Abstract
Reports regarding the changes in sperm concentration in different counties of the world are inconsistent. Furthermore, the reports that sprung up from specific epidemiological and experimental examinations did not include data of prior studies or geographical variations. The current study, following a previous report of massive fall in semen volume over the past 33 years, attempts to delineate the trend of altering sperm concentrations and factors responsible for this by reviewing article published from 1980 to July 2015 with geographic differences. The current study identified an overall 57% diminution in mean sperm concentration over the past 35 years (r = −.313, p = .0002), which, when analyzed for each geographical region, identified a significant decline in North America, Europe, Asia, and Africa. An increasing trend of sperm concentration was identified only in Australia. The association of male age with such a trend (R2 = .979) is reported. The authors also correlated male fertility with sperm concentration. Thus, this comprehensive, evidence-based literature review aims to concisely and systematically present the available data on sperm concentration from 1980 to 2015, as well as to statistically analyze the same and correlate male health with the declining pattern of sperm count in a single scientific review to serve the scientific research zone related to reproductive health. It points to the threat of male infertility in times ahead.
Introduction
There has been recent controversy regarding changes in sperm counts during the past 60 years worldwide (Sengupta, 2014a). It has been reported widely in last two decades that sperm count is declining (Table 1). Subsequently, Rolland, Le Moal, Wagner, Royère, and De Mouzon (2013) reported a 32% decline in sperm count from 1989 to 2005. The deterioration of semen qualities was first reported in 1974 by Nelson and Bunge. Since then, reports published regarding the changes in human semen parameters have been inconsistent. Nieschlag, Lammers, Freischem, Langer, and Wickings (1982) reported no changes, while Ng et al. (2004) revealed significantly different seminal volumes in different age groups. In 1992, Carlsen, Giwercman, Keiding, and Skakkebaek reported a worldwide decline in sperm counts in a meta-analysis of 61 studies between 1938 and 1990 evaluating the semen analyses of 14,947 presumably fertile men from 23 countries. Swan, Elkin, and Fenster (1997) published a reanalysis of the studies included by Carlsen et al. (1992). In that investigation, they reported significant declines in sperm count in the United States, Europe, and Australia, but no such decline in non-Western countries. Similar declines were also proclaimed by numerous other studies, but a clear cause was unable to be established (Auger, Kunstmann, Czyglik, & Jouannet, 1995; Swan et al., 1997). A recent article reported a decline in semen volume in aging males over the past 33 years (Sengupta, 2015). Because of reduced semen volumes, sperm concentrations were increased in older men. Moreover, significantly increased serum follicle-stimulating hormone levels sometime reflect in testicular spermatogenic function (Luetjens, Rolf, Gassner, Werny, & Nieschlag, 2002). Inhibin B as a possible marker for spermatogenesis identifies a moderate but significant decrease reflecting the aging process that possibly affects spermatogenesis (Mahmoud et al., 2003).
Studies on Changes of Sperm Concentrations in Different Age Groups (1980 to July 2015).

Note. Data are represented as mean (SD); ↓ = decrease; ↑ = increase; ↔ = no change; NS = not significant at p < .05; no p value indicates that no statistical testing was done.
Changes in sperm count can occur after occupational and environmental exposure to toxic agents (Dutta, Joshi, Sengupta, & Bhattacharya, 2013; Sengupta & Banerjee, 2014) or from predisposing factors of the host, such as age (Kidd, Eskenazi, & Wyrobek, 2001). The innumerable evidences, mostly from clinical studies, suggest that age is associated with all the concomitant factors, resulting in diminished sperm concentration (Spandorfer et al., 1998). Men at older ages (e.g., ≥50 years) were underrepresented in many clinical studies, which restricts statistical strength and prevents unveiling of the exact form of relationship between age and sperm concentration. In addition, potential confounders that might explain changes with age, such as smoking history or duration of abstinence, were hardly ever taken into consideration (Wyrobek et al., 1983).
A detailed scrutiny of diverse studies from specific cites reveals evidence of decline in sperm concentration, but a worldwide decline has not been demonstrated. It is arduous to execute a systematic, scientific study regarding the decline in human semen quality. Thus, the objective of the current review is to build up a substantial idea regarding alterations in sperm concentration in humans by picking the huge scattered reports of the past 35 years, molding them in sequential pattern, and statistically analyzing and correlating the worldwide declining sperm count trend with male fertility.
Data Extraction and Data Analysis
Research articles on humans published in English from 1980 through July 2015 were included in the current report. The authors also included 32 of 61 reports of the study by Carlsen et al. (1992), that is, reports from 1980 to 1992. The authors selected publications about sperm concentration with predefined criteria for inclusion and exclusion, as follows. (1) The non-Carlsen studies published during 1980 to July 2015 were identified by using Medical Subject Headings (MeSH) of electronic databases, which included Medline, National Library of Medicine, Bethesda, MD, with the following key words: sperm count, sperm density, sperm concentration, semen quality, male infertility, and semen analysis. (2) Relevant literature on changes of sperm concentration and its influence on future natural and assisted conception cycles were retrieved. (3) Data on subjects with clinical problems were been excluded. (4) Studies with insufficient numbers of subjects (n < 5) were excluded. In each case, sperm concentration and its outcome were evaluated. Analytic epidemiological studies were emphasized. Therefore, the current analysis was based on 138 studies published in from 1980 to 2015 (July). In the Results section, the relative changes in the outcome with age are represented. Whenever possible, the differences between younger men (i.e., ages ≤30 years) and older men (i.e., ages ≥50 years) were summarized. The authors also analyzed the correlation of age with mean sperm concentration obtained from the published studies with proper age data. For simple statistical analyses (calculation of mean sperm concentration, median values, and Box-and-Whisker plots), Microsoft Excel 2013 was used. Correlation and regression analyses of data were done using StatSoft (2011). Correlation coefficient was considered to be significant if p was <.05 or <.001 (Fisher & Yates, 1974). Mean sperm concentrations of all 138 reports were also analyzed with linear regression weighted by number of subjects included in the individual publications.
Description of Data
During the retrieval of relevant documents, the authors found a total of 138 studies that reported temporal decline in sperm concentration in the past 35 years. The outcomes of these studies are represented in Table 1. Most of the reports are based on andrology laboratories or assisted conception populations (44.20%) and epidemiological studies (28.26%), while others used volunteers recruited from sperm banks or advertisements (6.52%) and infertility clinics (16.67%). Among the 138 published research works discussed in this article from 1980 through 2015, most were carried out in Europe, North America, and Asia, while others were carried out in South America, Australia, and Africa. Most of the studies used sample sizes less than 500 subjects (68.12%) and ≥1,000 (21.01%), while a few studies used sample sizes between 500 and 1,000 subjects (10.87%). Five reports used an extraordinarily large sample size (>10,000; Andolz et al., 1999; Belloc et al., 2014; Eisenberg et al., 2014; Geoffroy-Siraudin et al., 2012; Rolland et al., 2013). Out of 138 reports, 80.43% provided data about the age of subjects. No significant alterations in sperm concentration was identified in 15.94% of the reports, only 3.62% reports identified a significant increase, and 80.43% reports identified a significant decrease in sperm concentration from 1980 to 2015. While only the reports identifying significant decrease were taken into consideration, it was observed that out of 111 studies, 23.42% of reports identified strong significance. The current report also enlisted interval studies published during the reporting period of the current article, which proclaims alterations in sperm concentrations during 1980 to 2015 (Table 2).
Summary of Interval Studies Reporting Decline in Sperm Concentration Published Within the Study Period of the Current Report.

State of Affairs: Past 35 Years
Worldwide Variations
Mean sperm concentrations were obtained either directly from the published articles or in some cases arithmetic mean was calculated from median or geometric mean. Linear regression analysis identified a significant decrease between 1980 and 2015 from 91.65 × 106/mL to 39.34 × 106/mL (r = −.313, p = .0002). This reflected almost a 57% decline in sperm count worldwide from 1980 (Figure 1A). The current study also identifies that recruitment of a larger population for this type of study increased predominantly after 1995.

(A) Temporal decline in sperm concentration (×106/mL) (r = −.313, p = .0002, R2 = .098); bubble size corresponds to the number of men in study. (B) Changes in sperm concentrations from 1980 to 2015 shown as Box-and-Whisker plots. White bands indicate the median values for each duration. The Y-intercept shows median (71.6 × 106/mL), and the regression band (y = −3.92x + 87.72, R2 = .460) indicates significant decline from 1980 to 2015.
Median values of sperm concentrations were calculated in seven different intervals (from 1980-1985 to 2010-2015). The median value was reported to be 71.6 × 106/mL during the study period. These values are plotted in the Box-and-Whisker plot in Figure 1B, which also identifies a significant decline from 1980-1985 to 2010-2015 (y = −3.92x + 87.72, R2 = .460).
Regional Variations
Recent studies on the male reproductive system bring conflicting evidence to the forefront regarding sperm counts, with some reporting significant decline while some have reported no change. North America, Europe, Asia, and Africa are more prone to a declining trend of sperm counts over the years, whereas studies based in South America and Australia do not depict such a trend (Figures 2 and 3). An overview of the sperm counts obtained from data produced by various regions of the world is reported in Figure 1, which identifies that there was a significant decline in sperm concentrations from 1980 to 2015. However, a striking feature of much of the data is the surfacing of regional differences in semen quality. It has been suggested that these regional differences in sperm counts possibly are biologically meaningful. Most of the controversies that have aroused from past clinical studies about semen quality may be partly due to involvement of only few selected groups of men. In many studies, historical data collected for other purposes have been used without close attention to important and specific factors relevant to an analysis of secular or geographical trends.
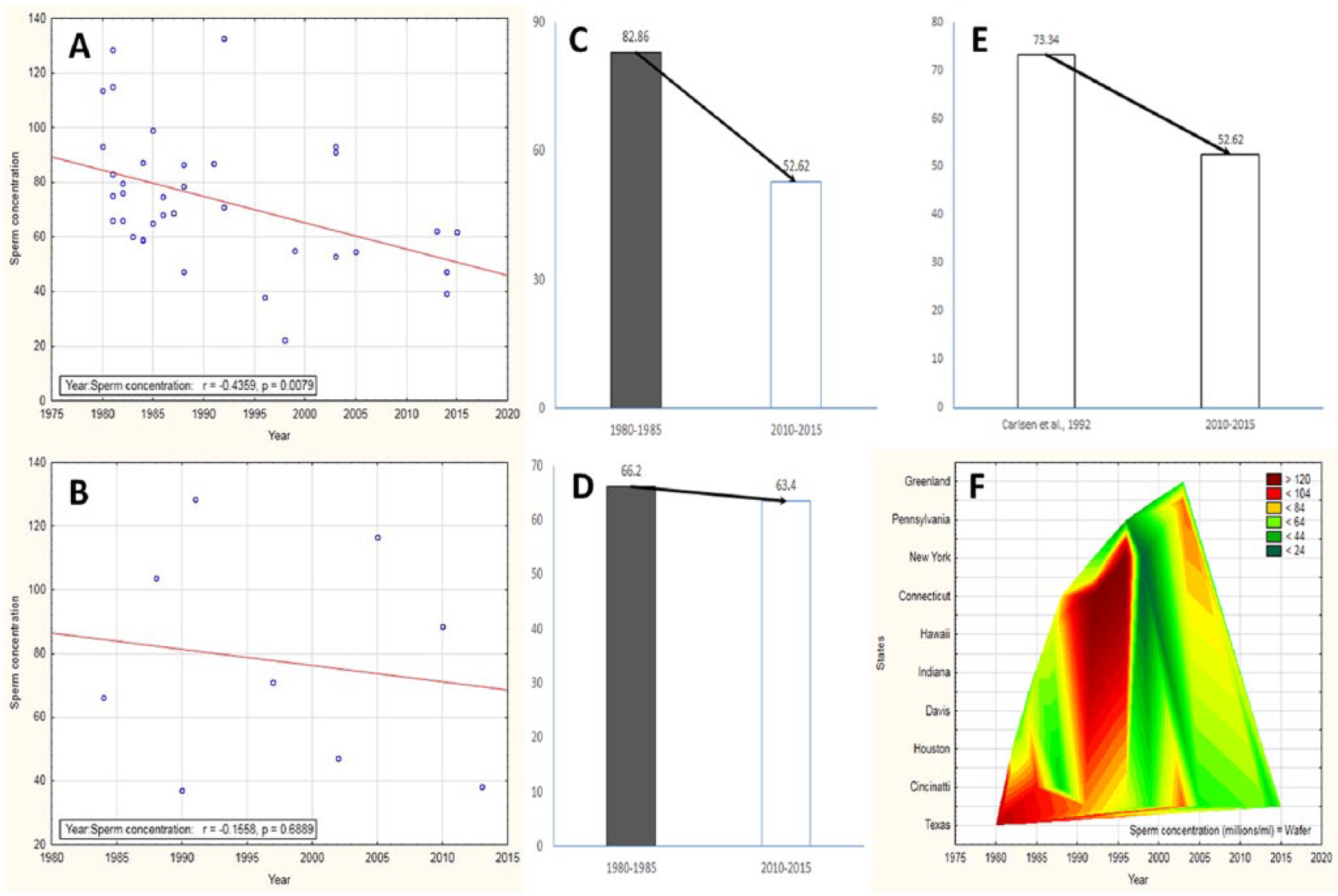
(A) Correlation plot shows significant decline in sperm concentration in North America (r = −.435, p = .007), but no such alteration in South America (B). It has decreased 36.49% in North America (C) and only 4.22% in South America (D) from 1980 to 2015. A comparative overview of Carlsen et al., 1992 and this study reveals a further 28.25% decline after that report in North America (E). Wafer plot reveals that New York and Connecticut have the highest sperm count (F).
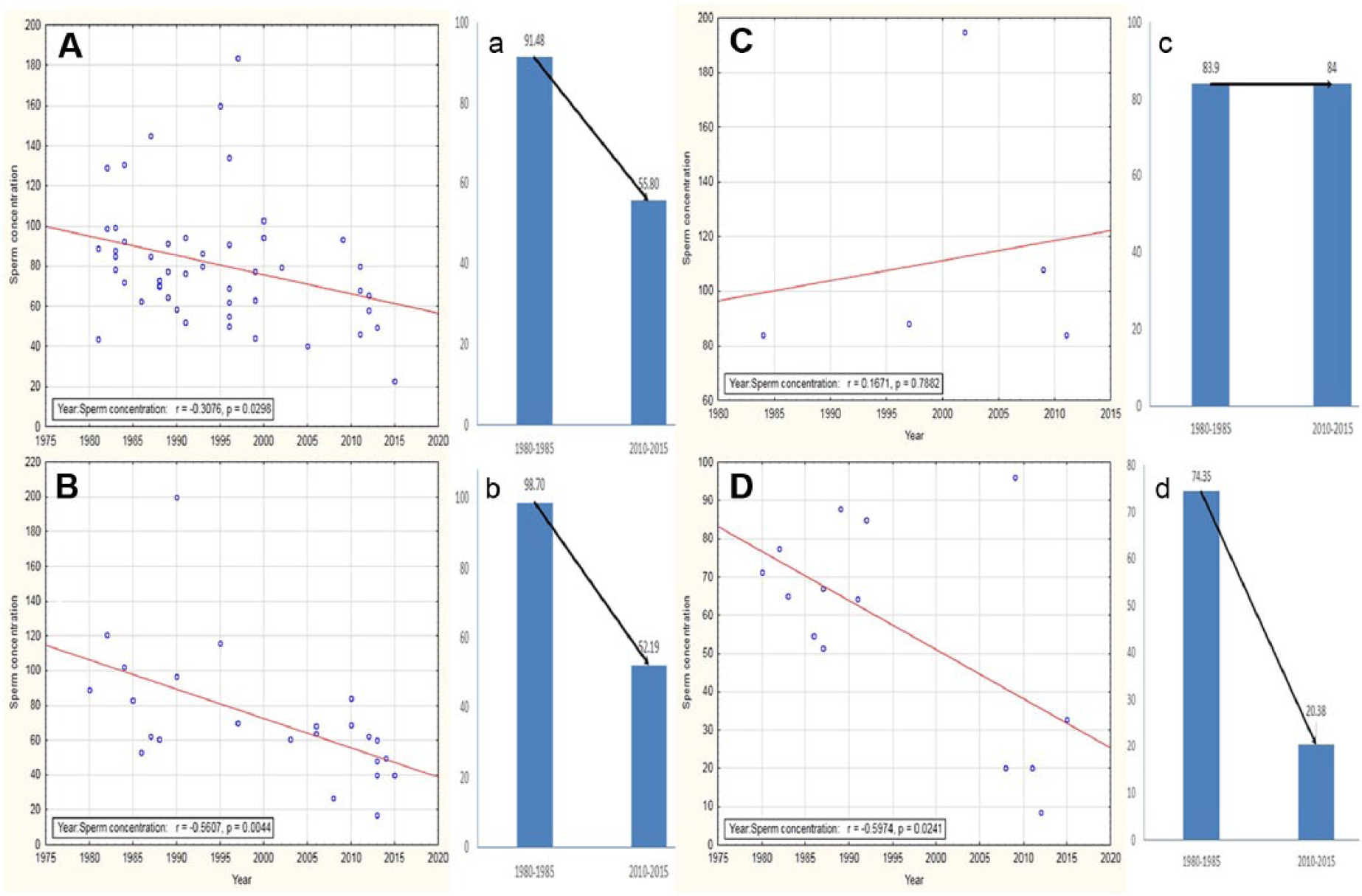
(A) Correlation plots of non-American continents showed significant reduction in sperm concentrations in Europe (r = −.307, p = .02) (A), Asia (r = −.560, p = .004) (B), and Africa (r = −.597, p = .02) (D), while Australia showed an increasing trend in sperm concentration over time (r = .167, p = .788) (C). Mean sperm concentrations reflected similar trends (a-d).
North America
Numerous studies conducted in the United States have demonstrated a declining trend of mean sperm concentration in different regions over the years 1980 to 2015 (r = −.435, p = .007; Figure 2A). Data on sperm counts procured from several studies from 1938 to 1980 (following the report of Saidi et al., 1999) also depicts a significant decrease in sperm count (r = −.635, p = .004) in North American men until 1980. Thus, from these two findings it can be resolved that North American states are going on with the trend of declining sperm counts from 1938 until 2015. The authors also evaluated the mean sperm concentrations in 1980-1985 and 2010-2015, which identified a 36.49% decline in sperm concentration over the last 35 years (Figure 2C). The current study also compares the mean sperm concentrations of 2015 with the report of Carlsen et al. published in 1992. It reveals an additional 28.25% decline in mean sperm concentration after 1992 in North American men (Figure 2E).
Various scattered reports were brought under one analytical review by Saidi et al. (1999) using 29 studies based in the United States from 1938 to 1996, which analyzed semen of 9,612 fertile men. The mean sperm concentrations of selected geographic locations were determined with analysis of variance (ANOVA), and any alterations were gauged with time using linear regression analysis. From that study, it was perceptible that New York had the highest value for mean sperm concentration as compared to the other sates of the United States (as also reported by Fisch & Goluboff, 1996). It can be explained through Figure 2F, which attempts to describe the variations in sperm count among states in North America and also indicates that New York and Connecticut have the highest sperm count in the United States. Thus, sperm concentrations were reported to vary with geographical areas in the United States, the maximum being that of New York whose exact reason is yet to be identified, but factors like climate, socioeconomic status, ethnicity, and other environmental and social factors should be considered.
South America
Only a handful of studies have been conducted involving regions of South America to assess the trend of declining, increasing, or static sperm counts as well as changes of the same from place to place across the continent (Borges et al., 2013; Oliva, Spira, & Multigner, 2001; Pasqualotto et al., 2005; Tortolero et al., 1999). A study in Venezuela, carried out on the male partners of infertile couples, suggested that the proportion of men presenting azoospermia or oligospermia showed no change from 1981 and 1995 (Tortolero et al., 1999). Studies also focused on the impact of chemical exposures on various characteristics of sperm among the populations of male partners of infertile couples during the period 1995 to 1998 in the southern coastal region of Argentina (Oliva et al., 2001).
Studies retrieved during the current analysis identified that most of the reports were from Brazil and Argentina. These reports indicated no such significant alteration in sperm count in South American men (r = −.155, p = .688; Figure 2B). The mean sperm concentrations have declined from 66 × 106/mL to 63.4 × 106/mL, which is only 4.22% relative to that of 1980 (Figure 2D). A presumption about the role of environmental and occupational factors on male reproductive health can be drawn through the above-mentioned studies (Oliva et al., 2001; Tortolero et al., 1999) and the declining trend of the sperm count (though not significant at p of .05). For example, agricultural zones of South America are cursed by exposure to organochlorine pesticides and other endocrine disruptors, which might be more detrimental to fertility in the coming years.
Europe
Europe is a continent of geographic diversities. The current report included 50 studies carried out from 1980 to 2015. Most of these were conducted in the United Kingdom, France, Denmark, and Finland. The analysis revealed a significant decline in sperm concentrations over time (r = −.307, p = .02; Figure 3A), and the mean sperm concentration have declined more than 39% compared to 1980 (Figure 3A). Among the European countries, the United Kingdom and Denmark had the maximum sperm concentrations, while Spanish men were reported to have the least sperm counts. Similar cross-sectional studies were performed by Møller and Skakkebæk (1999) and Skakkebæk et al. (1998) to investigate the possible geographical differences in sperm count, involving subjects who were male partners of pregnant women from Denmark, France, Scotland, and Finland and young men from the general populations in Denmark, Norway, Finland, Estonia, and Lithuania. These studies identified a significantly better semen quality in Finland, Estonia, and Lithuania from which an East-West gradient is revealed in the European area. A major factor that has an immense impact on sperm count is seasonal changes, which is evident from the fact that sperm count differed by about 30% in summer from that in winter in all the four countries. This fact is also supported by all other previous studies, some including men of known fertility and some of known subfertility, which observed seasonal variations in sperm count being lowest during the summer and highest during autumn or winter (Gyllenborg et al., 1999; Maier, Newbold, & McLachlan, 1985; Spira, 1984; Tjoa et al., 1982). There are a few studies that could not detect any seasonal variations (e.g., Mallidis, Howard, & Baker, 1991).
Time trends in semen quality are interestingly related with almost identical patterns in the occurrence of testicular cancer, which is rising worldwide. It was reported that the incidence of this disease is five times higher among Danish men than it is among Finnish men (Adami et al., 1994; Forman & Møller, 1994), while in the previously discussed study the former had a much better sperm count. Such inverse relationship between sperm count and the risk of testicular cancer is not only apparent from the cohort studies but is also observed in individuals (Møller & Skakkebæk, 1999). This could also be supported by the study that predicts that men born in Scandinavia during the Second World War had a comparatively lower risk of developing testicular cancer in adult life than men who were born before or after the war (Adami et al., 1994; Møller & Skakkebæk, 1999). Sperm counts are thought to decline with a more recent year of birth as is suggested by a couple of studies (Irvine et al., 1996; Skakkebæk et al., 1998), for which a possible causative agent could be exogenous factors that interfere with the functions and multiplication of the fetal Sertoli cells resulting in a syndrome of reduced sperm count, hypospadias, undescended testis, and testicular cancer (Bergman, Brandt, & Brouwer, 1996; Sharpe & Skakkebæk, 1993). In this respect, it is noteworthy that the gradient in the incidence of hypospadias between Denmark and Finland is apparently parallel to the gradient of the testicular cancer in these regions (Toppari et al., 1996). Thus, it can be concluded that European cities suffer from robust variations in sperm count, which is even related to sperm production and testicular cancer. These variations might be due to differences in life styles, environmental factors, endocrine disruptions, or other factors.
Asia
Most of the Asian studies were carried out in China (Diao et al., 2013; Nie et al., 2012; ; Tang et al., 2013; Tang et al., 2015; Wang et al., 1985) and India (Jajoo & Kalyani, 2013; Marimuthu et al., 2003; Mehta, Makwana, Ranga, Srinivasan, & Virk, 2006; Mukhopadhyay et al., 2010; Pal et al., 2006), and a few in Japan (Iwamoto et al., 2013), Israel (Benshushan et al., 1997; Homonnai et al., 1982; Laufer et al., 1985; Singer et al., 1990), and Saudi Arabia (el Shoura et al., 1995).
In 1980, Wang and Yeung first reported a decline in sperm count in Chinese men. Later, numerous studies reported similar observations (Diao et al., 2013; Nie et al., 2012). In Japan, studies on altering sperm concentrations in males are scarce. Thus, very limited information could be gathered regarding male reproductive status. A severe declining trend or variations in sperm quality has been reported by few cross-sectional studies in Japan (Iwamoto et al., 2013). India has a medley of cultures, religions, life styles, and most importantly geographic and climatic diversities according to which the physical attributes of the people belonging to each region have been molded. Therefore, variations in physicality and thereby reproductive health status of men are most likely to occur among different regions of this country. Not many studies have been done to analyze such predictions (Jajoo & Kalyani, 2013; Marimuthu et al., 2003; Mukhopadhyay et al., 2010; Pal et al., 2006). However, a relevant study was conducted in laboratories at five different cities, namely, Bangalore, Kurnool, Mumbai, Jalandhar, and Jodhpur, using sperm samples from male partners of infertile couples. Samples were analyzed for sperm concentrations using standardized methods recommended by the World Health Organization (WHO). The outcome of the study suggested that prevalence of both azoospermia and oligozoospermia was highest in Kurnool, being 38.2% and 51%, respectively. The observation from the mean sperm counts in normospermic men depicts lower values for the metropolitan cities like Mumbai and Bangalore than other small cities (Mehta et al., 2006).
Current analysis reveals a similar trend of declining sperm count in males based in Asia, which is similar to the U.S. and European countries. The current report identifies a significant decline in mean sperm concentration (47.12%) from 1980 to 2015 (r = −.560, p = .004; Figure 3B-b). This may be attributable to lifestyle, food habits, and the extensive use of fertilizers in cultivation. Gui-Yuan, Meng-Chun, Jin-Lai, and Wen-Qing (1989) demonstrated that a probable cause of such declining sperm concentrations in India might be due to extensive use of Gossypol (a phenol compound isolated from the seeds, stems, and roots of the cotton plant) and other pesticides.
Africa
In 1991, the WHO had estimated that almost 20 to 35 million couples were infertile in Africa. Nigeria is suggested to have been suffering from highest infertility problems among the other African regions, with the male infertility factor accounting for 40% to 50%. The degree of infertility and its cause vary from place to place. This is evident from the study pursued in mid-western Nigeria, which reported that about 50% of the 780 couples under evaluation differed in the causes of their infertility (Okonofua, Menakaya, Onemu, Omo-Aghaja, & Bergstrom, 2005). A study associated with south-western Nigeria had reported that 42.4% of infertility resulted from the male factor (Ikechebula, Adinma, Orie, & Ikegwuonu, 2003).
In the current analysis, the authors identified that most of the studies on sperm count in Africa was carried out in Nigeria (Akande et al., 2011; Jimoh et al., 2012; Lapido, 1980; Nnatu et al., 1991; Osegbe et al., 1986; Sobowale & Akiwumi, 1989; Ugwuja et al., 2008). A time-dependent decline in sperm concentration was observed from 1980 to 2015 (r = −.597, p = .02; Figure 3D) that reflected an overall 72.58% decrease in sperm concentration (Figure 3D-d). It is thus understandable that regional variations in reproductive status prevails in Africa and the high rates of male infertility in Nigeria is thought to be due to infections, sexually transmitted diseases, and hormonal abnormalities (Akinloye, Grommok, Nieschlag, & Simoni, 2009; Emokpae, Uadia, Omale-Itodo, & Orok, 2007).
The organochlorine pesticide DDT (1,1,1-trichloro-2,2-bis (chlorodiphenyl)ethane) is one of the most persistent organic pollutants that is known to be toxic, persistent, and bioaccumulative and has been used for malaria vector in South Africa since 1945. According to a cross-sectional study in an endemic malaria area (Limpopo Province, South Africa), DDT concentrations and sperm counts were negatively correlated among African men (Jager, Aneck-Hahn, Bornman, Farias, & Spanò, 2012). Therefore, sperm counts vary in different regions of Africa based on environmental factors, chemicals, and infections that each region offers.
Australia
There are very few reports on changes in sperm count of Australian men. In 1984, the report of Handelsman et al. about the semen quality of sperm donors first predicted that there is no alteration in the sperm count of Australian men. Later, several studies reported no alteration in semen quality (Costello et al., 2002; Stewart et al., 2009). In the current analysis, the authors identified a mild increase in sperm concentration from 1980 to 2015 (r = .167, p = .788; Figure 3C) that reflected 0.11% increase in sperm concentration in Australian men (Figure 3C-c).
Links With Possible Factors
These observed trends in sperm concentration might be linked to the effects of environmental factors, including endocrine disruptors. They might also be linked to other known semen altering factors, like an increase in body mass index (BMI), stress, nutrition, or sometimes systemic pathologies, such as diabetes, cancer, or systemic infection. In previous reports, the authors mentioned various environmental, occupational, and lifestyle factors that can alter sperm count (Dutta et al., 2013; Sengupta & Banerjee, 2014; Figure 4). In addition, aging increases the risk of oxidative stress and other systemic diseases, which can also contribute to a reduction in sperm count (R2 = .979, p > .001).

Possible mechanism of action of environmental toxicants, lifestyle factors, obesity, and systemic diseases in lowering sperm count in ageing male (red arrows show inhibition, blue arrows show stimulation, purple arrows indicate decrease, and blue arrows indicate increase in hormone levels).
Environmental and Lifestyle Factors: Direct Action on Spermatogenesis
Since 1961, dibromochloropropane (DBCP), a nematocide employed on various tropical crops, has been known to affect testicular functions. Torkelson (1961) reported that DBCP caused testicular atrophy in laboratory animals. Later, in 1977, Whorton, Krauss, Marshall, and Milby published an article that reported DBCP had rendered thousands of agricultural workers sterile in many countries worldwide. Since that report, many other chemical substances have been reported to have the potential to decrease sperm count in men (Bonde, 1996). These substances include pesticides such as ethylene dibromide (Ratcliffe et al., 1987) and carbaryl (Wyrobek, Watchmaker, et al., 1981), solvents such as glycol ethers (Welch et al., 1988), carbon disulfide (Lancranjan, 1972), and 2-bromopropane (Kim et al., 1996; Table 3), and heavy metals (Lancranjan, Popescu, Gavanescu, Klepsch, & Serbanescu, 1975; Sengupta, 2013; Sengupta, 2014b; Sengupta, Banerjee, Nath, Das, & Banerjee, 2015; Table 4). Along with these agents, thousands of chemicals should be included with experimental evidence that have demonstrated testicular toxicity in animals but for which data are not available for humans (Bonde, 1996; Łepecka-Klusek, Wdowiak, Pilewska-Kozak, Syty, & Jakiel, 2011; Sundaram & Witorsch, 1995). Rosenstock, Liptzin, Six, and Tomich (2013) reported a significant increase of pesticide use from 1980 to 2010 proclaiming that these chemicals play a pivotal role in declining sperm count in different age groups of males. Among thousands of chemicals that we are exposed to environmentally or occupationally, very few have been evaluated for reproductive toxicity, specifically in various age groups of men. It is known that the susceptibility to this chemical assault increases with age.
Studies on Pesticides and Sperm Concentration of Past Two Decades.

Note. MBP = mono-n-butyl phthalate; MEP = mono-ethyl phthalate; MMP = mono-methyl phthalate; MEHP = mono-2-ethylhexyl phthalate; BPA = biphenol-A; DBP = di-n-butyl phthalate; DEP = di-ethyl phthalate; p,p′-DDE = p,p′-Dichlorodiphenyldichloroethylene.
Studies on Heavy Metals and Sperm Concentration.

Besides chemical assault, researchers must also consider physical factors like ionizing radiation and heat. Spermatogenesis requires the temperature in the scrotum to be at least 3°C lower than body temperature. An increase in scrotal temperature disturbs spermatogenesis (Mieusset, Bujan, Mansat, Grandjean, & Pontonnier, 1991). Prolonged exposure to sources of radiant heat may lead to significant changes in sperm concentrations (Figa-Talamanca et al., 1992; Thonneau, Bujan, Multigner, & Mieusset, 1998). Other physical agents, such as high-frequency electromagnetic fields, may also affect testicular function (Weyandt et al., 1996; Table 5). As the use of cell phones, laptops, and Wi-Fi has increased tremendously in past three decades (Lenhart, Purcell, Smith, & Zickuhr, 2010), the possibility of radiation-induced decline in sperm count increases.
Reports on Cell Phone Usage and Sperm Concentration.
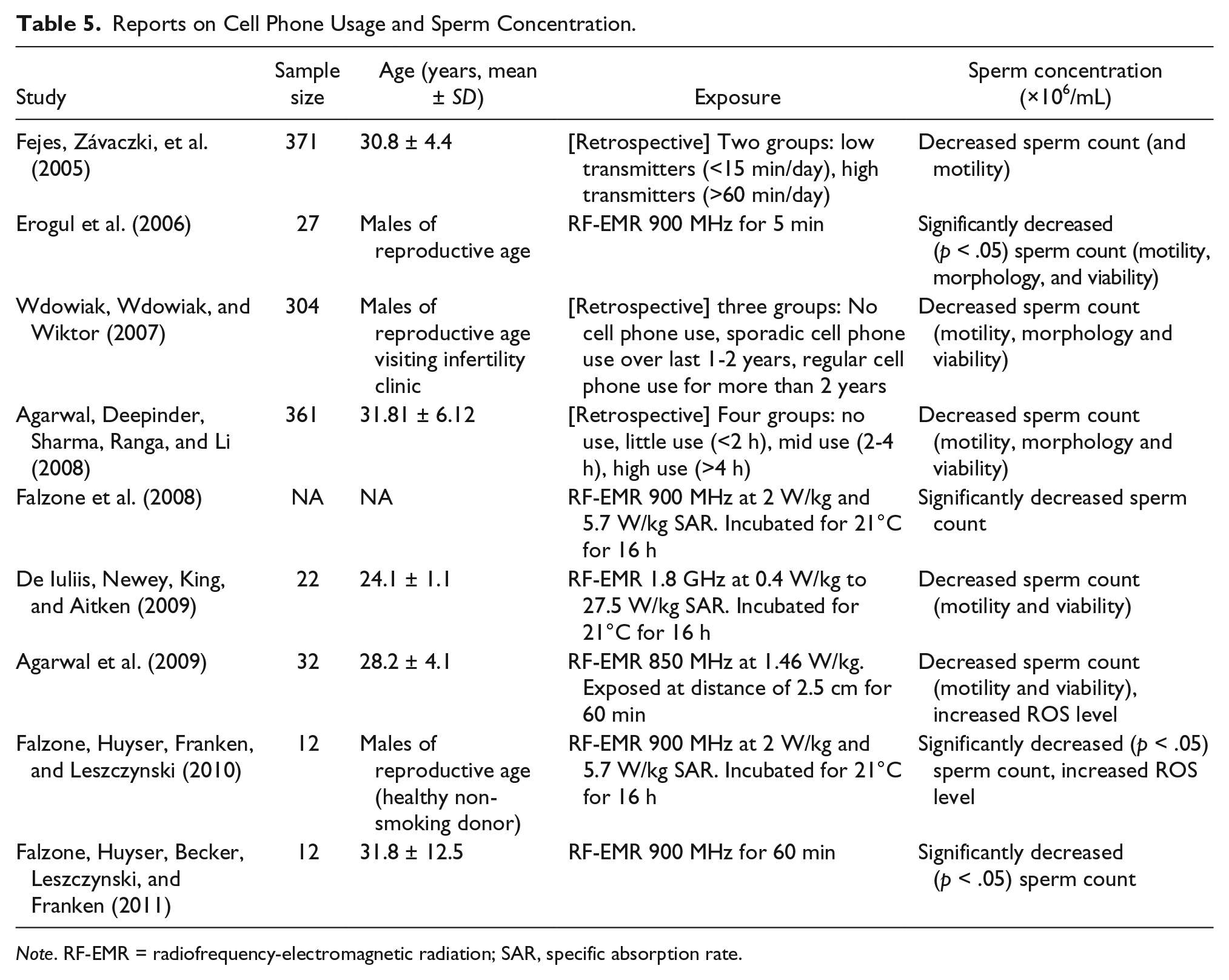
Note. RF-EMR = radiofrequency-electromagnetic radiation; SAR, specific absorption rate.
Finally, the effects of regular consumption of tobacco or alcohol, both of which are on the brink of disturbing spermatogenesis, should not be ignored (Bonde, 1996; Multigner & Spira, 1997). Stress, which is very hard to assess, has also been set forth as a factor that may have a negative impact on sperm production (Fenster et al., 1997; Negro-Vilar, 1993). In a report of the Royal College of Physicians (2012), researchers documented a massive change in consumption of tobacco from 1962 to 2007. They described an increase in tobacco consumption from 1962 to 2000 and a gentle fall from 2000 to 2012. They also recorded that this decrease is prevalent in the United Kingdom and other European countries, but in the rest of the world there are no data of decreasing active or passive consumption, specifically in developing countries, which could be related to the worldwide decrease in sperm count even in 2015. Another relevant Federal Survey (2013) reported that males in the 18 to 35 years age group are mostly addicted to tobacco consumption, followed by the age group of 35 to 60. This is of great concern in relation to declining sperm counts in aging males.
Major alterations in sperm counts have been observed in populations following changes in other diverse factors, such as catastrophes. The Kobe earthquake decreased sperm count of local men significantly (Fukuda, Fukuda, Shimazu, Yomura, & Shimizu, 1996). Clearly, a large number of environmental factors are likely to affect spermatogenesis in humans. However, most of the studies cited above were carried out in a professional environment in circumstances in which the level of chemical and physical exposure is generally high. This accounts for a nonnegligible proportion of the adult male population. In addition, given the widespread use of chemical substances in particular, it is legitimate to raise questions concerning the consequences for the general population of their accidental or deliberate release into the environment.
Effect of Endocrine Disruptors
There are numerous reports that identify the role of endocrine disrupting chemicals (EDCs) for secular changes in sperm count. These EDCs are liable to have adverse effects on individual organisms through primary effects on endocrine systems. These substances, via their estrogenic or anti-androgenic activities, are likely to hinder testicular development in the fetus and the postnatal functions of the testes (Sharpe, 1993). In 1938, estrogenic activity for a range of man-made chemicals was first documented (Dodds & Lawson, 1938). Since the 1960s, it has been known that synthetic compounds such as the chlorinated insecticides methoxychlor and DDT and polychlorinated biphenyls (PCBs) may have estrogenic activity in laboratory animals (Bitman, Cecil, Harris, & Fries, 1968; Tullner, 1961). It has been known for some time that some xenobiotics may act in a similar way to hormones (xenohormones), thereby affecting endocrine regulations. The list of chemical substances with hormonal activity in vitro or in vivo has not stopped growing in the past few decades. In addition to those already mentioned, they include insecticides (lindane), fungicides (vinchlozoline), surfactants (alkylphenols), plastics (bisphenol-A, phthalates), and industrial by-products (dioxins; reviewed in Colborn, Vomsaal, & Soto, 1993; Toppari et al., 1996). Experiments in vivo in laboratory animals have identified that the administration of methoxychlor, octylphenol, butyl phthalate, or dioxin during gestation or lactation causes a significant decrease in sperm production in the adult (Gray, 1982; Mably, Bjerke, Moore, Gendron-Fitzpatrick, & Peterson, 1992; Sharpe, Fisher, Millar, Jobling, & Sumpter, 1995). Several observations support the idea that EDCs may be involved in changes of sperm quality in humans and that these disrupting effects increase with the progression of age. Thus, the trend of declining sperm counts in aging males from 1980 to 2015 may be attributed to the persistently increased exposure of these EDCs.
Obesity Can Affect Spermatogenesis
The association between high adiposity and alterations in sperm count has not been clearly demonstrated in men. Data from large-scale epidemiological studies suggest an elevated risk for infertility among couples when the male partner is overweight or obese (Nguyen, Wilcox, Skjaerven, & Baird, 2007; Ramlau-Hansen et al., 2007; Sallmen, Sandler, Hoppin, Blair, & Baird, 2006). Several studies have reported an inverse correlation between BMI and sperm concentration or total sperm count (Jensen et al., 2004; Paasch, Grunewald, Kratzsch, & Glander, 2010), but other reports have failed to document this relationship (Aggerholm, Thulstrup, Toft, Ramlau-Hansen, & Bonde, 2008; Duits, van Wely, van der Veen, & Gianotten, 2010). The current report collected data from the past few decades regarding the association between BMI and sperm concentration and identified that overweight and obesity were associated with an increased risk of oligozoospermia or azoospermia (Table 6).
Body Mass Index (BMI), Obesity, and Sperm Concentration.

Note. IVF = in vitro fertilization; ICSI = intracytoplasmic sperm injection.
Regarding the correlation between obesity and alteration of sperm concentration, different hypotheses have been raised. First, alterations of the hypothalamo-pituitary-gonadal axis have been reported to be involved in this process. Aromatization of steroids to estrogens in peripheral tissues leads to the hypogonadotropic hyperestrogenic hypogonadism previously described in obese men (Schneider, Kirschner, Berkowitz, & Ertel, 1979), with a significant decline in total and free testosterone levels (with increased leptin) and increase in estradiol (E2), both leading to deleterious effects on spermatogenesis. Moreover, reports have identified a decrease of sex hormone-binding globulin among obese men, notably mediated by hyperinsulinemia, emphasizing the negative feedback effect of elevated total E2 levels (Stellato, Feldman, Hamdy, Horton, & McKinlay, 2000). Obesity is also associated with an increase of endorphins, leading to a both lower LH pulse amplitude and GnRH production (Bhattarai, Chaudhuri, Bhattacharya, & Sengupta, 2014; Bhattarai, Bhattacharya, Chaudhuri & Sengupta, 2014; Blank, Clark, Heymsfield, Rudman, & Blank, 1994; Dutta et al., 2013; Krajewska-Kulak & Sengupta, 2013). In an earlier report, the authors described that the role of thyroid hormones in men can also contribute to decreased sperm count (Krajewska-Kulak & Sengupta, 2013). A decreased level of circulating triiodo-thyronine (T3) may affect testicular production of testosterone, and thus affects spermatogenesis (Krajewska-Kulak & Sengupta, 2013). Some authors have also reported that obesity may directly alter spermatogenesis and Sertoli cell function (Winters et al., 2006) by the more severe diminution of inhibin B levels compared with the decrease of follicle-stimulating hormone. Another hypothesis is the increase of scrotal temperature caused by hip and abdominal fat tissue accumulation, or even scrotal fat deposition (Shafik & Olfat, 1981), would involve spermatogenesis disturbances. Preferential accumulation in fatty tissue of toxic substances and liposoluble EDCs would amplify those alterations, as indicated by serum organochlorine levels being correlated with BMI (Magnusdottir et al., 2005).
In 2007, Johnson et al. reported that obesity prevalence had increased tremendously in the second half of the 1900s, which can be correlated with the trend of declining sperm count in men. The WHO (2014) also reported the prevalence of obesity (BMI ≥30 kg/m2) had increased remarkably worldwide by 2014, and notably in the United States, where a decline in sperm count is more significant than in the rest of the world. The WHO reported worldwide obesity had more than doubled since 1980. They also reported, in 2014, that more than 1.9 billion adults 18 years and older were overweight. Of these, over 600 million were obese. In 2014, 39% of adults aged 18 years and over (38% of men) were overweight (WHO, 2014), and the prevalence of obesity increases with age. This is reassuring the data of declining sperm counts in aging males presented in this report.
Conclusion
The current study, with strong experimental evidences extracted by analyzing multitudinous studies, reports a declining trend in sperm concentration over the past 35 years with perceptions of the reasons of such deterioration in male reproductive health. These variations of sperm concentrations are observed by taking into account the different geographical regions. The outcome of this review is a systematic, concisely arranged scientific report on sperm concentration and the factors involved, from 1980 to 2015 from all over the world; statistical analysis of significant declining trend of sperm concentration over the said time period; and correlation of male health with the declining pattern of sperm count trend considering the age of an individual. With the development of more biomarkers to relate age with sperm concentrations and with upcoming studies investigating the causes of the decreasing quality of sperm parameters, greater knowledge could be developed to explore the possible remedies to overcome this expanding threat of infertility to the next generations.
Footnotes
Acknowledgements
The authors thank Dr. Sandeep Poddar, Research Manager, Lincoln University College, Malaysia, for his support in formulating this review. The authors are also thankful to him for his help in collecting information regarding data of sperm counts.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
