Abstract
Coronavirus disease 2019 (COVID-19) has been reported to decrease semen quality in reproductive-age men. Semen quality in vaccinated men after SARS-CoV-2 infection remains unclear. We recruited reproductive-age Chinese men scheduled for COVID-19 vaccination from December 2022 to March 2023. Among 1,639 vaccinated participants, an upward trend was found in sperm concentration (p < .001), progressive motility (p < .001), total motility (p < .001), total motile sperm count (TMSC) (p < .001), and normal morphology (p = .01) over time following COVID-19 recovery. Among men with an SARS-CoV-2 infection that lasted less than 30 days, men who received an inactivated vaccine booster had higher sperm progressive (p = .006) and total motility (p = .005) as well as TMSC (p = .008) than those without a booster vaccine, whereas no difference was found in semen parameters among men who received a recombinant protein vaccine. Similarly, an upward trend in semen quality was found among 122 men who provided semen samples before and after COVID-19. Higher risks of asthenozoospermia (odds ratio [OR] = 2.23, p < .001) and teratozoospermia (OR = 2.09, p = .03) were found among men who had an SARS-CoV-2 infection that lasted less than 30 days than among those without COVID-19. Collectively, after receiving SARS-CoV-2 vaccination, adverse but reversible semen parameters were observed in men recovering from COVID-19 over time. Recombinant protein vaccines and inactivated vaccine boosters should be recommended to all reproductive-age men.
Introduction
Since the end of 2019, coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and its variants, has presented a global health emergency (Chan et al., 2020). In December 2022, patients infected with SARS-CoV-2 variant Omicron rapidly increased in China. SARS-CoV-2 enters the host cell via the angiotensin-converting enzyme 2 (ACE2) receptor and transmembrane serine protease 2 (TMPRSS2). Previous studies identified that ACE2 and TMPRSS2 proteins are both highly enriched in the testis, especially in spermatogonia, Leydig cells, and Sertoli cells, and SARS-CoV-2 RNA is found in the ejaculate in symptomatic cases of COVID-19 (Ata et al., 2023; Qi et al., 2021; Saylam et al., 2021; Z. Wang & Xu, 2020). SARS-CoV-2 infection can directly affect spermatogenesis and sperm parameters, supporting an adverse correlation between male reproductive health and COVID-19 (Ata et al., 2023; Best et al., 2021; Gacci et al., 2021; Guo et al., 2021; Holtmann et al., 2020).
COVID-19 vaccines are the most effective and critical tools for controlling the ongoing pandemic (Z. R. Yang et al., 2023). Three types of COVID-19 vaccines, including adenovirus vector vaccines (e.g., Ad5-nCoV), inactivated vaccines (e.g., Sinovac and Sinopharm), and recombinant protein vaccines (e.g., ZF2001), are widely used in China (Dong et al., 2022). An Ad5-nCoV vaccine based on an adenoviral Ad5 vector expressing the S protein of SARS-CoV-2 was developed by CanSino Biologics (F. C. Zhu, Li, et al., 2020). Clinical trials of the vaccine were conducted in China, Argentina, Chile, Mexico, Pakistan, and Russia (Halperin et al., 2022; S. Wu et al., 2021; F. Zhu et al., 2022; F. C. Zhu, Guan, et al., 2020; F. C. Zhu, Li, et al., 2020). The vaccine was 57.5% and 91.7% efficacious against polymerase chain reaction (PCR)-confirmed and severe COVID-19 infection beginning 28 days postvaccination, respectively (Halperin et al., 2022). All phase trials reported that one dose of Ad5-nCoV was efficacious and safe in the general population. Inactivated viruses are often used for vaccine development, and neutralizing antibody responses have been found for several COVID-19–inactivated vaccines (Che et al., 2021; Z. Wu et al., 2021; Xia et al., 2020, 2021; Y. Zhang et al., 2021). CoronaVac, based on separate SARS-CoV-2 isolates from Chinese patients, was developed by Sinovac Life Sciences. The vaccine was well tolerated and induced humoral responses against SARS-CoV-2 in recruited participants aged 18 years and older (Z. Wu et al., 2021; Y. Zhang et al., 2021). In addition, CoronaVac has been reported to reduce COVID-19 infections, transmissions, and hospitalizations in phase clinical trials from Brazil, Indonesia, and Turkey (Hitchings et al., 2021; Tanriover et al., 2021). Another inactivated vaccine, BBIBP-CorV, was developed by the Beijing Institute of Biological Products and has shown an acceptable safety profile and immunogenicity in participants (H. Wang et al., 2020; Xia et al., 2020, 2021, 2022). A randomized clinical trial from the United Arab Emirates and Bahrain reported that the efficacy of BBIBP-CorV was 78.1% among participants aged 18 years and older (Al Kaabi et al., 2021). Similar to the above two inactivated SARS-CoV-2 vaccines, KCONVAC, developed by Shenzhen Kangtai Biological Products, was safe and well tolerated, inducing immune responses in adults (Pan et al., 2021). To date, inactivated vaccines have been widely used in China because of their advantages, including mature manufacturing processes and easy production and storage (Y. Wang et al., 2022). The receptor-binding domain (RBD) of the SARS-CoV-2 S protein is an attractive vaccine target against COVID-19. The recombinant protein vaccine ZF2001, developed by Anhui Zhifei Longcom Biopharmaceutical, is an RBD-based COVID-19 vaccine that was reported to be safe and effective against symptomatic and severe-to-critical COVID-19 for at least half a year (Dai et al., 2022; S. Yang et al., 2021).
During the early vaccine promotion stage, vaccine hesitancy due to the potential impact on reproductive health emerged (Rzymski et al., 2021). Notably, there has been no evidence of an adverse effect of SARS-CoV-2 vaccination on semen quality in the short term, and more research should focus on a long-term effect on male reproductive function after SARS-CoV-2 vaccination (Huang et al., 2023; Zace et al., 2022). The impact of SARS-CoV-2 vaccination on semen quality among males who recovered from COVID-19 remains unclear. As the number of vaccinated people increases, the confusion generated on social media about the effects of the COVID-19 vaccine needs to be addressed.
In this study, using a large data set from China, we focused on the effect of COVID-19 on semen parameters and sperm kinematics in men who underwent SARS-CoV-2 vaccination. To evaluate the positive effects of vaccines related to sperm quality, the association between COVID-19 and the risk of abnormal semen parameters in vaccinated men was also investigated.
Methods
This study was approved by The First Affiliated Hospital of University of Science and Technology of China Ethical Committee (Hefei, China; approval no. 2022-RE-417). All analyses were performed according to the Declaration of Helsinki and reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. Written informed consent was obtained from all participants.
Study Population
This single-center retrospective study at the First Affiliated Hospital of University of Science and Technology of China recruited reproductive-age men scheduled for COVID-19 vaccination from December 2022 to March 2023. The available vaccines were Ad5-nCoV, CoronaVac, BBIBP-CorV, KCONVAC, and ZF2001. Generally, the Ad5-nCoV vaccine was administered as a one-dose intramuscular injection. The inactivated vaccines were administered in at least two intramuscular injections. The recombinant protein vaccine was given in at least two doses intramuscularly. All participants underwent COVID-19 vaccination between January 2021 and January 2022. The inclusion criteria were as follows: (1) Chinese men; (2) aged >18 years; and (3) at least one semen sample available. The exclusion criteria included participants with azoospermia, genital surgery (testicular cancer, inguinal hernia, cryptorchidism, and testicular torsion), or genetic defects related to male reproduction. Unvaccinated men or those vaccinated with less than two doses of an inactivated vaccine were also excluded. Ultimately, 1,639 men were enrolled in this study, including 122 men who had semen analysis within 1 year prior to SARS-CoV-2 infection (Figure 1).

Flow Diagram of the Selection Process of the Eligible Study Population
Questionnaire
During the semen laboratory test, all participants completed a baseline questionnaire on lifestyle and reproductive histories, including alcohol consumption, smoking, and relevant confounders (cryptorchidism and genital surgery). Body weight and height were self-reported, and body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. Through the questionnaires, participants were asked if they had ever had a positive SARS-CoV-2 test by PCR or rapid antigen testing.
Semen Analysis
Semen samples were collected from all participants at least 6 months after the last dose of a vaccine. All participants were asked to ejaculate using masturbation. Specimen information, including the day of abstinence, was recorded. Semen parameters were measured after liquefaction at 37°C for at least 30 min. Semen volume was calculated by weighing the semen sample. Sperm concentration, progressive motility, and total motility were evaluated by computer-assisted sperm analysis (CASA, SAS-II system, SAS Medical, China). The total motile sperm count (TMSC, volume × sperm concentration × % progressive motility) was also calculated. The CASA system also provides several sperm motility kinematics: curvilinear velocity (VCL), straight-line velocity (VSL), average path velocity (VAP), frequency of beat cross (BCF), mean amplitude of lateral head displacement (ALH), linearity coefficient (LIN), straightness coefficient (STR), wobble coefficient (WOB), and mean angular displacement (MAD). According to the 2010 World Health Organization (WHO) laboratory manual for the examination and processing of human semen, sperm morphology was evaluated with a semen slide stained with Diff-Quick (Ankebio, Hefei, China). Leukocytes were stained using the Peroxidase Leukocyte Kit (Ankebio). Antisperm antibodies (AsAs) were measured by the mixed antiglobulin reaction (MAR) method (Ankebio). The sperm DNA fragmentation index (DFI) and high DNA stainability (HDS) were determined by sperm chromatin structure assay (SCSA, Celula, Chengdu, China).
Assessment of Semen Parameters
Since the full process of spermatogenesis takes approximately 74 days in humans, infected participants were further subdivided into three groups based on the day of semen analysis after SARS-CoV-2 infection: short term (infected less than 30 days, time point 1, T1), medium term (infected between 30 and 90 days, time point 2, T2), and long term (infected more than 90 days, time point 3, T3).
The data were classified into two cohorts. In cohort 1, semen analyses were performed for all participants and included semen volume, sperm concentration, sperm progressive motility and total motility, TMSC, sperm normal morphology, leukocytes, AsAs, sperm DFI, and HDS. In Cohort 2, semen parameters, including semen volume, sperm concentration, sperm progressive motility and total motility, TMSC, and sperm normal morphology, were collected from 122 participants who had semen analysis within 1 year prior to SARS-CoV-2 infection.
Statistical Analysis
Qualitative variables are reported as frequencies, and quantitative variables are reported as the mean values ± standard deviations (SD) or as the medians (interquartile ranges [IQRs]). For comparisons between the two groups, Student’s t-test and the Mannã Whitney U-test were used for parametric and nonparametric data, respectively, and Pearson’s chi-square test was used as appropriate. For comparison of more than two groups, the Kruskalã Wallis test was employed for nonparametric comparisons, and Pearson’s chi-square test was used for categorical variables. The Wilcoxon rank sum test was used to compare pre- and post-infection semen parameters. According to the WHO guidelines, the lower reference limits for semen parameters are as follows: sperm concentration <15 × 106/ml (oligozoospermia), sperm progressive motility <32% (asthenozoospermia), and normal morphology <4% (teratozoospermia). Covariates initially included factors possibly associated with semen parameters, including age, BMI, smoking, drinking, education, and types of participants. All tests were two-sided, and p < .05 was considered statistically significant. Statistical analyses were carried out using GraphPad Prism 9.0 (GraphPad Software, La Jolla, CA, USA).
Results
A total of 1,639 participants with a mean age of 31.0 ± 4.9 years and BMI of 24.5 ± 3.6 kg/m2 were enrolled in this study (Supplementary Table S1). Among the participants, 1,397 subjects (85.2%) suffered from COVID-19. In terms of lifestyles, 855 subjects (52.2%) consumed alcohol, and 702 subjects (42.8%) smoked tobacco. Regarding education level, 340 subjects (20.8%) had a middle school or lower education, 276 subjects (16.8%) had a high school education, and 1,023 subjects (62.4%) had a college or university education. The studied population included 867 healthy subjects (52.9%, men not selected regarding fertility status) and 772 subfertile subjects (47.1%, partners failed to conceive after 1 year of unprotected intercourse). The incidence rates of diabetes, hypertension, and dyslipidemia were 22 (1.3%), 84 (5.1%), and 74 (4.5%), respectively. Among participants, 1,065 subjects (65.0%) received inactivated vaccines (864 [52.7%] Sinovac, 186 [11.4%] BBIBP-CorV, and 15 [0.9%] KCONVAC), 498 subjects (30.4%) received recombinant protein vaccine ZF2001, 49 subjects (3.0%) received inactivated and recombinant protein vaccines (Sinovac/BBIBP-CorV/ZF2001), 23 subjects (1.4%) received viral vector vaccines (Ad5-nCoV), and four subjects (0.2%) received inactivated and mRNA vaccines (Sinovac/BBIBP-CorV/BNT162b2). Of these participants who received inactivated vaccines, 562 subjects (52.8%) received one or two booster doses.
Among all recruited men, the median sperm concentration and TMSC were 52.8 million/ml (IQR = 26.1–94.4) and 57.6 million (IQR = 21.0–134.4), respectively (Supplementary Table S2). Semen parameters were similar between the noninfected and infected groups (Table 1). An upward trend was found in sperm concentration (p < .001), progressive motility (p < .001), total motility (p < .001), TMSC (p < .001), and normal morphology (p = .01) over the study period following COVID-19. Compared with those without COVID-19, there were significant decreases in sperm progressive motility (p < .001) and total motility (p < .001), TMSC (p = .003), and normal morphology (p = .01) among men recovery from COVID-19 less than 30 days. Of those men recovery from COVID-19 more than 90 days, several semen parameters, including concentration (p = .001), progressive motility (p = .01), total motility (p = .01), TMSC (p = .001), and leukocytes (p = .02), were significantly increased when compared with noninfection values. We next investigated the relationship between COVID-19 and abnormal semen parameters according to the WHO lower reference limits among vaccinated men (Figure 2). Men recovery from COVID-19 less than 30 days were more likely to have sperm progressive motility (p < .001) and total motility (p < .001) limits below the WHO reference limits than men without COVID-19. In contrast, men recovery from COVID-19 more than 90 days had a lower probability of having a below-reference sperm concentration (p < .03), progressive motility (p < .02), total motility (p < .02), and TMSC (p < .01) than uninfected men. Consistent results were found in the healthy and subfertile subgroups (Supplementary Table S3).
Semen Parameters in Men With and Without SARS-CoV-2 Infection
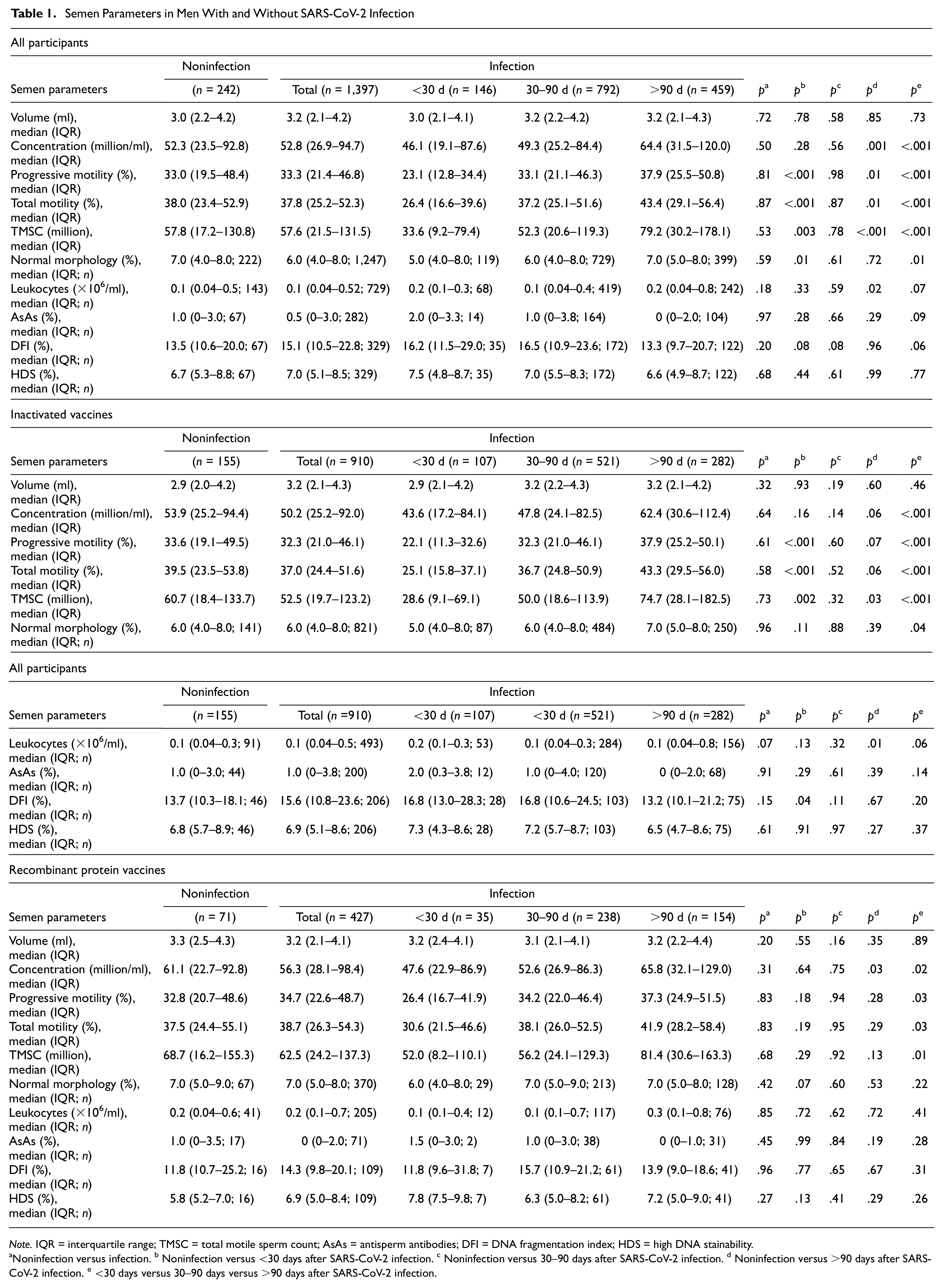
Note. IQR = interquartile range; TMSC = total motile sperm count; AsAs = antisperm antibodies; DFI = DNA fragmentation index; HDS = high DNA stainability.
Noninfection versus infection. b Noninfection versus <30 days after SARS-CoV-2 infection. c Noninfection versus 30–90 days after SARS-CoV-2 infection. d Noninfection versus >90 days after SARS-CoV-2 infection. e <30 days versus 30–90 days versus >90 days after SARS-CoV-2 infection.
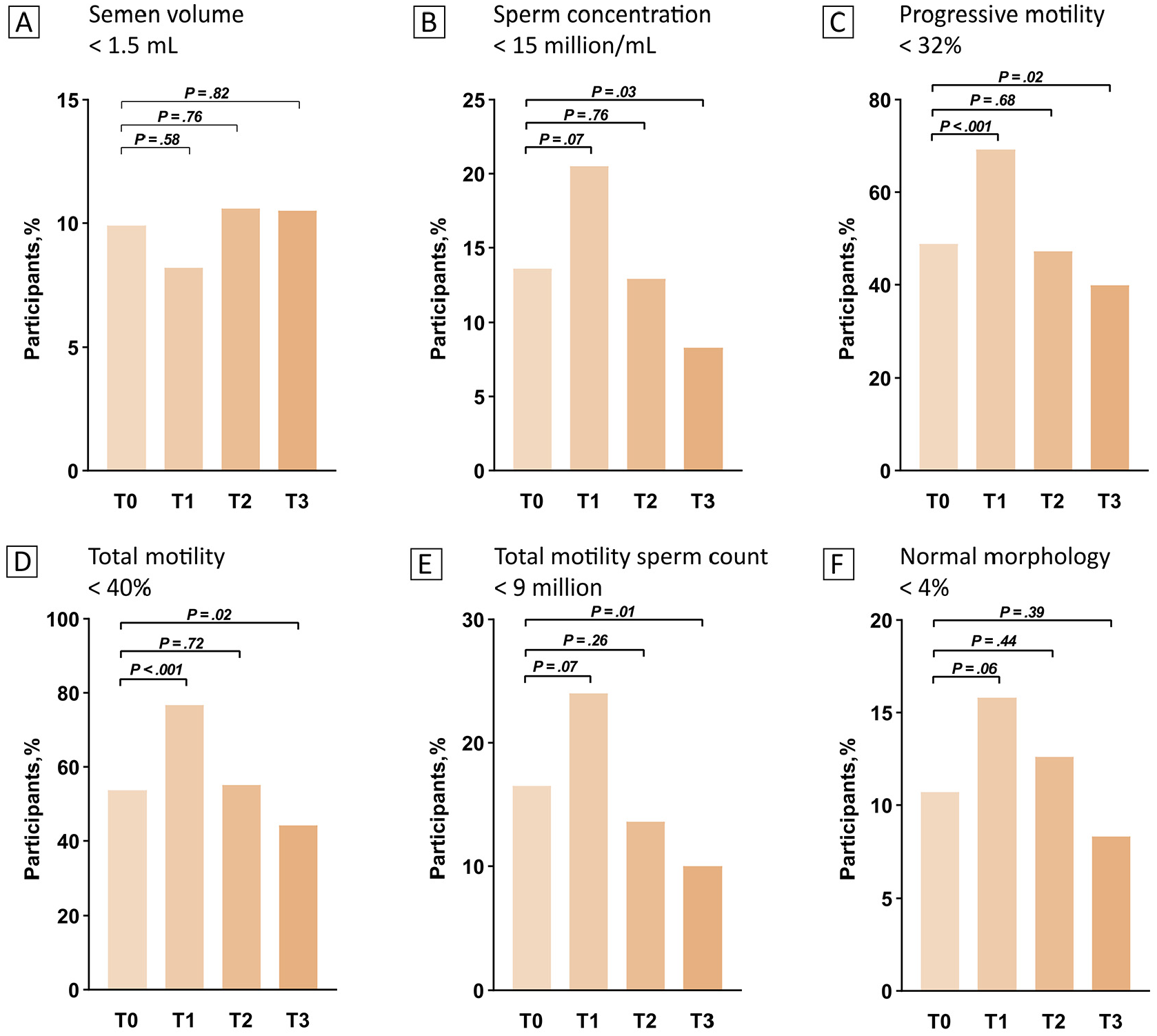
Abnormal Semen Parameters According to World Health Organization Lower Reference Limits Among Vaccinated Men With or Without COVID-19
The potential effects of different types of vaccines on semen parameters recovered from COVID-19 were further investigated (Table 1). Among men who received an inactivated vaccine, an improving trend of sperm concentration (p < .001), progressive motility (p < .001), total motility (p < .001), TMSC (p < .001), and normal morphology (p = .04) was observed among men who recovered from COVID-19 over time, and this trend was also found in both the Sinovac and BBIBP-CorV vaccine subgroups (Supplementary Table S4). In addition, similar semen quality results were observed for men who received recombinant protein vaccines (Table 1), adenovirus vector vaccines, and combined inactivated and recombinant protein vaccines (Supplementary Table S5). In the inactivated vaccine group, men recovery from COVID-19 less than 30 days were more likely to have lower sperm progressive motility (p < .001), total motility (p < .001), and TMSC (p = .002) and higher sperm DFI (p = .04) than those without SARS-CoV-2 infection, whereas no difference was found in semen parameters over the study period following COVID-19 among men who received a recombinant protein vaccine (Table 1). Similar relationships were observed in sperm kinematics between men with and without COVID-19 (Supplementary Table S6).
In terms of inactivated vaccine recipients, we observed that booster-vaccinated men had higher sperm progressive (p = .006), and total motility (p = .005) as well as TMSC (p = .008) than those who did not receive a booster dose during infection less than 30 days prior (Table 2). No significant changes were observed in semen parameters during later infection after 30–90 days followed by more than 90 days.
Semen Parameters in Men With and Without an Inactivated Vaccine Booster

Note. IQR = interquartile range; TMSC = total motile sperm count; AsAs = antisperm antibodies; DFI = DNA fragmentation index; HDS = high DNA stainability.
Comparison of booster and nonbooster vaccination among all men with SARS-CoV-2 infection. b Comparison of booster and nonbooster vaccination among men less than 30 days after SARS-CoV-2 infection. c Comparison of booster and nonbooster vaccination among men between 30 and 90 days after SARS-CoV-2 infection. d Comparison of booster and nonbooster vaccination among men more than 90 days after SARS-CoV-2 infection.
In Cohort 2, 122 men before and after COVID-19 were included in this study (Figure 3). When compared with the values of sperm parameters before COVID-19, a downward trend was found in sperm concentration and motility during SARS-CoV-2 infection less than 30 days prior, although these values were not significant. Compared with those without infection, lower sperm concentrations (p = .04) and TMSC (p = .01) were observed in men recovery from COVID-19 after 30–90 days, whereas an improved trend was found in sperm concentration, progressive motility, total motility, TMSC, and leukocytes in men recovery from COVID-19 more than 90 days, although only the difference in sperm concentration (p = .02) and leukocytes (p = .01) was significant. Similar trends were observed in semen quality before and after COVID-19 among healthy and subfertile men after undergoing SARS-CoV-2 vaccination (Supplementary Table S7).

Semen Parameters Before and After COVID-19 Among Men Who Underwent SARS-CoV-2 Vaccination
Associations between COVID-19 and the risk of abnormal semen parameters are shown in Table 3. Higher risks of asthenozoospermia( odds ratio [OR] = 2.23, 95% confidence interval [CI] = [1.41, 3.55], p < .001) and teratozoospermia (OR = 2.09, 95% CI = [1.08, 4.05], p = .03) were found among men recovery from COVID-19 less than 30 days than among those without COVID-19. In addition, men recovery from COVID-19 more than 90 days had decreased odds of asthenozoospermia after adjusting for confounders (OR = 0.69, 95% CI = [0.50, 0.96], p = .03). Similar associations were found in the subgroup analysis of men who received inactivated vaccines compared with those who received recombinant protein vaccines. In addition, consistent correlations were found in sensitivity analyses after adjusting for age, BMI, smoking, and drinking (Supplementary Table S8).
Odds Ratios (95% Confidence Intervals) for Abnormal Semen Parameters Across Time After SARS-CoV-2 Infection

Note. OR = odds ratio.
Discussion
Previous reports have suggested an increased risk of spermatogenesis defects among men with SARS-CoV-2 infection. Here, in a cohort of men who underwent COVID-19 vaccination, we found that semen quality was decreased in men with SARS-CoV-2 infection less than 30 days (short term) prior who recovered after 90 days (long term), suggesting adverse and reversible effects during SARS-CoV-2 infection over time. At the same time, men who received inactivated vaccines were more likely to have lower semen quality than those who received recombinant protein vaccines during short-term COVID-19. Men who received booster doses of an inactivated vaccine had better semen quality than those who did not receive booster doses. To our knowledge, this is the largest study to show the potential effects of COVID-19 vaccination on semen quality among men after SARS-CoV-2 infection.
Many studies have reported a decline in semen quality after SARS-CoV-2 infection when compared with uninfected controls (Ata et al., 2023; Leng et al., 2023). Several semen parameters, such as the sperm concentration, progressive motility, and total motility, were all significantly reduced in vaccinated men following short-term COVID-19 when compared with men without COVID-19. At 30–90 days after SARS-CoV-2 infection, semen parameters including sperm concentration, progressive motility, total motility, TMSC, and normal morphology in vaccinated men recovered to uninfected levels. A previous study reported that COVID-19 can lead to long-term (> 4 months) effects on semen quality (Hu et al., 2022; Mannur et al., 2021). In addition, our recent study reported that patients who recovered from COVID-19 at a median time of 76 days were more likely to have lower sperm concentration, progressive motility, and total motility than those without COVID-19 (Guo et al., 2021). Given that a spermatogenic cycle takes approximately 74 days in humans, these findings suggest that the after effects of COVID-19 may last for one spermatogenic cycle. However, in our study, although vaccination did not alleviate the decline in semen quality among men recovery from COVID-19 at the short-term stage, all semen parameters recovered or were even slightly higher in the medium-term (recovery from COVID-19 between 30 and 90 days) and long-term stages (recovery from COVID-19 more than 90 days) when compared with uninfected men. We further performed a retrospective cohort study among 122 vaccinated men to compare pre- and post-COVID-19 semen parameters and confirmed that the semen quality of vaccinated patients infected with COVID-19 decreased in the short term, although the difference was not significant, whereas the semen quality gradually improved in the long term. Thus, we can speculate that vaccination at least partly reduced the duration of poor semen quality after exposure to SARS-CoV-2.
Adverse effects on semen quality might be associated with direct effects and indirect effects (e.g., mental stress and adverse events). One of the most common symptoms in COVID-19 patients is fever, and previous studies have reported temporarily decreased semen parameters in recovered patients with fever symptoms during the course of COVID-19 (Guo et al., 2021; Holtmann et al., 2020; Hu et al., 2022). Although vaccination can reduce severe symptoms of COVID-19, mild and moderate symptoms still occur (C. C. Wu et al., 2022). Therefore, we cannot rule out the impact of several COVID-19-related symptoms on semen quality after SARS-CoV-2 infection among vaccinated men.
Several studies reported that COVID-19 vaccination had no effect on the semen parameters of the general population or infertile patients, whereas little is known about semen quality in vaccinated men recovering from COVID-19 (Huang et al., 2023). Al-Alami et al. (2022) performed a retrospective study among 354 men to investigate the effect of SARS-CoV-2 infection and/or vaccination on semen parameters. They reported that there was no difference between infected individuals and uninfected individuals regardless of COVID-19 vaccination status. However, the above study did not report the collection time of semen samples after COVID-19 or vaccination. In this study, among men who received different types of vaccines, we observed that semen quality was decreased in the short term and recovered after long-term COVID-19. The reduction in semen quality among men with recombinant vaccines after short-term COVID-19 was smaller than that among men with inactivated and viral vector vaccines, which may be related to the low adverse events and high effectiveness of recombinant vaccines (Dai et al., 2022; Halperin et al., 2022).
According to the technical guidelines for inactivated COVID-19 vaccines, two doses should be administered, and vaccine effectiveness against COVID-19 is greatest after boosting (Xu et al., 2023). A previous study reported that inactivated booster vaccination stimulated the response of long-term immunological memory and protected against symptomatic infection caused by SARS-CoV-2 in China (Tang et al., 2022). Our study also found that semen quality among men with an inactivated vaccine booster was significantly superior to that among men with nonbooster injection, thus indicating that boosters can improve the protective effect of the vaccine on semen parameters.
The major strength of our study is its relatively large sample size, affording high statistical power and precision. This study included information on semen parameters and sperm kinematics from vaccinated men before and after SARS-CoV-2 infection as well as the types and doses of vaccines, which provides more evidence that vaccination is a valuable and recommended tool for protecting the reproductive health of male COVID-19 patients. Several potential confounders (e.g., age, BMI, drinking, and smoking) that have been previously reported to be associated with semen quality were accounted for. Considering the time required for the full process of spermatogenesis and sperm maturation in humans, semen samples collected from COVID-19 patients after a follow-up of 3 months constituted a good analysis window, allowing the data to be analyzed according to short-term and long-term effects.
This study has several limitations. First, semen parameters from all participants were based on a single measurement. Previous studies have reported that single semen samples can be used to identify average differences in semen quality between individuals (Chiu et al., 2017; Q. X. Zhu et al., 2016). Second, the severity of the infection was not assessed in this study. However, data from 4,203 patients, mostly from China, identified that more than 80% showed fever symptoms (J. J. Y. Zhang et al., 2020). As the process of spermatogenesis is temperature dependent, an acute fever could cause changes in semen parameters (Sergerie et al., 2007). Third, we lacked data on serum hormones that might affect male reproductive health. Fourth, because of its retrospective design, our study was subject to potential selection bias. Finally, despite adjusting for a series of potential confounders, we could not exclude residual confounding.
Conclusions
In summary, adverse but reversible semen parameters were observed in vaccinated men recovering from COVID-19 over time. Recombinant protein vaccines and inactivated vaccine boosters are recommended for all reproductive-age men, particularly those in couples seeking a fertility evaluation.
Supplemental Material
sj-docx-1-jmh-10.1177_15579883241264120 – Supplemental material for SARS-CoV-2 Vaccination Improves Semen Quality in Men Recovered From COVID-19: A Retrospective Cohort Study
Supplemental material, sj-docx-1-jmh-10.1177_15579883241264120 for SARS-CoV-2 Vaccination Improves Semen Quality in Men Recovered From COVID-19: A Retrospective Cohort Study by Yuanqi Zhao, Yangyang Wan, Xuechun Hu, Xianhong Tong, Bo Xu, Xiaohua Jiang, Shun Bai and Cheng Cao in American Journal of Men's Health
Footnotes
Acknowledgements
The authors thank Xia Wu, BSc, Jing-Ru Xu, BSc, and Yan-Yan Shang, BSc (all with The First Affiliated Hospital of University of Science and Technology of China) for their valuable contributions to the data collection.
Author Contributions
Yuanqi Zhao, Shun Bai, and Cheng Cao designed the study. Yuanqi Zhao and Yangyang Wan contributed to the data acquisition. Yuanqi Zhao, Cheng Cao, Xuechun Hu, Xianhong Tong, Bo Xu, Xiaohua Jiang, and Shun Bai analyzed the data. Yuanqi Zhao, Shun Bai, and Cheng Cao wrote the original manuscript. Xiaohua Jiang, Shun Bai, and Cheng Cao have verified the underlying data. All authors contributed to the review of the final article and accept responsibility for the decision to submit for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (82303601, 82071709, 81901543), the Joint Fund for Medical Artificial Intelligence (MAI2022Q010), the Key Research and Development Project of Anhui Province (2022e07020014), the Joint Research Center for Genomic Resources (2017B01012-2021K001), and the Fundamental Research Funds for the Central Universities (WK9100000061).
Ethics Approval and Consent to Participate
This study was approved by The First Affiliated Hospital of University of Science and Technology of China Ethical Committee (Hefei, China; approval no. 2022-RE-417). All analyses were performed according to the Declaration of Helsinki and reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. Written informed consent was obtained from all participants.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
