Abstract
This study aims to investigate the effect and mechanism of cyclosporine A (CsA) on paclitaxel-resistant prostate cancer cells. Paclitaxel-resistant prostate cancer cell lines were established by gradual increment method. The proliferation of cells was tested using MTT and colony formation assay. Western blot was used to detect protein expression. Expression levels of gene mRNA were detected using real-time polymerase chain reaction (RT-PCR). Xenografts in nude mice were used to validate the conclusion in vitro. The results showed that CsA could increase the sensitivity of prostate cancer cells to paclitaxel. Treatment of paclitaxel-resistant prostate cancer cell lines with CsA gradients decreased metadherin (MTDH) protein expression. RT-PCR showed that CsA could decrease the mRNA level of MTDH. Overexpression of MTDH in prostate cancer cells increases paclitaxel resistance in prostate cancer cells. Conversely, knockdown of MTDH reduced paclitaxel resistance in prostate cancer cells. Treating cells with CsA failed to reduce paclitaxel resistance in prostate cancer cells when MTDH was overexpressed. Xenografts in nude mice yielded consistent conclusions with the in vitro results. In conclusion, CsA can reduce the resistance of prostate cancer cells to paclitaxel. In vitro and in vivo experiments have shown that CsA can reduce paclitaxel resistance in prostate cancer cells by decreasing MTDH expression. In clinical practice, CsA can be used in combination with paclitaxel to improve the therapeutic effect on prostate cancer. MTDH may serve as a novel target for treating paclitaxel resistance in prostate cancer.
Introduction
Prostate cancer is a malignant tumor that occurs in the prostate glands of men, and it is one of the most common cancers in men (Barsouk et al., 2020). Prostate cancer usually develops slowly and may have no obvious symptoms in the early stages, but symptoms such as dysuria, frequent urination, urgency, and hematuria may occur as the disease progresses (Wasim et al., 2022). The incidence of prostate cancer increases with age, particularly in men over 50 years of age. There are various treatments for prostate cancer, including surgery, radiation therapy, hormonal therapy, and chemotherapy (Desai et al., 2021).
Paclitaxel is an antineoplastic agent widely used in clinical practice, and it has a unique anti-microtubule mechanism of action. Paclitaxel inhibits normal dynamic reorganization of microtubules by stabilizing microtubule structure by promoting the combination of tubulin dimers and preventing their depolymerization. This results in cell cycle blockade in G2/M phase and prevents replication of tumor cells (Gouda et al., 2024; Lee et al., 2024). At present, paclitaxel is widely used in the treatment of a variety of cancers, including ovarian cancer, breast cancer, lung cancer, head and neck cancer, prostate cancer, esophageal cancer, gastric cancer, and soft tissue sarcoma (Demaziere et al., 2024; Lee et al., 2024; Wu et al., 2024). Resistance to paclitaxel, however, is an important issue in cancer therapy, and it limits the clinical use of paclitaxel (Tashakori et al., 2024; Yan et al., 2024).While docetaxel and cabazitaxel are the standard taxanes used clinically for advanced prostate cancer (Tannock et al., 2004), paclitaxel remains an important model compound for studying taxane resistance mechanisms (Orr et al., 2003). Paclitaxel shares the same core mechanism of action as docetaxel and cabazitaxel, stabilizing microtubules and disrupting cell division (Jordan & Wilson, 2004). As such, insights gained from paclitaxel resistance studies may inform our understanding of taxane resistance more broadly.
Metadherin (MTDH), also known as AEG-1 or LYRIC, has emerged as an important player in cancer progression and chemoresistance (Hu et al., 2009). Previous studies have implicated MTDH in resistance to various chemotherapeutic agents, including paclitaxel, in breast and ovarian cancers (Li et al., 2011).
Cyclosporine A (CsA) is a cyclic polypeptide produced by specific fungi and has a powerful immunosuppressive effect. CsA consists of 11 amino acids forming a ring structure. 1, 2, 3, 10, and 11 amino acids are binding regions to cyclosporine, while 3 to 9 amino acids are effector regions that interact with calcineurin (Ganugula et al., 2024). The main mechanism by which CsA acts is to form a complex with cyclosporine, which then interacts with calcineurin to inhibit the dephosphorylation of NF-AT so that it cannot enter the nucleus, thereby inhibiting the production of interleukin-2 (IL-2), which in turn inhibits the production of T lymphocytes (Saqib et al., 2024; Abduh, 2024). CsA is widely used in clinical practice as an immunosuppressive agent, especially in the field of organ transplantation, and plays an important role in the development of organ transplantation therapy (Rykowska et al., 2024). CsA has also been investigated in recent years to reverse resistance of tumor cells to certain chemotherapeutic agents, including paclitaxel. Clinical studies have investigated the safety and efficacy of CsA combined with paclitaxel. For example, in patients with metastatic breast cancer, oral paclitaxel combined with CsA treatment showed safety and was able to be adequately exposed to paclitaxel, thereby potentially reversing resistance (Parekh et al., 2020). However, the effect of CsA on paclitaxel-resistant prostate cancer cells and its molecular mechanism are still unclear. The aim of this study was to investigate the effect and mechanism of CsA on paclitaxel resistance in prostate cancer cells.
Method
Cell Culture
Prostate cancer cell lines PC-3 and LNCaP were purchased from the cell bank of Shanghai Cell Institute, Chinese Academy of Sciences. DMEM high glucose medium containing 10% fetal bovine serum was used and cells were cultured in an incubator containing 5% CO2 at 37°C, and the culture medium was changed every 2 to 3 days. 0.25% trypsin was used for digestion and passage.
Establishment of Paclitaxel-Resistant Cell Lines
Paclitaxel-resistant cell lines (PC-3-R, LNCaP-R) were established by treatment with increasing concentrations of paclitaxel at low doses (Daniel et al., 2024). At the beginning, paclitaxel was removed after three stable passages at a starting concentration of 0.1 nmol/L paclitaxel in the medium and routinely cultured for 1 week. Paclitaxel drug concentration was then increased to 1 nmol/L, and the drug was removed after three stable passages and cultured for another week. This process was repeated with gradually increased paclitaxel concentrations of 2, 5, and 10 nmol/L until the cells could grow stably in medium containing 10 nmol/L paclitaxel. Cells were then treated with high-dose paclitaxel pulses every 3 days for 24 hr using medium containing 20 nmol/L paclitaxel until cells could grow stably in medium at a concentration of 20 nmol/L.
MTT Assay
Cell proliferation rate was measured using MTT assay. First, cells were seeded into 96-well plates and MTT was added at different time points. Cells were treated with 0.5 mg/mL MTT for 4 hr and solubilized with dimethyl sulfoxide (DMSO) at the end of MTT treatment. DMSO treatment time was 30 min. Finally, measurements were performed at a wavelength of 490 nm using a microplate reader.
Quantitative Real-Time PCR
Total RNA was extracted using Trizol after cell culture until the cell density was 85% to 95% or after cell treatment. The extracted total RNA was reverse transcribed using the MonScriptTM RT II All-in-One Mixwith dsDNase kit. Reverse transcribed cDNA was subjected to RT-PCR. The mixtures were incubated at 50°C for 15 min, followed by 95°C for 5 min and then 35 PCR cycles performed profiles with the following temperature profiles: 95°C for 15 s, 60°C for 30 s, and 72°C for 1 min. The gene expression values were normalized to those of glyceraldehyde-3-phosphate dehydrogenase (GAPDH). PCR primer sequences were as follows: GAPDH: F: GGAGCGAGATCCCTCCAAAAT, R: GGCTGTTGTCATACTTCTCATGG.MTDH: F: AAATGGGCGGACTGTTGAAGT, R: CTGTTTTGCACTGCTTTAGCAT.
Western Blot
Western blot was used to detect protein expression. First, cell lysis was performed using cell lysate. Total intracellular protein was extracted following cell lysis. Total protein content was measured using the bicinchoninic acid (BCA) assay. Separation of total proteins was performed using polyacrylamide gel electrophoresis. After completion of electrophoresis, PVDF membranes were used for transfer and proteins were transferred to PVDF membranes. Blocking was performed with 5% bovine serum albumin solution after transfer of membranes was completed. Following completion of blocking, overnight incubations at 4°C were performed with the corresponding primary antibodies. Primary antibody information was as follows: GAPDH (2118, CST, MA, USA); MTDH (14065, CST, MA, USA). The next day, after completion of primary antibody incubation, the secondary antibody was incubated at room temperature for 30 min. Finally, a chromogenic reaction was performed.
Colony Formation Assay
Prostate cancer cells were seeded at 2,000 cells/well in 6-well plates and cultured for 14 days to form colonies. Cells were subsequently washed with phosphate-buffered saline (PBS), fixed using 4% paraformaldehyde for 15 min at 37°C, stained using 0.5% crystal violet staining solution for 1 hr, washed three times with ddH2O, and photographed.
Xenograft Experiments in Nude Mice
PC-3-R cells were trypsinized, washed with PBS, and resuspended in PBS. Trypan blue was used to determine viable cell number and viability. Fifteen 6-week-old female nude mice were randomly divided into three groups (five mice/group) and then subcutaneously injected with PC-3-R cells at a density of 1 × 106 cells/site. Treatment was then administered every 2 days with paclitaxel at 10 mg/kg. Tumor size was measured every 2 to 3 days. Tumor volume was calculated using the following formula: 1/2 (length × width2). Nude mice were sacrificed and tumor weights were measured after tumor dissection.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 8.0. Data were presented as the mean ±SD. Quantitative real-time PCR assay was analyzed by one-way analysis of variance (ANOVA) with Dunnett’s multiple comparisons post hoc test n = 3 replicates. Experimental results of MTT assay were analyzed by two-way ANOVA with Dunnett’s multiple comparisons post hoc test, n = 3 replicates. Experimental results of tumor weight and tumor volume were analyzed by one-way ANOVA with Dunnett’s multiple comparisons post hoc test, n = 5 mice. p values < .05 were considered to be statistically significant.
Results
CsA Reduces Paclitaxel Resistance in Prostate Cancer Cells
To investigate the effect of CsA on paclitaxel-resistant prostate cancer cells, two paclitaxel-resistant cell lines PC-3-R and LNCaP-R were constructed by low-dose increasing concentration method. Subsequently, MTT assay was used to detect the effect of paclitaxel on the proliferation of resistant cell lines and wild-type cell lines. The results showed that paclitaxel significantly inhibited the growth and proliferation of wild-type prostate cancer cells, while the inhibitory effect on paclitaxel-resistant cell lines was much less than that of wild-type prostate cancer cells (Figure 1A and B). Subsequent experiments using paclitaxel-resistant cell lines significantly inhibited the growth and proliferation ability of prostate cancer paclitaxel-resistant cell lines when treated simultaneously with CsA and paclitaxel (Figure 1C and D). The above results showed that CsA could reduce paclitaxel resistance in prostate cancer cells and improve the therapeutic effect of paclitaxel.
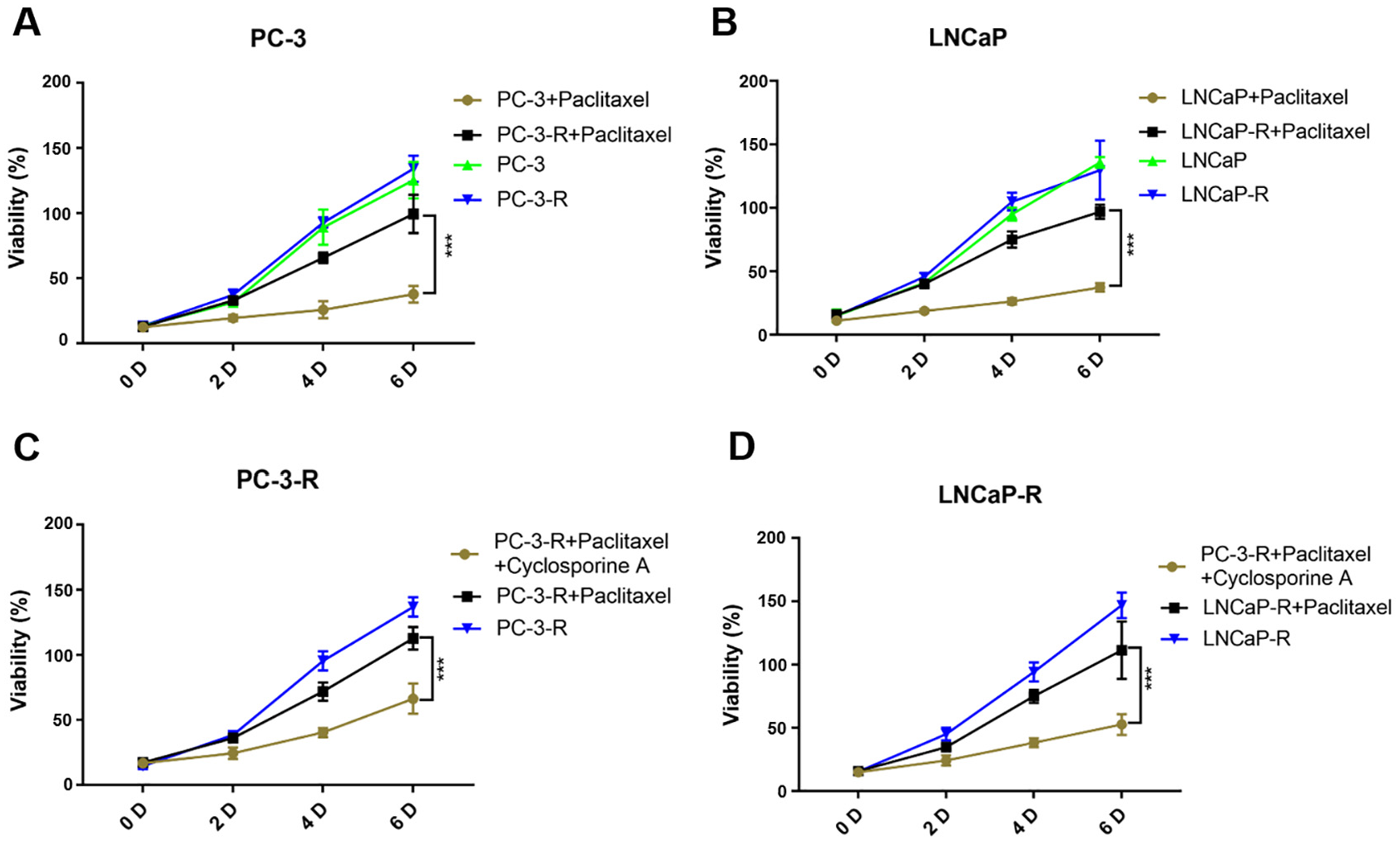
CsA Reduces Paclitaxel Resistance in Prostate Cancer Cells. (A, B) Establishment of Paclitaxel-Resistant Cell Lines PC-3-R (A) and LNCaP-3-R (B). (C, D) CsA Can Reduce Paclitaxel Resistance in PC-3-R (C) and LNCaP-3-R (D)
CsA Reduces MTDH Protein Expression
After discovering that CsA could reduce paclitaxel resistance in prostate cancer cells, we further explored the molecular mechanisms. By Western blot and RT-PCR, we found that prostate cancer cells treatment with paclitaxel reduced the expression of MTDH (Figure 2A and B). And we also found that the treatment of paclitaxel-resistant prostate cancer cells with CsA gradient resulted in a gradient decrease in MTDH protein expression, with the same results obtained in two different prostate cancer cell lines (Figure 2C and D). Subsequently, RT-PCR showed that treatment with CsA decreased the mRNA expression of MTDH (Figure 2E and F). In paclitaxel-resistant prostate cancer cells, CsA decreased the expression of MTDH protein.

CsA Can Reduce MTDH Protein Expression. (A) Western Blot Results for Expression of MTDH in Cells. (B) mRNA Expression of MTDH in Cells. (C, D) Treatment of PC-3-R (C) and LNCaP-3-R (D) Cells With a CsA Gradient for 48 hr Decreased MTDH Protein Expression. (E, F) Treatment of PC-3-R (E) and LNCaP-3-R (F) With a CsA Gradient for 48 hr Decreased the mRNA Expression Level of MTDH
Overexpression of MTDH Increases Paclitaxel Resistance in Prostate Cancer Cells
To further investigate the role of MTDH in paclitaxel-resistant prostate cancer cells, we constructed MTDH-overexpressed (MTDH-OE) prostate cancer cells. Western blot results showed that MTDH-OE cells were successfully constructed (Figure 3A and B). To confirm that the effect of CsA on prostate cancer cells was achieved by MTDH, MTT was used for validation. MTT results showed that treatment with CsA did not affect paclitaxel-resistant prostate cancer cells after overexpression of MTDH (Figure 3C and D). The results showed that CsA affected the resistance of prostate cancer cells to paclitaxel through the expression of MTDH protein. MTT results also showed that MTDH protein increased paclitaxel resistance in prostate cancer cells. The same results were obtained using colony formation assay (Figure 3E and F).

Overexpression of MTDH Increases Paclitaxel Resistance in Prostate Cancer Cells. (A, B) Validation of Western Blot Results for Overexpression of MTDH in PC-3-R (A) and LNCaP-3-R (B) Cells. (C, D) Overexpression of MTDH Increases the MTT Results of Paclitaxel Resistance in PC-3-R (C) and LNCaP-3-R (D) Cells. (E, F) Overexpression of MTDH Increases the Colony Formation Number of PC-3-R (E) and LNCaP-3-R (F) Cells
Knockdown of MTDH Decreases Paclitaxel Resistance in Prostate Cancer Cells
To fully confirm the effect of MTDH on paclitaxel-resistant prostate cancer cells, after completion of experiments overexpressing MTDH, we further knock down MTDH. Western blot results showed that we successfully constructed MTDH knockdown cell lines (Figure 4A and B). MTT results showed that prostate cancer cells had reduced resistance to paclitaxel after MTDH knockdown. Treatment of MTDH knockdown prostate cancer cells with paclitaxel effectively inhibited their growth and proliferation (Figure 4C and D). The same results were obtained using colony formation assay (Figure 4E and F).

Knockdown of MTDH Reduces Paclitaxel Resistance in Prostate Cancer Cells. (A, B) Validation of Western Blot Results for MTDH Knockdown in PC-3-R (A) and LNCaP-3-R (B) Cells. (C, D) Knockdown of MTDH Reduced the MTT Results of Paclitaxel Resistance in PC-3-R (C) and LNCaP-3-R (D) Cells. (E, F) Knockdown of MTDH Reduced the Colony Formation Number of PC-3-R (E) and LNCaP-3-R (F) Cells
MTDH Reduced Paclitaxel Resistance in Prostate Cancer Cells In Vivo
After in vitro experiments showed that CsA could influence paclitaxel resistance in prostate cancer cells by affecting MTDH, we further validated the results in vivo using xenografts in nude mice treatment with paclitaxel. Experiments were performed using PC-3-R cell lines, and paclitaxel treatment was administered to nude mice following overexpression and knockdown of MTDH. The results showed that knockdown of MTDH significantly reduced paclitaxel resistance in PC-3-R cell lines and significantly slowed tumor growth rate (Figure 5A). Tumor volume and tumor weight also changed significantly (Figure 5B and C). Subsequently, we treated PC-3-R cells using paclitaxel and CsA. The results showed that CsA treatment significantly increased the sensitivity of PC-3-R cells to paclitaxel (Figure 5D–F). The Western blot results showed that CsA treatment could reduce MTDH expression (Figure 5G). The above results showed that MTDH could also significantly affect the resistance of prostate cancer cells to paclitaxel in vivo.

MTDH Reduces Paclitaxel Resistance in Prostate Cancer Cells In Vivo. (A) Nude Mouse Xenograft Experiments Were Performed Using PC-3-R Cells Treatment With Paclitaxel. n = 5 Mice. (B) Statistics of Tumor Volume in Xenograft Experiments in Nude Mice. (C) Statistics of Tumor Weight in Xenograft Experiments in Nude Mice. (D) Nude Mouse Xenograft Experiments Were Performed Using PC-3-R Cells Treatment With Paclitaxel. n = 5 Mice. (E) Statistics of Tumor Volume in Xenograft Experiments in Nude Mice. (F) Statistics of Tumor Weight in Xenograft Experiments in Nude Mice. (G) Expression of MTDH in Tumors
Discussion
Paclitaxel is a natural antineoplastic drug extracted from plants, and its main mechanism of action is to bind to tubulin and enhance the stability of microtubules, thereby interfering with the mitotic process of cells, leading to cell cycle arrest and apoptosis. Paclitaxel is widely used in the treatment of a variety of cancers in clinical practice, including breast cancer, ovarian cancer, and non-small cell lung cancer (Noronha et al., 2023; Yu et al., 2023). The clinical use of paclitaxel is not limited to a single agent, but is often combined with other chemotherapeutic agents to improve the therapeutic effect. In the treatment of tumors, the use of paclitaxel is often prone to paclitaxel resistance, resulting in a decrease in the therapeutic effect of paclitaxel (H. Wang et al., 2022). At present, the molecular mechanism of paclitaxel resistance remains to be further investigated. In this study, we found that in prostate cancer, the use of CsA treatment can enhance the sensitivity of prostate cancer cells to paclitaxel and help enhance the therapeutic effect of paclitaxel. Through further molecular mechanism exploration, we found that CsA could affect the resistance of prostate cancer cells to paclitaxel by affecting the expression of MTDH protein. CsA can inhibit MTDH protein expression in prostate cancer cells. It was also further confirmed that MTDH could affect the resistance of prostate cancer cells to paclitaxel by overexpression and knockdown of MTDH in prostate cancer cells.
CsA is a widely used immunosuppressive agent in clinical practice. It is essentially a cyclosporine consisting of 11 amino acids (Matsushita et al., 2024). The main mechanism of action of CsA is to form a complex with cyclosporine, which in turn interacts with calcineurin to inhibit the dephosphorylation of NF-AT, thereby inhibiting the production of interleukin-2, and then inhibiting the activation of T lymphocytes (Ye et al., 2024). Because of its immunosuppressive effect, CsA is widely used in areas such as organ transplantation. In addition to its use in organ transplantation, CsA is also used to treat autoimmune diseases, such as rheumatoid arthritis and Behcet’s disease. In addition, it also has some efficacy against type I diabetes, psoriasis, and parasitic diseases such as malaria and schistosomes (Ishimaru et al., 2024; J. Wang et al., 2023). In cancer therapy, CsA also has the effect of anti-tumor multidrug resistance, which can act by binding to P-glycoprotein, inhibiting its transport function, or reducing its binding affinity to drugs (Lisak et al., 2023). CsA can be combined with paclitaxel to achieve better tumor treatment. CsA has been reported to reverse paclitaxel resistance by inhibiting Pg protein in the treatment of breast cancer, allowing paclitaxel to exert better results during the treatment of breast cancer (Vahedi et al., 2017). In addition, in breast cancer, CsA was reported to reverse paclitaxel resistance in breast cancer by enhancing paclitaxel-induced apoptosis by interfering with the PI3 kinase-AKT signaling pathway. CsA combined with paclitaxel can inhibit AKT activation and BAD phosphorylation, enhance the activation of caspase-9 and caspase-3, lead to the loss of mitochondrial membrane potential, and then promote apoptosis (Siddiqui et al., 2023). However, the role of CsA in prostate cancer is currently unknown. In this study, we found that in prostate cancer, CsA can also reverse paclitaxel resistance and make paclitaxel play a better role in the treatment of prostate cancer.
MTDH is a protein closely related to tumor development, metastasis, and resistance to therapy (Yang et al., 2024a). MTDH can promote the tumor cell high frequency and metastasis, at the same time, it has strong angiogenesis ability, and has immune escape ability. MTDH is able to help tumor cells abut distant blood vessels and is important for the spread and metastasis of cancer (Todorović & Amedei, 2024). In breast cancer, MTDH protein has been reported to be positively associated with poor patient prognosis, and inhibition of MTDH protein reduces metastasis and recurrence of breast cancer and can be used as a novel drug target or biomarker (Yang et al., 2024b). During chemotherapy, the high expression of MTDH leads to the phenomenon that patients are highly susceptible to chemotherapeutic drug resistance. As a membrane protein, it can be targeted by antibodies, which provides the possibility for the clinical development of therapeutic approaches against MTDH. At the same time, it has been found that the MTDH-SND1 complex plays a role in promoting tumors by binding and destroying Tap1/2 messenger RNA, reducing tumor antigen presentation, and inhibiting T-cell infiltration and activation (Shen et al., 2022). Development of paclitaxel resistance has also been associated with MTDH in multiple paclitaxel-resistant tumors. In breast cancer, lncRNA OTUD6B-AS1 promotes paclitaxel resistance in triple-negative breast cancer by regulating autophagy and genomic instability mediated by the miR-26a-5p/MTDH pathway (Li et al., 2021). In addition, it has also been reported that knockdown of MTDH in breast cancer decreased paclitaxel resistance in breast cancer cells (Yang et al., 2018). These studies have been consistent with the conclusions in this study. In the present study, we showed that treatment with CsA could decrease MTDH protein expression in prostate cancer cells, thereby reducing paclitaxel resistance. MTDH promotes paclitaxel resistance in prostate cancer cells as shown by overexpression of MTDH. Conversely, knockdown of MTDH reduced paclitaxel resistance in prostate cancer cells.
The differential response of endogenous and exogenous MTDH to CsA treatment observed in our study likely reflects the distinct regulatory mechanisms governing their expression. Endogenous MTDH is subject to the cell’s intrinsic transcriptional and post-transcriptional control mechanisms, which may be sensitive to CsA. In contrast, exogenous MTDH, typically expressed from a strong promoter on a plasmid vector, largely bypasses these regulatory pathways.
While our study focuses on MTDH’s role in taxane resistance, it is important to note that MTDH has been implicated in various aspects of prostate cancer progression. For instance, MTDH has been shown to promote epithelial-to-mesenchymal transition in prostate cancer cells (Liu et al., 2016), enhance angiogenesis through regulation of VEGF expression, and contribute to castration resistance by modulating androgen receptor activity (Kikuno et al., 2022). Our findings on MTDH’s role in taxane resistance add to this growing body of evidence highlighting MTDH as a key player in prostate cancer biology and a potential therapeutic target.
There are still some limitations in this study. First, molecular mechanisms regarding how CsA decreases MTDH expression remain to be further investigated. Second, the presence of high MTDH expression in samples from clinical patients with paclitaxel-resistant prostate cancer requires further verification by collecting samples from clinical patients. Finally, the conclusion of the study should be further validated using transgenic mouse models with MTDH knockout.
Conclusion
In this study, paclitaxel-resistant prostate cancer cell lines were constructed, and it was experimentally found that treatment with CsA could reduce paclitaxel resistance in prostate cancer cells. Molecular mechanism of CsA can affect paclitaxel resistance in prostate cancer cells by decreasing the expression of MTDH protein. In vitro and in vivo experiments have confirmed that MTDH can promote paclitaxel resistance in prostate cancer cells. Clinically, the combination of CsA and paclitaxel is beneficial to promote the therapeutic effect of paclitaxel, and at the same time, MTDH can be used as a new target for the treatment of paclitaxel-resistant prostate cancer.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Hebei Province 2019 Medical Science Research Project Plan (20190976).
