Abstract
Accumulating evidence suggests a link between vascular endothelial growth factor (VEGF), erectile dysfunction (ED), and metabolic syndrome (Mets), possibly because VEGF can alter the physiological pathways involved in the regulation of endothelial cell proliferation. This study aimed to investigate the genetic susceptibility of VEGF 2578C>A polymorphism to the development of ED and Mets. Collected data included five-item International Index of Erectile Function (IIEF-5), components of Mets, and VEGF 2578C>A polymorphism. A total of 596 subjects from Kaohsiung with a mean age of 55.5 years were enrolled, data collection was done at our hospital. Individuals carrying the VEGF 2578 A allele (CA+AA genotypes) demonstrated a higher prevalence of ED compared to those with the CC genotype, with an adjusted odds ratio (OR) of 1.582 (95% confidence interval [95% CI] = 1.123–2.227, p value = 0.009) in multivariate binary regression analysis. Similarly, individuals carrying the VEGF 2578 A allele showed a higher prevalence of Mets compared to those with the CC genotype, with an adjusted OR of 2.461 (95% CI = 1.491–4.064, p value < 0.001). Furthermore, A allele carriers had significantly lower IIEF-5 scores and a higher number of Mets components compared to those with the C allele (P value < 0.001, respectively). In conclusion, VEGF 2578 A allele carriers are at a greater risk of both Mets and ED, suggesting that the VEGF 2578C>A polymorphism may serve as a common genetic susceptibility factor in the development of both disorders. Further research is warranted to evaluate the mechanisms underlying this association.
Introduction
Metabolic syndrome (Mets) has gained global attention due to its widespread impact, encompassing hyperglycemia, hypertriglyceridemia, low high-density lipoprotein cholesterol (HDL-C), elevated blood pressure, and central obesity. Individuals with Mets are at a higher risk of the development of diabetes mellitus (DM), cardiovascular disease, and stroke (Saklayen, 2018). The prevalence of Mets in males surpasses that in females, with reported rates of 26.8% and 16.6%, respectively (Wilson et al., 2005). Moreover, this prevalence increases with advancing age (Ford et al., 2002). Therefore, it remains a significant concern for aging men.
Erectile dysfunction (ED), a prevalent disorder in males, is defined as the incapability to attain or sustain an erection adequate for satisfactory sexual performance. Multiple risk factors have been association with ED, encompassing both physical and psychological aspects (Bal et al., 2007; Billups et al., 2008; Esposito et al., 2005). The physical factors contributing to ED include the presence of DM, cardiovascular disease, hypertension, obesity, smoking, and hormonal imbalances, and the psychological factors include stress, anxiety, and depression (Bal et al., 2007; Billups et al., 2008; Esposito et al., 2005). Importantly, advanced age stands out as one of the foremost risk factors for ED, particularly affecting sexually active older men (Lewis et al., 2010).
The connection between Mets and ED was first noted by Gündüz et al. (2004). Subsequent research, such as the study by Besiroglu et al., demonstrated a 2.6-fold increase in ED prevalence among Mets patients (Besiroglu et al., 2015). Heidler et al., using the International Index of Erectile Function 5 (IIEF-5) score, emphasized Mets as an independent risk factor for ED (Heidler et al., 2007). A previous study reinforces a strong association between Mets and ED, with higher Mets components, such as obesity, hypertension, dyslipidemia, and DM, correlating with increased ED prevalence (Lee et al., 2010). In addition, there is a significant decrease in IIEF-5 scores with an increase in Mets components (p < .01; Lee et al., 2010). From different mechanism of metabolic disease such as insulin resistance, hypertension, obesity, dyslipidemia, and vascular disease. These conditions lead to ED through multiple mechanisms: (a) Endothelial Dysfunction: Mets is associated with endothelial dysfunction, which compromises the endothelial nitric oxide pathway, reducing nitric oxide bioavailability and impairing vascular smooth muscle relaxation. This adversely affects the hemodynamics of blood flow necessary for erection. (b) Atherosclerosis: Mets promotes atherosclerosis by inducing plaque formation in the vessel walls, which reduces blood flow to the penis, leading to ED. (c) Insulin resistance: Mets often involves insulin resistance and elevated insulin levels, which alter hormone levels, including testosterone. Lower testosterone levels can diminish libido and contribute to ED. (d) Inflammation: Chronic inflammation associated with Mets can damage blood vessels and affect the nerves involved in the erectile response. Despite accumulating evidence supporting the association between Mets and ED, limitations persist in the assessment of disease etiology, particularly regarding the elucidation of causal effects and underlying mechanisms.
Vascular endothelial growth factor (VEGF) is a transmembrane protein located on cell surfaces, and it plays a crucial role in human vasculogenesis and angiogenesis processes (Apte et al., 2019). The VEGF gene is located on the 6p21.3 region of the human chromosome, spanning approximately 14 kb and comprising eight exons and seven introns (Vincenti et al., 1996; Wei et al., 1996). Although the concept of relationships between VEGF with blood vessels, growth-stimulating factors, and tumor cells was first suggested in 1939 (Ferrara & Adamis, 2016), the isolation and cloning of VEGF A were only achieved in 1989 (Leung et al., 1989). Subsequently, research on VEGF has significantly expanded, especially in cancer and ophthalmology (Carmeliet, 2005; Pożarowska & Pożarowski, 2016). Evidence has identified an association between circulating VEGF levels and Mets markers such as obesity, poor glycemic control, and hyperlipidemia (Lee et al., 2017; Mazidi et al., 2017). Animal studies have also indicated that the intracavernous administration of VEGF can reverse ED (Lee et al., 2017; Rogers et al., 2003). Considering the close relationship of endothelial dysfunction to both Mets and ED, the attempt of searching for VEGF polymorphisms for common genetic factors influencing both disorders appears feasible.
The objective of this investigation was to explore correlations between VEGF polymorphisms and both ED and Mets. Specifically, we focused on evaluating the functional single-nucleotide polymorphism
Materials and Methods
Study Participants and Enrollment Process
A total of 596 male participants from Taiwan, all aged 40 years or older, were enrolled in this study, which was conducted as part of a health screening program at our institution. The screening program was open to men residing in Kaohsiung City. Detailed medical, surgical, and psychosexual histories of the participants were obtained, and comprehensive physical examinations were performed. These examinations included measurements of weight, height, waist circumference, blood pressure, and transrectal ultrasonography for the evaluation of prostate volume. The participants’ body mass index (BMI) was calculated by dividing their weight in kilograms by the square of their height in meters. Fasting blood samples were collected for the biochemical analysis of prostate-specific antigen (PSA), hormone profiling of testosterone, and genetic testing to identify individuals carrying the VEGF 2578 A allele. All data pertaining to the subjects were gathered in compliance with the applicable guidelines and regulations. Prior to their participation in this study, written informed consent was obtained from each subject. The experimental protocols employed in this study received approval from the Institutional Review Board of our hospital on September 15, 2011.
Variables
We collected data on variables related to ED and Mets, including age, BMI, physical activity, educational status, marital status, alcohol consumption (at least once per week), tobacco use (more than 10 cigarettes per week), and betel nut consumption (more than seven betel nuts per week) for a minimum duration of 6 months prior to the health screening (Ibrahim et al., 2019; Weber et al., 2013). The participants were also assessed for lower urinary tract symptoms using the International Prostate Symptom Score (IPSS) and quality of life (QoL) score questionnaires.
Definition of ED
ED was assessed through the five-item International Index of Erectile Function (IIEF-5) questionnaire, with a score below 22 indicating ED. Participants meeting this criterion were classified into the ED(+) group, while those not meeting it were placed in the ED(−) group.
Definition of Mets
The definition of Mets, as outlined by the Taiwan Department of Health, requires the presence of at least three of the following component risk factors: (a) waist circumference equal to or exceeding 90 cm for men, (b) systolic blood pressure of 130 mmHg or higher or diastolic blood pressure of 85 mmHg or higher, (c) fasting blood glucose level of 100 mg/dL or higher, (d) triglyceride level of 150 mg/dL or higher, and (e) HDL-C level below 40 mg/dL for men (Geng et al., 2022; Ou et al., 2023). According to this definition, we divided our cohort into participants with Mets, denoted as Mets(+) group, and participants without Mets, denoted as Mets(−) group.
Single-Nucleotide Polymorphism
QIAamp DNA Blood Mini Kits (Qiagen, Düsseldorf, Germany) were employed for DNA extraction, followed by allelic discrimination of VEGF 2578C>A (rs699947) alleles using validated TaqMan SNP genotyping assays (C_1647381_10 [rs833061], C_1647379_10 [rs1570360], and C_8311602_10 [rs699947]; Applied Biosystems) on an ABI PRISM 7900 SDS (Applied Biosystems). Polymerase chain reactions (PCRs) were conducted as per the manufacturer’s protocol, with SNP amplification assays performed using 10 ng of sample DNA in 10 mL of reaction solution, 5 mL 2X TaqMan Universal PCR Mix (Applied Biosystems), and 0.25 mL 20 probe-primer assay mix. PCR cycling was performed using an ABI Prism 7500HT sequence detection system. Direct sequencing was used to confirm genotypes post full-scale genotyping. In addition, 10% of the study sample was re-tested for quality control, showing 100% concordance.
Statistical Analysis
We initially assessed the association between the VEGF 2578C>A polymorphism and ED by dividing the participants into ED(+) and ED(−) groups. Clinical characteristics were presented as mean ± standard deviation (SD) for continuous variables and as number (percentage) for categorical variables. Differences in clinical characteristics between the ED groups were calculated using chi-square tests for categorical variables and independent t-tests for continuous variables. Further exploration of the association between the VEGF 2578C>A polymorphism and ED involved univariable and multivariable logistic regression analyses to calculate odds ratios (ORs) and 95% confidence intervals (CIs). Subsequently, participants were categorized into Mets(+) and Mets(−) groups, and similar methods were employed to determine differences between groups and explore the association between the VEGF 2578C>A polymorphism and Mets. Finally, analysis of variance (ANOVA) was conducted to compare the means of the IIEF-5 scores and the number of Mets components among different genotypes of the VEGF 2578C>A polymorphism. All statistical analyses were conducted using SPSS version 20.0 (IBM Corp, Armonk, NY, USA). A significance level of P value < 0.05 was considered indicative of a significant difference in this study.
Results
Individuals Carrying the VEGF 2578 A Allele Had a Significantly Higher Prevalence of ED
Table 1 outlines the characteristics of the 596 subjects who participated in this study. Among these subjects, 321 (54%) were diagnosed with ED, while 275 (46%) were not. Those with ED were notably older and had higher BMI, blood pressure, and a higher prevalence of Mets. In addition, their IIEF-5 scores and prostate volume were lower compared to those without ED. There was a significantly higher prevalence of ED among VEGF 2578 A allele carriers compared to noncarriers: 105 cases (50%) versus 159 cases (38%) respectively (P value = 0.006). There were no significant differences in other characteristics between the ED(+) and ED(−) groups.
Clinical Characteristics of Participants Classified by the Presence of Erectile Dysfunction (n = 596)
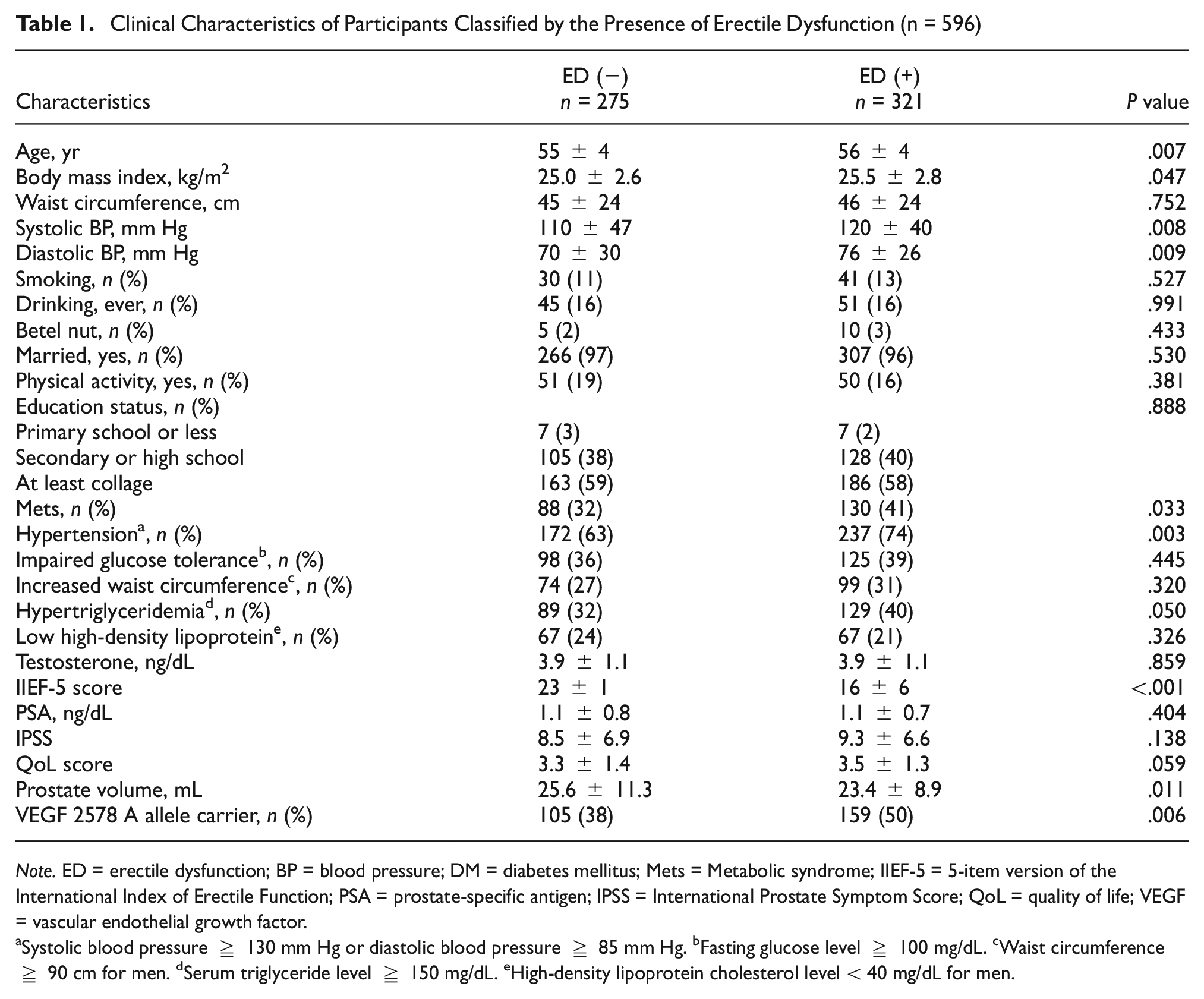
Note. ED = erectile dysfunction; BP = blood pressure; DM = diabetes mellitus; Mets = Metabolic syndrome; IIEF-5 = 5-item version of the International Index of Erectile Function; PSA = prostate-specific antigen; IPSS = International Prostate Symptom Score; QoL = quality of life; VEGF = vascular endothelial growth factor.
Systolic blood pressure ≥ 130 mm Hg or diastolic blood pressure ≥ 85 mm Hg. bFasting glucose level ≥ 100 mg/dL. cWaist circumference ≥ 90 cm for men. dSerum triglyceride level ≥ 150 mg/dL. eHigh-density lipoprotein cholesterol level < 40 mg/dL for men.
The VEGF 2578 A Allele Was Associated With an Elevated Risk of ED
In the univariate binary regression analysis, the presence of the VEGF 2578 A allele (CA+AA genotypes) demonstrated a 1.589-fold increase of ED comparing to those with VEGF 2578 CC phenotype (95% CI = 1.146 to 2.204, P value = 0.006; Table 2). Subsequent adjustment for variables showing significant associations with ED in the univariable binary analysis, including age, BMI, Mets, systolic blood pressure, diastolic blood pressure, and prostate volume (data not shown), revealed that the presence of the CA and AA genotypes of VEGF 2578 still demonstrated significant associations with an elevated risk of ED, with an adjusted OR of 1.582 (95% CI = 1.123–2.227, P value = 0.009; Table 2). Specifically, the CA genotype was present in 94 (34%) ED(−) participants and 130 (41%) ED(+) participants, with an unadjusted OR of 1.451 (95% CI = 1.031–2.042, P value = 0.033), and an adjusted OR of 1.455 (95% CI = 1.017–2.080, P value = 0.040) compared to the CC genotype. Similarly, the AA genotype was present in 11 (4%) ED(−) participants and 29 (9%) ED(+) participants, with an unadjusted OR of 2.767 (95% CI = 1.338–5.722, P value = 0.006), and an adjusted OR of 2.603 (95% CI = 1.235–5.488, P value = 0.012) compared to the CC genotype (Table 2).
Distribution and Association Between Genotypes of VEGF 2578C>A and Erectile Dysfunction (n = 596)

Note. ED = erectile dysfunction; VEGF = vascular endothelial growth factor; OR = odds ratio; 95% CI = 95% confidence interval. Multivariable model: adjustment for age, body mass index, metabolic syndrome, systolic blood pressure, diastolic blood pressure and prostate volume.
Individuals Carrying the VEGF 2578 A Allele Had a Significantly Higher Prevalence of Mets
We further examined the clinical characteristics of the 596 participants categorized based on the presence or absence of Mets. Among these participants, 218 (37%) were diagnosed with Mets, while 378 (63%) were not. Those with Mets were notably older, had higher BMI, waist circumference, blood pressure, smoking habits, and alcohol consumption, and lower education levels. In addition, they had a higher prevalence of ED and lower IIEF-5 scores. Importantly, individuals carrying the VEGF 2578 A allele exhibited a significantly higher prevalence of Mets compared to noncarriers: 117 cases (54%) versus 147 cases (39%) respectively (P value = 0.001; Table 3).
Clinical Characteristics of Participants Classified by the Presence of Metabolic Syndrome (n = 596)
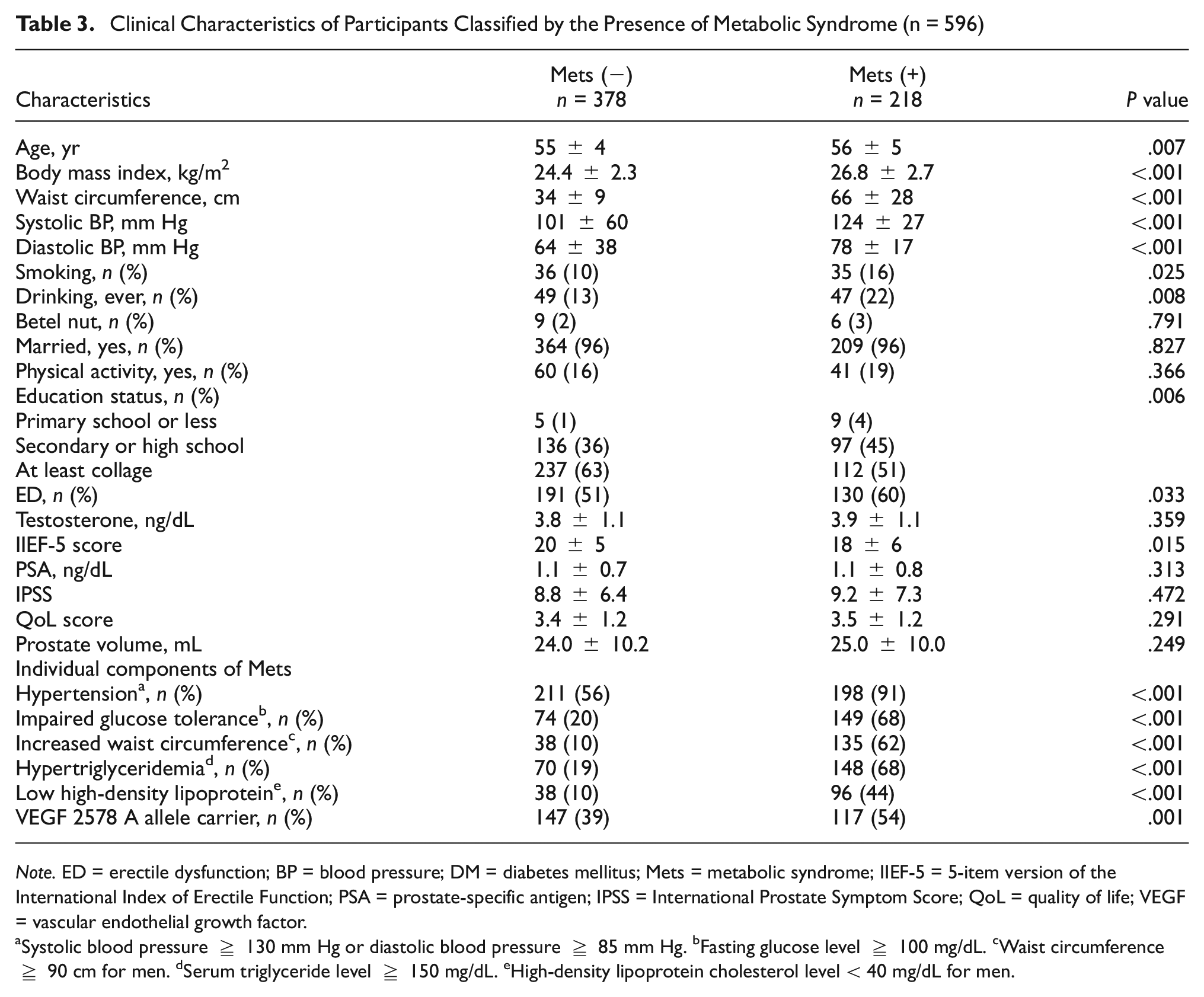
Note. ED = erectile dysfunction; BP = blood pressure; DM = diabetes mellitus; Mets = metabolic syndrome; IIEF-5 = 5-item version of the International Index of Erectile Function; PSA = prostate-specific antigen; IPSS = International Prostate Symptom Score; QoL = quality of life; VEGF = vascular endothelial growth factor.
Systolic blood pressure ≥ 130 mm Hg or diastolic blood pressure ≥ 85 mm Hg. bFasting glucose level ≥ 100 mg/dL. cWaist circumference ≥ 90 cm for men. dSerum triglyceride level ≥ 150 mg/dL. eHigh-density lipoprotein cholesterol level < 40 mg/dL for men.
The VEGF 2578 A Allele Was Associated With an Elevated Risk of Mets
The presence of the VEGF 2578 A allele (CA+AA genotypes) was associated with an increased risk of Mets, as evidenced by our univariate binary regression analysis, which revealed a 1.820-fold increase compared to individuals with the VEGF 2578 CC phenotype (95% CI =1.299–2.550, P value < 0.001; Table 4). Upon adjustment for significant variables identified in the univariable binary analysis (data not shown), including age, BMI, waist circumference, blood pressure, smoking, drinking, education status, and ED, the association remained significant, with an adjusted OR of 2.461 (95% CI = 1.491–4.064, P value < 0.001; Table 4). Specifically, the CA genotype was present in 122 (32%) Mets(−) participants and 102 (47%) Mets(+) participants, with an unadjusted OR of 1.912 (95% CI = 1.345–2.719, P value < 0.001), and an adjusted OR of 2.493 (95% CI = 1.479–4.203, P value = 0.001) compared to the CC genotype (Table 4).
Distribution and Association Between Genotypes of VEGF 2578C>A and Metabolic Syndrome (n = 596)

Note. Mets = metabolic syndrome; VEGF = vascular endothelial growth factor; OR = odds ratio; 95% CI = 95% confidence interval. Multivariable model: adjustment for age, body mass index, waist circumference, systolic blood pressure, diastolic blood pressure, smoking, drinking, education status, and erectile dysfunction.
The Association Between VEGF 2578C>A Polymorphism and Individual Components of Mets, and IIEF-5
Table 5 presents the results of the sensitivity analysis examining the frequency of VEGF 2578C>A polymorphism among the studied individuals, categorized by VEGF 2578C>A genotype and VEGF 2578 A allele carrier status. There was a significant decrease in IIEF-5 score corresponding to an increase in the number of A alleles (mean ±SD: 19.9 ± 4.9 for CC, 18.6 ± 5.9 for CA, and 16.8 ± 6.6 for AA, P value < 0.001). Similar trends were observed for the number of Mets components, individuals with a higher number of A alleles had a greater number of Mets components (mean ±SD: 1.7 ± 1.2 for CC, 2.2 ± 1.3 for CA, and 2.4 ± 1.6 for AA, P value < 0.001).
IIEF-5 Scores and Component Numbers of Metabolic Syndrome by VEGF 2578C>A Genotype

Individuals With Both ED and Mets Had the Highest Genetic Variance With the VEGF 2578 A Allele
Finally, we investigated the distribution of VEGF 2578C>A polymorphisms among individuals with both ED and Mets, comparing them to those without ED and Mets. Table 6 presents the frequency of VEGF 2578C>A polymorphism among the ED(−)Mets(−) and ED(+)Mets(+) groups. The frequencies of genotypes CC, CA, and AA significantly differed between these two groups. In the ED(−)Mets(−) group, the CC genotype predominated with 123 cases (69%), whereas in the ED(+)Mets(+) group, the CA and AA genotypes were predominant with 66 cases (53%) and 10 cases (63%), respectively, compared to the CC genotype with 54 cases (31%), indicating a significant association (P value < 0.001). In addition, there was a significantly higher prevalence of A allele carriers in the ED(+)Mets(+) group with 76 cases (57%) compared to the ED(−)Mets(−) group with 64 cases (43%), with a P value < 0.001.
The Frequency of VEGF 2578C>A Polymorphism Among ED(−)Mets(−) vs ED(+)Mets(+)

Discussion
Our findings reveal a significant genetic association between the VEGF 2578 A allele and the presence of Mets and ED. Individuals with the VEGF 2578 A allele demonstrated a 1.582-fold increased risk of ED and a 2.461-fold increased risk of Mets after adjustment for confounding variables. In addition, an increase in the A allele count corresponded to a significant decrease in the IIEF-5 score and an increase in the number of Mets components. We highlight that 2578C>A polymorphism may be a potential common genetic susceptibility factor for both Mets and ED. To our knowledge, this is the first study to propose that ED and Mets could share the same underlying biological mechanism, suggested by the presence of the VEGF 2578C>A polymorphism in both diseases.
Numerous studies have investigated the complex genetic polymorphisms associated with the development and severity of ED, including genes such as endothelial nitric oxide synthase (eNOS), angiotensin-converting enzyme (ACE), androgen receptor (AR), G-protein β3 (GNB3) subunit, methylenetetrahydrofolate reductase (MTHFR), TGFB1, VEGF, proprotein convertase subtilisin/kexin type 9 (PCSK9), ARG1, DRD2, DRD4, DDAH, and HNF4A (Eisenhardt et al., 2010; Mostafa & Taymour, 2020; Lee et al., 2012). Our findings underscore the remarkable influence of the VEGF gene on the intricate mechanisms underlying ED. One of the pivotal determinants in ED is vascular blood flow, with VEGF assuming a critical role in regulating this physiological process. Findings of the association between genetic variations in the VEGF gene and ED has consistently reported that VEGF 2578 A gene polymorphisms are a significant risk factor contributing to the complexity of ED (Lee et al., 2017). By shedding light on these genetic intricacies, our study contributes to the broader understanding of the genetic landscape of ED, paving the way for targeted interventions and personalized treatments in the realm of sexual health.
Research into Mets gene polymorphisms is an important field, which aims to identity the genetic factors contributing to this complex condition. In Mongolia, studies investigating SNPs in genes such as ADIPOQ, BDNF, and LPL have highlighted their influence on Mets susceptibility (Chuluun-Erdene et al., 2020). Jin et al. investigated VDR gene polymorphisms, and elucidated their impact on lipid profiles concerning Mets (Jin et al., 2021). In addition, various investigations on PON1, CETP (Dizaji et al., 2018), GNB3, PPARG, TCF7L2, APOA5, APOC3, APOE, and FTO (Povel et al., 2011) polymorphisms have revealed their significant associations with the risk of Mets. Of particular note, a pioneering study focusing on VEGF gene polymorphisms, specifically the VEGF −634G>C polymorphism, demonstrated a substantial link with Mets susceptibility (Kim & Hong, 2015). This aligns with our findings of an association between VEGF 2578 A gene polymorphisms and the risk of Mets, further confirming the role of VEGF polymorphisms in Mets.
Despite the findings of the significant influence of VEGF-related gene polymorphisms on Mets and ED (Kim & Hong, 2015; Lee et al., 2017), no study has explored the relationships between Mets, ED, and VEGF-related gene polymorphisms. To our knowledge, this is the first study to demonstrate that VEGF 2578 A gene polymorphisms act as a potential common genetic susceptibility factor for both Mets and ED. Previous research has indicated that individuals with Mets or ED often exhibit reduced VEGF levels and impaired VEGF signaling (Devaraj & Jialal, 2012; Erman et al., 2016; Rogers et al., 2003). This decrease in VEGF can lead to endothelial dysfunction in various parts of the body, including the cardiovascular system and corpus cavernosum penis, contributing to the development of these conditions (Devaraj & Jialal, 2012; Erman et al., 2016; Rogers et al., 2003). Some studies have suggested that therapies focused on increasing VEGF levels or enhancing VEGF signaling pathways could offer significant benefits in improving endothelial function and treating ED, particularly in individuals with Mets (Dall’Era et al., 2008; Gholami et al., 2003; Yamanaka et al., 2005). Our findings shed light on the intricate relationship between Mets, ED, and VEGF. These insights may pave the way for novel treatment strategies for individuals with these conditions.
The potential mechanism of the VEGF 2578C>A polymorphism in ED and Mets involves hypoxia-inducible factor 1 alpha (HIF1α). HIF1α binds to the hypoxia responsive element (HRE) to regulate VEGF gene expression (Forsythe et al., 1996; Hashimoto & Shibasaki, 2015; Levy et al., 1995). A high VEGF expression in hypoxic conditions can lead to the proliferation and migration of endothelial cells, resulting in neovascularization (Aiello & Wong, 2000; Cheng et al., 2017; Penn et al., 2008). Previous studies have demonstrated that VEGF 2578 A gene polymorphisms reduce the binding sites for HIF1α, a crucial component in the activation of VEGF gene expression induced by hypoxia. (Buroker et al., 2013; Ziello et al., 2007). Carriers of the A allele have a lower expression of VEGF due to regulation of HIFα, leading to the dysregulation of endothelial function (Buroker et al., 2013; Ziello et al., 2007). In contrast, the C allele facilitates the binding sites for HIF1α, potentially contributing to higher levels of VEGF and preserving endothelial function.
There are limitations to this study. First, our data were collected from a nonprobability sample of men, limiting the generalizability of our results due to the undefined population. Our data cannot represent the whole male population. The external validity of our findings relies on replication in subsequent studies. Second, the cross-sectional design restricts the establishment of causal relationships between the VEGF 2578C>A polymorphism, Mets, and ED, emphasizing the need for longitudinal investigations. Third, our exclusive focus on the VEGF 2578C>A polymorphism overlooks potential interactions with other genetic factors that could influence Mets and ED. This includes genes related to endothelial function such as eNOS (Lee et al., 2010) and lipid metabolism, including APOC3, APOB, LDLR, and LPL (Su et al., 2024). In addition, genes involved in hormonal regulation such as SHBG (Aleksandra et al., 2022), inflammation markers like C-reactive protein (CRP) and interleukin (IL)-6 (Das, 2007; Pradhan et al., 2001), and obesity-related genes such as OB (Moon et al., 2019) play significant roles. These genetic variations can impact cardiovascular health, hormone levels, inflammation, and fat distribution, thereby affecting both metabolic health and erectile function. Consequently, more comprehensive genetic studies are warranted to address this issue. In addition, the absence of specific details on crucial clinical parameters, such as lifestyle factors including physical exercise and alcohol consumption (Kalter-Leibovici et al., 2005), cardiovascular comorbidities (García-Cruz et al., 2013), and medication use, including antihypertensives, nonsteroidal anti-inflammatory drugs, and antacids (Razdan et al., 2018), impedes a comprehensive understanding of the relationships. Finally, the study only includes Taiwanese participants, limiting the applicability of the findings across different ethnic groups. Addressing these limitations in future research would significantly enhance the depth and reliability of our conclusions regarding the relationships between the VEGF 2578C>A gene polymorphism, Mets, and ED.
Conclusion
Our results demonstrated that VEGF 2578 A allele carriers were at a greater risk of both Mets and ED, suggesting that VEGF 2578C>A polymorphism may be a common genetic susceptibility factor in the development of both disorders. Further research is warranted to evaluate the mechanisms underlying this association.
Footnotes
Acknowledgements
The authors thank Ms. Chao-Shih Chen for her help to hold the healthy screening and Ms. Kai-Lin Huang for editing this manuscript.
Author Contribution Statements
(a) Conception and Design: Y.-C.L., S.-P.H., C.-C.L. (b) Drafting the Article: C.-I.L. (c) Revising It for Intellectual Content: C.-I.L., Y.-C.L., J.-H.G. (d) Final Approval of the Completed Article: Y.-C.L., J.-H.G.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Kaohsiung Medical University Hospital (grant no. KMUH101-1R45), Ministry of Science and Technology (112-2314-B-037-104-MY3 ), in part by Kaohsiung Municipal Siaogang Hospital (KMSH, H-112-03 and H-113-02), and in part by Kaohsiung Medical University (KMU, S112014).
Ethical Statement
This study received approval from the Institutional Review Board of Kaohsiung Medical University Hospital on September 15, 2011 (KMUH-IRB-980538).
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
