Abstract
The objective was to investigate the embryo morphokinitics using a time-lapse monitoring (TLM) system and assessment of clinical outcomes following intracytoplasmic sperm injection (ICSI) with zona pellucida (ZP)-bound sperm selection and conventional methods. A total of 371 metaphase II (MII) oocytes from 50 ICSI cycles were studied. Sibling oocytes were randomly divided into control (n = 199) and ZP-bound group (n = 172). All resulting zygotes were cultured and monitored in the TLM system up to Day 3 after ICSI. Fertilization rate, early embryo development, and clinical outcomes were evaluated. No significant differences were found in fertilization rate, time-lapse qualitative and quantitative measures, pronuclear fading time (PNF) t2, t3, t4, t5, t6, and t7 (times of cleavage to 2, 3, 4, 5, 6, and 7 cells), respectively. However, the t8 (time of cleavage to eight cells) and cc3 (duration of third cell cycle) revealed a significant difference between control and ZP-bound groups (p < .05). A significant difference between the two groups (p < .05) in the rates of Grade A embryos (according to Basile algorithm), chemical pregnancy, clinical pregnancy, and implantation was observed. Sperm selection using biological materials, such as ZP, improved both embryo quality and pregnancy outcomes, despite not affecting the early embryo development and morphokinetic parameters up to t8. This prospective randomized sibling oocyte trial was registered in October 2020 to January 2022 (IRCT20200705048021N1).
Introduction
Intracytoplasmic sperm injection (ICSI) is a well-established reproduction technology used for treatment of about half of the couples with male factor infertility (Nyboe Andersen et al., 2008). However, ICSI can lead to a significant decrease in both implantation and clinical pregnancy rates compared with conventional in vitro fertilization (IVF; Bhattacharya et al., 2001; Yoeli et al., 2000). In ICSI, sperms are subjectively selected by the embryologist, based on their motility and morphology criteria, bypassing the natural selection processes provided by biological interaction and physiological barriers (D. Y. Liu & Baker, 2000; Miller & Smith, 2001). Elimination of biological selection may result in selection of sperm with morphological and DNA abnormalities, thereby lowering the chance of success with ICSI (Mehta & Sigman, 2014). Several novel and traditional methods have been developed to improve sperm selection strategy, including membrane maturity of sperm (Beck-Fruchter et al., 2016; Parmegiani et al., 2010), motile sperm organelle morphology examination (MSOME; Bartoov et al., 2003), non-adherence of sperm to a glass wool column (Jeyendran et al., 1986) and assessing the surface charge of sperm (Ainsworth et al., 2005). However, the most efficient sperm selection method has not yet been determined (Said & Land, 2011). It has been reported that for fertile men, a small fraction (14%) of motile spermatozoa bind to the zona pellucida (ZP) of human oocytes (D. Y. Liu et al., 2003). In addition, several studies have demonstrated sperms that bind to ZP are mature with normal morphology (Garrett & Baker, 2004; Huszar et al., 1994; D. Y. Liu & Baker, 1992) and normal nuclear DNA compared with non-adhering sperm to ZP (Dadoune, 2007; Garrett & Baker, 2004; D. Y. Liu & Baker, 2007) in which potentially increase the mutational load carried by the embryos due to DNA defects (Aitken, 2017a, 2017b). Previous studies reported that the ZP has the ability to select higher quality sperm, which can be used for ICSI (Black et al., 2010; Braga et al., 2009; Casciani et al., 2014; Ganeva et al., 2019; Izadi et al., 2021; Jin et al., 2016). It seems that the human oocyte ZP can be used to help in selecting competent sperm to enhance ICSI clinical outcomes. Where a cohort of collection oocytes contains immature and mature oocytes, the ZPs are biologically similar, despite differing levels of cytoplasmic and genetic maturity (D. Y. Liu & Baker, 2007). As such, the ZP of the sibling immature oocytes are a potential source to be used for sperm selection in ICSI.
Specific morphological defects in the sperm head, midpiece, and tail have been reported to have significant effects on certain stages of embryonic development, from the appearance of pronucleus to the hatched blastocyst. These defects in sperm cells affect both embryo morphokinetic and assisted reproductive technology (ART) outcomes (Nikolova et al., 2020). Therefore, the present trial study was conducted to determine if there was a difference in embryo morphokinetics and clinical outcomes following sperm selected with ZP-bound sperm method versus conventional ICSI. To our knowledge, the morphokinetic of pre-implantation embryos has not been studied in ZP-bound sperm selection in ICSI setting.
Materials and Methods
This prospective randomized sibling oocyte trial was performed at the Reproductive Science Institute in Yazd, Iran from October 2020 to October 2022. This clinical trial was registered in the Iranian Registry of Clinical Trials (IR. CT20200705048021N1) and approved by the Ethics Committee of our institute (IR. SSU.RSI.REC.1398.022). The ICSI candidates were diagnosed with unexplained infertility and failure of fertilization after IVF provided written informed consent.
The inclusion criteria for females were age of <40 years, basal follicle-stimulating hormone (FSH) <10.0 IU/L with at least five to six mature and one immature oocytes (Table 1). The inclusion criteria for men were age of <50, having a semen sample with motile sperm concentration ≥5 × 106/mL and abnormal sperm morphology. Males with non-ejaculated spermatozoa or cryptozoospermia, were excluded. In addition, the cases in which the sperm cells failed to bind to the ZP were excluded. The couples did not have a history of Y chromosome micro-deletion in their diagnostic tests. The baseline laboratory characteristics were recorded before assigning the patients to the control and ZP-bound groups (Figure 1).
Baseline Laboratory Characteristics of the Patients

Note. SD = standard deviation; COC = cumulus-oocyte complexes; BMI = body mass index; FSH = follicle-stimulating hormone; MII = Metaphase II; PRL = prolactin.

Consolidated Standards of Reporting Trials (CONSORT) Diagram of Participants’ Allocation, Treatment, Follow-Up, and Analysis
Clinical Procedure
Seventy candidates for ICSI received the antagonist protocol for control ovarian stimulation, 20 cases were excluded due to the failure of spermatozoa not bounding to the ZP. In total, 371 MII oocytes were aspirated 34 to 36 hr after human chorionic gonadotropin (hCG) injection. Sibling oocytes from each patient were randomly and equally assigned to control (199 oocytes) and ZP-bound group (172 oocytes). After ICSI, incubation and confirmation of fertilization were performed. Afterward, the fertilized oocytes were transferred to time-laps culture dish. The dish was placed in a standard incubator equipped with time-lapse microscopy (TLM) system for morphokinetic assessment. Embryo transfer took place on Day 3 and clinical outcomes were recorded. All procedures were performed by the same blinded embryologist.
Controlled Stimulation of the Ovaries and Oocyte Retrieval
Ovulation stimulation protocol was performed through the long-term downregulation of pituitary and using agonist or antagonist of FSH (Gonal-F; Serono, Geneva, Switzerland) and gonadotropin-releasing hormone (GnRH). After appropriate growth of follicles using transvaginal ultrasound, hCG (Ovidrel; Merck-Serono) was injected to trigger the final follicle maturation and ovulation. Oocytes were then collected 36 hr post-hCG administration.
Collection of Oocytes
Cumulus-oocyte complexes (COCs) were isolated from follicular fluid, and incubated in SynVitro Flush medium (Origio, Cooper Surgical, Denmark) overlaid with oil (Ovoil; Vitrolife, Sweden). The COCs were transferred to universal IVF medium (Medicult, Copenhagen, Denmark) and incubated at 37oC, 6% CO2, 5% O2 for 2 hr. Following denudation, all oocytes were evaluated to determine the nuclear maturation stage. For the process of ZP-bounded sperm selection, the immature oocytes were selected, while mature oocytes were assigned to the ICSI.
Sperm Samples Preparation
Ejaculates were prepared using a discontinuous 80:40 density gradient (Nidacon Laboratories AB, Gothenburg, Sweden). The pellet was washed twice by resuspending in 1 mL, universal IVF medium and centrifuged at 300 g for 5 min. The ultimate sperm preparation was resuspended in 500 μL of the medium (World Health Organization, 2021).
ZP-Bound Sperm Selection
In the ZP group, 5-μL sperm sample (concentration: 1–2 × 106 motile sperm/mL) was added to a 5-μL droplet of universal IVF medium containing 1 immature oocyte (37°C, 6% CO2). After 2 hr, the immature oocytes were carefully washed by repeated aspirations in droplets of 5 μL (universal IVF medium) using a pipette fitted with a 200 to 250 µL tip for removal of loosely bound sperms. The immature oocyte with ZP-bound sperms was transferred to a 5μL droplet of SynVitro Flush medium (Origio, Cooper Surgical, Denmark) overlaid with oil in a plastic culture dish (Nunc153006). The culture dish was finally placed on the warm stage of an inverted microscope.
At ×400 magnification, the spermatozoa that were tightly bound to ZP were separated using a microinjection needle. The sperms isolated from immature oocytes were inserted in a made-droplet of polyvinylpyrrolidone (PVP) solution (Irvine Scientific). After sperm immobilization in the PVP, the sperms were injected into the oocytes allocated to the ZP-bound group (Braga et al., 2009; Casciani et al., 2014; F. Liu et al., 2011). In the control group, sperm selection and microinjection was performed based on the conventional method. In both groups, all injected oocytes were rinsed, then cultured in droplets of SAGE 1-Step medium (Origio, Cooper Surgical, Denmark) overnight at 37°C and 6% CO2.
Embryo Culture and Time-Lapse Monitoring
After incubation (16–18 hr), fertilization was confirmed when two pronuclei were present. Zygotes were transferred to culture dish (Primo Vision, Vitrolife, Sweden) containing 50 μL of pre-equilibrated SAGE 1-Step medium (Origio, Cooper Surgical, Denmark) covered with mineral oil at 5% O2 and 6% CO2, 37°C. Full traceability was provided for each zygote. The embryo culture dish was put in a standard incubator (5% O2 and 6% CO2, 37°C) equipped with TLM system. The images were captured at 3 to 11 focal plane of embryos every 10 min and full scan every 20 min (Faramarzi et al., 2017; Mangoli et al., 2020). Therefore, the embryo development was monitored in real time until Day 3.
Embryo Development Evaluating With TLM Technology
On Day 3, the embryos were evaluated by viewing time-lapse images and analyzed using Embryo Viewer TM (Vitrolife) software. Embryos with direct cleavage defined as a blastomere divided directly from 1 to 3 blastomeres <5 hr; Rubio et al., 2012), uneven blastomere and multinucleation (MN) at the 2 and 4-cell stage (Mn4) were excluded and all embryos with eligible morphology were subjected for scoring according to Basile criteria. Time points of early development stages were determined by Embryo Viewer TM (Vitrolife) software. Briefly, pronuclear fading time (PNF), time of cleavage to 2-cell (24–28 hr) (t2), to 3-cell (30–38 hr) (t3), to 4-cell (35–41 hr) (t4), to 5-cell (48–57 hr) (t5), to 6-cell (t6); to7-cell (t7) to 8-cell (50–59 hr) (t8). In addition, cc2 (8–12 hr) was defined as duration of the second cell cycle, cc3 was defined as duration of the third cell cycle, s2 was defined as second synchronous divisions, and s3 was defined as third synchronous divisions, which were calculated as follows: cc2 = t3 − t2, cc3 = t8 − t4, s2 = t4 − t3, and s3 = t8 − t5, respectively (Basile et al., 2015; Y. Liu et al., 2015; Meseguer et al., 2011; Tejera et al., 2017). The transferred embryos were graded according to Hill et al. (1989) that scored according to the rate of fragmentation and symmetry of the blastomeres A: symmetrical, no fragmentation; B: <10% fragmentation and/or asymmetric; C: 10% to 25% fragmentation; D: > 25% fragmentation with dark granules.
Embryo Selection and Transfer
Two embryos with high quality from one of the groups were selected and loaded by a blinded embryologist based on Hill indices (Hill et al., 1989). Embryo loading and transferring was performed with a Cook catheter (Cook Medical, USA) and SAGE 1-Step medium (Origio, Cooper Surgical, Denmark) (20–30 μL). Supernumerary embryos were cryopreserved for future use.
Outcome Parameters
Clinical pregnancy was defined when fetal heart beat and gestational sac were detected by ultrasound examination after 7 weeks. The secondary outcomes were fertilization rate (number of fertilized oocytes per number of injected oocytes), embryo morphokinetic scores include PNF, t2, t3, t4, t5, t6, t7, t8, cc2, cc3, s1, s2, and s3, implantation rate (ratio between the number of gestational sacs to number of transferred embryos) and chemical pregnancy rate (when b-hCG was positive 14 days after embryo transfer). Some time-points data in the time-lapse system were considered as missing values due to unfocused imaging and/or technical problem. The embryos dividing directly from 1 to 3 cells, 2 to 5 cells and/or MN at 2- and 4-cell stages were excluded.
Statistical Analysis
Normality of the data was assessed using the Kolmogorov–Smirnov test. Quantitative data are presented as mean ± standard deviation (± SD) for normal numeric data and proportions (%) for categorical variables. The unpaired t-test was used for the determination of differences between every two groups. Chi-square test was used to compare classification variables (χ2 test was used to determine the significance of differences between two groups). Values of two-tailed p < .05 were considered significant. SPSS version 22 was used for all statistical analyses.
Results
From a total of 417 collected COCs, 371 MII oocytes were assessed (maturation rate: 88.96%). One hundred seventy-two of the MII oocytes were assigned to the ZP-bound sperm selection group and 182 of the MII oocytes were assigned to control group (Table 1).
Fertilization Rate
In the ZP-bound sperm selection group, 129 oocytes out of 172 were normally fertilized (fertilization rate: 75%); while 133 oocytes from 182 were normally fertilized (73.0%) in controls. No significant differences were noted in the fertilization rates between the groups (t = 0.1, p = .68).
Morphokinetics of Embryos
There was no significant difference in the time points of embryo development (tPNf, t2, t3, t4, t5, t6, and t7) between two groups. However, t8 (t = 2.0, p = .04; Table 2, Figure 2A) and cc3d (t = 2.3, p = .02; Table 2, Figure 2B) in the ZP-bound group were statistically shorter, meaning that embryos in the ZP-bound group reached the 8-cell stages faster than embryos in the control group.
Individual Time Points and Calculated Interval Times in the Time-Lapse Monitoring-System in the ZP-Bound and Control Groups
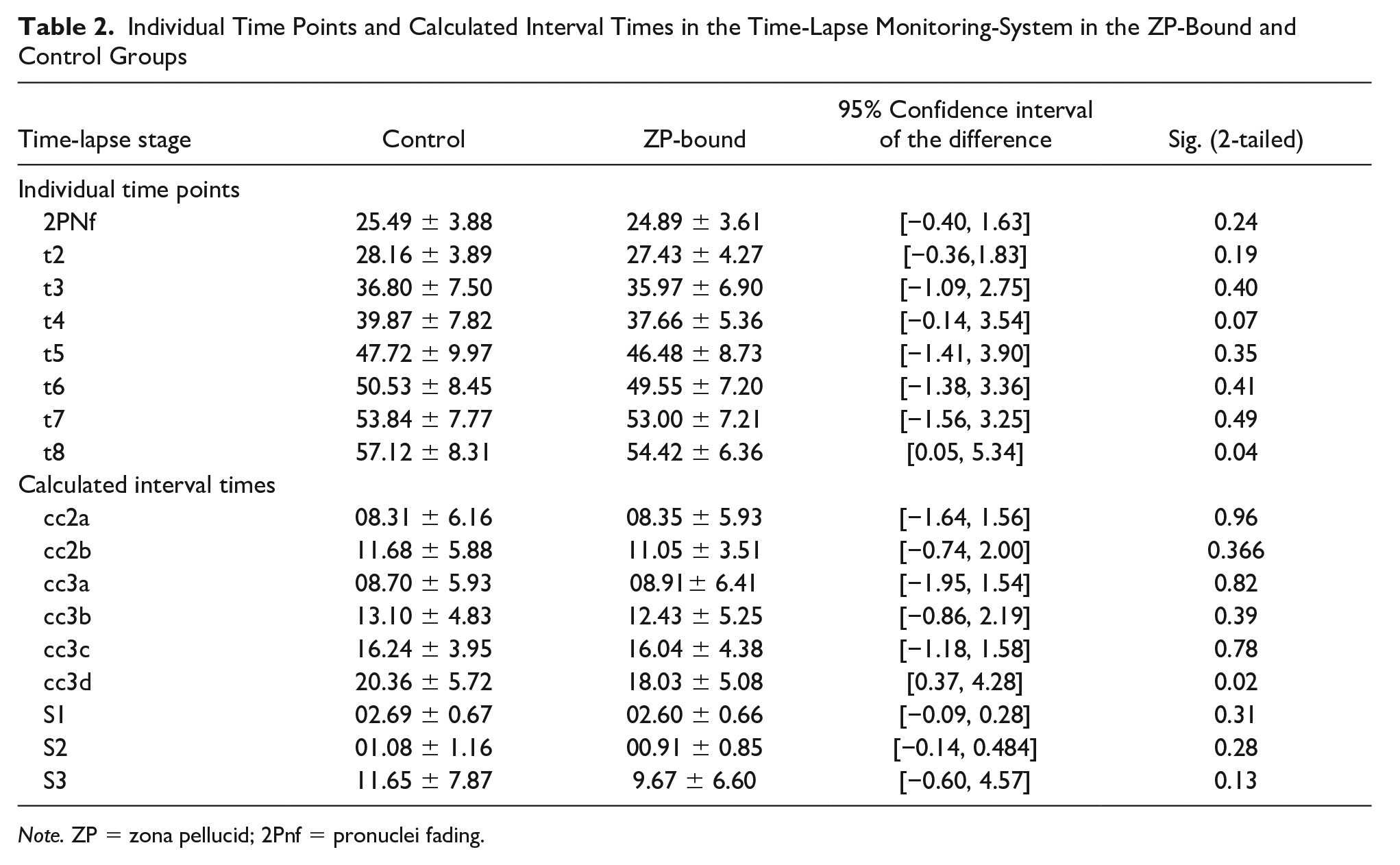
Note. ZP = zona pellucid; 2Pnf = pronuclei fading.
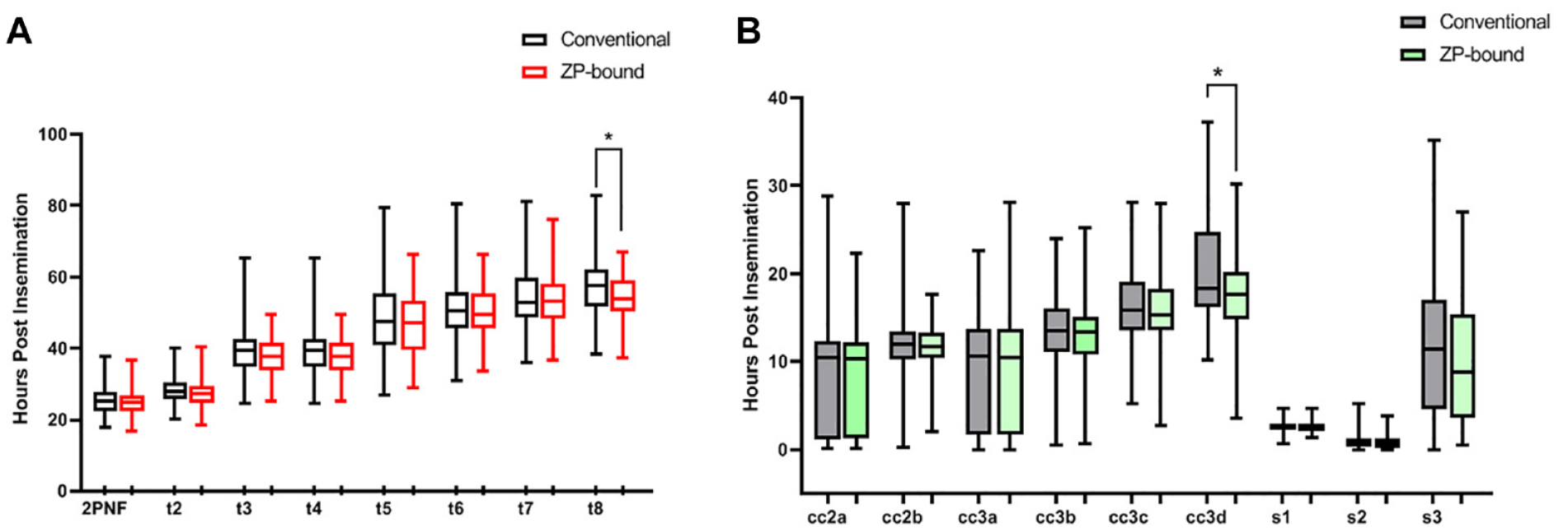
Time-Lapse Morphokinetic Parameters 2Pnf = Pronuclei Fading; t2 = First Cleavage (2-Cell Stage); t3 = Second Cleavage (3-Cell Stage); t4 = 4-Cell Stage; t5 = 5-Cell Stage; t6 = 6-Cell Stage; t8 = 8-Cell Stage. (A) Calculated Standard Intervals, Such as the Time to Complete First Synchronous Divisions s1 (t2–tPNf), Durations of the Second Cycle (cc2; t3–t2), the Time to Complete Second Synchronous Divisions (s2:t4–t3), Third Cell Cycle Duration (cc3; t8–t4) and the Time to Complete third Synchronous Divisions (s3:t8–t5) of Early Embryo Development After ICSI (B)
Embryo Quality
The rates of grade A embryos were significantly higher in ZP-bound group than control (t = 4.8, p < .05). The rate of grade C embryos was significantly lower in ZP-bound group (t = 5.6, p < .05; Table 3). In addition, according to Basile embryo classification algorithm, the rates of grade A were significantly higher in ZP-bound compared with the control (t = 3.8, p < .05). The rate of grade D embryos was significantly lower in ZP-bound compared with control group (t = 4.0, p < .05; Table 4).
Comparison Grade in ZP-Bound and Control Groups According to Hill.

Note. ZP = zona pellucid.
Comparison of the Rates of Embryo Grades in ZP-Bound and Control Groups According to Basile Classification
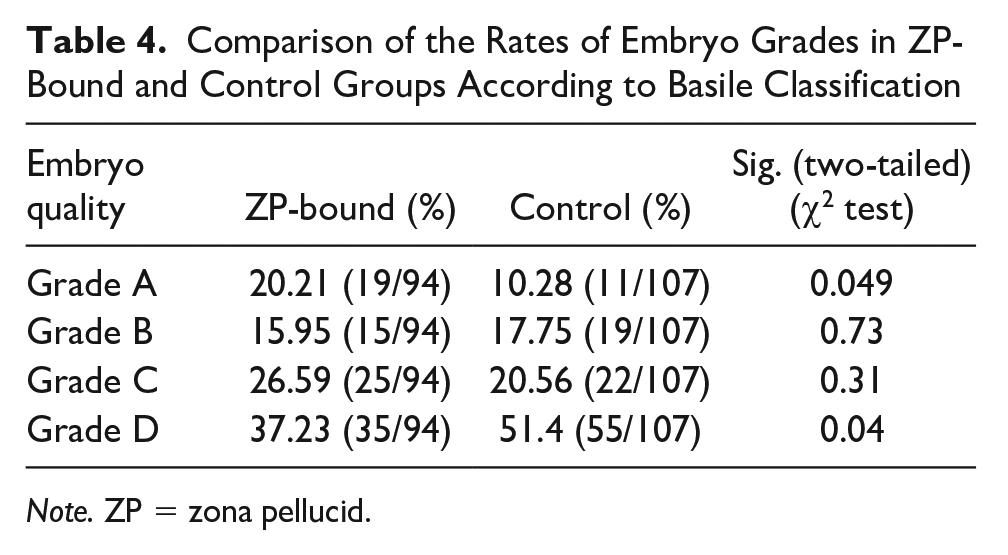
Note. ZP = zona pellucid.
As presented in Table 5, the chemical pregnancy rate in the control (4/16; 25%) was significantly lower than ZP-bound group (15/26; 57.5%) (t = 4.2, p < .05). The clinical pregnancy rate was significantly higher in ZP-bound group (14/26; 53.8%) compared with control (3/16; 18.8%) (t = 5.0, p < .01) and the implantation rate in the control group (4/32; 12.5%) was lower compared with the ZP-bound group (16/47; 34%), (t = 4.6, p < .05).
Rates of Clinical Outcomes in the Control and ZP-Bound Groups

Note. ZP = zona pellucid; hCG = human chorionic gonadotropin;ET = Embryo transfer; GS = gestational sac.
Discussion
Numerous suggestions have been proposed to improve sperm selection for ICSI, such as: surface charge of sperm (Ainsworth et al., 2005), non-apoptotic sperm (Grunewald et al., 2001), hyaluronic acid (HA)-based selection (Beck-Fruchter et al., 2016), nonadherence of sperm (Jeyendran et al., 1986), and MSOME (Bartoov et al., 2003; Teixeira et al., 2020). The ZP is the last checkpoint for natural sperm selection and ZP-binding has been used to select the suitable sperm for ICSI (Braga et al., 2009; Casciani et al., 2014; Jin et al., 2016; F. Liu et al., 2011). This method is cost-effective and additionally the sperm selection is almost identical to the sperm selection at the last checkpoint in the reproductive tract. However, there is inadequate evidence to support implementation of this method in clinical setting. Our study is the first report of different developmental dynamics and kinetics of ZP-bound sperm selection-derived embryos. The differences in embryo morphokinetics, high-quality embryos, and clinical pregnancy after ICSI were found for our randomized groups of sibling oocytes (ZP-bound sperm selection and control groups). There was no significant difference in fertilization rate between the groups. Therefore, it seems that the use of the ZP-bound sperm selection method cannot improve fertilization rate. The latest meta-analysis study is in line with our results (Izadi et al., 2021).
The use of time-lapse technology makes it possible to understand the kinetics of embryos and how they are influenced by external as well as internal factors (Bean et al., 2002). In ART, time lapse is applied most widely to grade embryos based on morphological features at different time intervals. Embryologists use this information to determine their treatment procedure to transfer or freeze the selected embryos. As a non-invasive apparatus, several embryo images can be collected in different focal planes every few minutes to assess the embryos without changing the culture medium. In addition, embryo morphological abnormalities can be analyzed, which can affect the clinical outcomes (Garcia-Belda et al., 2023). One of the internal factors that may affect the embryo development time and cleavage divisions is the sperm nucleus and/or cytoplasm (Lechniak et al., 2008; Tesarik, 2005). Given that, if the embryo development becomes too slow or too fast, it could be due to metabolic and chromosomal defects (Biezinova et al., 2006). For instance, Kakulavarapu et al. used time-lapse technology to investigate morphokinetic characteristics of embryos with cell exclusion (CE). It was reported that CE embryos showed a delay in specific time intervals including t2, t6, t7, t8. Earlier timings for t3 and t5 were observed in these embryos. Therefore, it can be concluded that this technology provides a wide range of markers to identify embryos with altered morphokinetics, such as CE embryos, which may be excluded from ET (Kakulavarapu et al., 2023). We observed non-significant differences in the tPNf, t2, t3, t4, t5, t6 and t7. The time-lapse parameters were almost the same until the 8-cell development stage between the two groups.
Although the sperm-derived genome is not completely silent between fertilization and the early stages of division (Schulz & Harrison, 2019; Tesarik, 2005), it has been identified that sperm quality plays a key role during fertilization (Hernández-Silva et al., 2022; Wakelam et al., 2007). In addition, embryonic development is influenced by sperm-derived genomic and cytoplasmic factors. The insignificance of the time-lapse parameters between the two groups in the embryo early development stages may be due to the lack of paternal genome activation before the 8-cell stage (Leng et al., 2019; Tesařék et al., 1988; Tesařík et al., 1986). The defects in the sperm nucleus are not detected before the 8-cell embryo, until the main expression of sperm-derived genes begins (Tesarik, 2005).
Other studies with different methods of sperm selection confirm our results. For example, it was identified that sperm selection by grade of sperm head vacuolization and IMSI methods had no effect on embryo outcomes until Day 3 (Hazout et al., 2006; Neyer et al., 2015). A previous study indicated no association between paternal factor infertility and embryo morphokinetics (Sacha et al., 2020). However, the relationship between sperm quality and embryo morphokinetic is not well understood (Knez et al., 2013; Neyer et al., 2015). A study, which replaced PVP with HA in the ICSI procedure, showed that embryo development was not affected up to Day 3 (Y. Liu et al., 2019). Thus, differences in the morphokinetics between the two groups may be apparent, if embryos were analyzed up to the blastocyst development (Y. Liu et al., 2016). This study identified significant differences between the two groups at time point 8, which is in line with former studies at this embryonic stage (Mangoli et al., 2020). Activation of the main expression of sperm-derived genes between the 4 and 8 cell stages (Leng et al., 2019) may be the reason for this event.
The data indicated the rates of biochemical and clinical pregnancies and implantation were significantly higher in our ZP-bound group compared with the control. In line with the results, some studies reported that significantly higher implantation rate acquired using ZP-bound sperm selection, and clinical pregnancy was insignificantly higher than the control group (Jin et al., 2016; F. Liu et al., 2011). Major expression of sperm-derived genes that occurs between the 4 and 8 cell stages (Leng et al., 2019) may play a role in the improvement of the pregnancy outcomes. Gamete fusion as a critical step in fertilization can cause the acrosome reaction and prevent the entry of enzymes into the ooplasm. This phenomenon in this sperm selection method may be a mechanism that ultimately improves the pregnancy results (Paes Almeida Ferreira de Braga et al., 2009).
A recent study reported that the use of sperm bound to isolated and immobilized ZP proteins from oocytes with a germinal vesicle (GV) can increase the pregnancy rate and reduce the risk of miscarriage (Ganeva et al., 2019). There are other studies demonstrating that the use of ZP-bound sperms resulted in high-quality embryos (Braga et al., 2009; F. Liu et al., 2011). In contrast, Casciani and colleagues reported that ZP-bound method provided equivalent outcomes than control groups with consideration of fertilization, implantation, and clinical pregnancy rates (Casciani et al., 2014).
ICSI-derived embryos in the ZP-bound group had a higher quality compared with the controls. So, the percentage of grade A embryos in the ZP-bound group was significantly higher than controls. In addition, the grade D embryos in the control were significantly higher than that of in the ZP-bound group. In line with our data, some studies reported that the percentage of high-quality embryos in the ZP-bound sperm selection method has significantly increased (Braga et al., 2009; F. Liu et al., 2011). In contrast, few studies have identified that ZP-bound sperm selection method has no effect on embryo quality (Black et al., 2010; Jin et al., 2016). These conflicting findings may be due to the differences in the inclusion criteria for selection of patients and other confounding factors, including seminal and oocyte qualities, stimulation protocols, and other factors within the IVF laboratory. The transferred embryos at our clinic were selected from one of the groups, either control or ZP-bound. The limitation of our study was related to performing ET on Day 3, such that it was not possible to evaluate the embryos at the blastocyst stage. The matching of couples between two groups was performed on the base of main restricted factors, such as similar female age and seminal profiles. In conclusion, our study showed that the selection of spermatozoa with human ZP-bound sperm method in cases with non-male factor infertility can improve the embryo quality and clinical outcomes without affecting early embryo morphokinetics in ICSI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This trial was approved by the Ethics Committee of our institute.
Participation
Informed consent forms for participation were signed by all couples.
Publication Consent
All couples signed informed consent to publish data.
