Abstract
Anejaculation (AE) has long been a challenging issue in andrology due to its low clinical reporting rate, leading to an insufficient understanding within the academic community. This case report discusses a 52-year-old AE patient with a long-term history of diabetes mellitus. His seminal vesicles ultrasound images revealed a notable absence of anechoic polycyclic areas, a characteristic not previously reported in AE patients. We hypothesize that this finding is attributable to diabetes-induced seminal vesicles secretory dysfunction, leading to AE in this patient. This case underscores the importance of anechoic polycyclic areas in the physiology of ejaculation and highlights the necessity for further research into the correlation between these areas and various types of ejaculatory disorders, aiming to provide valuable insights for the medical community.
Keywords
Introduction
Anejaculation (AE) is defined as the absence of normal antegrade ejaculation during orgasm, resulting from the lack of the emission and expulsion phases of the ejaculation reflex (McCabe et al., 2016). Historically, due to the lack of a clearly defined distinction, AE was often confused with delayed ejaculation (DE) and anorgasmia, making it difficult to ascertain its true prevalence. According to the latest report, the prevalence of DE is between 0.01% and 0.02% (Liao et al., 2024). However, this does not accurately reflect the prevalence of AE. In an epidemiological study conducted in the United States involving 988 patients with ejaculatory dysfunction (EjD), excluding those with premature ejaculation (PE), AE accounted for up to 37% (Paduch et al., 2015).
The precise pathophysiology of AE remains only partially elucidated. Researchers generally believe that AE is associated with psychosocial and endocrine factors (Corona et al., 2011; Waldinger & Schweitzer, 2005). In recent years, the high incidence of diabetes mellitus (DM) has increasingly drawn the attention of researchers to DM-induced EjD. Reports from various countries and regions indicate that more than 30% to 70% of diabetic patients suffer from some form of EjD (Burke et al., 2007; el-Rufaie et al., 1997; Getie Mekonnen et al., 2021). Regarding the two critical physiological processes in the definition of AE—emission and expulsion—DM can impair both the emission and expulsion of semen (Yonezawa et al., 2009). Seminal vesicle fluid accounts for approximately 70% of the total ejaculate volume (Aumüller & Riva, 1992). Insufficient production of seminal vesicle fluid can lead to EjD, manifesting as DE or even AE (Gao et al., 2024).
In streptozotocin-induced diabetic rat models, the volume of seminal vesicle fluid and the rate of spontaneous semen emission were significantly reduced (Yonezawa et al., 2009), suggesting that the loss of seminal emission due to a reduction in seminal vesicle fluid may contribute to the mechanism of EjD in diabetic rats. The diabetic models also exhibit significant changes in the sympathetic innervation of the seminal vesicles. Alterations in the reuptake, release, and synthesis of the neurotransmitter noradrenaline may contribute to changes in the concentration of this amine in the tissue. These observed changes may be related to the remodeling and regrowth of sympathetic nerve endings damaged in the early stages of hyperglycemia, potentially contributing to ejaculatory disorders in diabetic patients (Morrison et al., 2006).
Doppler ultrasound is helpful in diagnosing EjD, but there are currently few reports on the ultrasonographic characteristics of the seminal vesicles in patients with DE or AE (Forbes et al., 2018). In this unique case report, we describe a 52-year-old male patient with DM and AE. His seminal vesicles ultrasound images did not indicate the presence of anechoic polycyclic areas, providing a new observation parameter for the ultrasonographic characteristics of AE. This case highlights the significance of anechoic polycyclic areas in the seminal vesicles in the physiology of ejaculation, underscores the need for further research in this area, and aims to offer valuable insights into such cases for the medical community.
Case Presentation
A 52-year-old married and childbearing male has been experiencing AE for 3 years, along with an absolute absence of sensation of orgasm. This condition occurs during vaginal intercourse as well as all other forms of sexual activity. One year ago, he underwent a urinalysis at another medical institution following masturbation. During the process, even until his penis became flaccid, he did not experience a sexual climax, and no sperm was detected in the urine sample. Based on experience, the primary physician prescribed psychological and behavioral therapy, along with oral bupropion. Unfortunately, these treatments did not yield any positive results. A detailed medical history revealed that the patient has normal erectile function and a 20-year history of DM, currently managed with subcutaneous insulin glargine (6 IU at bedtime) and semaglutide (1 mg weekly). In addition, he has a 10-year history of hypertension, controlled with oral felodipine (5 mg daily), with typical blood pressure readings of 150/90 mmHg. The patient reported no urinary issues, such as difficulty urinating, frequent urination, urgency, or blood in the urine, and mentioned maintaining a good urinary flow. He has no history of major surgeries or any neurological, infectious, or endocrine conditions that could cause AE or loss of sexual pleasure. Furthermore, the patient has no history of multiple sclerosis, spinal cord injuries, radical prostatectomy, hypogonadism, or hypothyroidism—conditions known to cause AE and diminished sexual pleasure. In addition, the patient has not taken any medications that could cause AE or loss of sexual pleasure, such as α-methyldopa, thiazide diuretics, or antidepressants.
Physical examination revealed normal development of the external genitalia, no phimosis, and bilaterally 15 mL testes with no sensory dysfunction. Doppler ultrasonography of the urogenital system showed normal seminal vesicles size with no significant anechoic polycyclic areas in either gland (Figure 1A). The prostate measured 4.50 cm in transverse diameter, 3.34 cm in vertical diameter, and 3.42 cm in anteroposterior diameter, with a regular shape and uneven echo distribution. Multiple patchy hyperechoic areas with acoustic shadowing were observed, but no prominent protrusion into the bladder was noted. The testes exhibited a left vertical diameter of 4.15 cm and an anteroposterior diameter of 1.81 cm, and a right vertical diameter of 4.05 cm and an anteroposterior diameter of 1.66 cm, with homogeneous parenchymal echogenicity and symmetric color Doppler flow signals, indicating no abnormalities. No hydrocele was present in either testis. Both epididymides and their tails were of normal size with normal parenchymal echogenicity, though a cystic anechoic area measuring 1.47 × 1.07 cm was noted in the head of the left epididymis, displaying good acoustic transmission. No significant abnormal echoes were detected in the spermatic cords bilaterally. The laboratory test results are detailed in Table 1.
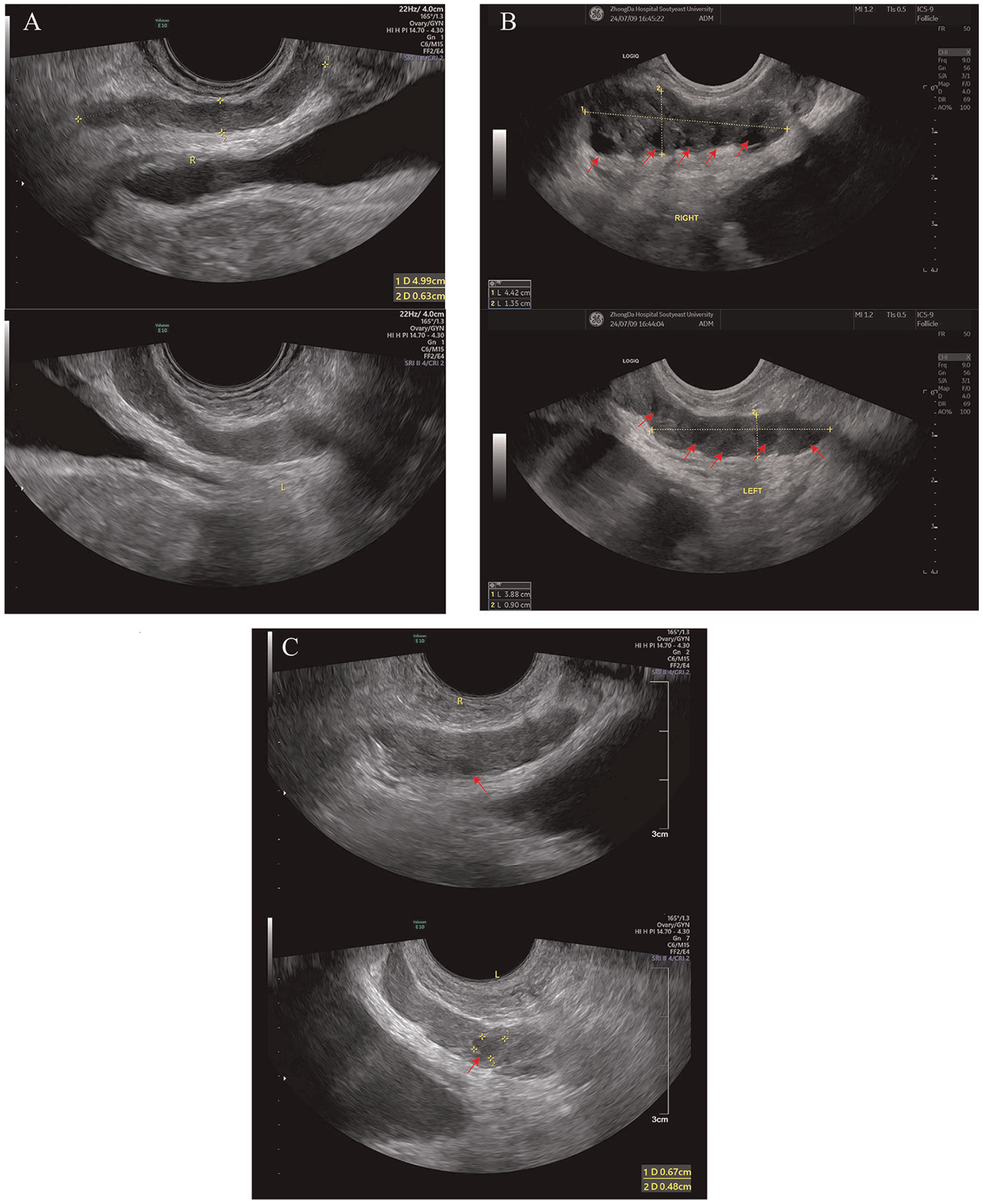
Doppler Ultrasound Images of the Bilateral Seminal Vesicles
Laboratory Test Results

Management and Outcome
In response to the patient’s primary condition, due to inadequate blood sugar control, consultation with an endocrinologist was sought. It was recommended to continue the current glycemic management plan and strictly adhere to a diabetic diet. For the patient presenting with the symptom of absent seminal emission, our approach integrates traditional Chinese medicine (TCM) theory and syndrome differentiation, identifying “kidney essence deficiency” as the underlying cause contributing to insufficient semen production. To address this, we have formulated a Chinese herbal prescription aimed at nourishing the kidney and replenishing the essence. The prescription includes Sheng Di Huang (Rehmannia glutinosa, radix) 10 g, Shu Di Huang (Rehmannia glutinosa, radix preparata) 10 g, Bie Jia (Trionyx sinensis, carapax) 20 g, Sheng Mu Li (Ostrea gigas, concha) 20 g, Dan Pi (Paeonia suffruticosa, cortex) 15 g, Tian Hua Fen (Trichosanthes kirilowii, radix) 15 g, Li Zhi He (Litchi chinensis, semen) 30 g, Yin Yang Huo (Epimedium brevicornum, herba) 10 g, Dan Shen (Salvia miltiorrhiza, radix) 10 g, Lu Lu Tong (Liquidambar formosana, fructus) 10 g, Chai Hu (Bupleurum chinense, radix) 15 g, Hong Hua (Carthamus tinctorius, flos) 6 g, Bai Hua She She Cao (Hedyotis diffusa, herba) 15 g, Zao Jiao Ci (Gleditsia sinensis, spina) 10 g, and Di Long (Pheretima aspergillum, lumbricus) 9 g. A total of 10 doses of herbal decoction were prescribed to the patient. Each dose is prepared daily by boiling twice; the water is first brought to a boil and then simmered for 30 minutes. After two rounds of boiling, a total of 400 mL of the decoction is obtained, to be taken in two 200 mL doses—one after breakfast and one after dinner. Unfortunately, this patient did not strictly adhere to the medical advice for follow-up visits, which hindered the assessment of treatment effectiveness.
Discussion
During the physiological process of ejaculation, the seminal vesicles play a crucial role in the secretion and accumulation of semen (Gao et al., 2024), with seminal vesicle fluid accounting for approximately 70% of the total ejaculate volume (Aumüller & Riva, 1992). As semen accumulates in the prostatic urethra, the local pressure effect, in conjunction with other sexual stimuli, activates the spinal ejaculation center, resulting in coordinated rhythmic contractions of the bulbospongiosus, ischiocavernosus, and pelvic muscles, which propel the semen anterogradely through the urethra (Alwaal et al., 2015; Coolen et al., 2004). Although ejaculation is a spinal reflex, and patients who have undergone prostatectomy may experience dry ejaculation (Alwaal et al., 2015), the contribution of the pressure chamber effect—induced by semen accumulation—in triggering the ejaculation reflex should not be underestimated. Peripheral nerve damage, commonly seen in diabetic patients, can impair both the secretion and contraction functions of the seminal vesicles (Tsounapi et al., 2017; Yonezawa et al., 2009), leading to a reduced volume of seminal fluid entering the prostatic urethra and a decrease in local pressure. This insufficient pressure fails to adequately stimulate the local nerves, thereby preventing the initiation of ejaculation. This proposed mechanism may help explain the reduced ejaculatory function observed in diabetic patients (Gao et al., 2024).
In the past, ultrasound examinations of the seminal vesicles in patients with DE and AE have not received significant attention from urologists and andrologists. Consequently, there is limited literature on case reports and clinical observational studies in the academic field. Although the treatment outcome for the patient we reported on was not effectively evaluated, this case report focuses on the abnormal seminal vesicles ultrasound findings—specifically, the absence of anechoic polycyclic areas in this patient with AE. The academic terminology for “anechoic polycyclic areas” is not standardized. The European Academy of Andrology (EAA) refers to these as “areas of endocapsulation” or “roundish anechoic areas” when describing the ultrasound characteristics of the seminal vesicles (Lotti et al., 2022). In addition, some studies using magnetic resonance imaging, computed tomography, and seminal vesiculoscopy have described the characteristic features of the seminal vesicles as exhibiting a “grape-like appearance” (Catania et al., 2020) or a “honeycomb appearance” (Chen et al., 2018). EAA’s research found that round anechoic areas are observed in one-sixth of men prior to ejaculation. However, their occurrence rate is halved postejaculation (p < .001) (Lotti et al., 2022). Furthermore, the detection rate of anechoic areas before ejaculation is positively correlated with ejaculate volume (Lotti et al., 2022). A similar result was also observed in another study by Lotti et al. (2012). Therefore, these areas should be understood as regions of seminal vesicle fluid expelled during ejaculation.
In this case, based on the patient’s medical history, examination results, and previous treatment plans, we excluded other potential causes of DE and AE, including psychological factors, multiple sclerosis, spinal cord injuries, radical prostatectomy, hypogonadism, hypothyroidism, and medications known to induce DE and AE. However, the patient’s history of DM for over 20 years warrants particular attention, as peripheral neuropathy caused by diabetes is a significant pathological basis for EjD (Desai et al., 2023). Therefore, we believe that DM is the culprit responsible for the patient’s AE.
A unique aspect of this case is the absence of anechoic polycyclic areas in the patient’s seminal vesicles ultrasound images. Typically, ultrasound images of the seminal vesicles in healthy males display a few anechoic areas (La Vignera et al., 2011; Lotti et al., 2022), indicating that the seminal vesicles store a certain amount of seminal fluid and suggesting normal secretory function. A study on the ultrasound characteristics of seminal vesicles in diabetic infertile patients has found that the number of anechoic polycyclic areas in their seminal vesicles is reduced compared to the control group (La Vignera et al., 2011). However, the sample size in this study was too small, with only 25 DM patients and 25 controls, which limits the power to detect significant differences.
Furthermore, clinical observations have consistently shown that semen volume in diabetic men is significantly lower than that in nondiabetic controls (Agbaje et al., 2007; Ali et al., 1993). Pei et al. (2013) discovered that the expression of aquaporin1 (AQP1) and AQP4 in the seminal vesicles of diabetic rats was significantly reduced compared to the control group. This reduction may contribute to the diminished secretory function of the seminal vesicles in diabetes, as AQPs are a family of water-transporting membrane proteins with important physiological roles, including water and salt homeostasis, epidermal hydration, and exocrine fluid secretion (Agre & Kozono, 2003; Pei et al., 2013; Verkman, 2012). In addition, DM can impair testosterone levels in males (Fushimi et al., 1989). Since the seminal vesicles are testosterone-sensitive organs, their secretory function is closely related to testosterone levels (Mann, 1974; Tsuji et al., 1998). Therefore, reduced testosterone levels are also a significant factor contributing to the insufficient production of seminal vesicle fluid.
For AE patients with a confirmed history of DM, proactive blood sugar control forms the cornerstone of treatment, aiding in disease progression prevention and averting other diabetes-related complications. However, the idea that blood sugar control can improve or reverse DE or AE in chronic diabetes, as suggested by animal studies, has been refuted (Yonezawa et al., 2009). Therefore, restoring ejaculatory function in DM patients remains a challenging topic for future research. Regarding the treatment for this case, the patient had previously undergone psychological and behavioral therapy, along with empirical treatment using bupropion for DE and AE, but without success. Consequently, the patient sought treatment with TCM. According to TCM theory, semen is derived from kidney essence (Shi et al., 2021), with seminal vesicle fluid considered a component of semen. Insufficient production of seminal vesicle fluid is also attributed to kidney essence deficiency (Shi et al., 2021). In the past, we successfully improved the ejaculatory function of patients with functional AE using Yangjing capsules, which are believed to nourish the kidney and replenish essence (Jin et al., 2012). The theoretical basis for this approach lies in stimulating seminal vesicle secretion, thereby increasing pressure in the prostatic urethra during ejaculation and triggering the ejaculation reflex (Jin et al., 2012). Animal studies have supported our hypothesis, showing that Yangjing capsules significantly enhance the secretion function of the seminal vesicles, increasing both the volume of seminal vesicle fluid and the size and surface density of secretory granules in the epithelial cells (Jin et al., 2014). Therefore, in this case, we attempted to use the same approach to improve the patient’s seminal vesicle secretion function. Unfortunately, due to the patient’s poor compliance and irregular follow-up visits, we were unable to adequately assess the efficacy of the treatment.
Limitations and Prospects
In the diagnosis and treatment of this case, several shortcomings were identified. Primarily, due to the influence of traditional Chinese culture, individuals of Chinese and East Asian descent tend to be relatively conservative about sexuality (Brotto et al., 2007). This cultural context made it challenging to persuade the patient to engage in masturbation during dynamic Doppler ultrasound examinations in a clinical setting. As a result, we were only able to perform transabdominal prostatic Doppler ultrasound. However, dynamic Doppler ultrasound could have more effectively elucidated aberrant patterns of muscular or structural dysfunction that co-occur with the sensations experienced during ejaculation (Forbes et al., 2018). Dynamic imaging techniques may also be valuable for assessing the coordinated events of the pelvic floor and associated anatomical structures during ejaculation, such as the contraction of the bulbospongiosus muscle and the flow of seminal vesicle fluid (Forbes et al., 2018). In regions with a more open sexual culture, combining dynamic Doppler color ultrasound could offer a better understanding of abnormal patterns in the pelvic floor muscles and related structures in patients with ejaculation disorders.
Moreover, due to personal reasons, the patient did not adhere strictly to the prescribed follow-up schedule or continue the treatment consistently. As a result, we were unable to assess the efficacy of the herbal decoction, which represents a major limitation of this case report. Therefore, future studies should include rigorously designed, large-scale, multicenter randomized controlled trials to evaluate the effectiveness of TCM in treating diabetic DE and AE. Such studies would help validate our hypothesis and provide new supplementary therapeutic options for managing diabetic DE and AE.
In addition, in other types of EjD ultrasound imaging studies, descriptions of anechoic polycyclic areas have also been observed, albeit using slightly different academic terminology. In the study by Hong et al., it was found that areas of endocapsulation in the seminal vesicles of PE patients were significantly higher compared to normal controls (Hong et al., 2017). These endocapsulated areas appeared as anechoic polycyclic regions on ultrasound images. PE, as another extreme form of EjD, exhibits an increase in areas of endocapsulation, in contrast to the absence of anechoic polycyclic areas observed in our reported patient. Thus, this suggests that anechoic polycyclic areas may be linked to seminal vesicle secretion function, seminal vesicle fluid volume, and prostatic urethral pressure. However, whether anechoic polycyclic areas can serve as a new independent influencing factor for EjD still requires confirmation through larger-scale studies.
Conclusion
In conclusion, anechoic polycyclic areas observed in seminal vesicles ultrasound images may be associated with the reserve of seminal vesicle fluid, reflecting the secretory function of the seminal vesicles. For DE and AE patients, assessing the quantity of these anechoic polycyclic areas can help evaluate seminal vesicles secretory function and guide the formulation of targeted treatment plans. Future research should involve large-sample, multicenter observational studies to explore the clinical significance of anechoic polycyclic areas in EjD, which would have important implications for addressing this global health challenge.
Footnotes
Author Contributions
D.G. was responsible for writing and revising the manuscript, Q.G. conducted the ultrasound diagnosis for patients, H.Y. participated in the consultation to adjust the patient’s glycemic control plan, B.J. provided research direction and manuscript revision suggestions, and D.S. provided the case information and participated in the patient’s consultation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 82074440) and Zhongda Hospital, Southeast University, Jiangsu Province High-Level Hospital Construction Funds (grant no. 2024GSPKY26).
Ethical Approval
Per the guidelines of our institution, no formal ethical approval or participant consent is necessary for a single case report if it contains no patient-identifying information. However, we have obtained the patient’s consent, and it can be requested from the corresponding author if needed. The methodologies employed in this study involving human participants align with the ethical norms of our institutional and national research committees and adhere to the principles outlined in the 1964 Helsinki Declaration, its subsequent amendments, or equivalent ethical standards.
