Abstract
Acute interstitial nephritis (AIN) is a relevant cause of acute renal failure. Drugs are the predominant cause, followed by infections and idiopathic lesions. AIN, as a form of hypersensitivity reaction, is an uncommon manifestation in the setting of human parasitic infections. We report a case of a polyparasitic infection (Giardia lamblia, Entamoeba coli, and Endolimax nana) resulting in a severe biopsy-proven AIN in a 61-year-old male patient. Despite the antiparasitic treatment followed by corticosteroid therapy, and during the 6-month follow-up period, the patient remained dialysis-dependent, and he developed autoimmune hemolytic anemia. Extensive search for another infection or neoplasia was negative. Immunological tests were also negative. The resulting hypersensitivity reaction to the triple parasite infection would have led to fatal evolution for the kidneys affected by this unusual type of AIN.
Introduction
Acute interstitial nephritis (AIN) accounts for 15% to 27% of renal biopsies performed because of acute kidney injury (Praga & González, 2010). It was first described by Biermer in 1860, and it was defined as a clinical entity by Councilman in 1898 (Councilman, 1898).
Many factors lead to AIN and diagnosis may be challenging due to the overlap of their clinical presentation. However, failure to distinguish and promptly treat AIN can lead to fibrosis and progression to chronic kidney disease (Raghavan & Eknoyan, 2014).
Considerable evidence implicates antigen-initiated cell-mediated injury in the pathogenesis of AIN. Drugs account for 70% of all cases, with more than 150 different incriminated agents. The remaining cases are due to infections and autoimmune diseases, and they are rarely idiopathic (Raghavan & Eknoyan, 2014).
In an infectious context, AIN, as a form of hypersensitivity reaction, is an exceptional manifestation during human parasitic infections (De Pascalis & Buongiorno, 2012).
Herein, we report a case of polyparasitic infection resulting in a particular AIN leading to terminal chronic renal failure. The diagnosis was challenging as the clinical biological picture was confusing with a DRESS. To the best of the authors’ knowledge, such a condition has never been previously reported.
Case Report
A 61-year-old male patient was admitted to the Nephrology Department in September 2021 for severe acute renal failure. He was a smoker with no medical history.
Three months earlier, he had been diagnosed with orchitis and was treated with Fluoroquinolone. Serum creatinine was at 64 µmol/l. Ten days before his hospitalization, he presented a urinary infection that was treated with Cephalosporin. On the fifth day of treatment, he developed a fever with a diffuse pruritic skin rash, and 3 days later, he developed watery diarrhea with oliguria and finally anuria.
Physical examination revealed a body temperature of 38.4°C, blood pressure of 120/60 mmHg, heart rate of 95 beats/min, respiratory rate of 19 breaths/min, oxygen saturation of 98% in room air, diffuse maculopapular lesions on the entire body except for the face, purpura in the lower limbs (Figure 1), and persistent anuria. No peripheral lymphadenopathy or uveitis was noted.

Massive Maculopapular Lesions Covering the Skin of the Entire Body, Except the Face
Laboratory tests showed serum creatinine of 1,700 µmol/l with metabolic acidosis requiring hemodialysis. Leukocytes were at 21,000/mm3 (normal range: 4,000–10,000) with hypereosinophilia of 1,300/mm3 (normal range < 500/mm3) and elevated count of Ig E at 4,048 UI/ml (normal range: <120 UI/ml). Hemoglobin was at 11 g/dl (normal range: >13 g/dl) and platelets at 210,000/mm3. Liver function tests were normal. There was an elevated rate of C-reactive protein at 240 mg/l (normal range: <8 mg/l) and procalcitonin at 8 ng/ml (normal range: <0.046 ng/ml), and serum albumin was low at 18 g/l (normal range: 36–48 g/l). Urinalysis revealed a hematuria (30 RBC/HPF), a leukocyturia (60 cells/HPF), a high rate of N-Acetyl-beta-D-glucosaminidase at 74 (normal range: 0–10 U/L), a high range of beta2-microglobulin at 0.38 (normal range: 0–0.25 mg/L) without proteinuria or eosinophils. Exploration of hemostasis revealed a low rate of prothrombin at 40% with a normal rate of factor V and a deficit of Vitamin K-dependent coagulation factors (II, VII, IX, X).
Immunological tests revealed negative antinuclear antibodies, antineutrophil cytoplasmic antibodies, and anti-glomerular basement membrane antibodies with a negative celiac serology. The level of C3 and C4 complement fractions was normal.
Cutaneous biopsy revealed an inflammatory infiltrate composed of lymphocytes and eosinophils around the vessels, destroying them in the dermis. Direct immunofluorescence revealed C3 and negative immunoglobulin A, G, or M.
The initial diagnosis was DRESS syndrome. No infection with viruses was confirmed. Indeed the real-time polymerase chain reaction (PCR) did not detect the viral DNA of CMV or human-herpes virus 6, and the serology of the hepatitis B virus (HBsAg, anti-HBs, anti-HBc Ig M, and IgG) was negative as well than hepatitis C, hepatitis A, and HIV (IgM and IgG antibodies negative), and the Epstein–Barr virus serology indicated a past infection (EBNA IgG +, VCA IgG+ and VCA IgM–).
The copro-parasitological examination of the stools, requested on the day of admission in the face of diarrhea, revealed Giardia lamblia, Entamoeba coli, and Endolimax nana.
Based on the overall clinical picture, and the current criteria of DRESS, this diagnosis was ruled out and digestive parasitosis was held responsible for the clinical and biological abnormalities.
Therapy with intra-venous metronidazole (1 g/day) was started, and it lasted 10 days. Apyrexia and regression of the inflammatory biologic syndrome (C-reactive protein and procalcitonin) were obtained within 2 days. Methylprednisolone 900 mg/day was therefore prescribed for 3 days, followed by prednisone per os (0.5 mg/kg/day). The parasites were not detected in stool during steroid therapy.
Within 3 days, diuresis was regained with 4 liters and the cutaneous rash changed to exfoliative dermatitis. An improvement in the patient’s diarrhea was also noted. Despite the resumption of diuresis, renal function did not improve, and creatinine remained around 1,800 µmol/l. We then decided to perform a kidney biopsy for essentially prognostic purposes.
After the administration of plasma derivatives containing the vitamin K–dependent clotting factors (PPSB), an improvement in the rate of prothrombin (at 80%) was obtained. A kidney biopsy was performed 3 days after beginning the treatment. Microscopic examination of the renal biopsy (Figures 2–4) did not show glomerular lesions. Particularly, the 12 glomeruli were preserved without features of expansion of the glomerular lobules, mesangial hypercellularity, or thickening of the glomerular basement membranes. Tubular changes were more prominent than glomerular alterations. In the lumen of the tubules, protein cylinders, erythrocytes, and sometimes granulocytes were present. Tubular atrophy was seldom found. Interstitial damage was severe, and extensive interstitial edema was present with an abundant mixed inflammatory infiltrate. The inflammatory infiltrate was rich in lymphocytes and granulocytes, without evidence of eosinophilic cells or granulomas. Arteries and arterioles were unaffected. The direct immunofluorescence study was negative.
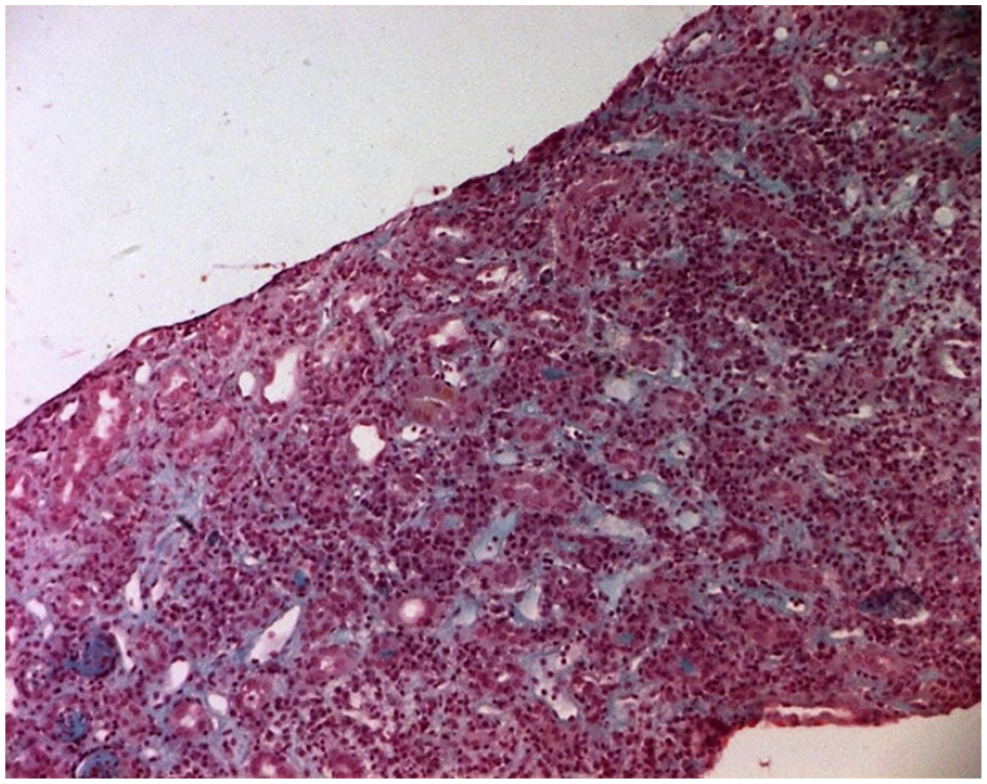
Microscopic Examination of Renal Biopsy Showing Prominent Interstitial Edema With a Severe Inflammatory Infiltrate (TM×100)

Tubulitis (Arrow) and Interstitial Damage in Contrast to Glomeruli With Normal Mesangial Cellularity (TM×200)

High Magnification of the Interstitial Infiltrate, Rich in Granulocytes and Lymphocytes (He×400)
Nine days after the patient’s presentation, a progressive deglobulization of 11 to 8 g/dl was noted without externalized bleeding. Fibroscopy showed erythematous and nodular antrofundic gastropathy. The anatomy pathology examination revealed chronic gastritis with Helicobacter Pylori +, without other abnormalities. Colonoscopy was normal. A positive Coombs test IgG ++ was detected, so we retained hemolytic anemia.
Despite corticosteroid therapy, renal function and hemolytic anemia did not improve. The patient has been on chronic hemodialysis for 9 months. The exhaustive search for underlying neoplasia came back negative.
Discussion
Diagnosis of AIN relies on performing a renal biopsy, and the typical findings are predominant in the tubulointerstitium. AIN is characterized by an infiltration of lymphocytes, macrophages, and eosinophils in the renal interstitium. Inflammatory cells in the renal tubules (“tubulitis”) are also present. Tubular injury and interstitial fibrosis often accompany this inflammatory infiltrate (Liapis et al., 2017).
Drug-induced AIN is the most common etiology of AIN, but AIN can also have infectious, autoimmune, or idiopathic causes. β-Lactam antibacterials, nonsteroidal anti-inflammatory drugs, and proton pump inhibitors are recognized as leading causes of AIN (Muriithi et al., 2015).
Drug-induced AIN is a delayed T-cell-mediated hypersensitivity reaction (Raghavan & Shawar, 2017). This AIN is usually mild, and it is cured without sequelae once treated on time. However, severe interstitial nephritis, acute tubular necrosis, or vasculitis have been reported, and they may lead to renal failure requiring hemodialysis (Cho et al., 2017).
DRESS is a potentially life-threatening drug-induced reaction (Cacoub et al., 2011). Clinically, it is classically characterized by a cutaneous eruption, fever, lymphadenopathy, eosinophilia, and visceral involvement (Schunkert & Divito, 2021) associated with a latency of 11 to 35 days post-stimulus (Wolfson et al., 2019).
Some of these manifestations were present in our patient, which led us initially to suspect an AIN in the context of a DRESS but the sufficient criteria to retain the DRESS were not met. Moreover, the patient presented other abnormalities, such as low prothrombin explained by a deficit of vitamin K–dependent coagulation factors without cholestasis or cytolysis, and particularly the absence of eosinophils in the kidney biopsy. These features supported the hypothesis of parasitic infection, resulting in malabsorption and AIN as a form of hypersensitivity reaction which is an uncommon and exceptional complication of parasitic infections (Cho et al., 2017).
Only a few cases of AIN associated with parasitic infection have been reported in the literature (Duvic et al., 1999). It has been noted previously as a consequence of Giardiasis, as observed in our case (De Pascalis & Buongiorno, 2012). However, our patient presented two other parasites, Entamoeba coli, and Endolimax nana, which have never been described to be associated with AIN.
In the reported cases, AIN represented an unusual manifestation of parasitic infection. Alongside the AIN, our patient presented another exceptional manifestation of parasitosis; these were spectacular skin lesions that constituted the main element initially suggesting a DRESS.
Indeed, intestinal parasites in particular Giardiasis, alone or in combination with other agents, may be the etiology of various skin diseases through inflammatory and allergic mechanisms or intestinal hyperpermeability. Mucocutaneous lesions are diverse: urticaria, angioedema, atopic dermatitis, erythema nodosum, Wells syndrome, and so on (Humbert et al., 2017).
The hemolytic anemia noted in our patient would also be secondary to parasitic infections. Indeed, some parasitosis have been identified in the literature associated with hemolytic anemia such as babesiosis (Santos et al., 2020) and malaria (Ghosh et al., 2017).
Polyparasitism increases morbidity and susceptibility to other infections (Wesołowska et al., 2018). It is common as reported in both theoretical and experimental studies (Alizon et al., 2013). The concomitant presence of these parasites in previously reported immunocompetent patients supported the performed neoplastic investigation that was negative in our patient.
Limitations: The exceptional association of pathologies reported in this article was about a single case. The causal relationship between it and polyparasitic infection would be better established if other cases are identified.
Conclusion
We, herein, reported an unusual case of a polyparasitic infection leading to a particular AIN in an immunocompetent male patient. The diagnosis was difficult in the presence of many elements suggesting DRESS in a patient who had received antibiotic therapy. Despite the antiparasitic treatment, and corticosteroid therapy, renal failure became chronic.
The resulting hypersensitivity reaction to the triple parasite infection would have led to fatal evolution for the kidneys affected by this unusual type of AIN in addition to spectacular skin lesions and persistent hemolytic anemia.
Footnotes
Author Contributions
All authors have contributed significantly to the manuscript, reviewed it, and agreed upon the presentation. S.M., W.R., and A.F. defined the research theme. S.M., R.B., A.Z., Y.G., and D.Z. designed the methods and experiments, carried out laboratory experiments, analyzed data, and interpreted the results. W.R., W.S., N.BA., N.A., M.M., and A.A. discussed the interpretation and presentation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Given the nature of the article, a case report, no ethical approval was required.
Informed Consent
Written informed consent was obtained from the patient for publication of this case and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
