Abstract
This study aims to analyze the registration information and outcome transparency for five common andrological diseases, as well as the factors influencing result availability. A comprehensive search was performed on ClinicalTrials.gov and the International Clinical Trials Registry Platform (ICTRP) to retrieve all clinical trial registration data related to the five defined andrological diseases from the inception of these databases up to September 1, 2024. The search extracted key trial details, including status, type, intervention, and result availability. Of 8,132 trials retrieved, 642 were analyzed. Among these, 259 trials (40.34%) reported results via ClinicalTrials.gov, ICTRP, or publications, while 113 trials (17.60%) made results publicly available on ClinicalTrials.gov or ICTRP. Among the five andrological diseases, male infertility had the highest rate of result availability (37/74, 50%), whereas benign prostatic hyperplasia exhibited the lowest (71/190, 37.37%). No significant differences were found across diseases (χ2 = 3.722, df = 4, p = .435). Factors such as study status, blinding, interventions, center type, location, and duration significantly influenced result availability, whereas study type, stage, funding, outcome indicators, and sample size did not. Clinical trials on andrological conditions show major gaps in registration and result disclosure, with low reporting rates and prevalent non-reporting and selective reporting. Developed countries dominate trial registration and result disclosure while developing countries have limited participation. Trial characteristics also influence result disclosure rates. These challenges compromise the integrity and credibility of research data, impede clinical practice, and hinder the progress of medical research. Measures are needed to improve transparency, reduce selective reporting, and enhance the rigor and credibility of andrology research.
Introduction
Andrological diseases are characterized by common conditions such as erectile dysfunction (ED), chronic prostatitis (CP), benign prostatic hyperplasia (BPH), premature ejaculation (PE), and male infertility (MI). These conditions are associated with high morbidity rates, poorly understood mechanisms, and complex diagnostic and treatment processes, profoundly impacting men’s physical health, exacerbating psychological burdens, and reducing overall quality of life (Kiani et al., 2024). Furthermore, with increasing social pressures and an aging population, the incidence of these conditions has risen annually, evolving into a significant public health concern. Diagnostic and therapeutic strategies face substantial challenges, including insufficient etiological research, limited treatment options, and inconsistent quality of clinical evidence (Agarwal et al. 2015; Nicol & Chung, 2023; Wang et al., 2024; W. Zhang et al., 2019; J. Zhang et al., 2020). Overcoming these challenges requires the implementation of high-quality clinical studies that prioritize transparency and openness in reporting results. Clinical trial registration and result disclosure are not only essential components of scientific research but also critical for optimizing diagnostic and treatment strategies and advancing evidence-based medicine (Garg et al., 2023). Studies have shown that selective reporting in clinical trials is particularly prevalent in industry-sponsored trials (Siena et al. 2023). This issue can result in biased trial outcomes, mislead scientific research and clinical practice, and ultimately influence patients’ treatment decisions and health outcomes. The availability of clinical trial registry results has received insufficient attention in the field of andrology. This study aims to systematically evaluate the current status of clinical trial registration and result availability for common andrological conditions using the ClinicalTrials.gov and International Clinical Trials Registry Platform (ICTRP) databases. The goal is to provide a scientific foundation for identifying existing challenges and improving trial transparency.
ClinicalTrials.gov is a database established in 2000 by the National Library of Medicine and the U.S. Food and Drug Administration to promote transparency in clinical trials. It is the largest database of its kind, containing information on more than 500,000 clinical trials conducted in over 200 countries (Tse et al., 2018). In addition to ClinicalTrials.gov, the ICTRP, managed by the World Health Organization (WHO), provides information on clinical trials conducted across various countries and regions that adhere to WHO registration standards. The ICTRP consists of 18 clinical trial registration sub-platforms operating globally and complying with WHO criteria (Negoro et al., 2019). As the primary global databases for clinical trial registration, ClinicalTrials.gov and the ICTRP offer extensive coverage of registered trials, providing a robust foundation for researchers to access comprehensive and systematic trial information. This study aimed to record fundamental information, including registration site, study status, study type, intervention, and result reporting to databases or academic journals. The analysis involved searching ClinicalTrials.gov and ICTRP for clinical trials focused on five common andrological conditions: ED, CP, PE, BPH, and MI. The findings aim to contribute to improving trial transparency and optimizing treatment strategies.
Materials and Methods
Data Sources
In this study, ClinicalTrials.gov and ICTRP were utilized as data sources to retrieve information on five common andrological diseases: ED, CP, PE, BPH, and MI (Agarwal et al. 2015; Garg et al., 2023; Negoro et al., 2018; Siena et al., 2023; Tse et al., 2018). The search was performed using an advanced strategy that combined subject terms and free-text keywords, such as “ED,” “erectile failure,” and “impotence” for ED. The database search began on September 1, 2024, marking its inception. A single researcher independently executed the searches, strictly adhering to a predefined search strategy.
Inclusion and Exclusion Criteria
The following inclusion criteria were applied:(a) Adult males are diagnosed with one of the following five diseases: CP, ED, BPH, PE, or MI. (b) Patients may receive any of the following interventions or a combination thereof: (i) western medicine treatment, (ii) surgical treatment, (iii) dietary supplements, (iv) device therapy, (v) behavioral therapy, (vi) diagnostic testing, (vii) biologics, or (viii) other therapies. (c) Patients may also receive any of the following control measures or a combination thereof: (i) Western drug therapy, (ii) surgical treatment, (iii) dietary supplements, (iv) device therapy, (v) behavioral therapy, (vi) diagnostic tests, (vii) biological agents, or (viii) other therapies. (d) The study design may include the following types: (i) randomized controlled trials, (ii) cohort studies, (iii) cross-sectional studies, or (iv) case-control studies. (e) The study status of all registered clinical trials must indicate completion, suspension, or termination as of September 1, 2022, or lack of reported results. Consistent with previous studies, a 2-year follow-up period was allowed to ensure that investigators had sufficient time to disclose the results of clinical trials (Huang et al., 2023).
The following exclusion criteria were applied: (a) Trials that did not fall into one of the five categories of andrological diseases—CP, ED, BPH, PE, or MI; trials involving patients who had already undergone surgical treatment related to andrological diseases; trials involving pediatric patients aged under 18 years; trials that included female patients or patients of other genders; and trials involving patients with two or more concomitant andrological diseases. (b) Studies whose design did not involve humans. (c) Trials with no specific completion date. (d) Trials that were completed, suspended, terminated, or not reported on after September 1, 2022. (e) Clinical trials with a study status other than “completed,” “suspended,” “terminated,” or “not reported.”
Extraction and Evaluation of the Transparency of Results
In the present study, two personnel independently extracted information from registered clinical trials related to andrological diseases in a consecutive manner, following the inclusion and exclusion criteria. The extracted information included registration number, study name, study site, number of sites, year of registration, study status, trial type, phase, number of participants, duration, source of funding, blinding, intervention, type of endpoints, and whether results were submitted. The “Results: Yes or No” entry in the ClinicalTrials.gov and ICTRP databases was used to determine whether the trial had reported results in the database. Based on the basic information provided in the databases, registered trials were sequentially searched in Google Scholar, PubMed, and Web of Science using the trial number or study title to determine whether corresponding articles had been published. Key information, including study samples, interventions, and primary outcome measures, was then verified to ensure the accuracy and consistency of the search results.
Statistical Analysis
The present study employed bibliometric and comparative research methodologies to organize and analyze the data, utilizing Excel 2021 and SPSS 27.0 (IBM Corp., USA) software. A comprehensive statistical analysis was performed to evaluate the overall reporting rate of clinical trial results, the reporting rate by disease, and the reporting rate by year (Supplementary Material). In addition, a comparative analysis was conducted to examine the positive rate of trial results published in academic journals versus those available only in databases but not published. The analysis stratified trials based on the availability of results, categorizing all included trials into two groups. A detailed statistical analysis was then performed on various influencing factors, including study status, study type, interventions, and trial phase, among others. Significant differences in trial characteristics between the two groups were assessed using univariate analysis, followed by multifactorial logistic regression to identify characteristics independently associated with the availability of clinical trial results. Results were expressed as effect sizes using odds ratios (OR) and their 95% confidence intervals (CI). Categorical variables were presented as frequencies and percentages and analyzed using Pearson’s χ2 test, while continuous variables were expressed as medians and interquartile ranges and compared using the non-parametric Mann-Whitney test. All statistical tests were two-sided, and differences were considered statistically significant at p ≤ .05.
Results
Search Results
A total of 8,132 clinical trials were identified in the initial search. After screening, the following exclusions were made: 4,087 were duplicates, 1,533 lacked a specific completion date, 926 did not meet the inclusion criteria for study status, 722 were completed after September 1, 2022, 170 involved females or minors, and 52 did not specifically focus on andrological diseases. Ultimately, 642 trials were included in the statistical analysis, consisting of 454 completed trials, 36 suspended or terminated trials, and 152 trials with an unreported status (Figure 1).
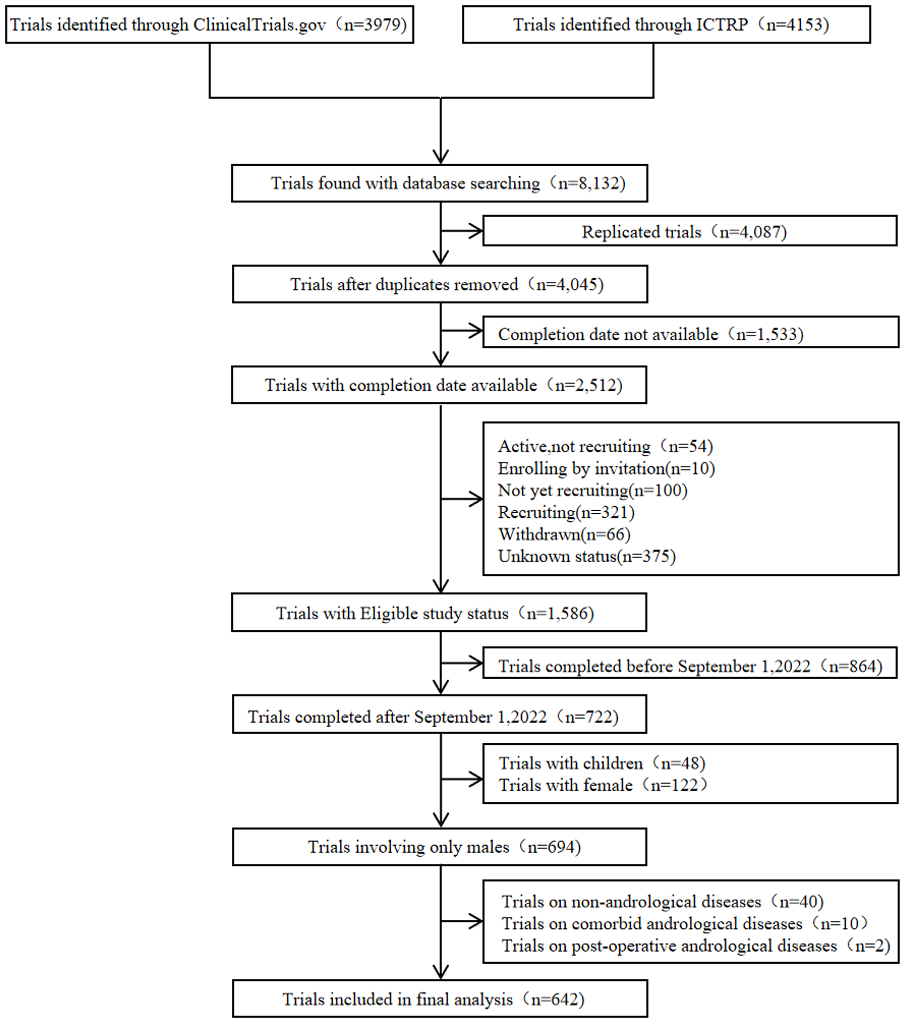
Flow Diagram of Clinical Trials Selection
Results Reporting Rate
Overall Results Reporting Rate
At the overall level, study results were available for 259 out of 642 trials. Of these, 44 trials (44/259, 16.99%) disclosed results on both the ClinicalTrials.gov and ICTRP platforms and were also publicly available in academic journals. In addition, 69 trials (69/259, 26.64%) disclosed results on either the ClinicalTrials.gov or ICTRP platform, while 146 trials (146/259, 56.37%) were publicly published in academic journals. The majority of clinical trials (336 out of 642, 52.34%) focused on ED. By contrast, the number of trials related to BPH and MI was significantly lower, with 17 trials (2.65%) and 25 trials (3.89%), respectively. Regarding the availability of outcomes, the highest rate was observed in trials on MI (37/74, 50%), while the lowest rate was recorded in trials on BPH (71/190, 37.37%). The difference in outcome availability between these five andrological diseases was not statistically significant (χ2 = 3.722, df = 4, p = .435) (Figure 2). The number of clinical trial registrations has steadily increased and stabilized in recent years, with the highest number of registrations recorded in 2019 (65 trials). The overall trend in the results availability rate showed a gradual increase, peaking in 2009 (18/25, 72.00%), followed by a slight decline and subsequent stabilization (Figure 3). Regarding the selective publication tendency of trial results, positive trial results were predominantly published in academic journals (159/190, 83.68%), which was significantly higher than the positive rate of accessible but unpublished trials in databases (35/62, 56.45%; χ2 = 19.56, df = 1, p < .001).

Histogram of Outcome Availability for Five Andrological Diseases

Trend Chart of the Number of Clinical Registrations and the Availability of Results for Andrological Diseases
The global distribution of clinical trial registrations for andrological diseases is highly concentrated, with the United States leading with 120 registrations, followed by Japan (62), South Korea (36), and China (29). Regional analysis reveals that most trial registrations in North America are concentrated in the United States (120), significantly outpacing other countries such as Canada (21), which, despite its smaller number of registrations, still makes a notable contribution to the region. In East and Southeast Asia, East Asia stands out, with Japan (62), South Korea (36), and China (29) as the primary contributors. In addition, countries such as India (15), Israel (10), and Singapore (6) contribute meaningfully to regional studies despite their relatively smaller numbers. In Europe, trial registrations are more dispersed, showing a pattern of multi-point contributions. Italy (41) and Germany (23) are the primary contributors, followed by France (11) and the United Kingdom (10). Notably, Switzerland, Spain, and Sweden each registered 11 trials, reflecting a balanced distribution. By contrast, Africa is significantly underrepresented, with only 15 trial registrations across the entire region. In Latin America, Brazil (21) leads, followed by Mexico (7) and Argentina (3), indicating limited research activity in the region. Furthermore, 66 trials did not report the location of their study sites (Figure 4(A)). The registration of clinical trials for andrological diseases demonstrates a significant global distribution but with marked regional concentrations. The United States again leads with 120 registered trials, followed by Japan (63), India (52), South Korea (42), Italy (41), the United Kingdom (40), Germany (37), and China (30). These figures highlight the strong participation of large, populous countries in this field. Conversely, some countries, such as Latvia, Lithuania, Nigeria, and Kazakhstan, recorded only 1–2 registrations. Geographically, North America stands out for its high number of registrations, with the United States far surpassing other countries, including Canada. Europe has been active in men’s health research, with notable contributions from Italy, the United Kingdom, Germany, France (40), and Spain (29). In Asia, Japan, South Korea, China, and India are the main contributors, while countries such as Thailand (11) and Israel (10) also participated. Africa had low participation, with only Nigeria, South Africa, and a few other regions contributing a total of nine registrations. In South America, Brazil (18) and Argentina (11) were the primary contributors, reflecting limited research activity in the region. In Oceania, Australia (11) was the major participant, with only one registration from New Zealand (Figure 4(B)).

Heat Map of the Geographic Distribution of the Number of Clinical Registrations for Andrological Diseases. A: Heat Map of the Geographic Distribution Based on the Country of Registration, B: Heat Map of the Geographic Distribution Based on All Trial Center Countries.
Outcome Reporting Rates Across Different Trial Characteristics
Specifically, based on the availability of trial results, all trials were categorized into two groups: the “results available group” and the “results unavailable group.” The differences between these two groups were systematically compared using one-way analysis, focusing on influencing factors such as study status, type of study, interventions, and phases of the study. In terms of study status, trials categorized as “completed” (204/454, 44.93%) were significantly more likely to disclose results compared to those classified as “unreported” (42/152, 27.63%) (p < .01). Regarding the type of blinding, “unreported” trials (12/81, 14.81%) were significantly less likely to report results (p < .001). For the number of study centers, trials with unknown study centers reported results less frequently (p < .0001) compared to single-center trials (105/238, 44.12%) or multi-center trials (102/214, 47.66%). Geographically, clinical trials conducted in the United States (65/120, 54.17%) were more likely to disclose results (χ2 = 10.92, p < .01). In addition, trials with longer duration were significantly more likely to disclose results (U = 15,489.5, 95% CI [Median range 10, 36 vs. 7, 25], p < .001). No significant differences in the availability of trial results were observed with respect to study type, intervention, phases of the study, funding source, type of clinical endpoint, or the number of participants in the trial (Table 1).
Availability of Results for the Included Trials

Abbreviation: NA = Not Applicable; NR = Not Reported.
Factors Influencing the Usability of Results
Based on the availability of trial results, all trials were categorized into two groups: the “results available group” and the “results unavailable group.” Influencing factors, such as study status, type of study, intervention, and phases of the study, were further examined using multi-factor logistic regression analysis. In terms of study status, the availability of results was significantly lower in “unreported” trials compared to “completed” trials (OR = 0.46, 95% CI [0.30, 0.70], p < .001). Regarding the type of blinding, results were significantly less available in “unreported” trials compared to “open-label” trials (OR = 0.16, [0.08, 0.33], p < .001). For interventions, trials using devices demonstrated a higher availability of results compared to those using Western medicines (OR = 1.63, [1.00, 2.65], p = .05). With respect to the number of study centers, trials with “unknown” study centers had significantly lower result availability compared to single-center trials (OR = 0.30, [0.20, 0.46], p < .01). Geographically, trials conducted in the United States showed significantly higher result availability compared to those conducted elsewhere (OR = 2.12, [1.36, 3.31], p < .001). Similarly, trials conducted in Europe exhibited higher result availability (OR = 1.57, [1.02, 2.41], p = .04) (Table 2).
Multifactorial Logistic Regression Analysis of Factors Influencing the Availability of Clinical Trial Results in Andrological Diseases

Discussion
In this study, 72.74% (467/642) of the clinical trials included in the analysis were registered on ClinicalTrials.gov, while 27.26% (175/642) were registered on the ICTRP. Both ClinicalTrials.gov and ICTRP are significant global clinical trial registration platforms supported by governing bodies that have established relevant laws and regulations on trial registration and results disclosure. Further improvements to the implementation process are needed. A key concern is the absence of clear legislation and regulations on trial results disclosure in certain sub-platforms across some countries or regions. This lack of enforcement weakens the binding force of such disclosures, directly affecting the transparency of registered trial results. In response to ClinicalTrials.gov, the United States passed the Food and Drug Administration Amendments Act (FDAAA) in 2007, a landmark piece of legislation aimed at improving transparency in clinical trials globally. Section 801 of the Act specifically mandates the registration and results disclosure of Applicable Clinical Trials (ACTs) conducted in the U.S. It requires that all ACTs be registered on ClinicalTrials.gov, with results disclosed within 12 months of trial completion (U.S. Congress, 2007). The disclosed results must include primary and secondary endpoints as well as fundamental subject characteristics. The introduction of this law marked the first instance in which the United States implemented mandatory regulations to comprehensively standardize the registration and results disclosure of clinical trials, addressing the issues of opaque trial information, selective reporting, and publication bias prevalent at the time. The implementation of FDAAA Section 801 is highly significant. First, it reduces the possibility of selective reporting and publication bias through mandatory registration and disclosure, thereby improving the transparency and credibility of clinical research. Second, it protects the public interest by ensuring that patients and clinicians have access to comprehensive and accurate trial information, supporting more scientific treatment decisions. Furthermore, it promotes research quality by encouraging researchers to design trials more rigorously and take greater responsibility for their results. Despite these clear legal requirements, significant compliance issues remain. Some investigators fail to register or disclose trial results in a timely manner, and the lack of regulatory oversight further diminishes the practical impact of the law (DeVito et al., 2020). By contrast, the ICTRP functions more as an advocate for transparency and ethical responsibility rather than as a platform enforcing mandatory legal requirements, such as those outlined in FDAAA Section 801. The necessity for mandatory reporting of trial results depends on the specific legal framework of the country or region where the trial is conducted. For example, The United States, through FDAAA Section 801, and the European Union, through Regulation (EU) No 536/2014, have established clear legal mandates for the registration and disclosure of clinical trial results (European Parliament and Council, 2014; U.S. Congress, 2007). Conversely, countries like China rely more on researchers’ ethical awareness and voluntary compliance, lacking mandatory regulations for trial transparency. This discrepancy highlights the uneven global standardization of clinical trial transparency management. While the 2007 legislation established a foundation for clinical trial registration and disclosure, challenges related to implementation and regulatory enforcement remain significant issues that require global attention. There is an urgent need for the international community to promote uniform standards and regulatory frameworks to ensure greater transparency and accountability in clinical research worldwide.
This study comprehensively analyzed the outcome reporting rates of clinical trials on andrological diseases and investigated the effects of various trial characteristics, including study status, study type, intervention, and study phase, on reporting rates. Several key findings emerged. The aggregate reporting rate of trial results on the ClinicalTrials.gov and ICTRP platforms was extremely low at 17.60%. Even when results from peer-reviewed journals were included, the rate increased only marginally to 40.34%, indicating that approximately three-fifths of the included trials did not report their results. Further analysis revealed significant publication bias, with a higher proportion of positive trials (159/190, 83.68%) publishing results in peer-reviewed journals compared to trials with results available in the database but not published (35/62, 56.45%). In addition, 63.89% of terminated or suspended trials failed to report results. While clinical trials in andrological diseases showed no significant differences in reporting rates based on study type, intervention, phases of the study, funding source, type of clinical endpoint, or number of participants, the overall reporting rate was significantly lower than in other disease areas. For example, a study by Postka et al. reported that 56% of 1,429 clinical trials in heart failure were published (Psotka et al., 2020). Similarly, Ren-Qian Huang et al. found that 73.13% of 856 clinical trials in pancreatic cancer disclosed results (Huang et al., 2023), and Shashank Garg et al. reported that 75.09% of 2,429 clinical trials in gastroenterology disclosed results (Garg et al., 2022). Furthermore, a study by DeVito et al. of 4,209 trials from 2018 to 2019 found that while only 40% of investigators reported results within 1 year of trial completion, 64% eventually disclosed results during the study period (DeVito et al., 2020). Consistent findings have been observed in other studies (Becker et al., 2014; Prayle et al., 2012; Ross et al., 2009, 2012; Saito & Gill, 2014). Despite variability in reporting rates across clinical trial domains, such as 22.8% for joint replacement trials (Smith et al., 2012), 26.9% for urology trials (Magnani et al., 2021), and 43.2% for orthopedic trauma trials (Gandhi et al., 2011), the reporting of results in andrological diseases remains suboptimal, underscoring the need for substantial improvement. The lack of outcome reporting has several important consequences. First, it can lead to publication bias, resulting in an overestimation of positive outcomes in the literature, which compromises the accuracy of systematic reviews and meta-analyses. Second, incomplete data prevent researchers from fully understanding the true effects of treatments, thereby affecting the design and implementation of subsequent studies. In clinical practice, selective reporting may mislead physicians’ therapeutic decisions and cause patients to accept treatments based on incomplete or biased information. In addition, underreporting of adverse events can lead to an underestimation of treatment risks, compromising patient safety. Beyond its impact on research and clinical practice, selective reporting also erodes patient and public trust. Failing to report trial results can be perceived as a lack of respect for patient participation, undermining trust in research and the healthcare system. This erosion of trust may hinder treatment progress, impede scientific innovation, and negatively affect the long-term development of the healthcare system. There is a compelling need to enhance the disclosure of trial results in andrological diseases to improve research transparency, promote scientific progress, and safeguard public trust.
The global distribution of clinical trial registrations is highly concentrated, with the United States leading in the number of registrations. The East Asian region demonstrates notable activity in andrological diseases research, the European region exhibits a more balanced and dispersed distribution, while the African and Latin American regions show a paucity of research activity. The geographical distribution of clinical trial registrations in andrological diseases worldwide is characterized by a pattern of “domination by developed countries and insufficiency by developing countries” (Figure 4(A)). This distribution may be closely linked to factors such as economic development, allocation of research resources, policy support, and the prioritization of disease research. In the future, enhancing international cooperation and optimizing resource allocation, particularly in regions with low trial registration activity, could help promote more balanced global development in andrological disease research. The global and regional distribution of trial registrations closely mirrors the trends observed in Figure 4(A) while further highlighting significant imbalances. There is a notable concentration of trial registrations in a small number of developed countries and research powerhouses, reflecting the current distribution of global research resources and the limited participation of developing countries in this field (Figure 4(B)). To address these disparities, future efforts should focus on fostering international collaboration and improving resource sharing. Such initiatives could facilitate more equitable development in global andrological disease research and strengthen the participation of underrepresented regions.
Univariate and multivariate logistic regression analyses of outcome reporting rates for different trial characteristics revealed several significant findings. Trials involving devices as interventions, those conducted at sites located in the United States and Europe, and trials with longer durations were significantly more likely to report outcomes. The low rate of outcome reporting for terminated or suspended trials remains a concern. The failure to report results from these trials can hinder the progress of medical science, as understanding the reasons for trial failures is essential for avoiding repeated mistakes and optimizing the design of future studies.
The present study aimed to systematically assess the transparency of registration information and results of clinical trials in andrological diseases. Several limitations and issues in the study design and implementation warrant further exploration. This paper highlights these issues to facilitate discussion among colleagues, provide a reference for future studies, and promote improvements in clinical trial transparency in andrological diseases. Unlike some previous studies focusing on clinical trial transparency (Garg et al., 2022; Psotka et al., 2020), this study included trials with a status of “not reported.” This decision was made to provide a more comprehensive assessment of enrollment rates, particularly given the high proportion of trials classified as “not reported” (152 out of 642, 23.67%). This approach offers a more realistic picture of the overall trial enrollment situation and may also introduce bias due to incomplete data, potentially affecting the interpretation of results. It is further hypothesized that a significant proportion of trials classified as “not reported” reflect delayed or incomplete data entry by researchers on registration platforms. Moving forward, it is anticipated that researchers will place greater emphasis on updating and managing trial information on these platforms to enhance data integrity and transparency. Second, despite thorough searches of multiple databases and independent verification of trials with unavailable results by another researcher, it is possible that some published articles were missed because certain journals were not included in the three databases used. This limitation highlights the need for future studies to broaden their search criteria to comprehensively capture trial results. Similarly, while this study relied on data from ClinicalTrials.gov and ICTRP, which provide broad coverage, relevant trials may still have been missed. In addition, this study relied on publicly available data and did not explore in depth the reasons for the unavailability of trial results. Factors such as ethical review processes, insufficient research funding, or the subjective willingness of investigators, may contribute to this issue and warrant further investigation. Furthermore, the study did not adequately account for the impact of differences in laws, regulations, cultural backgrounds, and research environments across countries and regions on trial registration and results disclosure. Future studies should aim to integrate a wider range of data sources and adopt a multidimensional analytical approach to better understand the underlying causes affecting the transparency of clinical trials in andrological diseases.
The transparency of clinical trial registration in andrological diseases is evidently inadequate. Improving clinical trial transparency is a complex and protracted process that cannot be achieved immediately and requires concerted efforts from multiple perspectives and at various levels. At the researcher level, it is crucial to strengthen awareness of the importance of trial registration and results disclosure, ensuring a full understanding of and strict compliance with relevant laws and regulations, such as FDAAA Clause 801, Regulation (EU) No 536/2014, and other country-specific requirements. In addition to providing technical support and incentives, greater emphasis should be placed on promoting ethical conduct in research. At the policy and regulatory level, it is necessary to improve relevant laws and regulations, clarify mandatory requirements for trial registration and results disclosure, and strengthen supervision and enforcement to ensure compliance. At the patient level, active participation in trials should be encouraged by fostering trust and willingness through transparent information disclosure. Public education initiatives should also be implemented to raise awareness of clinical trials, using science popularization and outreach efforts to foster social concern and promote public supervision of trial transparency. The enhancement of clinical trial transparency can only be progressively achieved through collective and coordinated efforts across all levels.
Supplemental Material
sj-xlsx-1-jmh-10.1177_15579883251325478 – Supplemental material for Analysis of Current Status of Clinical Trial Registrations in Andrological Diseases: Insights from ClinicalTrials.gov and ICTRP Databases
Supplemental material, sj-xlsx-1-jmh-10.1177_15579883251325478 for Analysis of Current Status of Clinical Trial Registrations in Andrological Diseases: Insights from ClinicalTrials.gov and ICTRP Databases by Li-Xing Lei, Ke-Cheng Li, Wen-Hao Yu, Jun-Long Feng, Hua-Nan Zhang, Mao-Ke Chen, Wen-Xuan Dong, Ji-Xuan Chen, Jie Li, Ji-Sheng Wang, Liang Han and Bin Wang in American Journal of Men's Health
Footnotes
Acknowledgements
All authors have made significant contributions to this study. As this paper reaches completion, I would like to express my heartfelt gratitude and extend my best wishes to everyone who has supported and guided me throughout this research and learning journey. Furthermore, this study utilized data from the ClinicalTrials.gov and ICTRP databases. We are sincerely grateful to these platforms for providing open and transparent clinical trial registration information and results data. These databases have been instrumental in facilitating the successful completion of this study and have significantly contributed to the transparency and standardization of global clinical research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Pilot Program for Upgrading Clinical Research and Achievement Translation Capabilities of Dongzhimen Hospital, Beijing University of Chinese Medicine (DZMG-QNZX-24012).
Ethical Statement
An ethics statement is not applicable because this study is based exclusively on the public database.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
