Abstract
Prostate cancer (PCa) is a major cause of illness and death in men of Sub-Sahara African origin. The study assessed the pattern of PCa, the effect of family history on PSA at diagnosis, and clinical characteristics of PCa in Nigeria. A cross-sectional survey of 200 participants was performed within a 12-month period in Nigeria. Data were collected through patients’ interview and hospital records and analyzed using SPSS version 25. Descriptive and inferential statistics were performed. P values <.05 were significant. Mean age of 68.5 years was observed among the 200 study participants. Only 64 (32.0%) had a positive immediate family history of PCa, and 61 (30.5%) were not aware of their family cancer history. Most patients 140 (70.0%) had lower urinary tract symptom (LUTS)/lower back pain/leg pain, and the average Gleason score was 7.55 (±0.876). Symptoms of LUTS/lower back pain mostly occurred in patients between 58 and 79 years, while LUTS/leg pain was more common in persons between 60 and 84. Average PSA differed among participants; persons with no family cancer history (M = 143.989; 95% confidence interval [CI] = 114.849–173.129), family history of PCa (M = 165.463; 95% CI = 131.435), family history of cervical cancer (M = 133.456; 95% CI = 49.335–217.576), and persons with no knowledge of their family cancer history (M = 121.546; 95% CI = 89.234–153.857). Univariate one-way (F-Tests) showed that family history of cancer had no significant impact on patients’ PSA (R2 = 0.017; adjusted R2 = 0.002; df = 3; F = 1.154; p = .329) at diagnosis. PCa mostly occurred in men within 60 to 70 years of age, and family history of cancer did not predict PSA at diagnosis. Patients presented to health facilities at advanced or metastatic stages. These findings highlight the need for policies and strategies that encourage early PCa screening.
Introduction
Prostate cancer (PCa) is a major cause of illness and death in men, particularly those of Sub-Sahara African origin. Previous studies of PCa reported that the tumor stage and grade at diagnosis are highest among men in Sub-Saharan Africa (McGinley et al., 2016; Rebbeck et al., 2013). Men of African origin also present with higher prostate-specific antigen (PSA) levels, less likely to receive curative therapy, and have higher rates of biochemical recurrence after surgery than men of other race (McGinley et al., 2016). A significant difference in primary treatment for PCa between African American and non-Africans with similar risk profiles have also been observed in a previous research (Moses et al., 2010). Socioeconomic, educational, cultural, and genetic factors, as well as variations in care delivery and treatment selection, contribute to this cancer disparity. Factors noted to strongly influence the risk of PCa-related mortality include; risk level, type of treatment, and health insurance (Moses et al., 2010). These risks play important roles in Sub-Saharan Africa as they may influence the disease characteristics and outcomes. Also, family history being a notably important factor in the development of cancers is sparsely researched among men in Sub-Saharan Africa, requiring enhanced understanding of its impact in the PSA of persons with PCa, which is mostly high at diagnosis (Ajape et al., 2010; Osegbe, 1997).
There is an upward trend in the incidence of PCa among men of Nigerian origin (Ajape et al., 2010), where it is the most diagnosed type of cancer in males. PCa burden is of utmost importance since it is a leading cause of cancer-related death among men in Nigeria (World Health Organization [WHO], 2020). Previous studies reported that most Nigerian men are diagnosed at very advanced and lethal stages of the disease, usually with locally advanced or metastatic, high-grade disease, and a 2- to 3-year high death rate (Ajape et al., 2010; Ekeke et al., 2012; Ikuerowo et al., 2013; Ofoha & Magnus, 2019; Osegbe, 1997; Yawe et al., 2006). A 2010 study of a Nigerian population by Ajape et al. (2010), observed a 10.3-month mean duration of symptoms prior to hospital presentation and high sensitivity of digital rectal examination (DRE); which probably accounted for the observed treatment without establishing the histologic diagnosis in majority of the cases. However, with increasing awareness and screening, the possibility of a change in the trend is anticipated. Therefore, this study assessed the current pattern, the effect of family history of cancer on PSA at diagnosis and the clinical characteristics of PCa in a Nigerian population.
Methods
Study Design and Setting
Cross-sectional method was employed to identify the pattern and clinical characteristics of PCa among the study population. The study was conducted at the Nigerian Navy Reference Hospital, Calabar, University of Calabar Teaching Hospital and Asi Ukpo Hospital. These are the cancer reference hospitals in Cross River State, Nigeria, which also had referrals from other regions of the Country.
Study Population and Eligibility Criteria
All persons with PCa who were treated at the health facilities formed the study population. The study included all new and previously diagnosed patients at any stage of the disease who visited the facilities within the study period. However, persons who did not give informed consent were excluded.
Sample Size
Total enumeration of all patients who visited the study facilities within the 1-year study period was adopted for the sample size. This method of sample size was adopted as a result of paucity of information for suitable sample size estimation in the region.
Data Collection
Using an appropriate proforma, patients were individually interviewed for relevant data during their visits to the health facilities between January 07, 2022 and January 03, 2023. Hospital records of the patients were also reviewed for extraction of relevant information. Data collected for the study included, medical history, illness history, social history, and demographic history. These included, date of presentation at the hospital, date of initial diagnosis, patients’ age at diagnosis, gender, family history of cancer, occupation, social history, co-morbidities, PSA values, and Gleason scores. Records were kept to prevent multiple enrollment of participants.
Outcome Measures
The study outcome measures were the current pattern, the effect of family history of cancer on PSA at diagnosis, and the clinical characteristics of PCa in Nigeria.
Data Analysis
Obtained data were analyzed descriptively and inferentially using SPSS version 25. Descriptive statistics such as frequencies, percentages, mean, standard deviation, median, range, and P value were used to highlight important and relevant features of the data. Chi-square tests of associations were also performed for socio-demographics and clinical data. Univariate one-way (F tests) was performed to investigate the effect of family history of cancer on patients’ PSA at diagnosis at 95% confidence interval. This test is based on the linearly independent pairwise comparisons among the estimated marginal means. P values <.05 were statistically significant.
Ethical Considerations
The ethical clearance was obtained from the Health Research and Ethics Committee of the University of Calabar Teaching Hospital (UCTH/HREC/33/582). Patients’ written informed consent and institutional permission were also obtained prior to the study.
Results
Study participants had a mean and median age of 68.5 and 68.00 years old, respectively, and age ranged between 50 and 92 years. Most frequently occurring age of participants was 65 years old. Majority were married (188; 94.0%), not enrolled in health insurance (144; 72.0%), had no previous or current history of smoking (146; 73.0% and 196; 98.0%, respectively), took alcohol (108; 54.0%), did not exercise (174; 87.0%) and were mostly retired (80; 40.0%) (Table 1).
Participants’ Socio-Demographic Information

Note. N = 200.
Several participants (64; 32.0%) had a positive immediate family history of PCa, (42; 21.0%) had an extended family history and (61; 30.5%) were not aware of their family cancer history. Most participants (140; 70.0%) had LUTS/lower back pain/leg pain during presentation at the hospital. Gleason scores ranged from 6 to 9, a mean score of 7.55 (±0.876) was observed, and scores between 8 and 7 were the most common (69; 34.5% and 62; 31.0%, respectively). Half of the participants had PSA >100 ng/mL, while mean PSA was 142. 58 ± 128.11, and a range of 7 to 657.7 was observed. Participants mainly experienced acute urinary retention (75; 37.5%) and obstructive uropathy (62; 31.0%). More than half of participants had hypertension >100 (>50.0%) and most co-morbidities were reported to have lasted 1 to 10 years (74; 37.0%). There was significant association between type of cancer in family history and patients’ age (X2 = 10.987; p = .015), patients’ age and extended family history of cancer (X2 = 16.545; p = .002), marital status and extended family history of cancer (X2 = 9.121; p = .046), patients’ and current history of smoking (X2 =13.545; p = .006), alcohol intake and educational status (X2 = 15.214; p = .003), educational status and frequency of alcohol intake (X2 = 55.155; p = .001), patients’ age and date of presentation at the hospital (X2 = 55.155; p = .001), patients’ age and date of initial diagnosis (X2 = 12.467; p = .008), patients’ age and use of other therapies (X2 = 57.164; p = .001), educational status and use of herbal drugs (X2 = 8.766; p = .055) and patients’ age and initial symptoms (X2 = 75.155; p = .000). See Table 2.
Participants’ Illness History

Note. N = 200.
Symptoms of LUTS and lower back pain mostly occurred in patients between 58 and 79 years, although it peaked at 66 years old. Symptoms of LUTS and leg pains was more common in persons between 60 and 84, but peaked at 65, 69, and 70 years old. Also, only little difference was observed for patients’ age and age at PCa diagnosis. See Figure 1.

Distribution of Patients’ Age at Diagnosis With Respect to Symptoms and Age-Difference Plot
More than 25% of persons who were not enrolled in health insurance and did not smoke had LUTS and lower back pain, over 20% had LUTS and leg pain, and almost 10% had only LUTS. See Figure 2.
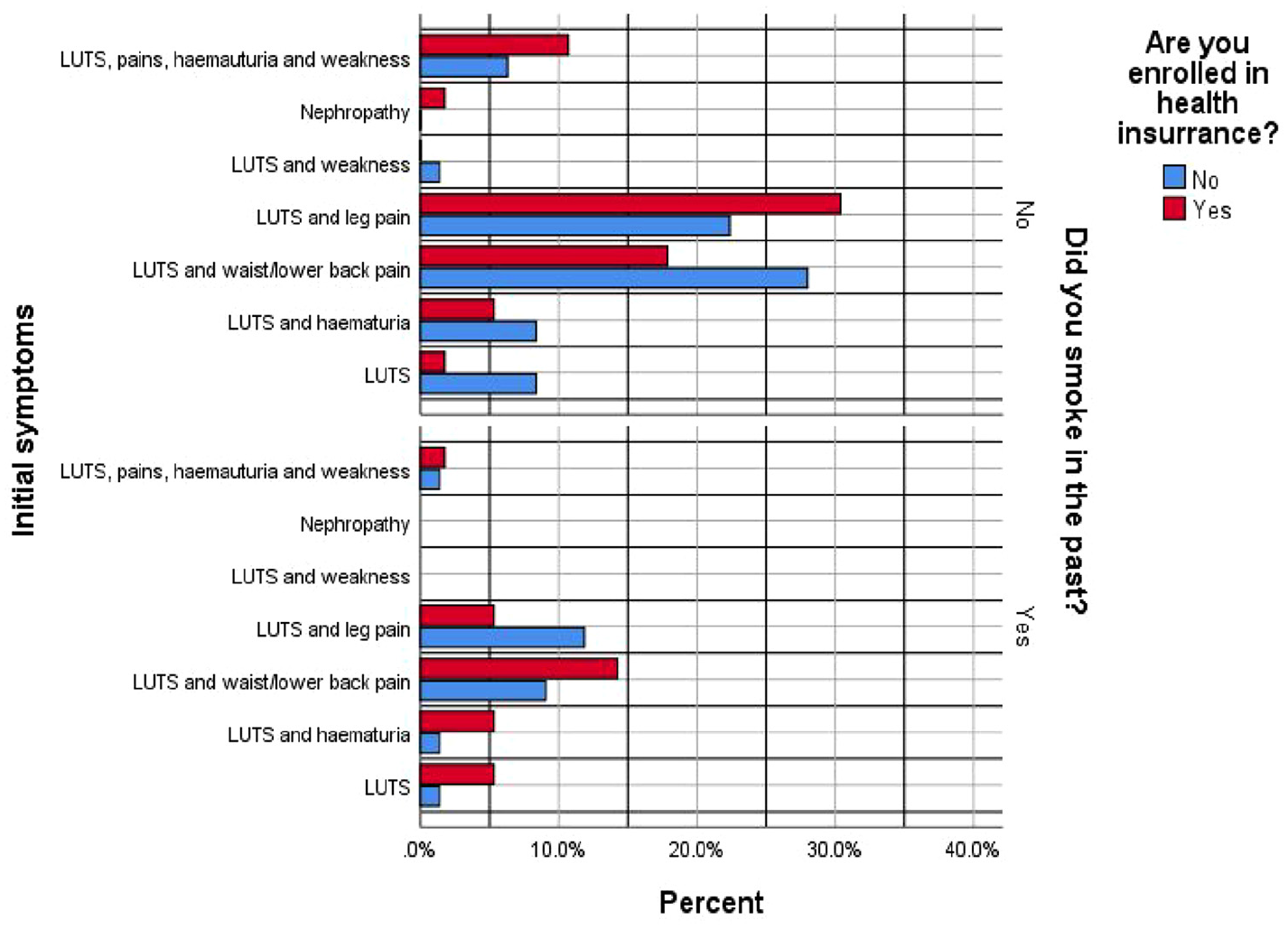
Distribution of Symptoms Based on Enrollment in Health Insurance and History of Smoking
The estimated marginal means across type of cancer in family history, was highest for patients who had a positive family history of PCa, and least for patients who did not have knowledge of their family history of cancer. The result shows the average PSA for persons with no positive history of cancer (mean = 143.989; 95% confidence interval [CI] = 114.849–173.129), positive family history of PCa (Mean = 165.463; 95% CI = 131.435), positive family history of cervical cancer (Mean = 133.456; 95% CI = 49.335–217.576) and persons who did not have knowledge of their family cancer history (Mean = 121.546; 95% CI = 89.234–153.857). Findings from the univariate one-way (F-Tests) showed that family history of cancer had no significant impact on patients’ PSA at diagnosis (R2 = 0.017; adjusted R2 = 0.002; df = 3; F = 1.154; p = .329). See Table 3.
Effect of Family Cancer History on PSA at Diagnosis
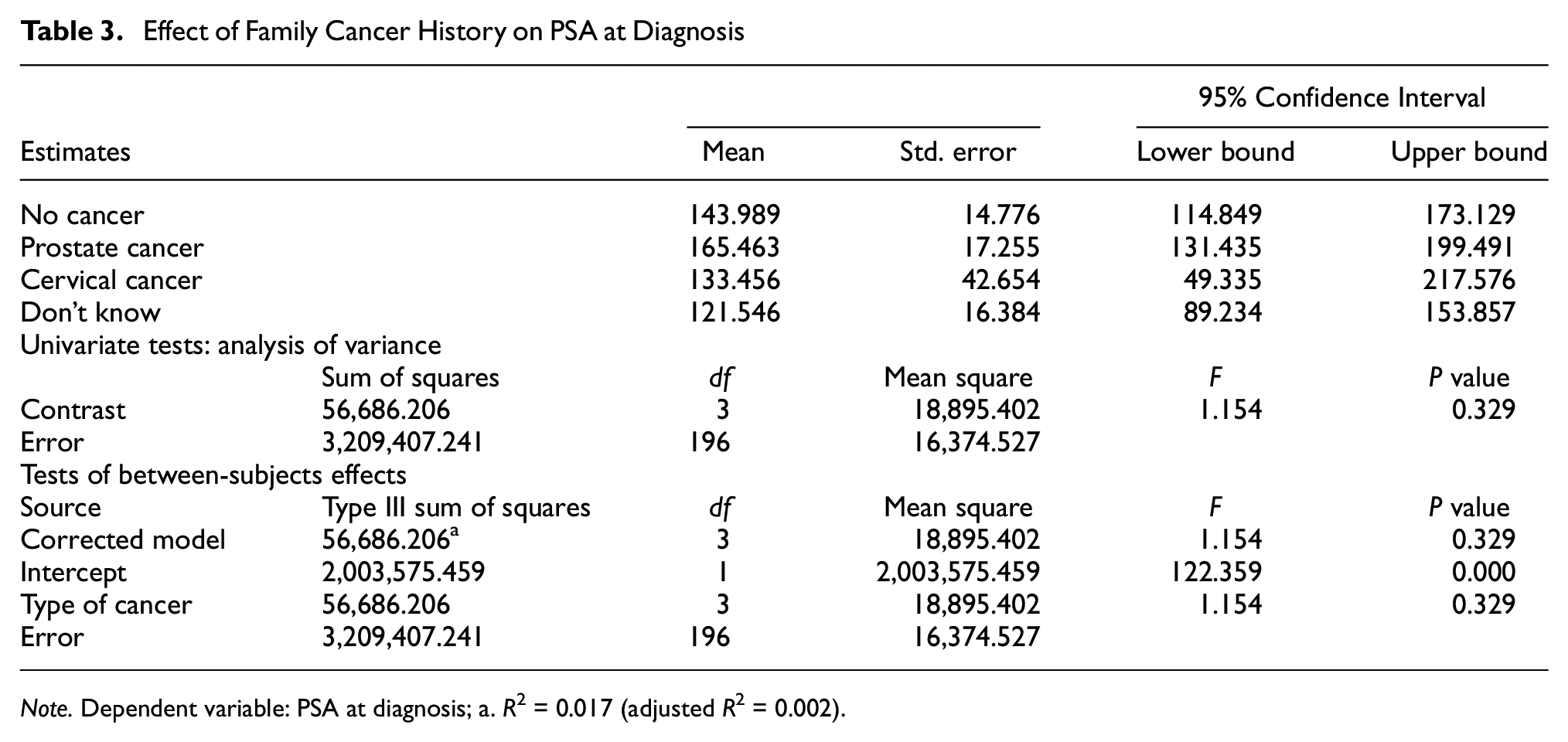
Note. Dependent variable: PSA at diagnosis; a. R2 = 0.017 (adjusted R2 = 0.002).
Discussion
Most study participants were married, retired (pensioners), or artisans. Being married has been demonstrated to have positive effects on PCa outcomes. For instance, previous large data analyses reported that marital status has a significant influence on the survival of postoperative and other PCa patients, as married PCa patients had longer survival time than unmarried, divorced, or widowed patients (Krongrad et al., 1996; Li et al., 2022). Similarly, a 10-year retrospective study reported that unmarried men with PCa had a greater risk of PCa-specific death and all-cause mortality after radical prostatectomy (Khan et al., 2019). Single men also appear to have elevated risks of high-grade PCa as identified in a study by Salmon et al. (2021). PCa patients’ marital status is an important factor to be considered during PCa-related counseling. Also, the observed retirement status of most participants is expected, as the observed mean age of participants is within the stipulated retirement age for workers in Nigeria. A study noted that retirement does not worsen LUTS or bowel symptoms (Bennett et al., 2018), and another study observed that quality of retirement experience was not related to PCa, although only few had access to health insurance (Bednarek & Bradley, 2005).
A large proportion of the study participants were not enrolled in a health care insurance scheme, and several percentage of these persons had symptoms of advanced/metastatic disease. This supports findings from a previous study in Nigeria where out-of-pocket payment for health services was observed as the primary means of access to health care services for cancer care in Nigeria (Mustapha et al., 2020). The perceived burden of incurred out-of-pocket costs for PCa is significant and has a potential negative relationship with PCa outcomes (Akin-Odanye et al., 2021). Therefore, policies that ensure wider coverage of health care insurance scheme should be explored for greater access.
Most of the participants did not smoke, but took alcohol occasionally and did not exercise. This agrees with findings from a previous study in Nigeria which also reported no association between smoking and PCa, as well as alcohol consumption and PCa (Agalliu et al., 2015). On the contrary, another study reported a significant relationship between higher alcohol intake and PCa risk (Rota et al., 2012), suggesting the need for cotrolled alcohol intake. It is also essential to note that physical activity is an important contributor to the risk and progression of PCa. A study reported that persons who had regular exercise had lower risks of PCa and lower risk of high-grade PCa, as well as less likelihood for fatigue (Keogh & MacLeod, 2012). Therefore, interventions such as strategic messaging on regular exercise are crucial for stakeholders.
Findings showed that study participants were mostly in their seventh decade of life and age was significantly associated with several clinical data. The observed prevalent age at diagnosis is consistent with findings from previous studies in Nigeria (Ajape et al., 2010; Ikuerowo et al., 2013; Ofoha & Magnus, 2019; Osegbe, 1997), but contrary to a national study in the United States where the mean age of most patients was within the sixth decade (Bechis et al., 2011). This difference in age at diagnosis reported in these studies may be unrelated to time of initial symptoms, but time of presentation at health facilities for diagnosis. Most patients in Nigeria present for diagnosis at later stages of the disease, implying at a later age (Ajape et al., 2010; Ekeke et al., 2012; Ikuerowo et al., 2013; Ofoha & Magnus, 2019; Osegbe, 1997). Findings imply that there is the need for greater focus on this age range, for routine monitoring and prompt treatments.
The age-line plot shows that age at diagnosis did not quite differ for current age in a large number of the patients, suggesting that most of the patients were recently diagnosed. According to previous studies in Nigeria, several patients are lost to follow-up (Yawe et al., 2006), and there is a high rate of PCa-associated mortality in Nigeria (Ajape et al., 2010; Ekeke et al., 2012; Ikuerowo et al., 2013; Osegbe, 1997). These may explain the presence of mostly newly diagnosed patients in our 1-year study. Interventions that target effective follow-up of patients, and approaches that enhance the life-span of PCa patients are very essential in Nigeria.
Also consistent with previous findings in Nigeria (Ekeke et al., 2012), few patients had a family history of PCa, while it was absent or unknown in the majority of patients. Also, the estimated marginal means across family history of cancer was highest for patients who had a family history of PCa, supporting findings by Barber et al. (2018), in the United States and Xu et al. (2021), in Sweden, which suggest that family history of PCa is a strong risk factor for the disease. However, our model estimates suggest that average PSA does not significantly differ for patients according to family history of cancer, which is consistent with reports from Grann which suggests that family history contributes only a small proportion of cases (Gann, 2002). These known risk factors probably contributed to the disease at varying degrees among our study participants. Although non-modifiable risk factors are implicated in PCa, attention should be channeled to modifiable risk factors such as exercise and so on to improve outcomes.
High Gleason scores and high PSA values at diagnosis were most common among the study participants. This corroborates findings from other studies in Nigeria (Ekeke et al., 2012; Ofoha & Magnus, 2019), suggesting that the trend has not changed over time. This findings implied that most patients presented late at the health facilities. PCa is mostly detected on the basis of elevated plasma levels of PSA; a glycoprotein normally expressed by prostate tissue. Gleason scores are suggestive of PCa prognosis, including its grade and aggressiveness. The observed high scores is likely a result of late-stage disease, resulting from delayed presentation of patients to the hospitals, as noted in previous studies in Nigeria (Ajape et al., 2010; Ekeke et al., 2012; Ikuerowo et al., 2013; Ofoha & Magnus, 2019; Osegbe, 1997). According to previous research, Nigerian men present to the care centers after several months of onset of symptoms (Ajape et al., 2010). A worldwide study of PCa in men of African descent also identified that tumor stage and grade were highest in Sub-Saharan Africa (Rebbeck et al., 2013). Policies that enhance access to PCa screening in Nigeria are needed for early detection and better disease outcomes.
LUTS/lower back pain/leg pain were the most common symptoms at presentation, which is consistent with a previous study in a Nigerian population (Ekeke et al., 2012), and contrary to a finding in the United Kingdom (Hamilton et al., 2006). Aside LUTS, pain was also a most frequently reported symptom of advanced PCa in 11 countries comprising European, American and Asian countries (Jefferson et al., 2020). Aside paraplegia, other observed clinical complications at diagnosis implied that most patients had advanced or metastatic PCa, which is consistent with the already overwhelming evidence that patients present for PCa diagnosis at advanced or metastatic stage of the disease in Nigeria (Ajape et al., 2010; Ekeke et al., 2012; Ikuerowo et al., 2013; Ofoha & Magnus, 2019; Osegbe, 1997). Low awareness, low access to health care/specialist care, high cost of PCa diagnosis and the prevailing low socio-economic status of persons in Nigeria are the most likely factors that contribute to this finding. Measures such as free PCa screening and other means that encourage early disease detection are necessary in the country.
Hypertension was the most common comorbidity and CCBs were the most used medications. This corroborates findings by Ofaha and Magnus (2019) in Nigeria and Jefferson et al. (2020), in the United States. Comorbidities have been observed to adversely affect PCa outcomes and is associated with late-stage PCa diagnosis (Ekeke et al., 2012; Xiao et al., 2016). Previous studies in Nigeria and Spain also noted that comorbidities significantly influenced quality of life of PCa patients (Lozano-Lorca et al., 2023; Mustapha et al., 2020).
The study provides evidence of the distribution of PCa among patients in health facilities, and the effect of patients’ family history of cancer on their PSA at diagnosis; however, findings may be limited by several factors. These factors include the inclusion of unconfirmed cases of PCa with substantial clinical diagnosis, which may have exaggerated the total number of cases. Limitations of self-reporting bias may also be associated with the results of this study.
Conclusion
PCa mostly occurred in men within 60 to 70 years of age, and patients mostly presented to health facilities at advanced or metastatic stages of the disease, with several complications. The study also suggests that family history of cancer may not determine patients’ PSA value at diagnosis. Findings have shown evidence of patients’ late hospital presentation, necessitating the need for policies and strategies that encourage early PCa screening and diagnosis.
Footnotes
Acknowledgements
Nil
Author Contribution
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
