Abstract
Acmella oleracea (L.) R. K. Jansen (Asteraceae) is a plant species widely used in traditional Amazonian medicine to treat sexual dysfunction. The use of this plant has gained popularity because of its sensory properties, such as a tingling sensation. In this study on patients with premature ejaculation, we evaluated the clinical action of a nano-formulation containing an ethanolic extract of A. oleracea inflorescences. Major constituents in the extracts were identified based on gas chromatographic analysis. Participants used a spray preparation based on the A. oleracea extract for 12 weeks, during which they were instructed to apply the product 5 min prior to sexual intercourse. To assess therapeutic efficacy, participants were required to record the mean intravaginal latency time for ejaculation (IELT). During the period of spray treatment, the nano-formulation of A. oleracea increased participant IELT values (M = 293 s) compared with the baseline values (193 s). This nano-formulation reported clinical action in patients with premature ejaculation. It is accordingly considered to have potential application as a therapeutic alternative with benefits for both patients and their partners. Given the small number of participants in this study, further multicenter studies involving a larger number of participants are needed to confirm these observations.
Introduction
Premature ejaculation (PE) is a male sexual dysfunction defined by poor ejaculatory control and associated problems. It can be classified as lifelong, when ejaculation occurs within the first 2 min after the initiation of penetrative sex, or as acquired, when the ejaculatory latency is markedly reduced as a consequence of prior sexual intercourse (Shindel et al., 2022). PE is a multidimensional disorder that affects approximately 5% to 36% of men worldwide, leading to psychological distress, such as anguish, annoyance, frustration, and a lower libido (Alghobary et al., 2020; Colonnello et al., 2021; Teixeira et al., 2018).
The therapeutic approaches available to treat this condition can be pharmacological or nonpharmacological. Nonpharmacological approaches include psychological counseling and behavioral techniques (Batista et al., 2021), whereas pharmacological treatment includes topical agents and oral drugs. The main oral drugs used for PE are selective serotonin reuptake inhibitors, tramadol, Type 5 phosphodiesterase inhibitors, α-adrenergic blockers, and clomipramine (Ciocanel et al., 2019). However, the administration of oral drugs can trigger systemic adverse effects, including serotonin syndrome, and even lead to suicide in young patients. As alternatives, topical agents can be applied to reduce penile sensitivity. These include anesthetic agents and preservatives based on lidocaine and prilocaine, which act locally, with minimal systemic effects, or at most the slight possibility of allergic reactions (Batista et al., 2021; Butcher et al., 2020; Gajjala & Khalidi, 2014).
Given the potential side effects of the available pharmacological options, which can compromise treatment, there is a need for new pharmacological agents with minimal or no adverse effects. In this regard, a potential natural alternative is the plant Acmella oleracea (L.) R.K. Jansen, commonly referred to as jambu, which has long been used in traditional medicine as a potent sexual stimulant (Batista et al., 2021). With respect to the chemical composition and pharmacological properties of A. oleracea, spilanthol, the main phytochemical compound of this species (da Rocha et al., 2018; Ramsewak, 1999), has been demonstrated to be an effect alternative that can be used to enhance sexual activity. Accordingly, in this study, we evaluated the clinical efficacy of a pharmaceutical formulation containing a standardized extract of A. oleracea as an aid in the treatment of PE.
Materials and Methods
Plant Material
A spray formulation based on A. oleracea (SEEAo) was prepared from the ethanolic extract of A. oleracea inflorescences collected in the Fazendinha District (0°02’56.4”S, 51°05’51.4”W) of Macapá-Amapá, Brazil, during the period from May 2020 to September 2020. An exsiccate of the plant material has been deposited at the IAN Herbarium (Embrapa Amazônia Oriental-Belém, Pará-Brazil, under registration number 196011).
Preparation of SEEAo
The spray preparation used in this study was produced at the Pharmaceutical Research Laboratory of the Federal University of Amapá. Inflorescences of A. oleracea were cleaned by rinsing in distilled water, blotted with paper towels to eliminate excess moisture, and then placed in a circulating air oven at 40°C for 48 hr to dry and stabilize the plant material. Subsequently, the dried inflorescences were pulverized in an industrial processor, and then macerated for 24 hr and percolated at room temperature in a 96% ethanolic solution at a plant material/extractant ratio of 1:10. The percolated extract thus obtained was filtered and concentrated in a micro-processed rotary evaporator (model Q344M2; QUIMIS) at 40°C in the heating bath rotating at 100 rpm. With a borosilicate condenser for the evaporation area, with circulating water for cooling in an ultra-thermostatic bath (SolidSteel) at 4°C. The nano-formulation was prepared using a base formulation of polysorbate surfactant (5%–10%) and deionized water (90%–95%) into which the standardized ethanolic extract of A. oleracea was incorporated (5%–10% extract and 90%–95% base formulation).
Qualitative Analysis of the Hydroethanolic Extract Using Gas Chromatography–Mass Spectrometry
The major constituent of the A. oleracea extract was identified using a gas chromatograph coupled with a mass spectrometer (GC/MS QP2010, Shimadzu, Japan). The analysis conditions were as follows: injector temperature, 210°C; detector temperature, 290 °C with electron impact ionization (70 eV); carrier gas, helium; flow rate, 1 mL/min; and injection rate, 1:20. The initial temperature was set at 120°C, at which it was held for 2 min, and was subsequently increased at a rate of 6°C/min to 290°C, at which it was maintained for 8 min. The total analysis time was 35 min. The column used was DB-5 (0.25 mm × 30 m × 0.23 µm). The injected sample concentration was 1 mg/mL of dichloromethane.
Study Design
This was a prospective, nonrandomized clinical study conducted with men suffering PE who had visited the Urology and Andrology outpatient clinic with logistical support from the Clinical Pharmacology Unit of the Federal University of Amapá (UNIFAP), Macapá, Amapá, Brazil, during the period from March 2019 to March 2022.
Ethics Statement
The study was conducted in accordance with the ethical principles of the Declaration of Helsinki (WMA, 2013) and complied with the ethical considerations of the Resolution of the National Health Council (CNS 466/12). It was approved by the Committee of UNIFAP Research Ethics, under opinion n. 3,799,796, as well as by the Brazilian Registry of Clinical Trials (ReBEC), with approval ID RBR-6qbx4h6. All participants provided written informed consent and were thoroughly informed as to the nature of the study protocol and ensured freedom to withdraw from the research at any time.
Participants
The study population included men aged between 18 and 60 years, with a clinical diagnosis of PE of at least 6 weeks duration (as established by a urologist). These participants had been in stable affective relationships with a single fixed heterosexual partner for a minimum period of 6 months. Generally, the diagnostic parameters for PE have been limited to heterosexual men engaging in vaginal intercourse because nonheterosexual men with PE have longer IELT and lower distress (McNabney et al., 2022). The participants had not received relevant treatment for at least six weeks. The exclusion criteria for participation were as follows: patients with genital anatomical deformities; erectile dysfunction (ED) secondary to spinal cord injury; concomitant diagnosis of other sexual dysfunctions, such as hypoactive sexual desire; prolactin levels greater than three times the upper limit of the normal range or total testosterone levels less than 300 ng/mL; uncontrolled psychiatric disorder; decompensated diabetes mellitus; a history of stroke, myocardial infarction, or significant cardiovascular disease within the past 6 months; poorly controlled arterial hypertension; major hematological, renal, or hepatic abnormalities; and substance abuse.
Clinical Trial Procedures
All participants underwent anamnesis, clinical examination, and determination of serum hormone levels and were assessed using the Hamilton Rating Scale (HAMA), International Index of Erectile Function (IIEF-5), and International Prostatic Symptom Score (IPSS). For the diagnosis of PE, we used the Premature Ejaculation Diagnostic Tool (PEDT). As a criterion for assessing treatment efficacy, participants were requested to record their intravaginal ejaculation latency time (IELT) at baseline before and during the treatment period. The participants were assessed over a 12-week treatment period, during which they were instructed to direct three sprays of the SEEAo preparation onto the penis glans 5 min prior to sexual intercourse. During the study period, participants were instructed not to use vacuum devices, intracavernous injection, intraurethral medication, testosterone gels or injections, or any other medications or therapies for treating PE. The participants recorded all data using individual forms specifically designed for this study.
Statistical Analysis
Data were analyzed using Jamovi software (Version 2.3). Data for clinical and laboratory variables of interest are presented as descriptive statistics, specifically the distribution of relative and absolute frequencies and mean and standard deviation (SD) values, with 95% confidence intervals (CIs). Therapeutic efficacy was assessed based on mean IELT values. Prior to analysis, the data were assessed for normality of distributions using the Shapiro–Wilk test. The normally distributed data were analyzed using a paired t-test to compare means to verify statistically significant differences in the group before and after treatment, with a p value < .05 being considered indicative of a significant difference.
Ethics Statement
The study followed all the ethical principles of the Declaration of Helsinki (WMA, 2013) and met the ethical considerations of the Resolution of the National Health Council, CNS 466/12 and was approved by the Committee of UNIFAP Research Ethics, under opinion n. 3,799,796, as well as by the Brazilian Registry of Clinical Trials (ReBEC), with approval ID RBR-6qbx4h6. Through an informed consent form, volunteers were thoroughly explained. They were informed about the study model, guaranteeing their freedom to withdraw from the research at any time.
Results
SEEAo Phytochemical Parameters
The chromatographic profile of SEEAo revealed a primary molecule at a retention time of 15.25 min (area 817447; Figure 1). Comparison of the spectrum with those in the NIST Mass Spectral Library indicated that the detected compound is the alkylamide spilanthol ([2E, 6Z, 8E]-N-isobutyldeca-2,6,8-trienamide), with characteristic molecular fragmentation, resulting from the homolytic breakage of the carbon–carbon bonds at m/z = 81 and m/z = 141. The relative purity of spilanthol after peak integration was 47% (Figure 2).

GC-MS Chromatogram of the Qualitative Analysis of the SEEAo Preparation

Characterization of the Main Chemical Constituent Present in the SEEAo Preparation
Participants’ Disposition and Baseline Characteristics
Of the original 74 respondents, 18 (24.3%) had ED and 12 (16.2%) had both ED and PE and were therefore excluded from study participation. The remaining 44 (59.5%) men with PE were deemed eligible for the study. Among these, however, only seven (9.4%) completed the entire 12-week course of treatment. The flowchart in Figure 3 displays the various reasons for participant withdrawal. The mean age of the final study group of seven men who completed the treatment was 42.6 ± 7.8 years, among whom four (57.1%) had completed high school and three (42.9%) were university graduates. Of these, one (14.3%) works in the business sector, three (42.9%) are employees, and three (42.9%) are laborers (Figure 3). With regard to marital status, six are married, and the seventh is in a stable relationship. On average, these relationships were of 14.2 ± 8.381 years duration and the partners have two children. The seven participants had an average body mass index of 26.9 ± 4.056 and the following serum hormone levels (ng/dL): prolactin, 8.69 ± 0.895; thyroid-stimulating hormone, 1.68 ± 1.213; triiodothyronine (T3), 2.51 ± 0.708; total tetraiodothyronine (T4), 4.64 ± 3.546; and total testosterone, 413.65 ± 105.365. The scores for the applied assessment indices PEDT, HAMA, IIEF-5, and IPSS were 15.9 ± 1.57, 18.4 ± 3.82, 23.7 ± 6.26, and 10.6 ± 8.46, respectively. In terms of pathological anxiety, three of the participants were classified as moderate, three as mild, and one as severe, and with regard to IIEF-5, three were normal, three were mild, and one was moderate. According to the recorded baseline characteristics, five of the participants were found to have secondary PE and two primary PE (Table 1).

Flowchart Illustrating the Different Steps of Participant Exclusion
Demographical and Clinical Data of the Studied Population
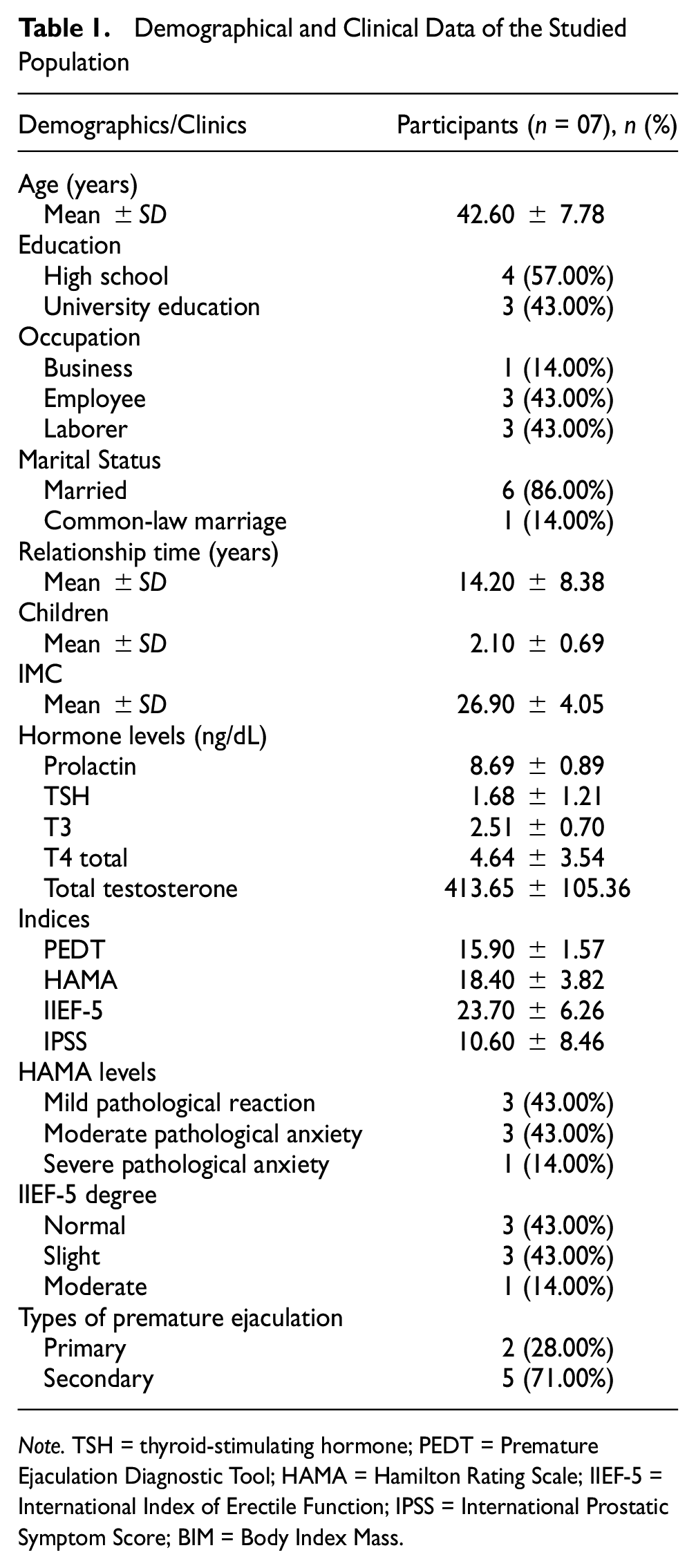
Note. TSH = thyroid-stimulating hormone; PEDT = Premature Ejaculation Diagnostic Tool; HAMA = Hamilton Rating Scale; IIEF-5 = International Index of Erectile Function; IPSS = International Prostatic Symptom Score; BIM = Body Index Mass.
Clinical Action
The action of the SEEAo preparation was assessed based on IELT values. Initially, the participants had an average time of 193 s (SE = 71.9) which increased to 293 s (SE = 76.8) after using the SEEAo spray; however, the difference was statistically non-significant, t(6) = −2.3, p = .061. Nevertheless, during the course of the 12-week treatment, none of the participants reported the occurrence of any side effects, such as allergic response or loss of erection, when using SEEAo (Figure 4).

Mode of Action of the SEEAo Preparation in the Treatment of Premature Ejaculation
Discussion
The results obtained in this study following treatment with SEEAo suggest a clinical action of this preparation as a therapeutic resource for the treatment of PE. Compared with baseline values, participants recorded an increase in IELT when using this spray, although these time increases were proved to be statistically nonsignificant, which may be attributable to the small number of participants included in the study. The commercially available pharmacological options for treating PE can trigger relevant side effects, which compromise the treatment. Therefore, there is a need for new pharmacological agents using natural extracts with better efficacy in reducing penile sensitivity to improve sexual satisfaction. Acmella oleracea has been widely reported to have anesthetic, anti-inflammatory, analgesic, anti-wrinkle, and vasorelaxant activities as well as weight loss, gastroprotective, antipyretic, and diuretic effects (Andrade et al., 2013; Bakondi et al., 2019; Chakraborty et al., 2010; Ekanem et al., 2007; Ferreira et al., 2004; Ratnasooriya et al., 2004; Wongsawatkul et al., 2008). The identification of spilanthol as the main constituent in the standardized extract of A. oleracea is consistent with the findings of previous studies, in which significant concentrations of this compound have been detected in extracts obtained from the inflorescences of this plant (da Rocha et al., 2018; Ramsewak, 1999). In addition to being a sexual stimulant, spilanthol has been demonstrated to penetrate the skin, increase the permeability of the dermis, raise serum testosterone levels, and enhance the release of nitric oxide (Boonen, Baert, Roche, et al., 2010; da Rocha et al., 2018; Sharma et al., 2011; Spiegeleer et al., 2013).
Alkylamides are a group of compounds formed by the union of a fatty acid with a chain of 8 to 18 carbons and an amine group (Boonen, Baert, Burvenich, et al., 2010; Nakatani & Nagashima, 1992). Alkylamides are primarily present in plants belonging to Asteraceae, Solanaceae, and Piperaceae families (Torres & Chavez, 2001). Spilanthol ((2E,6Z,8E)-N-isobutyldeca-2,6,8-trienamide) was identified as the major substance in the hydroethanolic extract of jambu inflorescences with an abundance of 80% in relation to the other compounds in the sample. The identification of spilanthol exhibited two characteristic fragmentation signals, resulting from the cleavage of the C-C bond, at 81 m/z and 141. Despite the variability of spilanthol quantification in the parts of A. oleracea, it is described in the literature that the highest concentration is found in the inflorescences, followed by the leaves (Ramsewak, 1999).
A. oleracea has several pharmacological activities, including sexual stimulant activity (Rocha et al. 2018). In a previous clinical trial, topical application of an A. oleracea-based product to the male genitalia was demonstrated to result in an improvement in sexual response, with the authors’ speculating that the observed increases in patients’ sexual desire and satisfaction were probably associated with the tingling sensation experienced following application of the cream to the glans (Regadas, 2008). Sharma et al. (2011) have described the aphrodisiac potential of an ethanolic extract of Spilanthes acmella (the basonym of A. oleracea), demonstrating that both in vitro and in vivo, the enhanced sexual potential was probably attributable to the presence of N-alkylamides in this species, among which, spilanthol was presumed to be the primary constituent contributing to the observed effects.
The baseline characteristics of the participants assessed in the present study proved to be consistent with the inclusion profile for the study. Regarding the diagnostic indices applied, participants were identified as having PE when answering the PEDT questionnaire, which rates respondents according to control, frequency, minimal sexual stimulation, anguish, and interpersonal difficulties. This self-reported measure is widely used in assessments of male sexual health, as it facilitates the systematic evaluation of patients throughout the course of treatments (McMahon et al., 2008; Symonds et al., 2007). We also assessed participants using the HAMA scale that scores respondents according to their degree of anxiety, which in the context of the present study is assumed to reflect the psychogenic component of the participants’ PE. Several factors can adversely influence the mechanisms underlying penile erection, among which are psychological anxiety disorders, which can significantly impair human health, with repercussions in practically all areas of interpersonal relationships (Codevilla et al., 2013; Sarris et al., 2016). The effects of emotional and relational problems on sexuality highlight to the need address individuals’ emotional behavior, to promote resilience, and to reduce the detrimental influence of stress on their relationships (Eleuteri et al., 2022).
The threshold considered mild based on scores obtained using the IIEF-5 scale indicate the need for early commencement of treatment, thereby minimizing the aggravation of problems faced by those suffering from PE, as well as the need to adopt a multidisciplinary approach to treatment, including the support of psychological therapies (Carteiro & Marques, 2010). According to the scores obtained for the IPSS, participants have moderate symptoms in the lower urinary tract, characteristic of benign prostatic hyperplasia. This questionnaire enables the tracking of symptoms, facilitates rapid diagnoses, and indicates treatments for potential health problems. Its application can serve as a valuable starting point for a more accurate investigation of a patient’s prostate function (Cam et al., 2003).
The use of a spray formulation in the present study is similar to the approaches adopted in previous studies, which have also opted to assess the application of topical treatments (Boonen, Baert, Roche et al., 2010; Freitas-Blanco et al., 2016; Regadas, 2008). Topical agents are simple therapeutic options based on local application (Gajjala & Khalidi, 2014). However, some reports have described certain disadvantages associated with the use of topical creams derived from natural products, including unpleasant colors and odors when applied to the penis, as well as undesirable effects, including local irritation, a burning sensation, delayed ejaculation, and dysuria (Choi et al., 2000; Shi et al., 2014). In contrast, when administered topically in mice, spilanthol has been demonstrated to inhibit mitogen-activated protein kinase signaling and improves allergic inflammation in 2,4-dinitrochlorobenzene-induced atopic dermatitis (Huang et al., 2019) and consequently does not elicit these localized adverse effects.
Using porcine buccal mucosa in an in vitro Franz diffusion cell configuration, Boonen, Baert, Burvenich et al. (2010) evaluated the transmucosal behavior of spilanthol administered in a gel formulation of S. acmella ethanolic extract. Accordingly, they demonstrated the promising local and systemic functionality of spilanthol in permeating the mucosa in a formulation-dependent manner, increasing penetration with both local and systemic effects on the mucosa after topical administration.
With respect to the pungency, paresthesia, and analgesia associated with the use of spilanthol, Freitas-Blanco et al. (2016) investigated the efficacy of applying a mucoadhesive film containing A. oleracea extract for topical anesthesia of the oral mucosa. This formulation was reported to show a high degree of permeation in vitro and an in vivo anesthetic effect similar to that obtained using a control comprising lidocaine (2.5%) and prilocaine (2.5%), which is used as a gold standard topical anesthetic. Thus, mucoadhesive films based on A. oleracea could serve as effective alternatives to the currently available topical anesthetics.
When evaluating the local antinociceptive and pronociceptive effects of natural alkylamides derived from A. oleracea, Dallazen et al. (2018) reported that compared with synthetic isobutylalkylamide, the intraplantar injection of Swiss mice with a hexane fraction rich in spilanthol derived from A. oleracea flowers promoted analgesia in the neurogenic and inflammatory phases based on a formalin test. This activity has been attributed to the modulation or blocking of V1 and A1 transient receptor channel subfamilies and an increase in the release of gamma-aminobutyric acid.
With respect to the application of SEEAo on the glans of participants in the present study, we speculate that the vasoactive and local anesthetic activities of a spilanthol formulation in inducing penile desensitization and increasing IELT are attributable to the unique property of alkyl amides in blocking the pores of KCNK potassium channels (KCNK3, KCNK9, and KCNK18), thereby activating mechanosensitive neurons and hence inducing a tingling sensation at the site of application (Albin & Simons, 2010).
KCNKs are channels that mediate the passage of K+ ions from cells and are open at resting potential (Patel & Lazdunski, 2004). Among these channels, KCNK3 encodes external rectifier channel protein that is sensitive to changes in extracellular pH and is inhibited by extracellular acidification (Dookeran & Paul, 2017). It is found in the brain and cerebellum, wherein it can be activated by anesthetics (Bittner et al., 2010). KCNK9 also generates external rectifier currents, modulated by diverse chemical and physical stimuli, including acidification and hypoxia. Similar to KCNK3, this protein occurs within the brain, particularly in the cerebellum, and responds to anesthetic agents (Luedi et al., 2007). In contrast, KCNK18 is related to an acid-sensitive subfamily of K+ channels that are expressed in the spinal cord. It is regulated by the calcium/calmodulin-dependent protein calcineurin phosphatase (Czirják & Enyedi, 2006).
It has been established that the penis glans contains dense clusters of free nerve endings that are innervated by A-fibers (Georgiadis & Holstege, 2005). Consequently, to corroborate the results obtained in the present study, it would be desirable in future studies to elucidate the neurophysiological mechanisms that underlie the ejaculatory behavior elicited in response to penile sensory stimulation with spilanthol-based topical products.
In their study designed to identify regions of the human brain that are activated during ejaculation, Holstege et al. (2003) monitored cerebral activity in heterosexual men based on positron emission tomography scans performed to measure increases in regional cerebral blood flow (rCBF) during ejaculation, which were compared with the effects of sexual stimulation performed manually by the volunteer’s partner. They accordingly detected substantial increases in rCBF, primarily in the cerebellum, thereby providing evidence in support of the supposition that neurophysiological mechanisms in the cerebellum play essential roles in emotional processing.
Ejaculation consists of several peripheral events that are controlled by autonomic (sympathetic and parasympathetic) and somatic divisions of the nervous system. Efferent and somatic autonomic signals originate in spinal nuclei within the thoracolumbar and lumbosacral segments. Coordinated activation of the autonomic and somatic spinal nuclei is modulated by a group of lumbar spinal interneurons defined as the spinal generator of ejaculation, which constitute a spinal network prominently influenced by sensory and supraspinal genital stimuli or inhibitors (Clement & Giuliano, 2016). This genital sensory stimulation or inhibition is closely associated with the high density of nerve receptors present in the penis glans, which, compared with other regions of the body, has a lower tactile threshold (Astbury-Ward, 2002; Eardley & Sethia, 2003). In this context, electrophysiological studies have identified that men with PE exhibit a heightened sensory response after penile stimulation. Thus, topical treatments with a local anesthetic reduce glans sensitivity, thereby delaying ejaculation (Wyllie & Hellstrom, 2011). We accordingly speculate that the use of SEEAo by participants in the present study prolonged sensory conduction, thereby reducing the hyperexcitability of the penis glans and thus inducing a certain degree of penile desensitization. These responses are assumed to be mediated via an afferent–efferent reflex mechanism, which contributes to prolonging the IELT and is associated with a potential improvement in ejaculatory control and the sexual satisfaction of participants. This is consistent with the findings of previous studies that have demonstrated the efficacy and safety of using local anesthetics to treat PE (Xia et al., 2013).
This study does, however, have certain limitations. Notably, a larger number of participants would be necessary to obtain more meaningful IELT results. In this regard, we experienced difficulty in recruiting a sufficient number of participants for the present study, which can probably be ascribed to our relatively stringent inclusion/exclusion criteria adopted, the reluctance of men to seek assistance or medical support related to sexual dysfunction (Britto & Benetti, 2010). The trial period of this study coincided with the COVID-19 pandemic, when the centers that normally conduct such clinical trials prioritized assistance for patients with COVID-19, owing to the urgent nature of the pandemic. Accordingly, given that clinical trials often necessitate a considerable number of participants to attain an adequate sampling power, further multi-center studies are warranted, which would thereby enable an assessment of the efficacy of the SEEAo to spray among a larger and more diverse participant population. Nevertheless, despite these limitations, the increases in IELT scores reported in this study provide evidence to indicate the beneficial clinical effects of a nano-formulation based on a standardized extract of A. oleracea in the treatment of PE.
Conclusion
In this study, we demonstrated that the application of the SEEAo spray in patients with premature ejaculation potentially increased the intravaginal latency times for ejaculation. The participants showed good tolerance to the spray and experienced no systemic side effects or incidence of local effects. This formulation is presumed to promote penile desensitization, which is attributed to the presence of spilanthol in an extract derived from the inflorescences of A. oleracea. Consequently, the SEEAo preparation could serve as an alternative therapy for the treatment of premature ejaculation. Further studies based on different treatment lengths and a larger more diverse range of participants are warranted to confirm these observations.
Footnotes
Acknowledgements
The authors would like to express their gratitude to the Universidade Federal do Amapá and to members of the Laboratório de Pesquisa em Fármacos.
Author Contributions
Conceptualization: L.L.B. and J.C.T.C.; writing-review and editing: L.L.B., R.C.R.K., and T.A.T.; methodology: L.L.B. and A.V.T.L.; data curation: L.L.B. and E.L.M.; investigation: L.L.B. and E.L.M. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
