Abstract
Breast cancer is rare in men and is managed using strategies similar to those for breast cancer in women. This study retrospectively analyzed the clinicopathological features, treatment, and survival of male breast cancer (MBC). A total of 66 patients with MBC admitted into Xijing Hospital from August 2006 to March 2024 were reviewed. Data were collected from patients’ hospital records and breast cancer database of Xijing Hospital. The incidence of MBC tended to increase from 2018, with affected individuals being older than those with female breast cancer (FBC). The most common histological type of MBC was invasive carcinoma, with positive hormone receptor status. A total of 62 (93.9%) patients with MBC underwent modified radical mastectomy. Chemotherapy was administered to 39 (59.1%) patients, while endocrine therapy was received by 14 patients (21.2%) and radiotherapy by nine patients (13.6%). Survival analysis indicated that the median overall survival (OS) of patients with MBC was 46.7 months (0.9–184.8 months). As of the latest data, 58 patients (87.9%) with MBC are alive. Factors significantly associated with survival included age (χ2 = 3.856, p = .050), estrogen receptor (χ2 = 10.427, p = .005), molecular types (χ2 = 10.641, p = .031), P63 (χ2 = 2.631, p < .001), and endocrine therapy (χ2 = 31.167, p < .001). These findings provide valuable insights into MBC within the Chinese population and serve as a reference for the standard treatment of MBC.
Introduction
Male breast cancer (MBC) is a rare malignancy, accounting for less than 1% of all cancers in men and less than 1% of all breast cancers (Gucalp et al., 2019). The lifetime risk of breast cancer for a man is approximately 1 in 1,000, compared with 1 in 8 for women (Giordano, 2018). Due to the lack of suspicion regarding breast cancer, men often experience delays in diagnosis when they discover a lump in the breast (Mattarella, 2010). Despite its rarity, the incidence of MBC has increased in recent years. Duso et al. (2020) have reported a steadily rising incidence of MBC. In 2023, J. Lee et al. (2023) found that the number of study registrants increased every 5 years from 1995 to 2016. Abdelwahab Yousef noted that the incidence of MBC is rising with some patient groups reaching a lifetime risk of 15%. Recently, knowledge about MBC is predominantly derived from Western countries, resulting in a lack of information regarding MBC in the Chinese population.
The biological differences, such as hormone regulation, may contribute to the distinct clinicopathologic characteristics and treatment responses observed in MBC. Recently, the causes, optimal treatments, and medical/psychosocial sequelae of breast cancer in men remain poorly understood (Ruddy & Winer, 2013). The major risk factors for the development of MBC were also unclear. Abdelwahab Yousef identified advancing age, hormonal imbalance, radiation exposure, and family history as potential risk factors (Abdelwahab Yousef, 2017). Fox noted that the risk factors for MBC include increased longevity, obesity, testicular diseases, and tumors, as well as germline mutations of BRCA2 (Fox et al., 2022). Furthermore, the treatment of breast cancer in men presents unique challenges. The management of MBC is often extrapolated from treatment protocols used for postmenopausal female breast cancer (FBC) patients (Giordano et al., 2004). Therefore, comprehensive guidelines for the management of MBC are necessary, which require further clinical evidence.
Given the low incidence of MBC, previous studies have often been hindered by small sample sizes, and short follow-up periods, which limit their interpretability (Yao et al., 2022). Due to the scarcity of clinical trials focused on MBC, real-world data analysis has become a common method for providing evidence for this patient population. In this study, we retrospectively reviewed real-world MBC data from Xijing Hospital, encompassing epidemiological data, diagnostic characteristics, pathological features, treatment strategies, and prognosis over an 18-year period, thereby contributing valuable information on MBC within the Chinese context.
Method
Patients
This is a retrospective, single-center study. Patients with MBC were searched from breast cancer database (n = 13,690) of Xijing Hospital from 2006 to 2024. Finally, 66 patients primarily diagnosed as MBC were enrolled, and the medical records of male patients with histologically confirmed breast cancer were retrospectively reviewed, such as demographics and pathologic characteristics, detailed treatment pattern, and clinical outcomes. The inclusion criteria were: (1) pathologically confirmed breast cancer and (2) male patients. Patients with unclear pathological characteristics were excluded. This study was approved by the ethical committee of Xijing Hospital, The Air Force Medical University (KY20232266-F-1).
Classification of Molecular Subtype
Estrogen receptor (ER) and progesterone receptor (PR) positivity was defined by American Society of Clinical Oncology/College of American Pathologists guideline (ASCO/CAP guideline). HER2-positive was defined as HER2 immunohistochemistry (IHC) 3+ or HER2 IHC 2+ with HER2 fluorescence or silver in situ hybridization positive. Luminal A subtype was defined as ER- and/or PR-positive and HER2-negative with Ki-67 ≤14%. Luminal B subtype was defined as ER- and/or PR-positive, HER2-negative with a Ki-67 index >14%. HER2-enriched subtype was defined as HER2 positive based on IHC of fluorescence in situ hybridization, irrespective of the ER or PR status. Triple-negative breast cancer (TNBC) was defined as ER-, PR-, and HER2-negative.
Survival Evaluation
Disease-free survival (DFS) was defined as the period from the time of primary treatment, such as surgical resection, to the date of disease recurrence or death from any cause. Overall survival (OS) was described as the period between the date of the first pathologic diagnosis and death or last follow-up.
Statistical Analysis
Continuous variables are presented as median values, and categorical variables are presented as percentages. Continuous variables were compared using the Mann–Whitney U test, while categorical variables were compared using the chi-square test and Fisher exact test. Survival analyses were performed using the Kaplan–Meier method and compared using the log-rank test. Hazard ratios for DFS and OS were estimated using the Cox proportional hazards model with a 95% confidence interval (CI). Two-sided p-values are presented for all analyses with p < .05 considered to be statistically significant.
Results
Baseline Characteristics
Finally, 66 patients with MBC were enrolled in the study from August 2006 to March 2024. Baseline patient characteristics are described in Table 1. The average age of the patients with MBC was 60.6 ± 12.63 years (range, 29–87 years), while the median age was 62 years. The average age of patients with FBC in our center was 49.5 ± 11.25 years. The number of MBC tended to increase from 2018 (Figure 1A). Most patients (55 patients, 74.2%) complained of a palpable breast mass in the first diagnosis, with a median symptom duration of 18.0 ± 45.78 months (range, 0.1 months to 30 years). The average body mass index (BMI) and body surface area (BAS) of patients were 24.0 ± 1.90 and 1.80 ± 0.122, respectively. All patients had unilateral lesion, with 27 (40.9%) in right and 39 (59.1%) in left breast. Two patients had a history of cancer (rectal cancer and esophageal cancer). Five patients had familial tumor history (one esophagus cancer, two gastric cancer, one breast cancer, and one thyroid cancer). In total, 17 (25.8%) and 11 (16.7%) of them had history of operation and smoking, respectively. 45.5% patients were diagnosed with stage II breast cancer, while 4.5% were initially diagnosed with stage IV MBC (Figure 1B).
Clinicopathologic Features of Male Patients With Breast Cancer

Note. HER2 = human epidermal growth factor receptor 2; TNBC = triple-negative breast cancer.

Incidence and Pathological Distribution of Male Breast Cancer in Real-World Data Analysis: (A) Incidence of Male Breast Cancer From 2006 to 2024, (B) TNM Stage, (C) Pathology, and (D) Molecular Subtypes
Pathological Characteristics of MBC
The histopathology and intrinsic subtype of enrolled patients were analyzed (Figure 2). Similar to patients with FBC, the most common histology of all 66 patients was invasive carcinoma (49 patients, 74.2%), including 38 cases of invasive carcinoma (non-specific type) and 11 cases of invasive ductal carcinoma. Percentage of ductal carcinoma in situ, mucinous carcinoma, and papillary carcinoma were 4 (6.1%), 5 (7.6%), and 8 (12.1%), respectively. The most common World Health Organization (WHO) grade of MBCs were grade II.
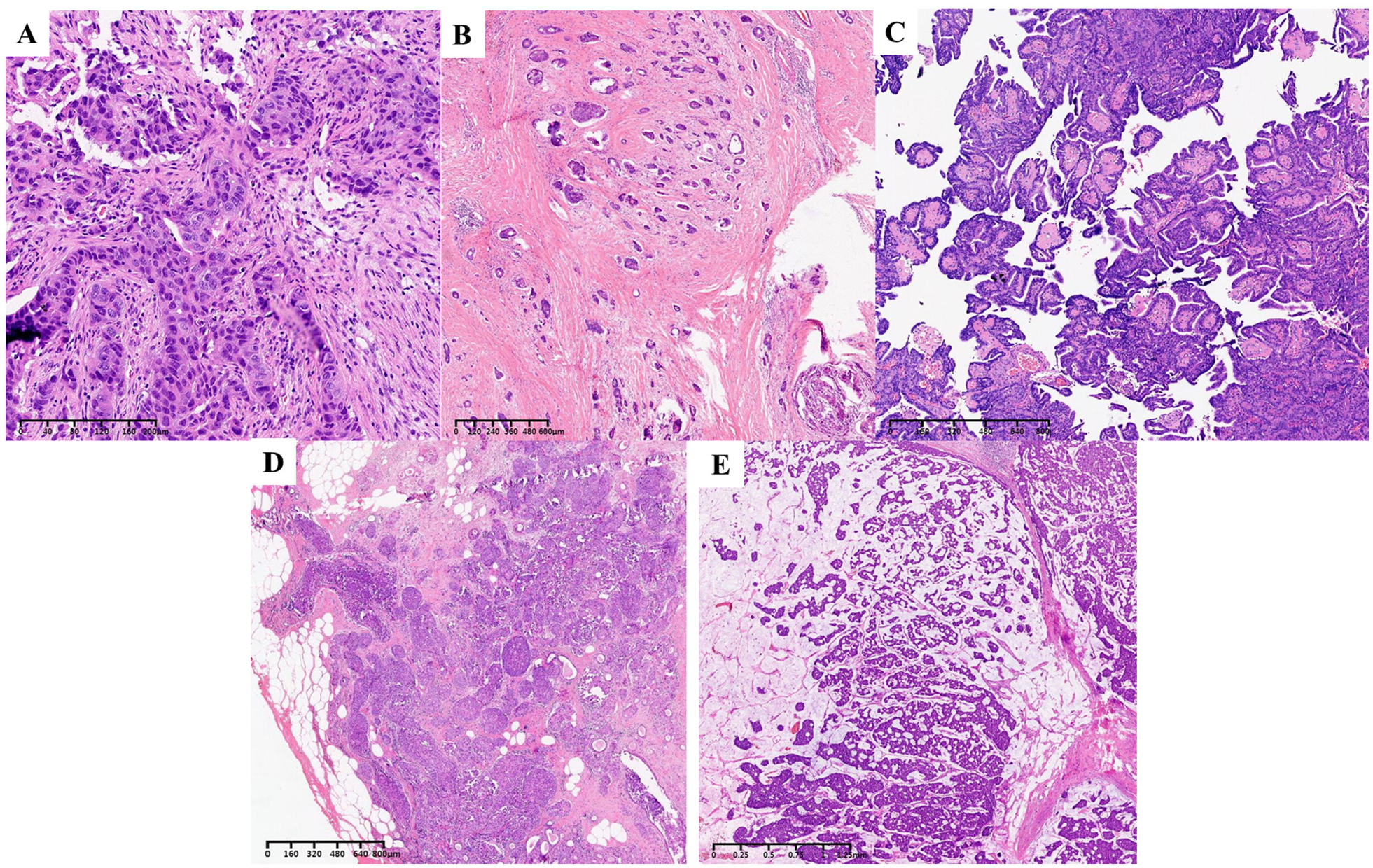
Macroscopic and Microscopic Features of Male Breast Cancer: (A) Invasive Carcinoma (Non-Specific Type), (B) Invasive Ductal Carcinoma, (C) Papillary Carcinoma, (D) Ductal Carcinoma In Situ, and (E) Mucinous Carcinoma
We achieved 48 patients with IHC results of ER, PR, HER2, and Ki67. IHC results showed that most patients were diagnosed with hormone receptor–positive MBC, most of which are detected with dual expression of the ER and PR. Among the 48 patients who underwent subtype classification, most of them were luminal A (24 patients, 50.0%) followed by 21 patients with luminal B disease (43.8%). The incidence of HER2-positive subtype was approximately 4.2% (two patients), and TNBC was the least expressed subtype (one patient, 2.1%; Table 1). Besides, we detected the positive expression of GATA3 (eight positive in eight patients), androgen receptor (AR; 24 positive in 24 patients), P53 (19 positive in 24 patients), P120 (26 positive in 26 patients), and E-cadherin (21 positive in 22 patients) in patients with MBC. Negative expression was found in CK5/6 (32 negative in 34 patients) and P63 (32 negative in 34 patients). Three cases of D2-40 or CD34-positive expression were found to indicate vascular invasion, while two cases of S100 positive expression were found to indicate neural invasion (Figure 3).

Marker Gene Expression by Immunohistochemistry Detection in Male Breast Cancer. Male Breast Cancer Was Often Detected With Positive Expression of ER (A), PR (B), and Negative Expression of HER2 (C). Positive Expression of AR (D), P53 (E), P120 (F), and E-cadherin (G) and Negative Expression of CK5/6 (H) and P63 (I) Were Often Found
Treatment of MBC
The current treatment guidelines are derived from research on FBC. Surgical options were commonly seen among male patients. As shown in Table 1, modified radical mastectomy is the mainstay of surgical management, which was selected by 93.9% of patients in our center. Importantly, 15 patients were accepted for second time surgery, due to the insufficient dissection of cancer lesion in local hospital. Three patients rejected the operation treatment. Sentinel lymph node biopsy (SLNB) and axillary dissection were proceeded in 11 (16.7%) and 50 (75.8%) patients, respectively. Lymph node metastasis was found in 18 patients, 10 of them were observed with more than three metastatic lymph nodes. Chemotherapy was proceeded in 39 (59.1%) patients, including 34 adjuvant chemotherapy, four neo-adjuvant chemotherapy, and one salvage chemotherapy. The chemotherapy strategy involves 30 chemotherapy regimen containing anthracycline (EC, EC-T, TE, TEC, TEC-GP) and nine regimen (T, TC, TCbHP) without anthracycline. Endocrine therapy was accepted by 14 patients, the drugs referring tamoxifen, goserelin, exemestane, leprerelin, and toremifene. Only one patient accepted targeted therapy, arising from the low incidence of HER2-positive subtype. Radiotherapy was accepted by nine patients. The area of exposure involved the chest wall, subclavicular area, and armpit.
Survival Outcomes of MBC
We followed up the survival outcomes of 66 patients with MBC, with the average follow-up time of 47.9 months. Data analysis showed that the median OS of patients with MBC was 46.7 months (range, 0.9–184.8 months). Up to April 16, 2024, there were 58 (87.9%) alive patients with MBC, while eight patients were dead. We conducted univariate analysis of factors that might influence the prognosis of MBC (Table 2). Results showed that age (χ2 = 3.856, p = .050), estrogen receptor (χ2 = 10.427, p = .005), molecular types (χ2 = 10.641, p = .031), P63 (χ2 = 2.631, p < .001), and endocrine therapy (χ2 = 31.167, p < .001) were significantly associated with the survival (Figure 4). Significant factors from univariate analysis of OS were included in a Cox proportional risk model for multivariate analysis, which identified no independent prognostic factors affecting OS of patients with MBC. The limited sample maybe the factor influencing result.
Factors Influencing the Prognosis of MBC


Survival Outcomes in MBC Patients: (A) Association of Molecular Subtype With MBC Survival, (B) Association of ER Expression With MBC Survival, (C) Association of Endocrine Therapy With MBC Survival, and (D) Association of Age With MBC Survival
Discussion
Male breasts are generally smaller than female breasts, which facilitates the detection of MBC. However, patients often fail to report their cases in a timely manner due to a lack of awareness (AlFehaid, 2023). MBC is recognized as an age-related disease, with incidence rates steadily increasing with age. Giordano reported that the average age at diagnosis for MBC is approximately 5 years older for men than for women, with figures of 67 years for men compared with 62 years for women (Giordano et al., 2004). E. G. Lee et al. (2020) found that the most common age range at diagnosis for MBC in Korea was 60 to 69 years, whereas for FBC, it was 40 to 49 years. In addition, J. Lee et al. (2023) reported a median age of 63 years for MBC. Consistent with the Korea population, the median age of MBC in our center was 62 years, which is 11 years older than that of FBC. Further data are needed to elucidate the differences in risk age among various races.
MBC is a rare malignancy that differs from FBC. In our study, the majority of breast cancer cases in men are invasive carcinomas, with invasive ductal carcinoma being the most prevalent histological type. Notably, approximately 10% of men diagnosed with breast cancer present with ductal carcinoma in situ (Anderson & Devesa, 2005). Less common histological subtypes in men include papillary cancers and mucinous cancers, while lobular carcinomas account for only 1% to 2% of MBC cases (Vermeulen et al., 1990). In 2022, G. Y. Lee et al. (2022) reported a rare case of male axillary accessory breast cancer, which was indistinguishable from lymphadenopathy and other malignancies, such as lymphoma and skin-derived tumors. Overall, most MBCs are characterized as estrogen receptor (ER) positive and human epidermal growth factor receptor 2 (HER2) negative (Fox et al., 2022). Giordano (2018) found that 99% of cases were ER positive, 82% were PR positive, and 97% were AR positive, with only 9% being HER2 positive. Most of the MBCs are classified into either luminal A-like (29%) or luminal B-like subtypes (71%) (Piscuoglio et al., 2016). Among Chinese population in our center, the pathological distribution was inconsistent with the reported outcomes among other population.
FBC has not been studied as extensively at the molecular level as FBC. Several tissue biomarkers have been reported to be associated with MBC, with BRCA being the strongest risk factor. Fox et al. (2022) claimed that BRCA2-associated MBCs tend to present at a higher stage and grade, and are more likely to be node-positive, as well as ER and PR positive. While somatic PIK3CA mutations are present in familial MBC, they are absent in BRCA2 carriers (Deb et al., 2013). Rizzolo et al. (2016) demonstrated that amplification of targetable oncogenes is common in BRCA1/2 mutation-negative MBCs. EMSY (Eleven-three homology, subdomain containing Y chromosome) amplification may represent a novel feature that could reveal underlying molecular pathways in MBCs, which are significantly associated with BRCA1/2 mutations (Navazio et al., 2016). GATA-3 appears to be effective for labeling MBC both in primary and metastatic setting (Biserni et al., 2018). MBCs exhibit recurrent somatic mutations in PIK3CA (36%) and GATA3 (15%), with TP53 (3%) somatic mutations occurring less frequent compared with ER-positive FBC (Moelans et al., 2019). Piscuoglio et al. (2016) reported that MBCs less frequently harbor somatic genetic alterations typical of ER-positive/HER2-negative FBCs, such as PIK3CA and TP53 mutations and losses of 16q. The correlation between PR negativity and p53 accumulation was associated with decreased 5-year survival rates and served as independent markers for the outcome of MBC (Kornegoor et al., 2012). Mutations in PALB2 have been documented in men with breast cancer, as well as in families with a history of breast cancer in men. However, the prevalence of PALB2 mutations in this population is reported to be only 1% to 2% (Blanco et al., 2012). In our study, we observed positive expression of AR, P53, P120, and E-cadherin, along with negative expression of CK5/6 and P63 in the majority of the MBC tumors examined. Nevertheless, the relationship between these gene expressions and MBC progression and clinical characteristics remains unclear. Recently proposed biomarkers for MBC are essential to establish a foundation for optimal patient management in the context of personalized medicine (Johansson et al., 2014). Given the distinct biology of breast cancer in men, promising new therapeutic targets are currently under investigation, including poly-ADP-ribose polymerase inhibitors and AR-targeted agents, either as monotherapy or in combination with other treatments (Gucalp et al., 2019).
Prospective data regarding the management of MBC are limited, and most treatment strategies are adopted from established guidelines for breast cancer in women. Recently, there have been very few well-conducted randomized controlled trials specifically addressing the treatment of MBC, with a limited number of registered clinical trials available (https://register.clinicaltrials.gov/). We identified only 10 registered clinical trials, comprising eight observational studies and two interventional studies (Table 3). It has been reported that the majority of men with early-stage breast cancer undergo mastectomy; however, breast-conserving surgery (BCS) with SLNB remains an alternative option in selected cases (Khan & Tirona, 2021). Compared with FBC, male patients are less likely to receive radiotherapy and chemotherapy (E. G. Lee et al., 2020). Nevertheless, since the majority of MBC are hormone receptor positive (HR+), adjuvant hormonal therapy is necessary. Furthermore, men are more likely to undergo total mastectomy and are less likely to receive radiotherapy. There was no significance in chemotherapy rates and only a minor difference in hormone therapy rates (Greif et al., 2012). An analysis of data from the SEER (Surveillance, Epidemiology, and End Results) registries indicated that mastectomy was performed in over 74% of patients, and overall 44.1% had >5 lymph nodes examined. Notably, only 18% of men with T1N0 tumors underwent BCS (Leone et al., 1990). Observational cohort studies have suggested that adjuvant therapy should be considered in male patients with primary breast cancer (Patel et al., 1989). In our practice, 93.9% of patients underwent modified radical mastectomy, 59.1% accepted chemotherapy, and 21.2% accepted endocrine therapy. In addition, radiotherapy is often underused in men with breast cancer. Diagnosis and treatment are attributed to the absence of standardized diagnosis and treatment protocols, highlighting the need for the further development of guidelines for MBC.
Registered Clinical Trials of Male Breast Cancer

In general, patients with MBC exhibit lower unadjusted OS rates compared with patients with FBC. Factors such as more advanced disease stage, older age at diagnosis, and generally shorter life expectancy may contribute to this disparity (Giordano, 2018). Miao et al. (2011) found that patients with MBC tend to have a later onset of disease and more advanced disease than their female counterparts, yet they face a lower risk of death from breast cancer. The differences in OS were highly significant, with rates of 83% for women compared with 74% for men (Greif et al., 2012). Kwok et al. (2022) suggested that late diagnosis and treatment, along with worsening morbidity and mortality, are attributable to limited public awareness. In Korea, poorer survival outcomes were associated with older age, inflammatory histological types, and advanced disease stage (E. G. Lee et al., 2020). While patients with MBC share similar risk factors with patients with FBC, PR and HER-2 status did not independently influence survival (Yao et al., 2022). Histologic grade was not significantly correlated with clinical outcome in this series, unlike what is seen in female patients (Vermeulen et al., 1990). Our study revealed that the median OS for patients with MBC was 46.7 months, with 87.9% of them remaining alive. Factors such as age, estrogen receptor status, molecular types, P63 expression, and endocrine therapy may serve as risk factors, which was similar with above reports.
This study presents single-center real-world data that address the clinical features and treatments of MBC during 18 years, providing references to explore clinical practice guidelines for this rare disease. However, this study also has several limitations. First, the participants from single center were still limited. Multicenter real-world evidences were needed to support a standard guideline. Besides, as a retrospective study, it relies on existing data, which may be incomplete, inconsistent, or inaccurately recorded. We have made significant efforts to ensure the accuracy and completeness of the data through telephone or artificial intelligence (AI) follow-up and multi-person verification. Despite these limitations, the assessment of treatments remains crucially important, contributing small amount of existing evidence to clinical oncologists.
In conclusion, the incidence of MBC has been observed to increase since 2018, with affected individuals generally being older than those with FBC. The most prevalent histological type of MBC is invasive carcinoma, which often exhibits positive hormone receptor expression. Current treatment guidelines for MBC are primarily based on research conducted on FBC; however, these guidelines should be modified to reflect clinical practice specific to MBC. The median OS for patients with MBC is reported to be 46.7 months, with a range extending from 0.9 to 184.8 months. Factors such as age, estrogen receptor status, molecular types, P63 expression, and endocrine therapy may contribute to the risk associated with MBC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by The Natural Science Foundation of Shaanxi Province (2023-JC-QN-0965).
Ethical approval and informed consent statements
As an observational study, this research was exempted from obtaining patients’ informed consent without therapeutic intervention, and this research was approved by the Research Ethics Board of Xijing Hospital and conducted in accordance with the Declaration of Helsinki as well.
Data Availability Statement
Data supporting the results presented in this study are available from the corresponding author upon reasonable request.
