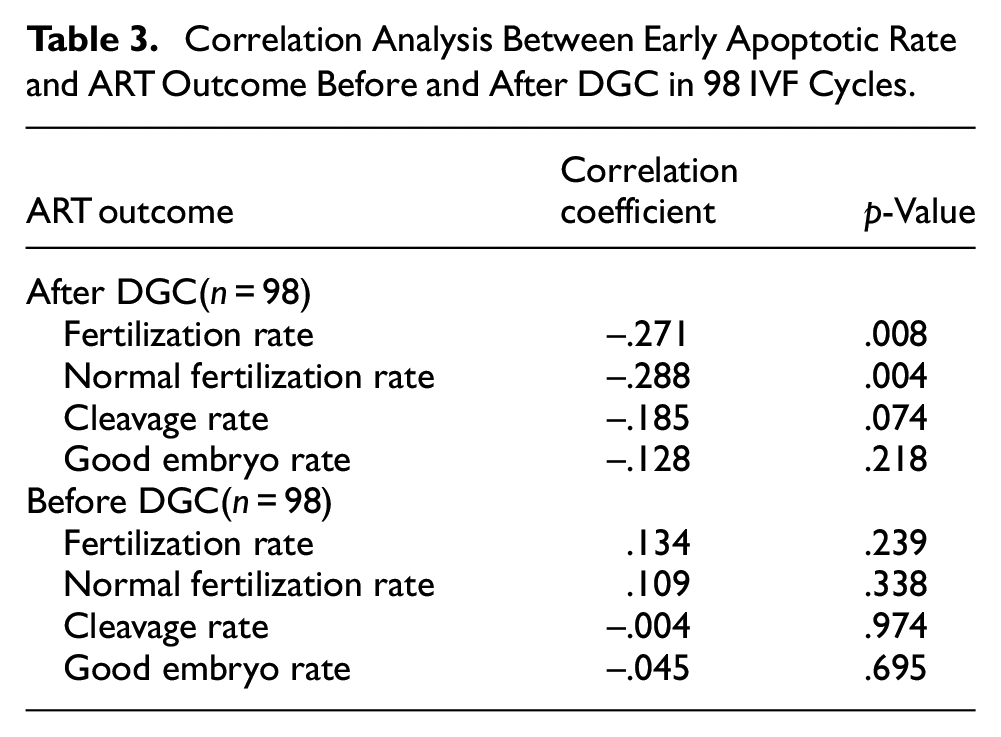
A correlation analysis was carried out to determine the relationships between the normal fertilization rate, the fertilization rate, the good embryo rate, the cleavage rate and the earlier apoptotic rate (syto16-/7AAD-) in IVF treatments before and after DGC. The earlier apoptotic rate after DGC was significantly negatively correlated with the normal fertilization rate (r = –.288; p = .004) and the fertilization rate (r = –.271; p = .008). Meanwhile, the rate of membrane integrity (syto16+/7AAD-) was significantly positively correlated with the fertilization rate (r = .318, p = .002) and the normal fertilization rate (r = .324, p = .001). Nevertheless, no correlation was found between the earlier apoptotic rate before DGC and ART outcome shown in Table 3.
Correlation Analysis Between Early Apoptotic Rate and ART Outcome Before and After DGC in 98 IVF Cycles.
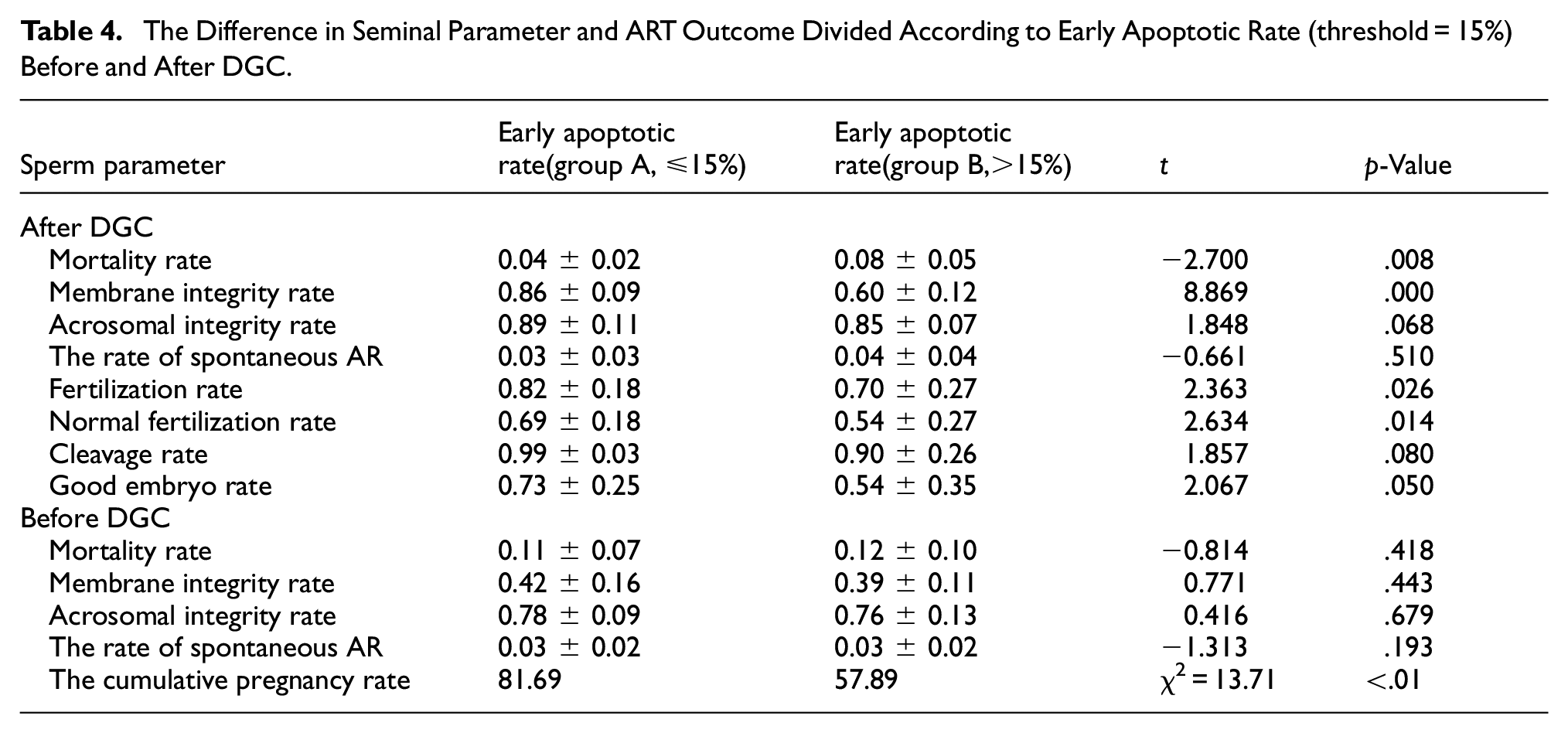
The Relationships of the Earlier Apoptotic Rate Divided by 15% with Seminal Parameter and ART Outcome
We evaluated the possible relationships of the earlier apoptotic rate (threshold = 15%) with ART outcome and conventional seminal parameters in IVF (Table 4). As regards the IVF group, a significantly lower cumulative clinical pregnancy rate was seen in the group with the earlier apoptotic rate >15% (57.89%) compared with that in the group with the earlier apoptotic rate ≤15% (81.69%, p < .01). The mortality rate (t = −2.700, p = 0.008) and membrane integrity rate (t = 8.869, p < .01) of sperm also exhibited statistically significant differences between group A (the earlier apoptotic rate ≤ 15%) and group B (the earlier apoptotic rate >15%) after DGC. Meanwhile, there were statistical differences in the fertilization rate (t = 2.363, p = .026) and the normal fertilization rate(t = 2.634, p = .014) between the two groups after DGC (Table 4). Yet, no statistical difference was found before DGC.
The Difference in Seminal Parameter and ART Outcome Divided According to Early Apoptotic Rate (threshold = 15%) Before and After DGC.
Discussion
The evidence (Romany et al., 2014) demonstrated that the non-apoptotic spermatozoa sorted with Annexin-V-conjugated magnetic-activated cell sorting (MACS) displayed increased oocyte penetration potential and sperm chromatin decondensation but not improved reproductive outcome for ICSI cycles. In contrast, the other two cases showed that applying Annexin-V-conjugated MACS technology to remove AV+ apoptotic spermatozoa increased sperm fertilization rate and clinical pregnancy rates in ICSI cycles(Grunewald et al., 2009; Grunewald & Paasch, 2013). Therefore, the effect of the earlier apoptosis of spermatozoa on fertilization rate and post-embryonic development is still a matter of debate.
In our study, Syto16, a cell-permeant nucleic acid stain that shows a large fluorescence enhancement upon binding nucleic acids RNA and DNA, is employed to distinguish apoptotic from non-apoptotic sperm. Changes in the fluorescence intensity of the Syto 16 dye correlate with the viability status of the cell in that live spermatozoa are seen as brightly fluorescent, while apoptotic spermatozoa show a dimmer fluorescence intensity that diminishes further with progression to terminal stages of spermatozoa death. Moreover, previous reports indicated that the combination of Syto16/7-AAD allowed the detection of apoptosis at an earlier stage than that of Annexin V/7-AAD (Sparrow et al., 2006; van der Pol et al., 2003). We introduce the new concept of “good embryo rate” defined as embryos graded 6C/2.2–12C/2.2 at day 3 to reflect the embryo developmental potential. We do not apply “blastocyst formation rate” to predict the embryo quality in that not all embryos are cultured for blastulation, and the definition is not objective. Nevertheless, good embryos are developed from all fertilized oocytes, and the “good embryo rate” is believed to, in part, reflect the early embryo development potential objectively.
In this study, the percentage of the earlier apoptosis in raw sperm was not correlated with the fertilization rate, the normal fertilization rate and the good embryo rate for IVF. The predictive ability of the earlier apoptosis test of sperm, performed on raw samples, diminishes. However, the predictive ability of the earlier apoptosis test of sperm is enhanced when spermatozoa are prepared using techniques such as DGC. The reason why pre-preparation sperm parameters have little prognostic value in terms of fertilization and pregnancy in ART may be the “normalizing” effect of the semen preparation procedure: it is likely that, regardless of the initial sample, a degree of homogenization occurs after sperm preparation. This suggests a need for assessing the sperm apoptotic status in the appropriate context, that is, sperm apoptosis in raw semen with reference to natural conception and sperm apoptosis in post-preparation samples in relation to ART.
Results of the present study pointed first to a highly significant negative correlation between the earlier apoptotic rate(syto16-/7AAD-), detected by FCM in semen samples after DGC and the fertilization rate, as well as the normal fertilization rate in IVF patients. Our data have already shown that the rate of membrane integrity in semen samples after DGC was significantly positively correlated with the fertilization rate and the normal fertilization rate. Therefore, this suggests that the earlier apoptotic rate of spermatozoa could be responsible in part for the fertilization rate. Existing data regarding the relationship between apoptosis and the fertilization rate are conflicting (Borges et al., 2019; Herrero et al., 2013; Martínez et al., 2021; Romany et al., 2014; Simon et al., 2017; Tavalaee et al., 2014; Wang et al., 2022), whereas, in our study, the earlier apoptotic rate was significantly negatively correlated to the fertilization rate and the normal fertilization rate in IVF. In our test, the sperm in the earlier apoptotic phase exhibited a decreased syto16 fluorescence, as suggested that the DNA/RNA structure in the nucleus had already been destroyed subtly and the probe could not be combined with DNA/RNA structure. The change in DNA/RNA structure may reduce fertilization, and this is in line with data shown by Menkveld (1991) and van Dyk et al. (2000). In IVF, spermatozoa with RNA/DNA disorder, have low fitness in oocyte fertilization, and the zona pellucida may be able to ‘identify’ genetically altered spermatozoa.
Generally, it is thought that DNA fragmentation adversely affects the embryo development/quality and hence pregnancy outcome (Borini et al., 2006). There have been fewer investigations regarding the effect of the earlier apoptosis of spermatozoa on embryo development/quality. Our data seemed to indicate that the earlier apoptosis of spermatozoa did not affect embryonic development and high-quality embryo formation during IVF. However, patients with a high proportion of the earlier apoptotic spermatozoa had lower pregnancy rates compared to patients with a low proportion of the earlier apoptotic spermatozoa (threshold = 15%).
Extensive data have already verified that the first stages of embryo development mainly depend on maternal transcripts; the paternal influence (such as sperm nuclear anomalies) begins at the six- to eight-cell stage when embryo transfers are generally performed(Vallet-Buisan et al., 2023). Spermatozoa at the earlier apoptotic stage exhibit the disorder of nuclear DNA/RNA structure by Syto16 stain, as an unfavorable paternal factor. However, the natural selection barriers that occur during IVF prevent fertilization with the earlier apoptotic spermatozoa containing RNA/DNA disorder, and only spermatozoa with intact DNA can complete sperm-egg fusion followed by embryonic development. Therefore, the process of embryonic development would not be blocked because effective conception only occurs following the fertilization of an oocyte by sperm with intact DNA. However, based on the negative correlation between the earlier apoptosis of spermatozoa and fertilization, the lower fertilization rate results in a smaller number of available embryos and the lower cumulative clinical pregnancy rate seems to be reasonable.
Conclusions
Our data supported a strong correlation between the earlier apoptosis of spermatozoa and sperm concentration, fertilization rate and clinical pregnancy rate, although no preferential effect on sperm morphology seemed to be present. In addition, to depict the inadequacy of sperm selection based mainly on morphology, our data offered a clear demonstration of the earlier apoptotic sperm that might affect fertilization but not embryo development.
However, due to the limitations of the present study, the larger sample size and further prospective investigations are still needed to obtain more accurate information on the correlation between the earlier apoptotic rate of spermatozoa and the fertilization rate as well as the cumulative clinical pregnancy rate, to outline the predictivity of the earlier apoptosis of spermatozoa in recurrent pregnancy loss and repeated implantation failure cases. Furthermore, the mechanisms by which the earlier apoptosis of spermatozoa affects fertilization should be highlighted. Until such evidence is provided, there is sufficient evidence to recommend the routine use of the earlier apoptosis test of sperm for sperm selection in IVF and for the interpretation of the cause of male infertility.
