Abstract
This before-and-after design study, which was conducted during Ramadan 2023, aimed to compare salivary flow rate (SFR), potential hydrogen (pH), and electrolyte concentrations before Ramadan (BR), at the end of Ramadan (ER), and after Ramadan (AR). Men aged 20 to 30 years who were fasting during Ramadan were recruited from the University of Monastir (Tunisia). Unstimulated saliva was collected over a 5-minute period between 10 and 11 a.m. at BR, ER, and AR. After recording the SFR, the pH was measured immediately using a pH meter. Concentrations of several salivary electrolytes (e.g., calcium, potassium, sodium, chloride, and phosphate) were determined using inductively coupled plasma-optical emission spectroscopy. Twenty-seven participants completed all three sessions. Ramadan intermittent fasting (RIF) caused significant changes only in calcium levels and pH. The mean (M)±standard deviation (SD) salivary calcium concentration decreased during ER compared to BR and AR (0.10 ± 0.08 vs. 0.21 ± 0.18 [p = .041] vs. 0.22 ± 0.20 [p = .026], respectively). The M±SD salivary pH increased during ER compared to BR (6.88 ± 0.23 vs. 6.69 ± 0.35 [p = .049], respectively). To conclude, RIF decreased salivary calcium, increased salivary pH, and did not significantly affect potassium, sodium, chloride, or phosphate. The pH change, although statistically significant, was clinically insignificant, as values remained normal. The lack of reference values for salivary calcium limits assessment of its clinical impact. There is a need for further research on the effects of RIF on saliva secretion.
Introduction
Saliva is an acidic biological fluid secreted by the three major salivary glands (i.e., parotid, submandibular, and sublingual) and by minor glands (i.e., labial, buccal, lingual, and palatal tissues) (Kaczor-Urbanowicz et al., 2017). Saliva contains gingival crevicular fluid, cell debris, plaque, bacteria, nasal and bronchial secretions, lining cells, blood, and exogenous substances (Kaczor-Urbanowicz et al., 2017). Saliva is composed of water (99%), proteins, and both inorganic and organic substances (Liu & Duan, 2012). The most prevalent inorganic components include sodium, potassium, calcium, magnesium, chloride, and carbonates, while the organic components comprise amylases, peroxidase, lipase, mucins, lysozyme, lactoferrin, kallikreins, cystatins, hormones, and growth factors (Chiappin et al., 2007). Saliva protects the oral mucosa from biological, mechanical, and chemical factors, as well as from bacterial, viral, and fungal infections (Kaczor-Urbanowicz et al., 2017). Saliva prevents enamel demineralization and enhances remineralization due to its buffering capacity (Garcia-Godoy & Hicks, 2008). Salivary flow rate (SFR) and composition play a crucial role in the caries process (Garcia-Godoy & Hicks, 2008). The components of saliva work to eliminate harmful by-products of bacterial metabolism in the dental biofilm (Garcia-Godoy & Hicks, 2008). One study reported that salivary components could inhibit the growth of Candida albicans (Salvatori et al., 2016). A reduction in SFR and changes in salivary composition could potentially increase oral yeast carriage rates, predisposing individuals to opportunistic infections such as oral candidiasis (Nadig et al., 2017). Furthermore, SFR and salivary potential hydrogen (pH) levels are inversely proportional to caries activity; their decrease may be a risk factor for caries (Singh et al., 2015). The pH of saliva is influenced by its electrolyte composition (Bragazzi, 2015). Any change in salivary components can affect the pH, which, in turn, may influence the function of saliva and its protective role in the oral cavity, leading to adverse effects on oral health (Bragazzi, 2015). Salivary composition is known to vary depending on factors such as the type of stimulation, short-term acute mental stress, taste, smell, and seasonal or circadian rhythms (Besbes et al., 2022), including the effects of Ramadan intermittent fasting (RIF) (Qasrawi et al., 2017).
Ramadan is the ninth lunar month of the Islamic Hijra calendar (Fernando et al., 2019). RIF is one of the five pillars of Islam and is obligatory for all healthy Muslims from puberty onward (Fernando et al., 2019). Hundreds of millions of people observe Ramadan, which involves abstaining from eating and drinking from sunrise to sunset (Hassanein et al., 2017). Depending on geographic location, the duration of fasting can range from 9 to 22 hr/day (Hassanein et al., 2017). This modification in lifestyle for one lunar month may influence oral health (Hassanein et al., 2017). Among the numerous studies analyzing the effects of RIF on general health (Al-barha & Aljaloud, 2018; Ben Saad, 2018, 2022; Kammoun et al., 2022; Latiri et al., 2017; Miladi et al., 2020; Mrad et al., 2019; Rejeb et al., 2018; Singha Roy & Bandyopadhyay, 2018; Zouari et al., 2018), only a few (Al-Rawi et al., 2020; Bahammam et al., 2013; Dehaghi et al., 2020; Develioglu et al., 2013; Illahi et al., 2016; Khaleghifar et al., 2017; Rahim & Yaacob, 1991; Sariri et al., 2010) have investigated its impacts on whole saliva. A 2021 systematic review examining the effects of RIF on SFR and metabolic parameters (Besbes et al., 2022), which included six studies (Al-Rawi et al., 2020; Bahammam et al., 2013; Dehaghi et al., 2020; Develioglu et al., 2013; Khaleghifar et al., 2017; Sariri et al., 2010), concluded that fluctuations in salivary parameters during Ramadan were not as significant as changes in blood markers. The methodological quality of the six studies included in that systematic review (Besbes et al., 2022) was low, making it difficult to draw strong clinical conclusions regarding the effects of RIF on saliva secretion. The authors of the systematic review (Besbes et al., 2022) offered recommendations for future research on the effects of RIF on salivary parameters.
The aim of this before-and-after design pilot study was to compare SFR, pH, and electrolyte concentrations before Ramadan (BR), at the end of Ramadan (ER), and after Ramadan (AR) in healthy men who fasted during Ramadan. Since women do not fast for the entire month of Ramadan (Mahesh et al., 2014), since hormonal fluctuations during the menstrual cycle can affect salivary parameters (Mahesh et al., 2014), and since female sex has been reported to lower SFRs (Fenoll-Palomares et al., 2004), the present study included only men. We hypothesized that during fasting, the mouth does not engage in normal eating and drinking activities, leading to reduced salivary gland activity, a decrease in SFR, and changes in saliva acidity (Illahi et al., 2016).
Population and Method
The present study is part of a larger project comprising two parts. The first part, which is the focus of this study, examines the effects of RIF on SFR, salivary pH, and electrolyte concentrations. The second part will evaluate the effects of RIF on salivary cortisol, glucose, and protein concentrations.
Study Design
This was a longitudinal (before-and-after design) pilot study conducted at the Department of Oral Physiology, Faculty of Dental Medicine, University of Monastir (Tunisia), following the guidelines of the Strengthening the Reporting of Observational Studies in Epidemiology statement (von Elm et al., 2014).
Approval for the study was obtained from the Institutional Ethics and Research Committee of the University Hospital Farhat Hached (IORG 0007439, reference: ERC 02082024). The study was conducted in accordance with the World Medical Association’s (WMA, 2013) Declaration of Helsinki for research involving human subjects. All participants were enrolled voluntarily without receiving any direct benefits. Participants were individually informed about the purpose of the study, and each signed an informed consent form after receiving a detailed explanation of the study’s characteristics. No fees were charged for the oral tests. The study was carried out during the spring of 2023, and Ramadan lasted from March 23 to April 20, 2023. The elapsed time between dawn and sunset was approximately 14.9 hr.
Population
Healthy men aged between 20 and 30 years who were fasting during Ramadan were recruited by convenience from the students of Monastir University (Tunisia). The specific 20- to 30-year age range was selected because SFR rate is affected by age, with old age corresponding to lower the SFR (Khemiss et al., 2017; Narhi et al., 1999). Based on the World Health Organization definition of health as “a state of complete physical, mental and social well-being and not merely the absence of disease or infirmity” (International Health Conference [IHC], 1948) and focusing on pathologies that disturb salivary parameters, the following non-inclusion criteria were applied: history of systemic diseases or use of medications known to affect oral health (e.g., diabetes mellitus, topical or systemic corticosteroids, or antibiotics), smoking (cigarette or narghile), regular fasting on Mondays and Thursdays throughout the year, engagement in other intermittent fasting (e.g., for weight loss or protest), periodontal treatment within the last 6 months, genetic or acquired dental anomalies (e.g., fluorosis, amelogenesis imperfecta), overweight or obesity (i.e., body mass index ≥25 kg/m2), and prior orthodontic treatment. Participants were instructed to maintain their regular diet during non-fasting hours and to avoid altering their habitual physical exercise levels BR, at the ER, and AR.
Sample Size
The sample size was calculated using the following formula (Serhier et al., 2020): n = (Zα/2)2s2/d2, where:
“s” is the standard deviation (SD);
“d” is the accuracy of the estimate (how close to the true mean of the primary outcome, i.e., SFR); and
“Zα/2” is the normal deviate for a two-tailed alternative hypothesis at a significance level of 0.05 (Zα/2 = 1.96).
Given the pioneering nature of this study, the values for “s” and “d” were collected from a previous Malaysian study involving 41 healthy participants (Rahim & Yaacob, 1991). That study compared the SFR and concentrations of calcium, phosphate, protein, and nitrite in whole unstimulated saliva under Ramadan fasting (i.e., after at least 6 hr without food and water) versus control conditions (i.e., 30–60 min after eating). The SFR for fasting saliva was reported to be half that of the control (0.098 ± 0.023 vs. 0.208 ± 0.020 ml/min, respectively), and “s” was set at 0.0215 [= (0.023+0.020)/2], and “d” was estimated at 0.0080.
The sample size was calculated as n = (1.96)2× 0.02152/0.00802, yielding 27 participants. Assuming a 15% dropout rate for the second or third session, the corrected sample size was 33 participants.
Protocol Design
The protocol design consisted of three sessions: 10 days BR (March 13–22, 2023), 4 days at the ER (April 17–20, 2023), and 10 days AR (April 21–30, 2023). The ambient temperature and humidity (M±SD) during the BR, ER, and AR sessions were 14.0 ± 0.3°C and 63.0 ± 4.8%, 17.8 ± 0.3°C and 36.0 ± 2.0%, and 19.5 ± 0.6°C and 78.0 ± 4.0%, respectively.
During the BR session, all participants completed a medical questionnaire, and saliva samples were collected. In the ER and AR sessions, only saliva collection was performed.
General Characteristics
Age (years) and oral habits (e.g., frequency of tooth brushing and number of dental visits) were collected using a French-language medical questionnaire.
Saliva Collection
Unstimulated whole saliva was collected by expectoration into sterile 15-mL centrifuge tubes over a 5-min period after participants rinsed their mouths with distilled water (Develioglu et al., 2013). The collection was conducted between 10 and 11 a.m. during BR, ER, and AR, as this is when circadian rhythms influence saliva production (Illahi et al., 2016). All samples were stored at −20°C and later transferred to a biological laboratory for biochemical analysis.
Participants were instructed to:
Rinse their mouths with water before saliva collection,
Avoid coughing or throat clearing into the collection tube, and
Refrain from brushing their teeth or using oral hygiene products before collection.
SFR
SFR represents the amount of saliva produced by the salivary glands, expressed in ml/min (Edgar, 1992). The volume of unstimulated saliva was divided by the duration of collection.
Salivary pH
After recording SFR, the salivary pH was measured immediately to prevent sample degradation. A 0.5-mL portion of each saliva sample was placed in a tube (Moritsuka et al., 2008). The pH was measured using a pH meter (Eutech’s Cyberscan pH510, Singapore®). The accuracy of the pH meter was checked daily using standard pH 4, pH 7, and pH 10 buffers (Moritsuka et al., 2008).
Salivary Electrolytes
The concentrations of salivary electrolytes were determined using inductively coupled plasma-optical emission spectroscopy. A 0.1-mL saliva sample was diluted with 4.9 ml of 2% (w/v) nitric acid to detect concentrations (Lan et al., 2020). Standard curves for calcium, potassium, sodium, chloride, and phosphate ions were generated using standard solutions (Sigma-Aldrich, St. Louis, MO, USA) before each detection run (Lan et al., 2020). The coefficients of variation were 34.1% for chloride, 42.5% for sodium, 50.5% for potassium, 57.4% for phosphate, and 97.7% for calcium.
Salivary Cortisol, Glucose, and Proteins
These parameters will be analyzed in a subsequent study.
Statistical Analysis
The Kolmogorov–Smirnov normality test was used to assess the normality of quantitative data. Normally distributed data with equal variances were expressed as M±SD (95% confidence interval [CI]), and ranges for pH, while non-normally distributed data were expressed as median (interquartile range, IQR).
Comparisons of SFR, pH, and electrolyte concentrations between the three sessions (BR, ER, and AR) were made using one-way analysis of variance (ANOVA). Where appropriate, significant differences between means were further analyzed using the Tukey honestly significant difference (HSD) post hoc test. If statistical differences were found, median (IQR) delta changes were calculated as follows: 100 × (ER value − BR value)/ER value; or 100 × (ER value − AR value)/ER value. Statistical analysis was performed using Statistica software (Statistica Kernel version 6; StatSoft, Paris, France). A significance level of .05 was set for all analyses.
Results
Of the 33 participants initially enrolled in the study, 27 completed all three sessions (Figure 1). The remaining participants dropped out due to absence during the second session (n = 4) or the third session (n = 2).
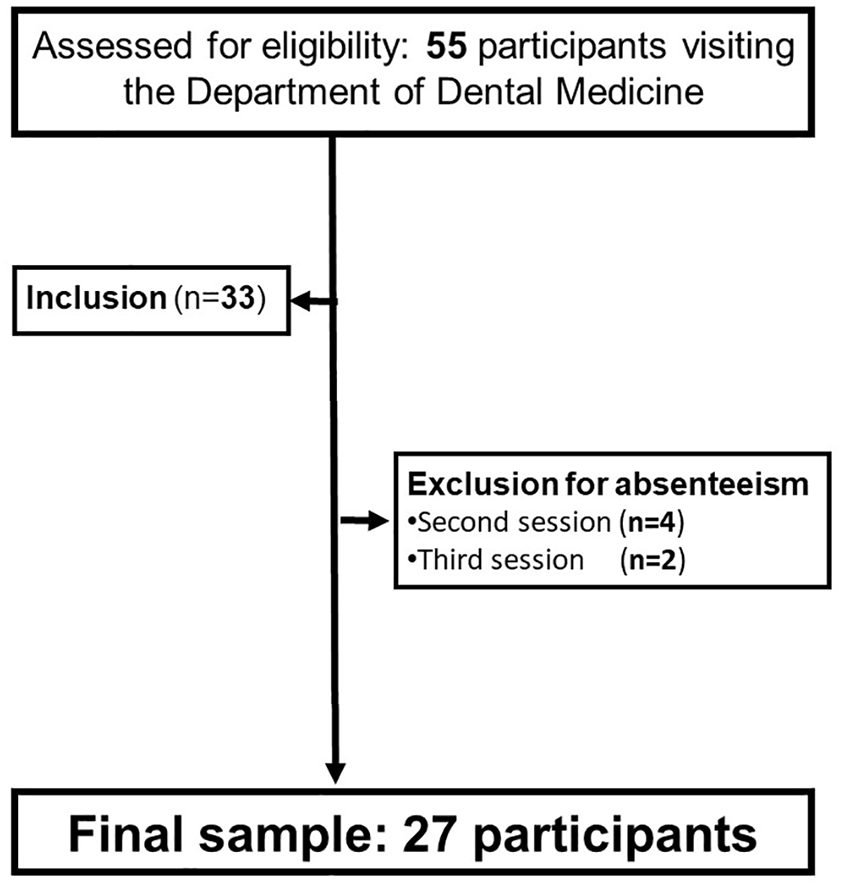
Study Flowchart
Participant Characteristics
The M±SD (95% CI) age of participants was 23 ± 4 [21, 24] years, and the frequency of tooth brushing was 2.0 ± 0.6 [1.7, 2.2] times per day. The median (IQR) number of visits to the dentist was 0.00 (0.00 to 1.00).
SFR and pH
Table 1 presents the SFR and pH data for participants. While the SFR remained largely unchanged across sessions, the pH values were significantly affected by RIF. The pH during the ER session was significantly higher compared to the BR session (p = .049), with a median (IQR) delta change of 1.7% (−0.3% to 6.4%) (Figure 2). The pH [minimum–maximum] values for the BR, ER, and AR sessions were [5.75–7.78], [6.28–7.46], and [6.02–7.12], respectively.
Saliva Flow Rate (SFR) and Potential Hydrogen (pH) of the 27 Healthy Adult Men

Note. ANOVA: analysis of variance between the three sessions. AR: after Ramadan. BR: before Ramadan. ER: end of Ramadan. Data were M±SD (95% confidence interval).
BR versus ER (p = .049).
p < .05 (one-way ANOVA): comparison between the three sessions; p < .05 (Tukey HSD post hoc test).

Effects of Ramadan Intermittent Fasting (RIF) on Saliva Potential Hydrogen (pH) (n = 27 Healthy Men)
Salivary Electrolytes
Table 2 presents the concentrations of salivary electrolytes for participants. Among these, only calcium concentration was significantly impacted by RIF. The calcium levels during the ER session were significantly lower than in both the BR (p = .041) and AR (p = .026) sessions, with median (IQR) delta changes of −75% (−300% to 14%) and −100% (−200% to 40%), respectively (Figure 3).
Saliva Electrolytes Data of the 27 Healthy Adult Men

Note. ANOVA: analysis of variance between the three sessions. AR: after Ramadan. BR: before Ramadan. ER: end of Ramadan. Data were M±SD (95% confidence interval).
p < .05 (one-way ANOVA): comparison between the three sessions; p < .05 (Tukey HSD post hoc test): ER versus BRa (p = .041) or versus ARb (p = .026).
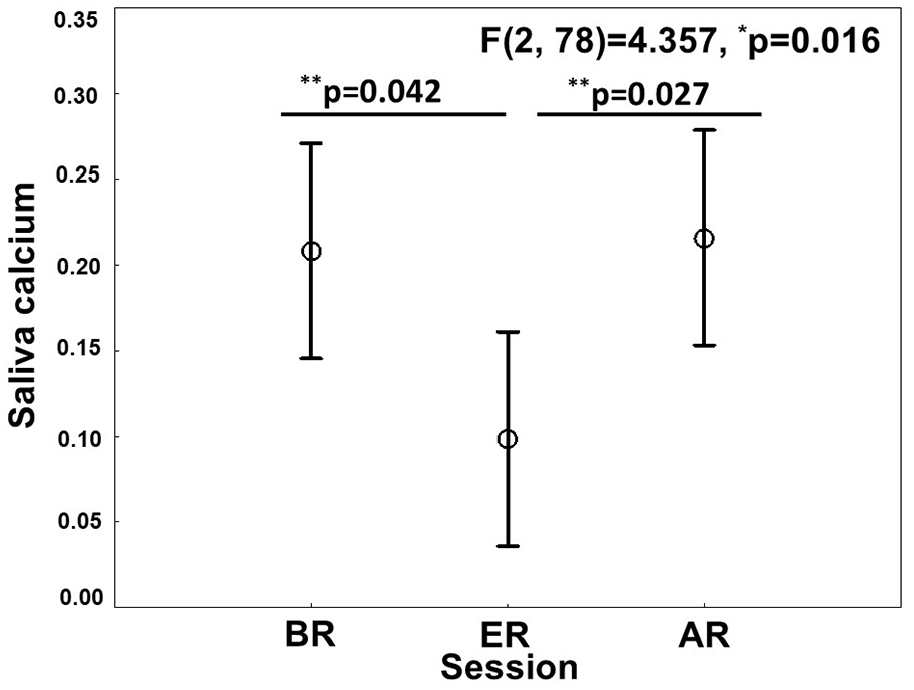
Effects of Ramadan Intermittent Fasting (RIF) on Saliva Calcium (n = 27 Healthy Men)
Discussion
The main findings of this pilot study revealed that RIF significantly influenced the salivary calcium concentration and pH in healthy men fasting during Ramadan 2023. Specifically, the salivary calcium concentration decreased significantly during the ER by 75% compared to BR and by 100% compared to AR. In addition, salivary pH increased by 1.7% during ER compared to BR. RIF did not cause any significant changes in SFR or the concentrations of other electrolytes such as chloride, phosphate, potassium, and sodium.
It is important to distinguish RIF from other types of fasting (Poursalehian et al., 2024). RIF is a unique form of religious intermittent fasting that significantly alters both eating and sleep patterns for an entire month, creating a specific fasting model that demands detailed physiological studies (Ben Saad, 2019; Poursalehian et al., 2024). While RIF has been associated with health benefits (Ben Saad, 2019), prolonged fasting (continuous fasting over multiple days) may have detrimental effects on health (Dote-Montero et al., 2022). The literature on the effects of RIF on salivary parameters is sparse (Al-Rawi et al., 2020; Bahammam et al., 2013; Dehaghi et al., 2020; Develioglu et al., 2013; Illahi et al., 2016; Khaleghifar et al., 2017; Rahim & Yaacob, 1991; Sariri et al., 2010). Only eight studies have examined these effects (Al-Rawi et al., 2020; Bahammam et al., 2013; Dehaghi et al., 2020; Develioglu et al., 2013; Illahi et al., 2016; Khaleghifar et al., 2017; Rahim & Yaacob, 1991; Sariri et al., 2010), each using different methodologies (Table 1S, Supplementary Appendix), and producing conflicting results (Table 2S, Supplementary Appendix).
Effect of RIF on SFR
There was no statistically significant effect of RIF on SFR (Table 1). Our findings contradict previous studies (Rahim & Yaacob, 1991; Sariri et al., 2010) (Table 2S, Supplementary Appendix). First, Rahim and Yaacob (1991) reported a 50% decrease in SFR in the fasting group (n = 22, abstinence from food and water for at least 6 hr before saliva collection) compared to the control group (n = 19). Second, Sariri et al. (2010) observed a 10% reduction in SFR due to RIF.
It is important to differentiate between hyposalivation and the sensation of dry mouth (Tenovuo, 1997). Hyposalivation is characterized by low saliva production, but it is not always associated with dry mouth symptoms (Tenovuo, 1997). Even individuals with normal SFR may experience oral dryness (Tenovuo, 1997). Saliva can evaporate, which may lead to a dry mouth sensation, particularly in mouth-breathers, a phenomenon that may occur during RIF (Tenovuo, 1997). Since saliva plays a crucial role in moistening the mouth, neutralizing acids produced by bacterial plaque, and clearing bacteria and food debris, any changes in saliva can create an environment conducive to the growth of bacteria, especially on the dorsum of the tongue (Alqumber & Arafa, 2014). Such changes may contribute to malodor during RIF, although other factors are involved, too (Bragazzi, 2015). In addition, the absence of gustatory stimulation during fasting decreases the activation of salivary glands, potentially reducing SFR (Besbes et al., 2022), as observed in two prior studies (Rahim & Yaacob, 1991; Sariri et al., 2010). While normal SFR is essential for maintaining oral health (Besbes et al., 2022; Singh et al., 2015), its reduction can lead to dental caries (Singh et al., 2015) and oral candidiasis (Besbes et al., 2022). SFR from major and minor salivary glands is primarily regulated by the autonomic nervous system (Leicht et al., 2018; Proctor & Carpenter, 2007; Tenovuo, 1997). Both parasympathetic and sympathetic stimulations depend on the nature of the stimulus, which may affect the secretion of saliva and its composition (Leicht et al., 2018). Parasympathetic stimulation generally produces large amounts of watery saliva, while sympathetic stimulation enhances protein production and secretion (Proctor & Carpenter, 2007; Tenovuo, 1997).
The discrepancies between our findings (no effect of RIF on SFR) and those of previous studies [which reported a reduction in SFR due to RIF (Rahim & Yaacob, 1991; Sariri et al., 2010)] could be attributed to several factors (Poursalehian et al., 2024): differences in fasting duration [e.g., 13.5 hr in Sariri et al. (2010) vs. 14.9 hr in our study], ambient temperature [e.g., 20°C in Sariri et al. (2010) vs. 18°C in our study], and season [e.g., autumn in Sariri et al. (2010) vs. spring in our study]. Physical activity levels may likewise play a role (Chamari et al., 2023). During Ramadan, a more sedentary lifestyle with reduced orofacial movement and slower metabolism in body tissues, including the oral cavity, may result in lower stimulation of the autonomic nervous system (Khaleghifar et al., 2017). This reduced SFR could lead to malodor, especially during fasting (Eldarrat et al., 2008). In our study, the stability of SFR may be explained by the fact that our young adult participants maintained regular physical activity throughout the study.
Effect of RIF on Salivary pH
Salivary pH values were significantly influenced by RIF, with pH levels being notably higher during the ER compared to BR (Table 1 and Figure 2). Our findings differ from a prior study by Illahi et al. (2016), which observed a decrease in salivary pH after participants broke their fast compared to while participants were fasting. In their study of 16 fasting students and 19 non-fasting controls, Illahi et al. (2016) reported that pH levels during fasting were significantly lower than after breaking the fast (7.26 ± 0.24 vs. 7.66 ± 0.23) (Table 2S, Supplementary Appendix). Illahi et al. (2016) suggested that fasting decreases salivary gland activity due to the lack of food and water intake, which subsequently reduces saliva production and alters the mastication process that normally influences salivary pH.
Our results are somewhat unexpected, as SFR typically affects pH levels, with higher SFRs usually resulting in a more alkaline pH (Abdul Khader & Dyasanoor, 2015). Under normal conditions, salivary pH generally ranges from 6.20 to 7.60 (Cichonska et al., 2022). In the present study, pH levels ranged from 5.75 to 7.78 BR and from 6.02 to 7.12 AR. Salivary pH is regulated by several buffering systems, including carbonic acid/bicarbonate, phosphate, and protein systems (Abdul Khader & Dyasanoor, 2015), which help maintain near-neutral pH levels even during periods of rest (salivary pH does not dip below 6.3 during rest) (Cichonska et al., 2022). Our findings are contrary to those of Illahi et al. (2016), who reported a pH of 7.26 during fasting, compared to our finding of 6.88 during fasting. An alkaline pH environment can promote the deposition of calcium and phosphate, which favors the mineralization of plaque and the formation of calculus (Wong et al., 2002). In addition, an alkaline environment is associated with increased proteolytic activity of Porphyromonas gingivalis, a gram-negative anaerobic bacterium commonly found in the gingival sulcus and linked to the development of periodontitis (Mysak et al., 2014). The optimal pH for the growth of P. gingivalis is around 7.5 (Zilm et al., 2010), suggesting that the higher pH observed during fasting may potentially enhance conditions for this pathogen, thus increasing the risk for periodontal diseases. This observed increase in salivary pH during RIF, despite stable SFR, might indicate other mechanisms at play, possibly influenced by changes in diet, oral habits, or metabolic responses during fasting. Further research is needed to clarify these interactions.
Effect of RIF on Salivary Electrolytes
Saliva consists of approximately 99% water, with the remaining 1% made up of proteins, organic compounds, and inorganic substances (Tjahajawati et al., 2021). The predominant electrolytes in saliva include calcium, magnesium, potassium, sodium, chloride, bicarbonate, and phosphate (Tjahajawati et al., 2021). Among the salivary electrolytes analyzed in our study, only calcium concentration led to significant changes. To the best of the authors’ knowledge, only Rahim and Yaacob (1991) have explored the effects of RIF on salivary electrolytes such as calcium and phosphate.
First, the salivary calcium concentration during the ER session was significantly lower than BR and AR sessions (Table 2 and Figure 3). This result contrasts with the findings of Rahim and Yaacob (1991), who reported that the calcium concentration in fasting saliva was similar to that of non-fasting controls (Table 2S, Supplementary Appendix). Calcium concentrations in saliva can vary throughout the day, even when the SFR is controlled, with differences observed between parotid and submandibular saliva (Dawes, 1972, 1975). Importantly, in our study, calcium levels returned to nearly BR values in the AR session, suggesting a temporary effect of RIF on salivary calcium (Table 2). Salivary calcium plays a crucial role in tooth demineralization and remineralization processes (van Santen et al., 2023). A reduction in calcium levels, such as that observed during the ER session, could increase the risk of dental caries (Borzuei et al., 2023). It is well established that calcium concentrations are typically lower in unstimulated saliva compared to stimulated saliva (Baumann et al., 2017). Given that RIF involves prolonged fasting periods with reduced salivary gland stimulation, it is possible that this decline in calcium concentration contributes to heightened vulnerability to tooth decay.
Second, we found no significant impact of RIF on salivary phosphate concentrations (Table 2). This finding aligns with Rahim and Yacoob’s report (1991) (Table 2S, Supplementary Appendix). The results of Rahim and Yaacob’s (1991) should be interpreted with caution due to methodological limitations, such as the lack of specificity regarding the timing of saliva collection during Ramadan. Besbes et al. (2022) excluded Rahim and Yaacob’s (1991) study from their systematic review for this reason. Phosphate is essential for enamel remineralization (Humphrey & Williamson, 2001), as it aids in the replenishment of minerals through the organic matrix to the crystals of enamel (Humphrey & Williamson, 2001). Phosphate has antimicrobial properties, inhibiting the binding of proteins to bacterial cell surfaces and contributing to the bactericidal effect of apolactoferrin (Almstahl & Wikstrom, 2003).
Finally, the lack of significant changes in sodium, potassium, chloride, and phosphate concentrations during RIF may reflect the body’s adaptive capacity during fasting periods (Poursalehian et al., 2024). Sodium, for instance, plays a role in saliva production (van Santen et al., 2023). Since RIF reduces salivary gland stimulation, leading to stable or decreased SFR, it is plausible that sodium levels remained unchanged throughout Ramadan. The ability of the human body to maintain electrolyte balance, even in fasting conditions, likely contributes to this stability.
Discussion of Methodology
Saliva collection for this study took place at 11:00 a.m. to avoid the potential confounding effects of circadian rhythms (Illahi et al., 2016), which are the natural “up and down” cycles of the body over a 24-hr period (Rudney, 1995). Circadian variations can impact SFR and the concentrations of electrolytes and proteins in saliva (Rudney, 1995). Since meal timing plays a critical role in regulating circadian rhythms (Iraki et al., 1997), RIF is expected to influence these biological cycles, including salivary variables. Some researchers have underestimated the effect of meal timing on circadian rhythms (Iraki et al., 1997).
Environmental factors play a significant role in altering salivary parameters (Besbes et al., 2022). These factors include temperature and humidity, which can affect the body’s hydration levels during fasting hours (Besbes et al., 2022). Their impact varies depending on geographic location, seasonal conditions, dietary habits, and cultural practices (Bilto, 1998) and may account for discrepancies between different studies.
Unstimulated saliva was chosen for collection in this study, as it is considered more reproducible than stimulated saliva (Pandey et al., 2015). Mechanical or gustatory stimulation can alter both the quantity and composition of saliva (Pandey et al., 2015). Saliva collection is non-invasive, easy to perform, safer compared to blood plasma sampling, and it has been used to measure various biological markers such as metabolites, hormones, and enzymes (Besbes et al., 2022). In this study, saliva was collected via the “spitting method,” where participants gently expelled saliva into a test tube (Develioglu et al., 2013), a technique found to be more convenient for participants (Pandey et al., 2015). Besbes et al. (2022) suggested that unstimulated saliva is preferable when saliva collection is performed at least once during Ramadan (Gomar-Vercher et al., 2018). Stimulated saliva, in contrast, is collected by chewing sterile paraffin (Gomar-Vercher et al., 2018). To preserve the integrity of the saliva sample and prevent degradation due to the loss of carbon dioxide when exposed to air, pH measurements were taken immediately after sample collection using a glass-electrode pH meter (Migliario et al., 2021). This device has the advantage of being able to measure the pH of a small volume of saliva, as little as 0.5 ml.
Several inclusion criteria were carefully applied to minimize confounding factors that could influence salivary characteristics (Besbes et al., 2022). Participants who smoked, consumed alcohol, had systemic diseases, took medications, or were obese were not included. First, studies reported that obese individuals tend to have a lower SFR compared to non-obese individuals (Modeer et al., 2010). Second, participants who regularly fast outside of Ramadan (e.g., fasting on Mondays and Thursdays) were not included, as their routine fasting could influence saliva parameters (Besbes et al., 2022). Finally, participants were instructed not to eat or drink for 3 hr before saliva collection and to maintain their usual diet during non-fasting hours.
Study Limitations
This study has some limitations. First, the lack of a non-fasting control group could introduce bias, as the observed variations in the assessed parameters may not be solely attributable to RIF (Besbes et al., 2022). Including non-fasting participants presents challenges, particularly due to religious considerations in Muslim countries (Besbes et al., 2022). For practical reasons, we opted to use participants’ own measurements outside the Ramadan period (e.g., BR and AR) as a control (Besbes et al., 2022). This approach allowed us to more easily manage variables than arranging a separate non-fasting group. Second, the use of convenience sampling presents a potential confounder (Sousa et al., 2004). This non-probability sampling method relies on the investigator’s judgment to select participants (Sousa et al., 2004). Although it is cost-effective and practical, making it an appealing option for researchers, it can result in the overrepresentation or underrepresentation of certain groups. This limitation can affect the generalizability of findings to the broader population (Sousa et al., 2004), and the results obtained from our convenience sampling should be interpreted with caution. Third, the collection of whole saliva, which comprises not just pure secretions from the salivary glands, is a subject of debate (Dawes et al., 2015). Whole saliva is a mixture that includes various non-salivary constituents such as gingival crevicular fluid, expectorated bronchial and nasal secretions, intraoral bleeding (serum and cells), non-adherent microorganisms and their products, desquamated epithelial cells, and food debris (Dawes et al., 2015). The composition of this mixed saliva can vary considerably depending on the contributions from these different sources. Despite this complexity, whole saliva remains the most frequently studied type for salivary analysis due to its ease of collection and non-invasive nature (Dawes et al., 2015). Fourth, evaluating participants’ hydration status during the three sessions would have been beneficial (Besbes et al., 2022). The observed differences in salivary calcium concentration could be partly attributed to variations in hydration status, which can affect salivary composition and SFR (Allgrove et al., 2014). Adjusting the results for factors such as total protein content, saliva osmolality, SFR, and saliva secretion rate would have provided a more comprehensive understanding of the data (Lindsay & Costello, 2017). Fifth, it was preferable to evaluate objectively (e.g., via valid questionnaires) the diet regime of participants for at least two reasons: (a) RIF involves modifications in eating schedules (i.e., eating one large meal after sunset and at least one lighter meal before dawn, food quantity and quality, nocturnal food consumption, and meal frequency) (Barkia et al., 2011) and (b) dietary abnormalities significantly influence both SFR and salivary parameters, with effects ranging from reduced production to changes in pH and protective properties (Javaid et al., 2016; Sheiham, 2001; Yee, 2017). Sixth, it was desirable to estimate objectively (via valid questionnaires) the physical activity status of the participants, since physical activity has been associated with variations in SFR and salivary composition (Ligtenberg et al., 2015, 2016). Finally, when comparing multiple variables, it is probable that some will appear statistically significant purely by chance (Bland & Altman, 1995). Implementing stricter significance criteria than the usual 5% (e.g., Bonferroni adjustment) would be advisable (Subhan, 2008; Subhan et al., 2006). For example, with seven variables compared in our study, achieving statistical significance would require a p-value of .05/7 = .0071 (Subhan, 2008). If this method had been employed in our study, our conclusions regarding pH and calcium would have been changed, suggesting that RIF does not affect these parameters. The Bonferroni method is often criticized for being overly conservative, potentially missing real differences (Bland & Altman, 1995; Perneger, 1998). Given that our findings are exploratory in nature, we chose not to apply this adjustment. Future confirmatory studies will be necessary to validate our results (Bender & Lange, 1999).
Conclusion
Among healthy young men fasting during the holy month of Ramadan in 2023, RIF was found to decrease salivary calcium, increase salivary pH, and have no significant effect on the salivary concentrations of potassium, sodium, chloride, or phosphate. Although the change in pH was statistically significant, it was not clinically significant, as the pH values remained within the normal range. Regarding salivary calcium, the absence of reference values precludes determination of the clinical impact of the findings. The limited existing literature on RIF underscores the importance of future research to explore its impacts on saliva secretion, particularly regarding immunological parameters and salivary hormones.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883241312396 – Supplemental material for Impact of Ramadan Intermittent Fasting on Salivary pH, Flow Rate, and Electrolyte Levels in Healthy Adult Men
Supplemental material, sj-docx-1-jmh-10.1177_15579883241312396 for Impact of Ramadan Intermittent Fasting on Salivary pH, Flow Rate, and Electrolyte Levels in Healthy Adult Men by Sinda Yacoub, Mehdi Khemiss, Amira Besbes and Helmi Ben Saad in American Journal of Men's Health
Footnotes
Acknowledgements
The authors wish to express their sincere gratitude to all participants for their cooperation and Ms Ibtissem Jelizi for the biochemical analysis. They also would like to express their sincere gratitude to the two reviewers for their excellent feedback, which has substantially improved the quality of this work. Their insightful comments and constructive suggestions were invaluable in refining our manuscript.
Author Contributions
SY, MK, AB, and HBS contributed to the literature search, data collection, study design, data analysis, preparation, and review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors wish to disclose that an artificial intelligence tool (ChatGPT ephemeral) was utilized to enhance the clarity and coherence of the manuscript writing. The tool was utilized for language refinement purposes only, ensuring the text was clear and coherent without altering the scientific content or generating any new text (Dergaa et al., 2023).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Approval for the study was obtained from the Institutional Ethics and Research Committee of the University Hospital Farhat Hached (IORG 0007439, reference: ERC 02082024). The study was conducted in accordance with the World Medical Association’s Declaration of Helsinki for research involving human subjects.
Informed Consent/Patient Consent
Each participant signed an informed consent form after receiving a detailed explanation of the study’s characteristics.
Trial Registration Number/Date
Not applied.
Grant Number
Not applied.
Data Availability Statement
Data will be available upon request from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
