Abstract
The tRNA-derived fragments (tRFs) are a new class of regulatory noncoding RNAs and have different biological functions in cancer. This article investigated the expression and clinicopathological significance of tRF-Glu-TTC-2 in prostate carcinoma (PCa), and its effect on tumor growth. Expression profiles of tRFs and tiRNAs were analyzed by tRF and tiRNAs microarray in PCa samples, and then the expression was confirmed by qRT-PCR; RNA in situ hybridization was used to detect the positive expression of tRF-Glu-TTC-2 and to analyze the correlation between the expression level of tRF-Glu-TTC-2 and clinicopathological parameters. CCK-8 experiment was used to detect the effect of tRF-Glu-TTC-2 on the proliferation of PCa cells, and nude mice subcutaneous tumor model was used to detect the effect of tRF-Glu-TTC-2 on the growth of PCa cells. The results showed that tRF-Glu-TTC-2 was mainly positive and its expression level increased in PCa. The high expression was closely related to the tumor size (p < .05). Overexpression of tRF-Glu-TTC-2 promoted the proliferation of PCa cells, and decreased expression of tRF-Glu-TTC-2 inhibited the proliferation of PCa cells (p < .05). The results of subcutaneous tumor transplantation in nude mice showed that the tumor volume and weight of the knockdown group were smaller than those of the control group(all ps < .05). Ki-67 staining showed that the proportion of Ki-67-positive cells in the reduced tRF-Glu-TTC-2 group was lower than that in the control group (p < .05). The tRF-Glu-TTC-2 may be a new oncogene that can promote growth and proliferation of PCa. It provides a new idea for the treatment of PCa.
Worldwide, prostate cancer (PCa) is the second most common cancer after lung cancer (Bray et al., 2018). PCa is one of the most vulnerable cancers in men (Daniyal et al., 2014), and its incidence rate accounts for 13.5% of the new male cancer cases in the world, while its mortality accounts for 6.7% of the total male cancer deaths (Bray et al., 2018). Although the incidence rate of PCa in China is lower than that in Western countries, it has increased year by year in recent years; it has become the most common malignant tumor of the urinary system (Dy et al., 2017). In eukaryotes, the ratio of protein-coding gene sequences is less than 2%, and the rest are non-coding RNA (ncRNA) gene sequences. ncRNA includes transport RNA (tRNA), ribosomal RNA (rRNA), small nuclear RNA (snRNA), microRNA (miRNA), and other RNAs (Fujimoto et al., 2016). In 2021, studies have reported that tRNA-derived fragments (tRFs) are a new class of regulatory ncRNA, which have different biological functions in cancer and stress diseases. Meanwhile, the high abundance of tRNAs and tRFs in patients’ body fluids makes it a new biomarker for clinical application (Yu et al., 2021). At present, some studies claim that tRFs are involved in the proliferation and metastasis of tumor cells in colorectal cancer, cervical cancer, lung cancer, breast cancer, and liver cancer (Goodarzi et al., 2015; Huang et al., 2017; Keam et al., 2017; Kim et al., 2017; Pekarsky et al., 2016), but the role of tRFs in PCa has not been reported in the literature. The expression level of tRF-1:31-Glu-TTC-2 (tRF-Glu-TTC-2) in PCa was increased and verified by tRFs and tiRNAs expression profile. This study will explore the clinical significance of tRF-Glu-TTC-2 in PCa and its effect on growth and proliferation.
Materials and Method
Specimens and Data Collection
Seventy-seven pairs of PCa tissues and adjacent normal prostate tissues were obtained from urological surgery at Yangzhou University Affiliated Hospital from January 2018 to December 2021. The diagnosis of PCa was confirmed by the pathologist. The use of specimens and the collection of clinical data were approved by the ethics committee of the Affiliated Hospital of Yangzhou University (2022-YKL2-21-003), and all participants provided written informed consent to participate in the research study. The clinicopathological parameters of all patients were collected, including age, sex, smoking, tumor size, grade, lymph node metastasis, and tumor node metastasis (TNM) stage. After the collection of fresh samples, except for a small part of total RNA, the rest of the tissues were put into the liquid nitrogen tank and stored in the refrigerator at −80 for a long time for later experiments.
Cells and Plasmids
PCa PC-3 and DU145 cell lines were purchased from Shanghai Cell Biobank of Chinese Academy of Sciences (Shanghai, China). The cell lines were cultured in RPMI-1640 medium (Hyclone company) containing 10% fetal bovine serum and 1% penicillin / streptomycin (Invitrogen company) in a humid incubator at 37℃ and 5% CO2. Normal human prostate epithelial cells RWPE-1 were donated by X.Y. Dong (University of Yang Zhou, China) and cultured in defined keratinocyte series-free medium with 5 ng / mL epithelial growth factor.
tRF-Glu-TTC-2 mimic / inhibitor plasmids and control groups were purchased from Shanghai Sangong Biotechnology Co., Ltd. Lipofectamine2000 reagent (Invitrogen) was used to transfect the related plasmids into PCa cells. The test groups include the overexpression tRF-Glu-TTC-2 group and scramble group, knockdown tRF-Glu-TTC-2 group, and vector group.
Quantitative Reverse Transcription Polymerase Chain Reaction
Total RNA was extracted using Trizol reagent (Invitrogen). The optical density (OD) 260/230 ratio of RNA was >1.8 that was obtained by ultraviolet/visible spectrophotometer (Yiipu Instruments, China). Primescript first-strand complementary DNA (cDNA) synthesis Kit (Takara) was used to reverse transcribe RNA into cDNA according to the Kit Proposal. Quantitative reverse transcription polymerase chain reaction (qRT-PCR) experiments were performed on the Applied Biosystems 7500 PCR instrument according to the steps of the real-time fluorescence quantitative Kit (Takara). The upstream primer of tRF-Glu-TTC-2 was acatggttagcggttagga, and the downstream primer was ttcccacaccgggagtcgaa. The internal reference of tRF is U6. The upstream primer of U6 was tgcgggtgctcgcttcggcagc, and the downstream primer was attaaccaggtgcagggtcgaggt.
TRF and tiRNA Microarray Detection of tRF and tiRNA Expression Profile in PCa
Three pairs of total RNA from PCa tissues and adjacent normal tissues were extracted, respectively, and sent to Shanghai Kangcheng biotechnology company (aksomics) to study the expression profile of Arraystar nrStar tRNA PCR Arrays (H/M) in human PCa. TRF and tiRNA microarray can detect the tRNA of 185 individuals, covering all the nuclear anticodon isoacceptors in the GtRNAdb database, and providing two levels of expression detection: isoacceptor and isodecoder. Aksomics provides data analysis for expression profiling and image acquisition.
Detection of tRF-Glu-TTC-2 Expression by RNA In Situ Hybridization
Seventy-seven fresh specimens were made into PCa paraffin specimens, which were sectioned and dewaxed to water. At room temperature, hydrogen peroxide treatment can eliminate endogenous peroxidase. Tissue sections were digested with pepsin. The tissue sections were incubated with prehybridizing solution in the incubator and then incubated with hybridizing solution for 12 hr. Sealing fluid was added after cleaning and then biotinylated digoxin was added. SABC joined the organization. Biotinylated peroxidase was added. The tissues were stained with diaminobenzidine (DAB), counterstained with hematoxylin, washed, dehydrated, transparent, and sealed. The prehybridized solution was used to replace the hybridized solution containing the probe as the blank control. The tRF-Glu-TTC-2 probe sequences were tcccacatggtctagcggttaggatcctg and ggttcgactcccggtgtggga. Five high-power visual fields were taken from each sample to calculate the proportion of positive cells: number of positive cells / total number of cells.
CCK-8 Experiment to Detect Cell Proliferation Activity
The PCa cells (about 4-5 × 103 cells) were suspended in each hole of the 96-well plates, and there are three multiple wells. The 96-well plates were put into a humid incubator at 37℃ and 5% CO2 until the whole hole bottom is covered. Ten microliter CCK-8 solution was added to each hole, and the culture plate was gently hit to mix the cells and the solution and incubated in the incubator for 4 hr. The absorbance value was calculated on 450 nm visible light using a microplate reader (the absorbance value of pores with medium and CCK8 solution but without cells should be subtracted from the results), and the proliferation activity of knockdown/overexpression tRF-Glu-TTC-2 on cells was calculated. The CCK-8 experiment requires a total of 9 duplicate holes (numbered 1–9), of which holes numbered 1–3 are used for 24-hr detection, holes numbered 4–6 are used for 48-h detection, and holes numbered 6–9 are used for 72-hr detection. The relevant absorbance value data are obtained through the microplate reader.
Subcutaneous Transplanted Tumor Model in Nude Mice
BALB/c nude mice were from the animal experiment center of Yangzhou University. The nude mice weighed 23 to 27g and were 5 to 7 weeks old. Sixty nude mice were divided into experimental group (n = 30) and a control group (n = 30). Sixty nude mice were all raised in SPF-level animal incubators. On the fourth day, 100 μL (about 5 × 106) PCa cells were subcutaneously inoculated into the right axillary abdominal wall of nude mice. The life and growth of nude mice and subcutaneous tumor tissues were monitored every day after inoculation. After 50 days, the nude mice were sacrificed by neck pulling, the tumor tissues were completely stripped, the volume and weight of tumor were measured, and tumor photos were taken. The volume of the stripped tumor is approximately equal to the length × wide × high. The nude mouse study was approved by Affiliated Hospital of Yangzhou University (2022-YKL2-21-003).
Immunohistochemistry Assay
The subcutaneous transplanted tumor was stripped and fixed in 10% neutral-buffered formalin and then they were paraffin-embedded. For immunohistological analyses, we used 4-μm paraffin tumor sections with heat-induced antigen retrieval, tumor sections were blocked with 5% BSA for 1 hr at room temperature, followed by incubation with mouse monoclonal human Ki-67 Antigen (Kit-0005-2, Maixin Technology Co., Ltd, China) 1:300 dilution overnight at 4℃. The horse radish peroxidase-conjugated secondary antibodies (1:1000 dilution) were purchased from Maixin Technology Co., Ltd. (FuZhou, China). Peroxidase substrates were DAB (Maixin Technology Co., Ltd, China). The nucleus was counterstained with hematoxylin (Shanghai Huzhen Biotechnology Co., Ltd, China) for 2 min. The stained tumor sections were further analyzed by Nikon microscope and JDRMS software (Jetta Technology Co., Ltd, China). A minimum of 10 views per slide were captured and analyzed.
Statistical Analyses
Data processing and statistical analysis was done using SPSS16.0 statistical analysis package. The variance test was used for measurement data (Mean ± standard deviation), and Chi-square (χ2) test was used for counting data. T-test was used between two groups and analysis of variance test between multiple groups. p < .05 means the difference is statistically significant.
Results
Analysis of tRF and tiRNA Expression Profile in PCa
The tRF and tiRNA expression profiles of three PCa tissues and three normal prostate tissues were detected by Arraystar nrStar tRNA PCR Arrays (H/M) microarray. The results showed that the hierarchical cluster analysis (Figure 1A) showed that hundreds of tRF and tiRNAs were abnormally expressed, showing upregulation and downregulation. Scatter plot screening was performed between PCa and control groups, with threshold multiple changes ≥3.0, p < .01 (Figure 1B) to identify statistically significant differentially expressed tRF and tiRNA.

(A) Hierarchical Cluster Analysis was Used to Analyze the Differential Expression of tRF & tiRNAs Between PCa and NC. (B) The Scatter Plot Shows the Differential Expression of tRF & tiRNAs.
Validation of tRF and tiRNAs in PCa
Screening conditions for upregulation of TRF & tiRNAs included log2FC > 2, p < .01, and control CPM is >5; the screening results showed that there were 10 upregulated tRF & tiRNAs between PCa and normal tissues, and their relative expression levels are shown in Table 1. qRT-PCR verified that only tRF-1:31-Glu-TTC-2 (tRF-Glu-TTC-2) and tRF-1:32-Gly-TCC-2 were upregulated in 77 pairs of PCa specimens and normal tissues (Figure 2). According to the results, tRF-Glu-TTC-2 with a high upregulation level was selected as the experimental object in this study.
Upregulated tRF and tiRNAs in PCa.
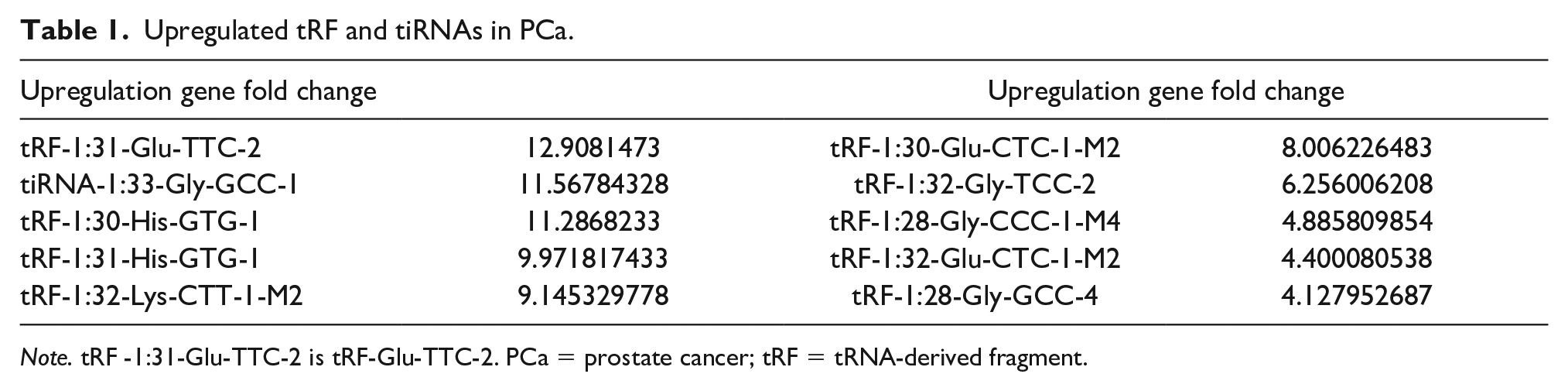
Note. tRF -1:31-Glu-TTC-2 is tRF-Glu-TTC-2. PCa = prostate cancer; tRF = tRNA-derived fragment.

qRT-PCR Analysis of 10 Upregulated tRF and tiRNAs.
Positive Expression of tRF-Glu-TTC-2 in PCa
RISH was used to detect the RNA expression of tRF-Glu-TTC-2 in 77 pairs of PCa tissues and adjacent normal tissues. As shown in Figure 3, compared with the proportion of positive cells in adjacent normal tissues (9.12% ± 1.45%), the proportion of tRF-Glu-TTC-2 positive cells in PCa tissues was 90.38 ± 8.56%, t = 19.769, p = .003 < .05. The cytoplasm of tumor cells containing brownish-yellow granules was positive cells.

The Positive Expression of tRF-Glu-TTC-2 in PCa.
Correlations Between Positive Expression of tRF-Glu-TTC-2 in PCa and Clinicopathological Parameters
The correlation between the positive expression of tRF-Glu-TTC-2 and the clinical parameters of PCa patients through the experimental results of RISH detection was explored. The age of 77 patients ranged from 53 to 88 years, with a median age of 67 years. PCa patients were divided into high-expression group (positive cell percentage>10%, n=53) and low-expression group (negative cell percentage ≤ 10%, n=24) according to the percentage of tRF-Glu-TTC-2-positive cells in RISH test results. Then the correlation between the positive expression of tRF-Glu-TTC-2 and the clinical parameters of PCa patients was statistically analyzed. As shown in Table 2, tRF-Glu-TTC-2 was closely correlated with tumor size and Gleason score (all p < .05) but not with age, lymph node metastasis, and TNM stage (all p >.05). As we all know, the size of the tumor indicates the growth and proliferation of the tumor, and Gleason score reflects the malignant degree of prostate cancer. Therefore, this result shows that the highly expressed tRF-Glu-TTC-2 in PCa reflects the growth and malignancy of the tumor.
Correlation Between tRF-Glu-TTC-2 Expression and Clinicopathological Parameters.

tRF-Glu-TTC-2 Promotes the Proliferation of PCa Cells
The above experiments suggest that tRF-Glu-TTC-2 is closely related to tumor size. Therefore, we further studied the effect of tRF-Glu-TTC-2 on the proliferation of prostate cancer cells through the CCK-8 experiment. First, the efficacy of PCa cells overexpressing or reducing tRF-Glu-TTC-2 expression was verified by qRT-PCR (p < .05; Figure 4A). CCK-8 data (Figure 4B, Table 3) showed that overexpression of tRF-Glu-TTC-2 promoted the proliferative activity of PCa PC3 cells while decreasing the expression of tRF-Glu-TTC-2 inhibited the proliferative activity of prostate cancer cells (p < .05). To further study the role of tRF-Glu-TTC-2 in the proliferation of PCa, PC3 cells with reduced tRF-Glu-TTC-2 expression were injected subcutaneously into nude mice. The experimental results showed that the tumor volume and weight of the knockdown group were significantly smaller and lighter than those of the control group (Figure 5, all p < .05). Ki-67 staining (Figure 6) of the cell proliferation index of the subcutaneously transplanted tumor showed that the proportion of Ki-67-positive cells in the reduced tRF-Glu-TTC-2 group (27.6 ± 2.5%) was lower than that in the control group (51.3 ± 6.5%, p < 0.05). The above results showed that tRF-Glu-TTC-2 plays a role of an oncogene in PCa and promotes the growth and proliferation of cancer cells.

CCK-8 Assay Results. (A) Validation of Overexpression or Reduction of tRF-Glu-TTC-2 Expression; (B) CCK8 Shows the Promoting Effect of tRF-Glu-TTC-2 on Prostate Cancer PC3 Cells, p < .05.
Effect of tRF-Glu-TTC-2 Expression on Proliferation of Prostate Cancer Cells (± s, n=3).

Note. OD = optical density.
Compared with the control group, *p < .05.
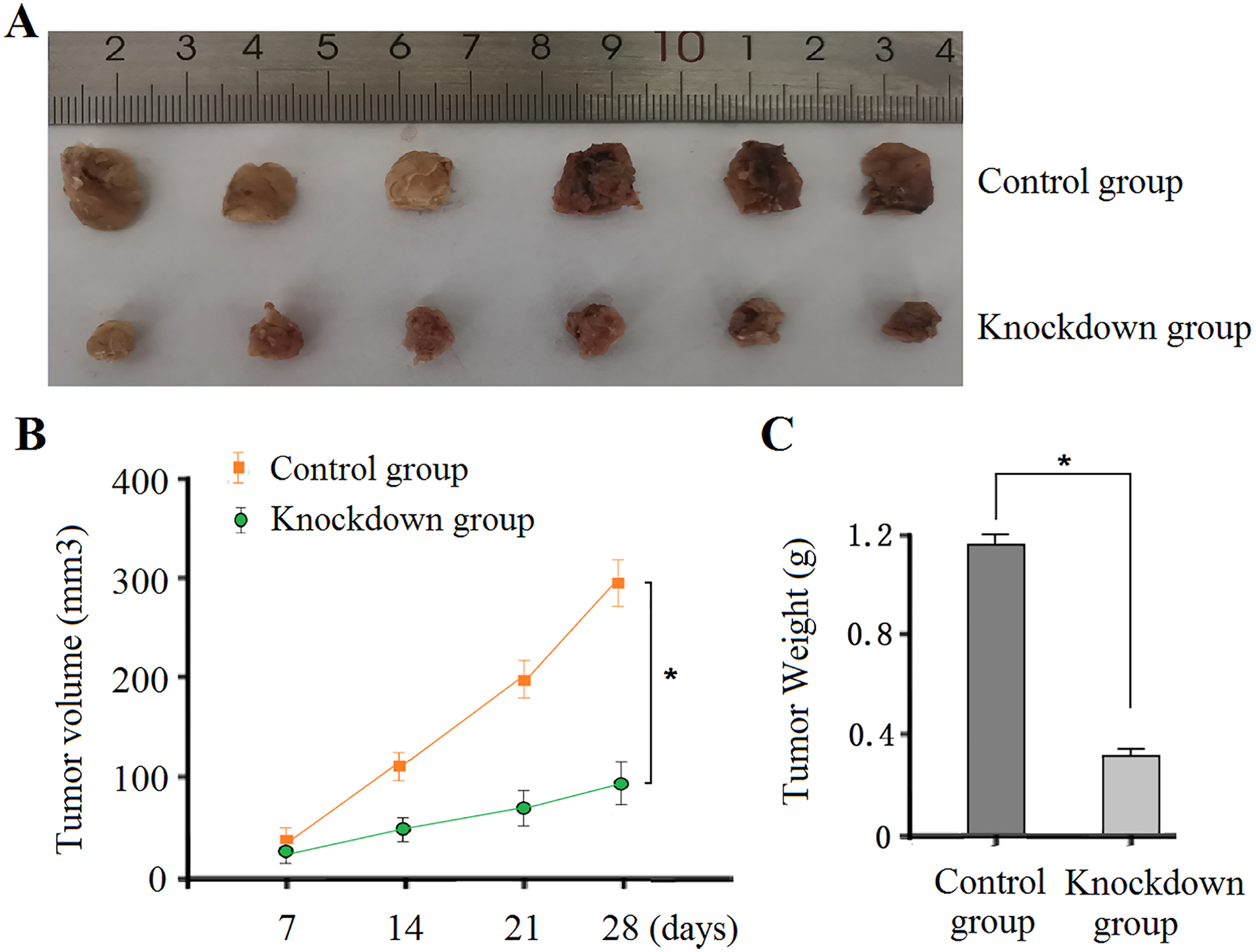
(A) The Volume of Transplanted Tumors After Silencing tRF-Glu-TTC-2 Expression was Smaller than that of Control by the Naked Eye. (B-C) The Growth Volume and Weight of Transplanted Tumor After Silencing tRF-Glu-TTC-2 Expression Were Smaller and Lighter than Those in the Control Group, p < .05.

The Proportion of Ki-67-Positive Cells in the Knockdown tRF-Glu-TTC-2 Group was 27.6 ± 2.5%, While the Positive Rate of Ki-67 in the Control Group was 51.3 ± 6.5%, p < .05
Discussion
tRFs and tiRNAs are a class of small ncRNA, which are small fragments derived from mature or precursor tRNA. According to the source, tRFs can be divided into tRF-1, tRF-2, tRF-3, tRF-5, and i-tRF and tiRNAs can be divided into 5′tiRNA and 3′tiRNA. The tRFs and tiRNAs reported in the literature are derived from nucleus-encoded tRNAs. Their main functions include inhibiting protein synthesis, regulating gene expression, priming viral reverse transcriptase, and regulating DNA damage response (Jiang & Yan, 2019). Some tRFs and tiRNAs promote cell proliferation and cell cycle progression by regulating the expression of oncogenes, while others inhibit cancer progression. For example, tRFs and tiRNAs bind to Y-box binding protein 1 (YBX1) and block YBX1 transcription, inactivating the initiation factor eif4g/a, thereby promoting the translation of ribosomal proteins or activating mitotic regulator Aurora kinase A (Zhu et al., 2019). It was reported that the expression level of tRF-Glu-TTC-027 was significantly downregulated in gastric cancer (GC). Overexpression of tRF-Glu-TTC-027 in vitro inhibits the malignant activity of GC cells. Mechanism studies confirmed that tRF-Glu-TTC-027 plays a role in GC through a mitogen-activated protein kinase signaling pathway (Xu et al., 2021). The expression levels of tRF-Gly-CCC-046, tRF-Tyr-GTA-010, and tRF-Pro-TGG-001 were downregulated in breast cancer, which can be used as new biomarkers for early diagnosis of breast cancer (Zhang et al., 2021). tRF-phe-GAA-031 and tRF-VAL-TCA-002 were identified to be significantly upregulated in colorectal cancer tissues. They were significantly correlated with distant metastasis and clinical stage. They might play an important role in the metastasis of colorectal cancer. So they may be potential biomarkers and intervention targets in the clinical treatment of colorectal cancer (Chen et al., 2022). It was confirmed that the expression level of tRF-03357 was significantly increased in serum samples of high-grade serous ovarian cancer (HGSOC), and the mechanism study identified that tRF-03357 may promote the proliferation, migration, and invasion of ovarian cancer cells by regulating HMBOX1 (Zhang et al., 2019). The expression of tRF-Leu-CAG was significantly increased in non-small cell lung cancer (NSCLC), which could promote the proliferation and cell cycle progression of lung cancer cells, and the expression of tRF-Leu-CAG was upregulated in the serum of patients with advanced NSCLC (Shao et al., 2017).
So far, some physiological functions of tRFs and tiRNAs have been reported in PCa. The tRF-1001 is derived from tRNA-Ser precursor and is related to the proliferation of PCa cells (Lee et al., 2009). The short 5′-tRFs (18-20 nt) were frequently expressed in normal prostate samples, while 29- and 30-nt 5′-tRFs were highly expressed in PCa samples (Magee et al., 2018). In another further study, it was confirmed that tRF-315 can regulate the expression of p53 and apoptosis-related proteins in PCa cells. In addition, the induction of cytochrome c by tRF-315 suggests that mitochondria may be the target of tRF regulation in cells (Yang et al., 2021). There is no literature report on the effect and role of tRF-Glu-TTC-2 in regulating the occurrence and development of PCa, which is the research of this project.
In this study, tRF-Glu-TTC-2 was positive in most cases of PCa by RNA in situ hybridization (RISH); through qRT-PCR array, it was found that the expression level of tRF-Glu-TTC-2 in PCa tissues was higher than that in adjacent normal tissues, indicating that tRF-Glu-TTC-2 played a role of an oncogene in the occurrence and development of PCa. When analyzing the relationship between the positive expression of tRF-Glu-TTC-2 and the clinicopathological features of PCa patients, it was found that the high expression of tRF-Glu-TTC-2 was closely related to the tumor size and Gleason score of PCa but not to the age, lymph node metastasis and TNM stage of patients. These results suggest that tRF-Glu-TTC-2 may be related to tumor growth and proliferation, that is, the higher the expression level of tRF-Glu-TTC-2 in PCa, the more the tumor cells can proliferate and grow, which further indicates that tRF-Glu-TTC-2 may be a cancer promoting gen and could be related with PCa progression and prognosis. Maybe further experiments in the future can verify that tRF-Glu-TTC-2 may be a biomarker for the growth of prostate cancer, it can be used as a molecular marker for the diagnosis of PCa. In TNM staging, T represents the size and depth of invasion of the primary tumor. The statistical results of this study suggest that the expression of tRF-Glu-TTC-2 has no relation to TNM staging of prostate cancer. It may be that the diameter divided in this experiment is inconsistent with the size of the primary tumor in TNM staging. In addition, the number of samples in this study may be too small, which will also affect the statistics of this result.
At present, the deficiency of this study is that it only studies the function of tRF-Glu-TTC-2. Although biological information also predicts some downstream target genes of tRF-Glu-TTC-2, the target genes of tRF-Glu-TTC-2 have not been identified and verified, so the specific mechanism of tRF-Glu-TTC-2 on the growth and proliferation of PCa cells has not been studied. In the near future, we will deepen the research on the specific regulatory mechanism of tRF-Glu-TTC-2 in proliferation, providing a new diagnostic and judgment basis for prostate cancer patients.
Conclusion
tRF-Glu-TTC-2 could be a new oncogene, which can promote the growth and proliferation of PCa cells and provide a new idea for the future research and treatment of PCa.
Footnotes
Author Contributions
LS and LW conceived and designed the study. LW,C H-H, Z Y-D, J J-Y, and S W-J provided contributions to research design, data analysis, and article writing. Y X-L and W Q-Y helped to perform the animal experiment. All authors contributed to the article and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by International Science and Technology Cooperation Project of Yangzhou Science and Technology Bureau (YZ2020200), Instructional Research Project of Jiangsu Commission of Health (Z2019046), Social Development Scientific Research Project of Yangzhou Science and Technology Bureau (YZ2022104).
Ethics Approval and Consent to Participate
Written informed consent for publication of this paper was obtained from The Affiliated Hospital of Yangzhou University and all authors. All participants provided written informed consent to participate in the research study.
