Abstract
Chronic prostatitis (CNP) is a prevalent inflammatory disorder among men. The Xialiqi capsule has been reported to alleviate the clinical symptoms of CNP patients, which may be related to its anti-inflammatory effect; yet, its exact mechanism of action remains unclear. In this study, human normal prostate epithelial cells (RWPE-2 cells) were categorized into a control group, a model group, an inhibitor group, along with high, medium, and low drug-containing serum groups (5%, 10%, and 15%, respectively). With the exception of the control group, cell pyroptosis models were created by stimulating with lipopolysaccharide (100 ng/mL) and adenosine triphosphate (5 mM). Subsequently, drug-containing serum and the NOD-like receptor 3 (NLRP3) inhibitor (MCC950) were utilized to intervene with the model cells according to their respective groups. Post-administration of MCC950 and drug-containing serum, an improvement in cell viability was noted in the inhibitor group and medium-high dosage groups (by 20.5%, 38.2%, and 73.2%). Transmission electron microscopy indicated a reduction in the features characteristic of cell pyroptosis. Levels of nitric oxide, interleukin-18 (IL-18), and tumor necrosis factor-α in the cellular supernatant decreased significantly (60.7%, 21.6%, 33.7%, 41.8%; 49.2%, 54.8%, 53.5%, 69.3%; 31.3%, 44.4%, 38.1%, 51.2%). Immunofluorescence showed reduced fluorescence intensity of NLRP3 and Cysteine aspartate protease-1 (Caspase-1) proteins, and Western Blot analysis revealed a significant decline in the expression of NLRP3, pro-Caspase-1, and gasdermin D (20.5%, 45.9%, 58.1%, 74.8%; 23.2%, 36.7%, 51.6%, 51.9%; 15.4%, 28.6%, 33.1%, 39.2%). In vitro experiments suggest that the Xialiqi capsule may treat CNP by regulating prostate epithelial cell pyroptosis and reducing inflammatory factor release via inhibiting the NLRP3/Caspase-1 signaling pathway. This study offers a novel approach for future CNP treatment with traditional Chinese medicine preparations, deserving further promotion.
Introduction
Chronic prostatitis (CNP) is a condition characterized by the persistent inflammation of prostate tissue, stemming from diverse etiological factors. The prostatitis is categorized into four distinct types by the National Institutes of Health (NIH) in the United States. Chronic non-bacterial prostatitis/chronic pelvic pain syndrome, also referred to as Type III CNP, accounts for more than 90% of prostatitis cases (Mi et al., 2012; Schaeffer et al., 2002). Type III CNP is characterized by a spectrum of symptoms, a protracted course, and challenging treatment, with pain being the primary clinical manifestation of type III CNP. This pain is a significant contributor to the decreased quality of life experienced by patients, persisting chronically and proving resistant to treatment (Zhang et al., 2016). Presently, the deficiency of effective treatment options in western medicine has emerged as a major issue of concern for public health in contemporary society. Consequently, the primary focus of current research is to effectively relieve the symptoms related to CNP, which presents a formidable challenge.
The Xialiqi capsule, previously known as the Qianlieshu capsule, is a commonly used medication for the management of moderate-to-severe prostatic hyperplasia. Similarly, previous clinical studies have demonstrated the remarkable capacity of the Xialiqi capsule to considerably improve the clinical manifestations experienced by individuals with CNP (Wang et al., 2020, 2021). Animal experiments have demonstrated that the administration of the Xialiqi capsule effectively inhibits the pathophysiological alterations observed in rats with CNP models. This treatment results in a reduction in the number of white blood cells found in the prostate fluid, restrains the proliferation of bacterial colonies, and curtails the release of pro-inflammatory cytokines in the rat’s prostate (Meng, 2018). In recent years, the role of pyroptosis-mediated inflammation in the pathogenesis of various diseases has gained significant attention from experts and scholars. Classical pyroptosis triggers the activation of Cysteine aspartate protease-1 (Caspase-1) via NOD-like receptor 3 (NLRP3) inflammasomes. Once activated, Caspase-1 cleaves the gasdermin D (GSDMD) protein. The cleaved GSDMD protein then releases its N-terminal domain, which can bind to the cell membrane and form pores in the cell membrane, ultimately resulting in the occurrence of pyroptosis (Shi et al., 2017). This process results in the release of numerous intracellular pro-inflammatory cytokines into the extracellular milieu, thereby triggering a severe inflammatory response across various body systems (Ketelut-Carneiro and Fitzgerald, 2022). The findings revealed that the expression levels of NLRP3 and Caspase-1 in the prostate tissue of CNP model rats was significantly higher compared to those of blank control rats, suggesting that NLRP3 inflammasome can be activated within the prostate tissue of CNP rats (Lu et al., 2019). The NLRP3 inflammasome is capable of mediating the inflammatory response in the prostate. This results in an elevation of prostatic inflammatory factors, subjecting the prostate to a prolonged inflammatory environment, which in turn exacerbates the damage to prostate tissue. Inhibition of NLRP3 inflammasome can effectively mitigate prostate inflammation (He et al., 2021). Consequently, the regulation of NLRP3 pathway holds promise as a novel therapeutic target for CNP treatment. Currently, only a limited number of studies have delved into the anti-pyroptosis effect of the Xialiqi capsule and other Chinese herbal preparations on prostate epithelial cells. Given its well-established clinical efficacy, this study specifically aimed to verify, through in vitro experiments, whether the Xialiqi capsule could regulate the pyroptosis of prostate epithelial cells. This would fulfill the role of treating CNP and offer a novel perspective for the treatment of CNP using traditional Chinese medicine.
Materials
Reagents
RWPE-2 cells (iCell-h185, Sebacon); Trypsin-EDTA Solution (T1300, Solarbio); FBS (10099-141, Gibco); 1 × PBS (0.01 M, pH7.4) (KGB5001, KEYGEN BIOTECH); RWPE-2 special medium (iCell-h185-001b, Sebacon); NLRP3 inhibitor (HY-12815A, MCE); CCK8 cell proliferation detection kit (KGA317, KEYGEN BIOTECH); nitric oxide (NO) colorimetric test kit(E-BC-K135-M, Elabscience); TNF-α kit (E-EL-H0109c, Elabscience); IL-18 kit (E-EL-H0253c, Elabscience); lipopolysaccharide (LPS; HY-D1056, MCE); adenosine triphosphate (ATP; HY-B2176, MCE); Tissue cell fixative solution (P1110, Solarbio); BSA solution (A8020, Solarbio); cy3 (AS007, ABclonal, 1/200); DAPI (KGA215-50, KEYGEN BIOTECH); BCA protein quantitative kit (E-BC-K318-M, Elabscience); Supersensitive Luminescent Liquid (RJ239676, ThermoFisher); Mouse Anti-β-Actin (HC201; TransGen Biotech, 1/2000); HRP conjugated Goat Anti-Mouse IgG (H + L) (GB23301, Servicebio, 1/2000); Rabbit Anti NLRP3 (Ab263899, Abcam, 1/1000); Rabbit Anti Pro-Caspase-1 (Ab179515, Abcam, 1/1000); Rabbit Anti Cleaved-Caspase-1 (AF4005, Affinity, 1/500); HRP conjugated Goat Anti-Rabbit IgG (H + L) (GB23303, Servicebio, 1/2000).
Instruments
CO2 Incubator (BPN-80CW, Shanghai Yiheng Scientific Instrument Co., LTD., Shanghai,China); Inverted Fluorescence Microscope (MF53, Guangzhou Mingmei Optoelectronics Co., LTD., Guangzhou,China); Clean table (BBS-SDC, BIOBASE); Medical centrifuge (TD4A, Changsha Yingtai Instrument Co., LTD., Changsha,China); Multifunctional Enzyme Label Analyzer (SuPerMax3100, Shanghai Flash Spectrum Biotechnology Co., LTD., Shanghai,China); Ultra High Sensitivity Chemiluminescence Imaging System (Tanon-5200, Shanghai Tianneng Technology Co., LTD., Shanghai,China).
Methods
Preparation of Drug Serum
To rule out the non-specific effects of the Xialiqi capsule extract directly on cell culture, we specially selected the drug-containing serum of the Xialiqi capsule for cell intervention experiments. This approach aimed to mimic the specific action process of the active components of the drug following their absorption into the bloodstream and subsequent interaction with prostate epithelial cells. A total of 12 male Sprague-Dawley rats, aged 49 days, obtained from SiPeiFu (Beijing) Biotechnology Co., LTD. (license number: SCXK [Beijing] 2019-0010), were divided into two groups: a blank control group and a drug-containing serum group. The rats were housed in an environment with a temperature ranging from 20°C to 26°C and the humidity ranging from 40% to 70%. After an adaptation period, the rats in the drug-containing serum group were given an intragastric dose of 0.36 g/kg/d, while the control group received an equal volume of normal saline once a day for a continuous period of seven consecutive days. The rats were euthanized 1 hr after the final administration on the seventh day, and blood was subsequently collected via the abdominal aorta. The collected blood was allowed to stand for a period of 1 to 2 hr, followed by centrifugation at a speed of 3,000 revolutions per min for 15 min. The supernatant, which appeared as a yellowish liquid, was carefully extracted using a pipetted to obtain the serum. When employed, the Dulbecco's Modified Eagle Medium(DMEM) was appropriately diluted to achieve the desired concentration. This study was reviewed by the Animal Ethics Committee of the fourth Clinical Medical College of Guangzhou University of Chinese Medicine (Ethics number: K2023-112-05).
Cell Culture
RWPE-2 cells were placed in serum-free keratinocyte medium and cultured in an incubator containing 5% CO2 at 37°C, and then passed when the cell growth and fusion reached about 80% to 90%.
Cell Models
It has been reported that stimulation with LPS and ATP can induce the activation of the NLRP3 inflammasome, which leads to Caspase-1/GSDMD-mediated pyroptosis (Lv et al., 2021). According to the literature reports (Gao et al., 2022), a cell-damage model was established by stimulating RWPE-2 with 100 ng/mL LPS for 4 hr, followed by a 30-min stimulation with 5 mM ATP. We can verify the occurrence of pyroptosis by multidimensional and multiple detection methods. After model establishment, we can observe the expression levels of NLRP3 and Caspase-1 proteins in the model group, detect the levels of interleukin-18 (IL-18) and other inflammatory factors, and use transmission electron microscopy to observe specific morphological changes such as cell-membrane rupture and membrane-pore formation to verify the success of model.
CCK-8 Detection of the Concentration of NLRP3 Inhibitor
RWPE-2 cells were then seeded into 96-well plates at a density of 8 × 103 cells per well and 5 wells per group. To investigate the effects of the NLRP3 inhibitor (MCC950), a 10 mM stock solution was prepared by dissolving MCC950 in Dimethyl Sulfoxide(DMSO). The cells were treated with various concentrations of MCC950 (0, 0.01, 0.1, 1.0, 10 μM) for 24 hr. The appropriate treatment concentration was determined using the CCK-8 method. The cells in 96-well plate cells to be tested were replaced with the same medium, with 100 μL per well, and 10 μL of CCK-8 reagent was added to each well. The plate was then incubated in an incubator for 2 hr. The absorbance value of each well at a wavelength of 450 nm was measured using a microplate reader.
CCK-8 Detection of Cell Viability
RWPE-2 cells were seeded into 96-well plates at a density of 8 × 103 cells per well and 5 wells per group. After a 4-hr treatment with LPS, ATP was subsequently added for 30 min to establish the model. Subsequently, following model establishment, the MCC950 and drug-containing serum were added to the experimental groups. The cells were then incubated for 24 hr. The appropriate treatment concentration was determined using the CCK8 method. The medium in the 96-well plate was replaced with the same medium, at a volume of 100 μL per well. Then, 10 μL of CCK-8 reagent was added to each well, and the plate was incubated in an incubator for 2 hr. The absorbance value of each well at a wavelength of 450 nm was measured using a microplate reader.
Transmission Electron Microscope Detection
The cells adhered to the coverslip were fixed with a 2.5% glutaraldehyde solution for 2 hr. Subsequently, the cells underwent dehydration using a gradient ethanol solution, followed by embedding, curing, slicing, and staining with a 3% uranium acetate and lead citrate solution. The resulting samples were then examined and imaged using transmission electron microscopy, 3 holes per group and 5 × 105 cells per hole.
Biochemical Detection of NO Level
After collecting the liquid, centrifugation was performed at 2°C to 8°C, 1,000 × g for 20 min to remove impurities and cellular debris. The supernatant was then collected for analysis. Following the instructions provided with the NO colorimetric test kit, the optical density (OD) values of each well were measured at 530 nm using an enzyme-labeled instrument, 3 holes per group.
Elisa Detection of TNF-α and IL-18 Level
After collection the liquid, it was subjected to centrifugation at a temperature of 2°C to 8°C and a speed of 1,000 × g for 20 min to remove impurities and cellular debris. The resulting supernatant was then used for analysis. The OD values of each well were measured at a wavelength of 450 nm using a microplate reader, following the instructions provided by the Tumor Necrosis Factor-α (TNF-α) kit and IL-18 kit, 3 holes per group.
Immunofluorescence Detection of NLRP3 and Caspase-1 Proteins
For cell fixation, a 4% histopathological cell fixation solution was employed for a duration of 15 min. This was followed by treatment with a 5% bovine serum albumin (BSA) solution at 37°C for 30 min. The diluted primary antibodies (NLRP3 and Caspase-1) were incubated overnight at 4°C. The next day, after a 45-min re-warming period, the diluted fluorescent secondary antibody, Cy3, was introduced and incubated at 37°C for 30 min. Subsequently, 4',6-Diamidino-2-phenylindole(DAPI) was added and incubated in the dark for 3 min. Finally, the Petri dish was sealed with a 50% glycerol solution, 3 holes per group, and each hole had 3 slides taken.
Western Blot Detection of the NLRP3, Pro-Caspase-1, and GSDMD Protein Expression
The cells were gathered, and the medium was discarded. The total protein was extracted using Radio-Immunoprecipitation Assay(RIPA) lysis buffer. Subsequently, the extracted protein was subjected to high-speed centrifugation at 12,000 rpm in a centrifuge set at 4°C for 10 min. The resulting supernatant was collected, and the total protein content was quantified using the BCA protein assay kit. Following denaturation, the protein samples were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) for 1.5 hr, followed by transfer to a membrane at a constant current of 300 mA for 1 hr. Subsequently, the Poly(vinylidene fluori) membrane (Millipore) was then blocked with skim milk powder, and the primary antibody was incubated overnight at 4°C. The next day, the secondary antibody was incubated at room temperature for 2 hr. The PVDF membrane was then immersed in a supersensitive luminescent liquid and positioned within an ultrasensitive chemiluminescence imaging system. As an internal reference, β-Actin was used to calculate the relative expression level of target protein, 3 holes per group, and each hole was tested independently for three times.
Statistical Analysis
The GraphPad Prism 9.0.0 software (GraphPad Software Inc.,California,USA) was used for statistical analysis and graphing. Quantitative results were expressed as mean ± standard deviation (
Results
CCK-8 Method Screens the Optimal Concentration of NLRP3 Inhibitors
RWPE-2 cells were cultured and exposed to various concentrations of MCC950 (0, 0.01, 0.1, 1, 10 μM). The optimal dose concentration was determined at 24 hr using the CCK8 assay. The results indicated that 1 μM of MCC950 significantly enhanced cell proliferation (130 ± 8.86) compared to the 0 μM concentration (100 ± 10.31). This concentration was subsequently chosen for further experiments. Please refer to Figure 1 for more detailed information.
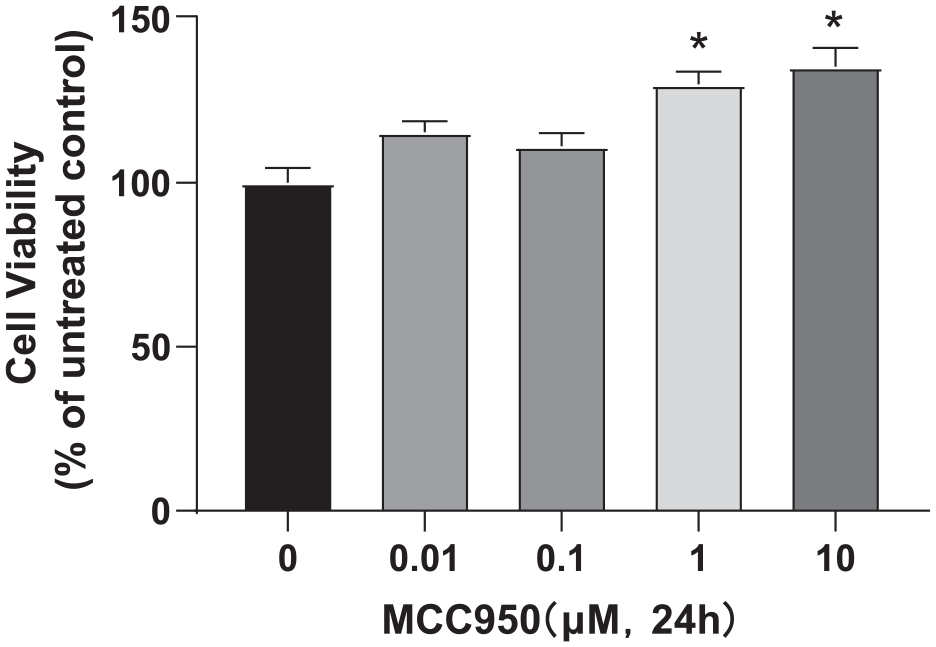
The optimal dose concentration of MCC950 was measured using the CCK-8 assay. Data are expressed as the mean ± SEM. Multiple comparisons were conducted using one-way ANOVA, and Tukey’s test was used for pairwise comparison. Differences with p < .05 were considered statistically significant
CCK-8 Analysis of Cell Viability
According to the CCK-8 test results, compared with the control group (100 ± 6.11), the cell vitality of the model group was decreased (52.21 ± 2.91, p < .05). However, the cell vitality was restored after the intervention of MCC950 (62.91 ± 3.12, p < .05). Furthermore, the application of 10% and 15% Chinese medicated serum resulted in a significant enhancement of cell vitality and promotion of cell proliferation (72.14 ± 4.09, 90.44 ± 6.30, p < .05). Detailed information can be found in Figure 2.

Cell viability of each group was detected using the CCK-8 method. Data are expressed as the mean ± SEM. Multiple comparisons analysis was conducted using one-way ANOVA, and Tukey’s test was used for pairwise comparison. Differences with p < .05 were considered statistically significant
Cellular Ultrastructure Utilizing Transmission Electron Microscopy
In RWPE-2 cells, transmission electron microscopy findings indicated that the ultrastructure of cells in the control group exhibited normal characteristics, including intact cell membranes, abundant organelles, and a large number of mitochondria. Conversely, the model group displayed disorganization, cellular swelling, reduced organelles, compromised cell membranes, the formation of pyroptosis cells, atrophied mitochondria, and abnormal morphology, as compared to the control group. The inhibitor group exhibited a relatively normal state, characterized by mild cell swelling, abundant organelles, intact cell membranes, and an increased number of mitochondria, with a small number of autophagosomes observed following the administration of serum containing a Chinese medicine drug at concentrations of 5%, 10%, and 15%; improvements were noted in cell swelling, restoration of cell membrane integrity, restoration of mitochondrial morphology, and a reduction in autophagosomes. The above improvements were more pronounced with the increase in serum concentration, indicating that the inhibitor and the drug-containing serum improved the pyroptosis state of cells. See Figure 3 for details.

Ultrastructure of cells was observed by transmission electron microscopy. (a) Control group, abundant mitochondria (blue arrow); (b) Model group, cell membrane rupture (yellow arrow); (c) Inhibitor group, a small number of autophagosomes (red arrow); (d, e, and f) were the low-dose, middle-dose, and high-dose serum groups, respectively. In panel (d), there were more autophagosomes and cytoplasmic vacuoles (black arrow)
Assay of NO in Cell Supernatant
Following cell modeling and drug treatment, the supernatant from each group was collected. Compared to the control group (1.017 ± 0.193), the culture supernatant of the model group exhibited an increase in NO content (3.127 ± 0.126, p < .05), which was subsequently reduced after treatment with NLRP3 inhibitor and 5%, 10%, 15% drug serum (1.228 ± 0.127, 2.452 ± 0.193, 2.072 ± 0.263, 1.819 ± 0.263, p < .05). Refer to Figure 4 for further details.

The content of NO in cell supernatant of each group was detected by biochemical detection. Data are expressed as the mean ± SEM. Multiple comparisons were analyzed using one-way ANOVA, and Tukey’s test was used for pairwise comparison. Differences with p < .05 were considered statistically significant
Elisa Analysis of TNF-α and IL-18 in Cell Supernatant
The levels of IL-18 and TNF-α in the culture supernatant of the model group (20.21 ± 2.41, 1381.30 ± 37.72) were found to be significantly increased compared to those in the control group (7.78 ± 0.93, 485.95 ± 67.52), with a statistically significant difference (p < .05). Upon treatment with the NLRP3 inhibitor and 5%, 10%, and 15% drug serum, the levels of IL-18 and TNF-α were significantly reduced compared to the model group, with a statistically significant difference (10.27 ± 0.58, 9.14 ± 0.57, 9.39 ± 0.75, 6.20 ± 1.71; 949.56 ± 99.32, 767.75 ± 68.68, 855.33 ± 52.38, 674.66 ± 54.01, p < .05). Further details can be found in Figure 5.

The levels of inflammatory factors TNF-α and IL-18 in the cell supernatant of each group were detected by Elisa. Data are expressed as the mean ± SEM. Multiple comparisons were analyzed using one-way ANOVA and Tukey’s test was used for pairwise comparison. Differences with p < .05 were considered statistically significant
Immunofluorescence Detection of NLRP3 and Caspase-1 Proteins
Adherent cells were harvested and subjected to immunofluorescence analysis. Compared to the control group, the model group indicated a notable increase in the positive fluorescence intensity of NLRP3 and Caspase-1. Subsequently, the fluorescence intensity was observed to decrease to varying degrees following treatment with MCC950 and 5%, 10%, or 15% serum containing Chinese medicine. Further details can be found in Figure 6.
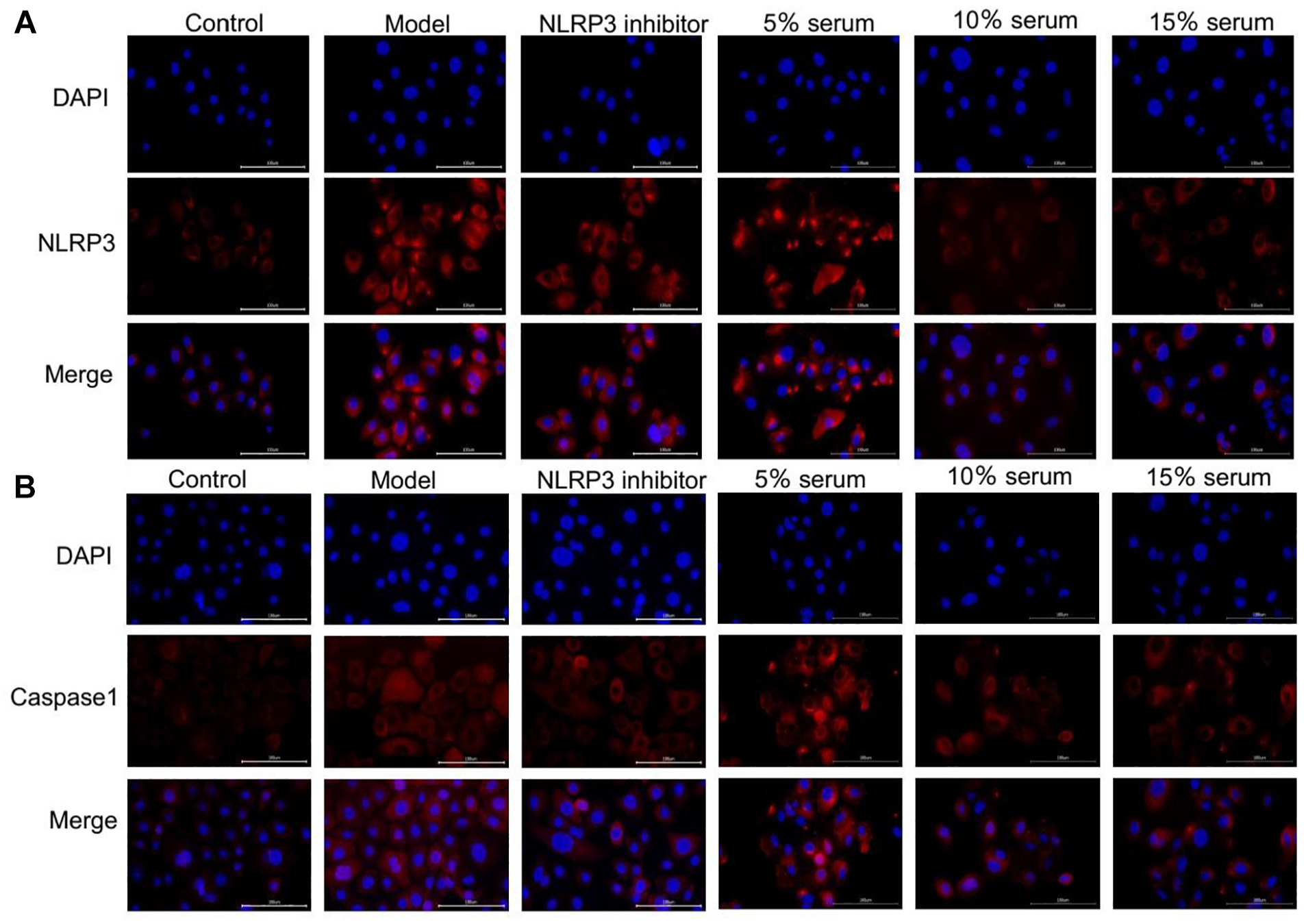
(a) The expression of NLRP3 was detected by immunofluorescence, and the NLRP3 protein appeared red in the figure. In the control group, the red fluorescence was almost invisible, whereas in the model group, distinct red fluorescence was apparent. (b) Immunofluorescence detection of Caspase-1 expression showed that Caspase-1 protein appeared red fluorescence in the figure, and the fluorescence in the control group was almost invisible. The red fluorescence in the model group was significantly enhanced compared with that in the control group, and the red fluorescence in the inhibitor group and the drug-containing serum group was diminished to varying extents.
Western Blot Analysis of NLRP3, Pro-Caspase-1, and GSDMD Protein Expression
Following cell manipulation and drug treatment, the total cellular protein was extracted, and the expression of relevant proteins was assessed. The contents of NLRP3, pro-Caspase-1, and GSDMD were found to be increased in the model group (1.212 ± 0.077; 1.168 ± 0.048; 1.264 ± 0.145) compared to the control group, (0.260 ± 0.026; 0.290 ± 0.023; 0.661 ± 0.029) with the difference being statistically significant (p < .05). After treatment with MCC950 and serum containing 5%, 10%, and 15% of the drug, the protein levels of these markers were decreased (0.963 ± 0.074, 0.655 ± 0.121, 0.508 ± 0.083, 0.306 ± 0.035; 0.897 ± 0.058, 0.739 ± 0.046, 0.565 ± 0.012, 0.561 ± 0.012; 1.069 ± 0.072, 0.902 ± 0.133, 0.845 ± 0.154, 0.769 ± 0.126) compared to the model group. The differences were also statistically significant (p < .05). See Figures 7 and 8 for details.

Electrophoretic images of pyro-related proteins in the control group, the model group, the inhibitor group, and the drug-containing serum group (NLRP3, GSDMD, pro-Caspase-1)

Western Blot analysis of pyroptosis-related proteins (NLRP3, pro-Caspase-1, GSDMD) in the control group, the model group, and the drug-containing serum group. Data are expressed as the mean ± SEM. Multiple comparisons analysis were conducted using one-way ANOVA. Tukey’s test was used for pairwise comparison. Differences with p < .05 were considered statistically significant
Discussion
CNP is a urogenital disorder that is prevalent among adult males. It is characterized by persistent lower abdominal and perineal pain, abnormal urination, and, in severe cases, potential sexual dysfunction. The etiology of type III CNP in western medicine is multifaceted, encompassing theories such as urine regurgitation, pathogenic infections, lower urothelial dysfunction, oxidative stress, psychological factors, and abnormal neuroregulatory mechanisms (Nickel, 2020). Recent research suggests that pyroptosis leads to the release of various cytoplasmic components, such as lactate dehydrogenase, inflammatory cytokines IL-1β and IL-18, which in turn initiate an inflammatory cascade. An imbalance of cytokines (Minton, 2020; Shi et al., 2017), particularly the excessive release of pro-inflammatory cytokines, plays a significant role in the development of CNP (Liang and Hao, 2019).
In a study conducted by Zuo et al. (2021) the authors observed that the combination of the Xialiqi capsules and the Tamsulosin capsules led to a significant improvement in the clinical symptoms of type III CNP, as evidenced by a comparison of NIH-CPS scores before and after treatment. Furthermore, this combination therapy outperformed the use of alpha-blockers alone. Additionally, animal studies demonstrated that the Xialiqi capsules effectively suppressed the pathophysiological changes in rat models of nonbacterial prostatitis, and there was an increase in rat urine output (Hong et al., 2010a). The above results show that the Xialiqi capsules have a definite curative effect on CNP. As pharmacological research has deepened, the pathogenesis of CNP has become increasingly linked to immunity and inflammation. Zeng and Liu (2020) demonstrated a substantial increase in the serum levels of the inflammatory factors TNF-α and Interleukin-4 (IL-4) in clinical patients. Moreover, IL-4 exhibited an immune activation effect, suggesting that higher concentrations of TNF-α and IL-4 are associated with greater cell damage in prostate tissue among patients. Additional research has indicated that the Xialiqi capsules effectively ameliorate pathological inflammatory infiltration (Hong et al., 2010b). This may be related to the improvement of the Xialiqi capsule on pyroptosis and the eventual reduction of inflammatory response; hence, we carried out in vitro experiments to further verify the mechanism.
In this study, the optimal dose concentration of the MCC950 was screened using the CCK-8 assay. Based on the results, a concentration of 1 μM of MCC950 was selected for subsequent experiments. RWPE-2 cells were selected and induced by LPS and ATP to create a model of cell pyroptosis injury. The CCK-8 assay results indicated that cell vitality in the model group was significantly lower than in the control group. Transmission electron microscopy revealed that the ultrastructure of RWPE-2 cells in the control group was normal, featuring a complete cell membrane, numerous organelles, and abundant mitochondria. Conversely, the model group exhibited disrupted ultrastructure, including cell swelling, reduced organelles, ruptured cell membranes, and pyroptotic changes such as mitochondrial atrophy and abnormal morphology. These findings confirmed the successful establishment of the pyroptosis cell model. Subsequent intervention with MCC950 and serum showed that the inhibitor group and the groups treated with 10% and 15% Chinese medicine-containing serum significantly enhanced cell viability and promoted cell proliferation.
Pyroptosis is an inflammatory form of cell death characterized by the involvement of gasdermin family proteins in the formation of plasma membrane pores. Its morphological manifestations include nuclear contraction or rupture, cytoplasmic swelling, and the formation of an incomplete cell membrane and inflammasomes (Galluzzi et al., 2018). Additionally, NLRP3, a crucial member of the NOD-like receptor family (NLRs), plays a significant role in pyroptosis. Upon activation, NLRP3 inflammasome facilitates the activation of intracellular interleukin-1β (IL-1β) and its precursor through the activation of Caspase-1. This leads to the activation of GSDMD, which subsequently triggers pyroptosis (Guo et al., 2023; Zheng et al., 2023). The results of transmission electron microscopy in this study showed that after the intervention with an inhibitor and drug-containing serum, the swelling of model cells was improved; the integrity of the cell membrane and the mitochondrial morphology were restored; and the number of autophagosomes was reduced. These changes were more pronounced with increasing serum concentration, indicating that the drug-containing serum improved the pyroptotic state of prostate epithelial cells.
NO, a free radical in vivo, exhibits the properties of a second messenger, neurotransmitter, and cytotoxic agent. Our research has shown a significant increase in serum NO levels in the model group compared to the normal control group, indicating a robust inflammatory response within the cells of the model group. Conversely, the inhibitor group and each drug-containing serum group exhibited significantly lower NO levels than the model group, indicating the ability of the Xialiqi capsule to effectively reduce NO levels within these cells and thereby alleviate the inflammatory state. Furthermore, we assessed the concentrations of pro-inflammatory factors TNF-α and IL-18 in the cellular supernatant. The model group had higher levels of IL-18 and TNF-α in the culture supernatant compared to the control group. However, after the administration of MCC950 and drug-containing serum, the levels of IL-18 and TNF-α decreased. These changes in the levels of NO, IL-18, and TNF-α suggest that the Xialiqi capsule has the potential to ameliorate inflammation in prostate epithelial cells. These results suggest that the Xialiqi capsule may reduce the inflammatory damage of RWPE-2 cells and enhance cell vitality by improving the pyroptosis state of RWPE-2 cells in vitro.
To further investigate the potential mechanism of action of the Xialiqi capsule in improving pyroptosis and exerting an anti-inflammatory effect, we employed the immunofluorescence method to assess the expression of pyroptosis-related proteins. Compared to the control group, the model group exhibited a significant increase in the positive fluorescence intensity of NLRP3 and Caspase-1. After treatment with MCC950 and drug-containing serum, the fluorescence intensity was reduced to varying degrees. Western Blot detection results indicated an increase in the levels of NLRP3 and pro-Caspase-1 in the model group compared to the control group. Following treatment with MCC950 and drug-containing serum at concentrations of 5%, 10%, and 15%, the protein levels decreased significantly. GSDMD was also activated in the model group, and the GSDMD content decreased after the intervention of drug-containing serum, which was consistent with the molecular trend of the NLRP3/Caspase-1 pathway. Previous studies have confirmed that the NLRP3 pathway can mediate prostate inflammation and cause prostate tissue damage, and inhibiting the NLRP3 pathway can effectively reduce prostate inflammation (He et al., 2021; My et al., 2024). The results of this study prove that the activation of the NLRP3 pathway plays an important role in the pathogenesis of CNP. The regulation of the NLRP3 pathway may be an important target for improving the inflammatory state of CNP and alleviating clinical symptoms, which is consistent with previous research results. Therefore, the regulatory role of pyroptosis in the future treatment of CNP cannot be ignored. The prospect of improving CNP by regulating pyroptosis with Chinese medicine preparations is also worth further exploration. The ideas and significance of this study are shown in Figure 9.

Overall process and significance of this research
Limitations
In vitro experiments were used in this study. The quality and stability of the medicated serum may be affected during the storage and thawing process, thus affecting the experimental results. Therefore, in the future research process, we will store the medicated serum in the refrigerator at −20°C, and it will be necessary to conduct programmed thawing before use to prevent excessive precipitation due to excessive temperature difference during the thawing process. At the same time, the RWPE-2 cell line is not fully representative of the complex physiological environment of prostate epithelial cells in vivo. In vivo, prostate epithelial cells interact with stromal cells, immune cells, and other cell types to participate in the physiological and pathological processes of prostate. Next, we will conduct in vivo experiments to further explore these mechanisms of action. Although this study verified the inhibitory effect of the Xialiqi capsule on cell pyroptosis, the effective monomer component of the specific action is not clear, so this is also the focus of our further research.
Conclusions
The aforementioned findings indicate that the Xialiqi capsule has the potential to mitigate pyroptosis in prostate epithelial cells by inhibiting the NLRP3/Caspase-1 signaling pathway and its associated molecules in an in vitro setting. This suppression results in a reduction of inflammatory mediators, thereby alleviating the inflammatory response exhibited by prostate epithelial cells. These findings suggest that this mechanism may be a contributing factor to the therapeutic effectiveness of the Xialiqi capsule in the management of CNP. At present, the regulation of cell pyroptosis provides a new direction for the prevention and treatment of CNP, and the use of Chinese medicine to treat CNP holds broad prospects and far-reaching significance. We should further verify through vivo experiments and clinical trials, and explore and transform the results of TCM preparations for CNP treatment. In clinical work, the classification and treatment plan of prostatitis is complicated. The results of this study can be further applied to the treatment of various types of prostatitis through the combination of traditional Chinese and western medicine in the future, and even for prostatitis caused by bacterial infection, it can also be combined with antibiotics to achieve better clinical efficacy.
Footnotes
Acknowledgements
All the authors of the manuscript are immensely grateful to the foundations for their valuable support.
Author Contributions
Z.M.H. and J.G. conducted research design, participated in the experimental operation, and drafted the manuscript. W.S.C. and Z.L.C. performed CCK8 analysis, biochemical assay, Elisa analysis, and statistical analysis. J.F.Q., S.J.W., and Y.W. performed transmission electron microscopy observation, immunofluorescence detection, and Western Blot analysis and participated in the experimental operation. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Science and Technology Innovation Project of the Chinese Academy of Traditional Chinese Medicine (NO. C12021A02208) and the Shenzhen Key Specialty Construction Project of Traditional Chinese Medicine (NO. 2019-2023(14)).
Ethical Approval
This study was reviewed by the Animal Ethics Committee of the fourth Clinical Medical College of Guangzhou University of Chinese Medicine (Ethics number: K2023-112-05).
Consent for Publications
The final manuscript was approved and read by all authors.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
