Abstract
Childhood sexual abuse (CSA) is associated with autobiographical memory (AM) disturbances. AM is important for future thinking, sense of self, and coping with negative emotions. CSA is under-researched among men, with research examining long-term neural correlates limited even further. This study explored the neural correlates of re-experiencing traumatic/negative memories to examine the influence of CSA on AM into adulthood. Fifteen males who experienced CSA, with and without posttraumatic stress disorder (PTSD; CSA+PTSD, n = 6; CSA–PTSD, n = 9) and control males without CSA histories nor PTSD (n = 11) completed a script-driven imagery paradigm during functional magnetic resonance imaging (fMRI). Males with CSA histories, with and without PTSD, processed their negative autobiographical memories with less activation compared to control males. The CSA+PTSD group of males had less activation in the left superior occipital, left superior parietal and left parahippocampal gyri compared to control participants. The CSA–PTSD group had reduced activation in the same regions to a lesser extent. This study provides preliminary empirical evidence to suggest CSA impacts AM for traumatic experiences, and the impact is notable even for men who experienced CSA but do not have PTSD. This study highlights the need for more research with men who have experienced CSA, so that, we can fully understand the neural correlates of emotional memories, and better support the mental health and continued wellness of men who experienced CSA.
Experiencing childhood adversity can predispose individuals to detrimental and potentially long-lasting consequences impacting cognitive functioning and well-being. Childhood sexual abuse (CSA) is one of several adverse childhood occurrences experienced by approximately one in six men (Dube et al., 2005; Finkelhor et al., 2014; Gartner, 1999; Romano & De Luca, 2001). Some men with histories of CSA will develop a range of difficulties later in life, such as substance use (Butt et al., 2011) or posttraumatic stress disorder (PTSD; Tolin & Foa, 2006); however, some men with CSA histories appear to experience resilient functioning (Ressel et al., 2018). Despite the prevalence of male experienced CSA and the potential consequences for the mental health of men with CSA histories, neuroimaging research related to CSA has been conducted mostly with female samples. Innate differences in brain structure and function exist between males and females, for example, in males compared to females, greater cortical thickness is observed in the orbitofrontal cortex and the insula, regions important for decision-making and emotion (Ritchie et al., 2018). It is of interest to investigate why there are such diverse outcomes from CSA in men and the potential mechanisms which may contribute to varying mental health outcomes. In the current study, we examined the neural correlates of recalling early trauma to determine the influence that the experience of CSA has on memory in adult men. This research can guide clinicians and researchers to evidence informed practices which can positively influence the well-being of men who experienced CSA.
Autobiographical memory (AM) is an explicit form of memory for personally relevant historical information, including details of traumatic experiences that occurred in childhood. Adverse childhood experiences, such as CSA, are associated with an increased prevalence of childhood AM disturbances (Brown et al., 2007). CSA can include a sexual act, abusive sexual contact, or noncontact sexual abuse (Leeb et al., 2008). CSA is one potential stressor that can lead to a trauma and stressor-related disorder, such as PTSD. As such, understanding how AM is impacted in men with CSA histories, with and without a diagnosis of PTSD, can give insight into how men process their CSA memories. This is especially relevant since AM is important for future thinking, sense of self, and coping with negative emotions (D’Argembeau, 2012; Kross et al., 2009; Prebble et al., 2013). An integrated model of memory describes that specific (i.e., episodic, AM), abstract (i.e., personal semantic), and emotional information can integrate in maladaptive ways leading to psychopathology. Revisiting maladaptive memories, and incorporating/reinforcing new emotional experiences can be of therapeutic benefit (Lane et al., 2015). AM is central to the formation of a person’s self-concept. Emotion, relevant for specific and abstract AMs, is a common theme which can provide insight into identity representation and self-continuity (Grilli & Ryan, 2020). Recently, a meta-analysis provided preliminary support for autobiographical episodic memory training, which aims to modify processing biases (e.g., overgeneralizations), as an effective and accessible treatment option for mood disorders, such as depression (Hitchcock et al., 2017).
AM is achieved through a distributed brain network. The dorsal-medial subsystem (including prefrontal and temporal areas) underlies the processes of retrieval, such as abstract or oversimplified representations of cognitive information. The medial-temporal subsystem (e.g., the parahippocampal gyrus and retrosplenium) underlies the process of re-experiencing AM and is related to the perceptual imagery of AM (Thome et al., 2019). Thome et al. (2019) proposed that studies related to AM should distinguish between scanning during a traumatic imaging paradigm (likely related to retrieval) and after the traumatic exposure (likely related to re-experiencing), since the temporal order of events are related to different neural correlates as described above.
AM related to traumatic events can be studied with a script-driven imagery task. Script-driven imagery protocols involve recording physiological reactions as participants are prompted to think about a personally relevant event in their lives (an AM). Lanius et al. (2001) adapted the trauma-script-driven imagery task for functional magnetic resonance imaging (fMRI) and reported that participants with PTSD (from CSA or motor vehicle accident) had reduced activation in the thalamus, and medial prefrontal cortex (e.g., the anterior cingulate, and medial frontal gyrus) while re-experiencing their traumatic event, compared to participants who experienced similar trauma but did not have PTSD. This reduction in activity is thought to underlie disturbances in emotion regulation (Lanius et al., 2001, 2003) Commonly experienced symptoms of PTSD like re-experiencing, dissociation and avoidance are important to consider when performing fMRI studies with trauma script-driven imagery tasks. For example, dissociative PTSD (D-PTSD) was associated with greater limbic and frontal activation during traumatic memory recall in women with D-PTSD from CSA (n = 7) compared to control participants (n = 10; one male) who experienced similar trauma but did not have PTSD. Compared to participants with D-PTSD, control participants had significantly more activation in the left parahippocampal gyrus, right middle frontal lobe, and left superior temporal gyrus (Lanius et al., 2002).
Use of the Responses to Script-Driven Imagery (RSDI; Hopper, Frewen, Sack, et al., 2007) scale can provide insight into the commonly experienced PTSD symptoms of re-experiencing, dissociation, and avoidance. Understanding the neural correlates of these symptoms during or following a script-driven imagery task is important given that different symptomology is linked with different patterns of activation, and the RSDI scale can provide important insight on the subjective experience of participants during the protocol. A study with participants who had PTSD (N = 27, seven men) using the script-driven imagery paradigm reported that re-experiencing was positively associated with right insula activity and negatively associated with right anterior cingulate cortex activity (Hopper, Frewen, van der Kolk, & Lanius, 2007). Commonly experienced symptoms of re-experiencing, avoidance, and dissociation are thus important considerations for the script-driven imagery paradigm.
Consistent with previous findings using script-driven imagery, Shin et al. (2004) conducted a study using positron emission tomography (PET) and reported that combat veterans with PTSD (compared to combat veterans without PTSD) exhibited a regional cerebral blood flow (rCBF) decrease in medial frontal regions and an increase in rCBF in the amygdala, while re-experiencing a traumatic AM (compared to neutral AM) script. Driessen et al. (2004) studied traumatic compared to nontraumatic AM with fMRI in a sample (n = 12) of women diagnosed with borderline personality disorder, half with comorbid PTSD. The women without PTSD had more bilateral prefrontal cortex activation while the women with PTSD were more activated in temporolimbic areas (e.g., parahippocampal gyrus and amygdala), with more activation in the right hemisphere. Clinical disorders, such as major depressive disorder (MDD), can influence responses to script-driven imagery (Lanius et al., 2007). Ludäscher et al. (2010) used the script-driven imagery task with women who had borderline personality disorder, some with comorbid presentations of PTSD. The scripts utilized by the study contained experiences that previously induced a dissociative state in the women and experiences that were emotionally neutral. Differences were reported in activation patterns for the whole group of participants (n = 15) compared to a subgroup of women with PTSD (n = 10) for dissociative state inducing, compared to neutral experience scripts (Ludäscher et al., 2010). Lanius et al. (2007) reported that participants with PTSD and MDD (n = 15, four males) had different patterns of neural activity during traumatic script imagery compared to participants with PTSD but no MDD (n = 11, four males) and controls without PTSD nor MDD (n = 16, three males). Both groups with participants with PTSD had reduced activation in the anterior cingulate cortex compared to controls. Participants with PTSD without MDD were associated with greater activation in the left insula compared to participants with PTSD+MDD. Taken together, these studies provide evidence to support the idea that populations with mental health disorders (e.g., MDD) have varied neural activation patterns during script-driven imagery tasks, and that co-morbid diagnoses need to be considered.
Another fMRI study imaged six participants (n = 5 males) within 3 months of a traumatic accident (e.g., motor vehicle) with the script-driven imagery paradigm. This study utilized personal narrative scripts from the traumatic incident, personal narrative scripts related to a negative memory that was not related to the trauma (i.e., a nontraumatic negative memory) and a neutral memory. Results indicated that the traumatic and nontraumatic negative memory were processed similarly. Compared to the neutral memory, the combined effect of the memory of the traumatic accident and the negative nontraumatic memory was associated with increased activation bilaterally in the retrosplenial cortex, left middle temporal gyri, temporal poles, and other regions important for memory and emotion, such as the amygdale, hippocampus, and left parahippocampal gyrus. Similar regions and activation patterns were noted for separate comparisons of the traumatic/neutral and negative nontraumatic/neutral comparisons (Piefke et al., 2008). More recently, a case-controlled twin study with 26 pairs of male twins (some with combat exposure, and some with associated PTSD) utilized the script-driven imagery paradigm with stressful (trauma unrelated) events compared to neutral events. Males with PTSD from combat trauma exhibited diminished medial prefrontal cortex (e.g., the anterior cingulate cortex and the medial frontal gyrus) activation for stressful events compared to neutral events, when compared to males without PTSD (Dahlgren et al., 2018).
Notably, many studies have employed the script-driven imagery paradigm with participants with and without PTSD. Previous studies identified that AM processing in participants with PTSD is different than that among non-PTSD participants. Moreover, potential symptoms of PTSD (e.g., dissociation) and other clinical disorders (e.g., MDD) can influence the neural correlates of traumatic AMs. Although this literature has involved studies with participants who have experienced CSA, they have been predominantly conducted with females, or with mixed trauma groups, including participants with PTSD from CSA, and participants with PTSD from an accident (Hopper, Frewen, van der Kolk, & Lanius, 2007; Lanius et al., 2001, 2002, 2007; Ludäscher et al., 2010; Piefke et al., 2008). Understanding how AM for negative experiences is influenced in men, specifically, with CSA histories is important because it will increase understanding of how one’s sense of self and coping with negative emotions may be impacted by CSA. This is important since CSA has been linked with AM disturbances (Brown et al., 2007), AM is important for sense of self and negative emotion coping (D’Argembeau, 2012; Kross et al., 2009; Prebble et al., 2013) and revisiting maladaptive memories, while incorporating/reinforcing new emotional experiences may be of therapeutic benefit (Lane et al., 2015).
In the current study, we examined traumatic AM processing in a sample of adult males with CSA histories with and without a diagnosis of PTSD, and control participants with no CSA histories and no PTSD. The objective was to provide insight into how CSA affects the neural basis of processing traumatic AMs for those with and without PTSD. It was hypothesized that differences in activation while experiencing traumatic/negative compared to neutral life events would be found between males with CSA histories and those without. In particular, it was expected that men without CSA histories would process the negative memory with more posterior cortices, related to perceptual information of their memory. Most studies that have used the script-driven imagery paradigm have imaged participants with trauma histories and PTSD compared to participants with similar trauma histories without PTSD (controls). Our control group was different so the hypothesis related to our control group is exploratory. It was expected that there would be significant differences in activation within the CSA group, depending on the presence or absence of a PTSD diagnosis. The CSA group was divided into those with (CSA+PTSD) and without PTSD (CSA–PTSD). We expected, as previous studies identified (Dahlgren et al., 2018; Driessen et al., 2004; Lanius et al., 2001, 2003; Shin et al., 2004) that trauma exposed participants without PTSD would re-experience their traumatic AM with more prefrontal activation compared to those with PTSD. Investigating these neural correlates of AM in a sample of only men with CSA is unique. This imaging study may help characterize the impact of CSA on men and potentially provide empirical evidence of a neural consequence that demands more attention from clinicians and more support for these men.
Method
Participants
This study was part of a larger project examining psychological and adaptive functioning among adult males who experienced CSA. The recruitment and neuroimaging protocols were approved by the University of Ottawa Research Ethics Board (REB) and by the REB for the Royal Ottawa Mental Health Center (REB # 2017028) where the neuroimaging tasks were completed. Participants were recruited through posters at local community centers (e.g., local YMCA) and through online advertising (e.g., Kijiji and Facebook).
A subset of CSA males from the larger sample (n = 69), and control males (n = 40) were selected based on their interest in the neuroimaging study and their fMRI compatibility. Potential participants were excluded from the neuroimaging study for moderate to severe substance use disorder from alcohol, cannabis, or opioid use (CSA, n = 27; controls, n = 10), a recent concussion (i.e., within the past 6 months), postconcussion syndrome, or attention deficit hyperactivity disorder (CSA, n = 8; controls, n = 3), left-handedness (CSA, n = 3; controls, n = 1), size/weight (CSA, n = 6; controls, n = 4), and lack of interest (CSA, n = 5; controls, n = 7). A few participants could not complete the task due to technical difficulties (CSA, n = 1; controls, n = 2). Additional factors, such as a glass eye, nontitanium metal implants, heart condition, or high blood pressure excluded several more potential participants (CSA, n = 6; controls, n = 2). The final sample consisted of 26 participants, 15 with CSA histories (n = 9 CSA–PTSD, n = 6 CSA+PTSD) and 11 control participants.
Participant Demographics
On average, the control participants were 35.3 (SD = 8.9) years old and the participants with CSA histories were 43.2 (SD = 10.9) years old. The distributions of race, education, and income were similar between the groups. All participants in this study were cisgender male, predominantly white (CSA, n = 11, 73.3%; control, n = 8, 72.7%), and college or university educated (CSA, n = 12, 80.0%; control, n = 7, 63.6%). Fewer participants in the control group (n = 11, 27.3%) than the CSA group (n = 9, 60.0%) were married or in a common law relationship.
Measures
A socio-demographic questionnaire was administered to obtain information on the participant characteristics (e.g., age, education). The Childhood Trauma Questionnaire (CTQ; Bernstein & Fink, 1998), a sound measure (Spinhoven et al., 2014), was administered to retrospectively assess physical/emotional abuse and physical/emotional neglect from their childhood. The CTQ was not used to assess sexual abuse. Sexual abuse was assessed with a modified version of the Sexual Victimization Survey (SVS; Finkelhor, 1979), that has test–retest reliability and good inter-rater reliability (Lyons & Romano, 2019). Participants were assessed for psychosis, mood symptoms, substance use, trauma, and anxiety symptoms with the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5; American Psychiatric Association, 2013; SCID-5; First et al., 2015), which is commonly used in clinical settings to measure psychological outcomes, including PTSD.
Script-Driven Imagery Task
The script-driven imagery task was unique to each participant. Days prior to the neuroimaging session, all participants were asked to record a brief neutral life event that was personally relevant. Participants with histories of CSA were asked to write briefly (i.e., something that would take around 30 s to say at a regular speaking pace) about a traumatic memory related to their abuse (e.g., recalling a particular CSA experience). Control participants were asked to write briefly about a negative/adverse memory from their childhood (e.g., death of a loved one). The memories were then turned into audio recordings with a neutral male voice, which was the same for each participant. During the task, a crosshair was visible on the screen and participants were instructed to keep their eyes open for the whole task. A 3-s beep signaled the start of their life event audio, which lasted 33 s and was then followed by 33 s of quiet. The participants were asked to think about or remember the event as best as they could while listening to the audio, and during the 33 s of rest that followed each presentation of the audio recordings. Three repetitions of the neutral memory condition (66 s × 3) were presented first, followed by three repetitions of the negative/adverse memory condition (66 s × 3) (e.g., control males) or traumatic CSA memory (e.g., CSA+/– PTSD males).
Responses to Script-Driven Imagery Scale
Participants were assessed for symptoms that may have arisen during the traumatic script task using the RSDI (Hopper, Frewen, Sack, et al., 2007) scale immediately following the neuroimaging session. The RSDI is an 11-item scale, with items measured on a scale from 0—not at all to 6—a great deal, intended to assess self-reported symptoms of re-experiencing, avoidance, and dissociation that may occur during the script-driven imagery task.
Procedure
Prior to the neuroimaging session as a part of the larger study, written consent was provided, and a series of questionnaires was administered to gather information on socio-demographics, childhood maltreatment (CTQ, SVS), and mental health functioning (SCID-5). The preliminary sessions were conducted by a qualified clinical psychology PhD student, and participants were compensated US$40 for the first session. The researchers were not blind to the participants’ group membership. At a later date, participants attended one neuroimaging session and were compensated with US$30 for travel/parking expenses. All imaging was performed on the 3T Siemens Biograph MRI-PET scanner at a local hospital using the 12-channel head coil. The imaging followed the same order for each participant. These included a T1-weighted structural scan, followed by the responses to script-driven imagery task. The pulse sequence for the fMRI scans was an echo planar imaging sequence with TR = 3,000 ms, TE = 34 ms, 48 slices, 3 mm slice thickness, 3 × 3 × 3 mm voxel acquisition size, flip angle 90°, FOV 64 × 64.
For the duration of the scanning session, participants lay supine on the MRI bed and looked at a mirror where the fMRI stimuli were projected. Participants were provided with noise reduction headphones which allowed the auditory communication to be heard while reducing noise from the scanner. They were given an emergency ball, which when squeezed would alert the technologist of any problems and they were also equipped with a pulse oximeter that allowed for the detection of signs of physiological stress. During the script-driven imagery fMRI task, participants were instructed to keep their eyes open for a crosshair on the screen. Three repetitions of the neutral memory condition were presented first, followed by three repetitions of the negative/adverse memory (for control males) or traumatic CSA memory (for men with histories of CSA+/– PTSD). The participants were asked to think about or remember each event as best as they could while listening to the audio, and for a period of rest after each recording. The RSDI scale was administered immediately following the neuroimaging session. A clinical psychologist was available following neuroimaging sessions if participants were distressed. Similarly, phone numbers for help-lines and counseling services were available in the information forms presented to the participants at the start of the study.
Data Analyses
For the script-driven imagery task, images were postprocessed with Statistical Parametric Mapping 12 (SPM12). This included realignment to correct for motion by employing the procedure outlined by Friston et al. (1996). Images were spatially normalized to match the echo planar imaging template provided in SPM12. The 2 × 2 × 2 spatially normalized images were smoothed with a 8 mm full-width at half-maximum Gaussian filter. Motion correction was applied using six regressors (x, y, z, pitch, roll, yaw) for all first-level analyses, and the canonical hemodynamic response function (HRF) was estimated in the first-level GLM. First-level, fixed effects analyses were performed for each participant using these images, representing the different conditions of the task. Contrast images were produced to identify the neural activation related to re-experiencing AM by subtracting the average activation during the three baseline condition blocks (the 33 s following the neutral memory script) from the average activation for the 33 s following each of the three blocks of the traumatic memory condition. Data were analyzed using a general linear approach to identify voxels with activity that co-varied in time with the re-experiencing of traumatic/negative relative to neutral AMs. The resulting activation was attributed to re-experiencing traumatic AM.
Whole brain second-level, random-effects analyses, with these contrast images, were then conducted with two-sample t tests between groups of participants: control, n = 11, CSA–PTSD, n = 9; control, n = 11, CSA+PTSD, n = 6; and CSA–PTSD, n = 9, CSA+PTSD, n = 6. A set threshold of puncorr = 0.05, with a cluster-wise correction at pFWE = 0.05 and a set cluster size larger than 100 voxels was used. To control for potential confounding effects of age and childhood physical abuse, participants’ age and physical abuse scores (CTQ) were included in the fMRI analyses as covariates. Each additional covariate added lowers power by one degree of freedom, and covariates of no interest should be distributed randomly and balanced across groups (Jenkinson et al., 2018). As such, emotional abuse was not included as a covariate in the analyses; the distribution of emotional abuse scores in the control group was not random. Childhood emotional and physical abuse scores were correlated (r = .36, p = .053).
Results
Within the sample, all CSA occurred by male perpetrators. The age where CSA began was between 5 and 13 years old for the participants with histories of CSA+PTSD (M = 8.17 years old) and between 5 and 12 years old for the participants with histories of CSA–PTSD (M = 7.56 years old). Mental health status, childhood trauma history, and average RSDI responses differences are denoted in Table 1.
Trauma History, Mental Health, and Results from the RSDI Scale.
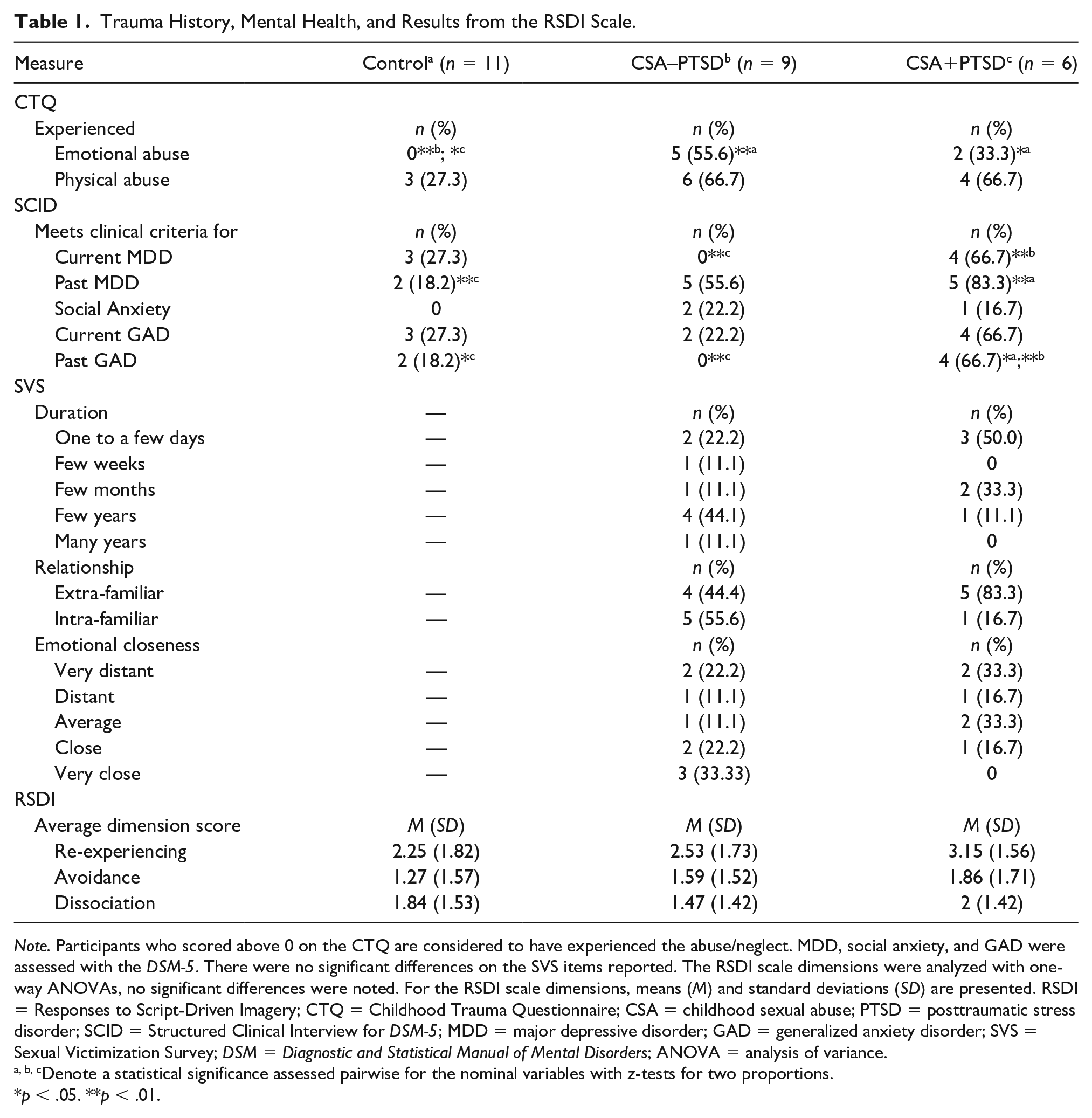
Note. Participants who scored above 0 on the CTQ are considered to have experienced the abuse/neglect. MDD, social anxiety, and GAD were assessed with the DSM-5. There were no significant differences on the SVS items reported. The RSDI scale dimensions were analyzed with one-way ANOVAs, no significant differences were noted. For the RSDI scale dimensions, means (M) and standard deviations (SD) are presented. RSDI = Responses to Script-Driven Imagery; CTQ = Childhood Trauma Questionnaire; CSA = childhood sexual abuse; PTSD = posttraumatic stress disorder; SCID = Structured Clinical Interview for DSM-5; MDD = major depressive disorder; GAD = generalized anxiety disorder; SVS = Sexual Victimization Survey; DSM = Diagnostic and Statistical Manual of Mental Disorders; ANOVA = analysis of variance.
Denote a statistical significance assessed pairwise for the nominal variables with z-tests for two proportions.
p < .05. **p < .01.
Script-Driven Imagery fMRI Task
CSA+PTSD participants had significantly less activity than control participants in the posterior cortices (Figure 1A) while re-experiencing their traumatic AM, as well as less bilateral dorsolateral prefrontal cortex activation (Figure 1B; Table 2). Similarly, CSA–PTSD participants had less activation in the posterior cortices while re-experiencing their traumatic AM compared to control participants (Figure 2). When compared to CSA+PTSD participants, the CSA–PTSD participants had more bilateral dorsolateral and right middle prefrontal cortex activation while re-experiencing traumatic AM (Figure 3). One-way analyses of variance (ANOVAs) of the RSDI subscales determined that there were no significant differences in self-reported symptoms of re-experiencing (see Table 1 for mean scores and standard deviations from the RSDI scale dimensions by group). The overall patterns of activation for all analyses remained the same when age and childhood physical abuse were included as covariates.
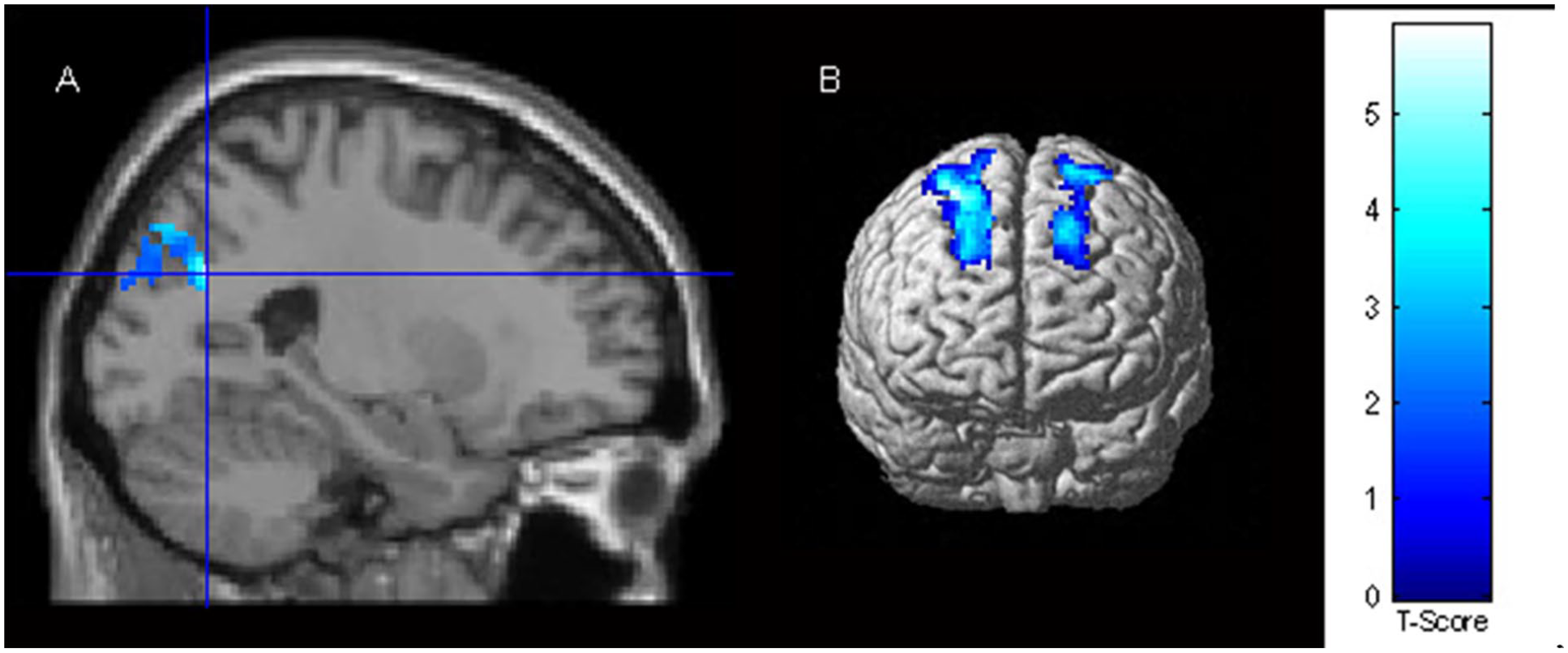
CSA+PTSD < Controls. Regions of the Brain Where Participants With CSA+PTSD Histories Had Significantly Less Activity Than the Control Participants During Negative Autobiographical Re-Experiencing. (A) Blue Crosshairs Are Located on the Left Superior Occipital Gyrus and (B) Rendered Image of Bilateral Prefrontal Cortex.
Significant Activation Differences for Re-Experiencing Traumatic > Neutral.
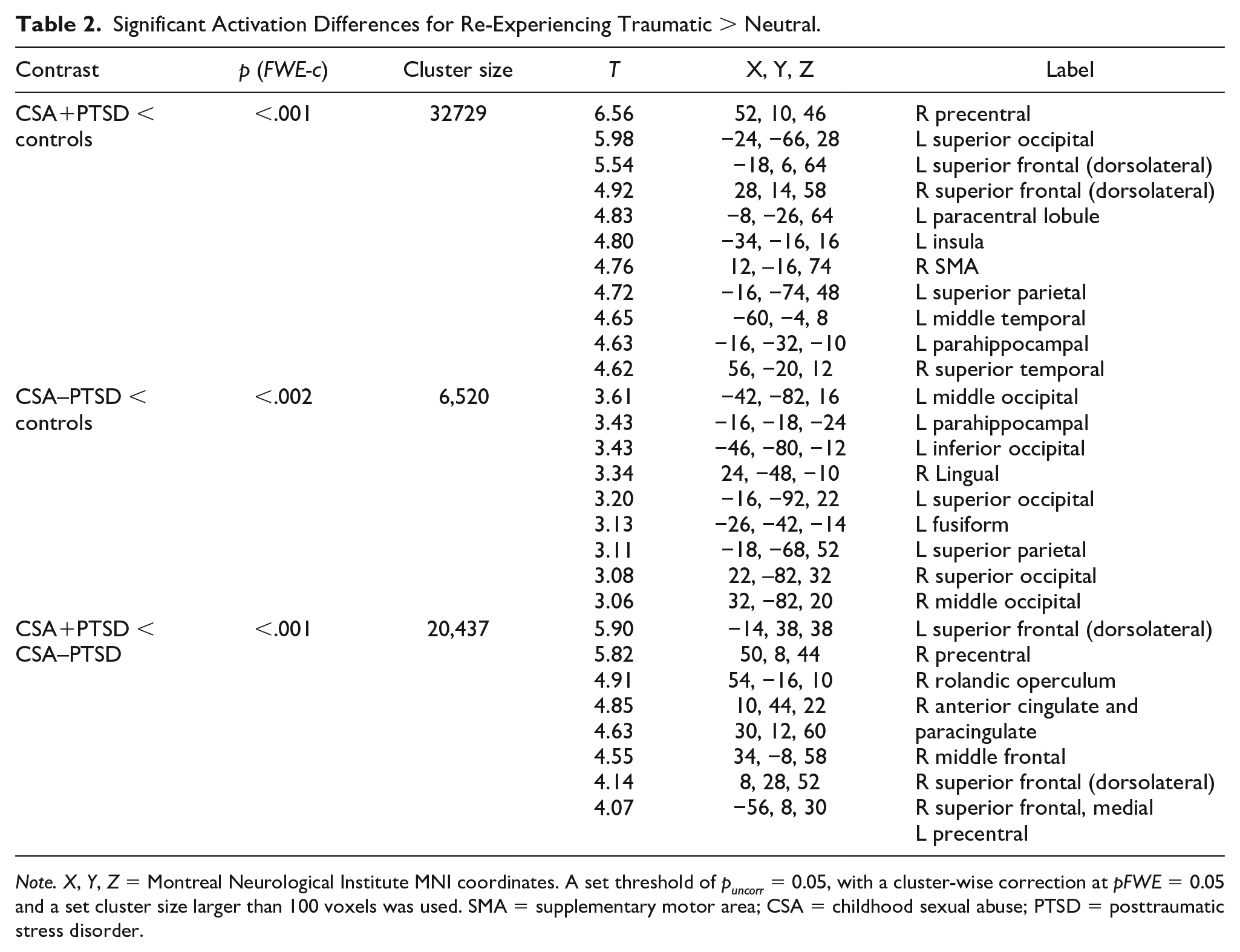
Note. X, Y, Z = Montreal Neurological Institute MNI coordinates. A set threshold of puncorr = 0.05, with a cluster-wise correction at pFWE = 0.05 and a set cluster size larger than 100 voxels was used. SMA = supplementary motor area; CSA = childhood sexual abuse; PTSD = posttraumatic stress disorder.
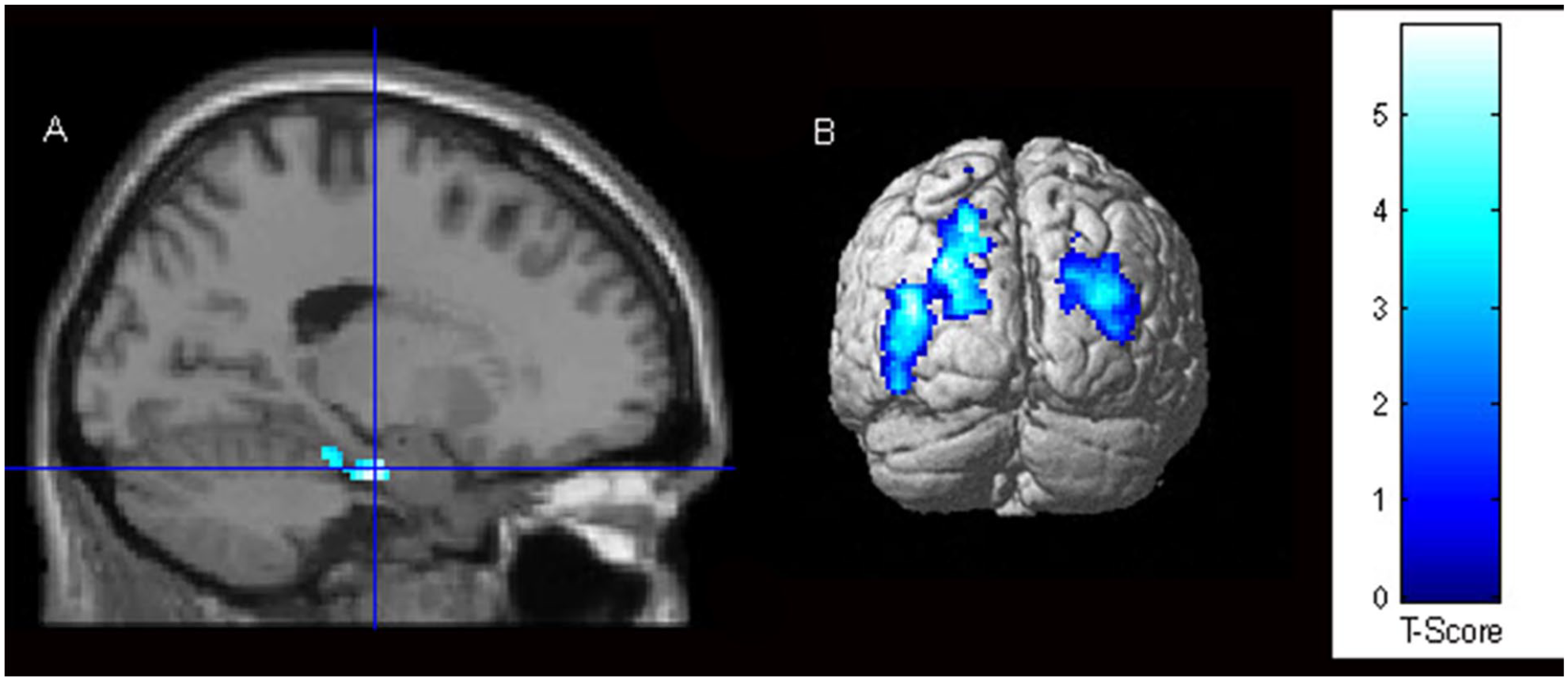
CSA–PTSD < Controls. Regions of the Brain Where Participants With CSA–PTSD Histories Had Significantly Less Activity Than the Control Participants During Negative Autobiographical Re-Experiencing. (A) Blue Crosshairs Are Located on the Parahippocampal Gyrus and (B) Rendered Image of the Superior, Middle And Inferior Occipital Gyri and Left Superior Parietal Lobule.

CSA+PTSD < CSA–PTSD. Regions of the Brain Where Participants With CSA+PTSD Histories Had Significantly Less Activity Than the Participants With Histories of CSA–PTSD During Negative AM Re-Experiencing. (A) Blue Crosshairs Are Located on the Anterior Cingulate And (B) Rendered Image of Bilateral Prefrontal Cortex Activity Differences
Discussion
Most trauma literature for men has focused on combat-related experiences. With the prevalence of CSA and the lack of research into its neural impact on men specifically, it was important to perform this investigation. Similarly, it was important to assess emotional memory re-experiencing as it may be impacted by the experience of CSA. Previous studies using the script-driven imagery paradigm have focused on the process of retrieval, re-experiencing, or both. This study focused on understanding the neural correlates of CSA in males while re-experiencing traumatic compared to neutral autobiographical memories. Men who experienced CSA with and without PTSD and control men without CSA histories completed the script-driven imagery paradigm, and provided subjective information about their experience during the task with the RSDI scale. Differences were found in activation between males with and without CSA histories (e.g., less activation in the anterior cingulate for men with histories of CSA+PTSD during the traumatic > neutral contrast compared to men with histories of CSA–PTSD). This finding provides preliminary empirical evidence to demonstrate that experiencing CSA has a long-term impact that may help explain a propensity for mental health consequences later in life. As stated in the introduction, CSA can have a negative impact on AM (Brown et al., 2007), AM is important for self-concept, coping with negative emotion, and so on (D’Argembeau, 2012; Kross et al., 2009; Prebble et al., 2013). Given the small sample size included in this study it is important for future research to continue to investigate the impact of CSA on AM in men who experienced CSA.
Participants with CSA histories, with and without PTSD, processed their negative AMs with less activation distributed widely across both hemispheres when compared to control males. In more posterior brain regions, the CSA+PTSD participants had less activation in the left superior occipital, left superior parietal and left parahippocampal gyri compared to control participants. The CSA–PTSD participants had reduced activation in the same regions, the left superior occipital, left superior parietal and left parahippocampal gyri, although to a lesser extent. Similarly, in a separate fMRI task (emotional working memory n-back), with the same group of participants, the males with CSA histories, even without a PTSD diagnosis, were impacted when working memory was influenced by negative emotion. The study also reported differences in working memory processing between participants with CSA histories with and without PTSD (Chiasson et al., 2021).
The medial temporal lobe is important for several aspects relevant to memory. Anterior components of the parahippocampal gyrus are connected to occipital regions through dorsal streams important for object recognition. Posterior components of the parahippocampal gyrus are connected to the parietal cortex through ventral streams important for spatial recognition (Raslau et al., 2015). Reduced activation in these regions among males with CSA histories, with and without PTSD, demonstrates that control males are processing their negative memory in regions relevant to object and spatial details more so than both groups of participants with CSA histories. One possibility is that these neurological differences could underlie disruptive processing patterns (e.g., overgeneralizations, Hitchcock et al., 2017) commonly experienced by individuals with mood and anxiety disorders. It is also possible that this disrupted processing is protective, but further research is required to identify this relationship.
Interestingly, all brain regions where there were significant differences between the CSA+PTSD and CSA–PTSD groups were localized to the frontal lobe. Specifically, while re-experiencing traumatic AM, men with histories of CSA–PTSD had greater activation bilaterally in the prefrontal cortex than those with PTSD. Previous studies have documented similar results with females. Participants (n = 10, nine females) with trauma histories, but no PTSD were more activated in the right middle frontal lobe compared to participants with trauma histories and PTSD (n = 7, all female) while re-experiencing traumatic compared to neutral AM (Lanius et al., 2002). Similarly, female participants with borderline personality disorder without PTSD were more activated bilaterally in the prefrontal cortex when compared to participants who also had borderline personality disorder and accompanying PTSD while re-experiencing traumatic more than neutral AM (Driessen et al., 2004). These similarities between results with females and males suggest that although men’s experiences of CSA, including their long-term consequences, may be different from females, their neural responses are somewhat similar. Similarly, males with PTSD from combat trauma exhibited diminished medial prefrontal cortex activation for stressful events compared to neutral events, when compared to males without PTSD (Dahlgren et al., 2018). As such, our finding during re-experiencing traumatic AM contributes to the literature by documenting reduced bilateral prefrontal activation in men who experienced CSA+PTSD compared to those with histories of CSA–PTSD. Future studies should attempt to recruit a large, diverse sample of people who experienced CSA so further comparisons in terms of gender, mental health profiles and adverse childhood experiences can be conducted to strengthen our understanding of the impact of CSA on AM.
The dorsolateral and ventromedial prefrontal cortices are important for emotion regulation and attention. The dorsolateral prefrontal cortex is important for cognitive processes (e.g., attention redirection) related to the valence of emotional stimuli, while the ventromedial prefrontal cortex is important for cognitive processes (e.g., extinction) related to emotional control (Nejati et al., 2021). Bilateral dorsolateral prefrontal cortex activation was reduced in the CSA+PTSD participants relative to both the controls and the CSA–PTSD participants. Furthermore, there were no significant differences in dorsolateral prefrontal activation between the control participants and CSA–PTSD participants. Similarly, there were medial prefrontal cortex differences in the medial frontal gyrus between CSA+PTSD and CSA–PTSD participants, as well as differences in activation in the anterior cingulate cortex. The anterior cingulate cortex is an important brain region for emotion and cognition, as it is functionally connected to both limbic and prefrontal regions (Stevens et al., 2011). Perhaps, this prefrontal activation is what helps men with CSA histories avoid PTSD. Thus, training the prefrontal cortex, by capitalizing on its neuroplasticity, through cognitive behavior therapy, mindfulness or other lateral frontal attention network interventions may be of importance to investigate further for males with CSA histories. These interventions may harness protective factors, including temperament, personality, basic attitudes toward one’s self and the environment, as well as effective coping skills and use of social supports (Campbell-Sills et al., 2006; Guay et al., 2006; Yehuda & Flory, 2007; Yuan et al., 2011).
The subjective experience of negative/traumatic AMs during the script-driven imagery task is an important consideration. The traumatic script of participants with and without PTSD from CSA was a memory of sexual abuse during childhood. The groups of participants who experienced CSA were compared to the control group, who listened to an otherwise negative memory, unrelated to CSA as they did not experience CSA. Previous research has indicated that traumatic and nontraumatic negative memories are processed similarly (Dahlgren et al., 2018; Piefke et al., 2008). The current study was one of the first to focus on AM with a sample of males who experienced CSA. Given the importance of AM for the formation of a person’s self-concept and that emotional memories can provide insight into identity representation and self-continuity (Grilli & Ryan, 2020), studying traumatic AM in males with CSA histories should be a priority for researchers. Especially since autobiographical episodic memory training could be an effective and accessible treatment option for mood and anxiety disorders (Hitchcock et al., 2017).
Limitations
The two main limitations for this study are the small size and reliance on self-report measures. Participants self-reported their experiences of CSA and this information could not be validated. Also, the amount of time that elapsed since the CSA varied between participants. It is important to consider that each of the participants who experienced CSA would have experienced their trauma uniquely, and the amount of trauma experienced within the group varied. The variability in abuse experiences may have contributed to the results, for example, there was a significant difference in the amount of physical and emotional abuse experienced between the controls and the participants who experienced CSA. Although the participant’s age and childhood physical abuse did not influence the results, the influence of childhood emotional abuse could not be examined due to a lack of distribution in the emotional abuse scores of the control group (i.e., all scores were null). The low number of participants in the study means there is a possibility that some significant activation differences were not captured (i.e., possibility of a Type II error). Likewise, the lack of statistically significant differences in the RSDI subscales for symptoms related to re-experiencing, dissociation and avoidance could potentially be contributing to a type II error, given that a small sample size would only allow differences with large effect sizes to be captured as significant. Nonetheless, this study importantly provides preliminary evidence that suggests AM for traumatic events is impacted in men with CSA histories, with and without PTSD.
Conclusion
AM is important for future thinking, sense of self, and coping with negative emotions (D’Argembeau, 2012; Kross et al., 2009; Prebble et al., 2013). This study provides preliminary empirical evidence to suggest CSA impacts AM for traumatic experiences, and the impact is notable even for men who experienced CSA but do not have PTSD. This evidence can provide men with validation of their lived experiences, be a step toward removing stigma related to CSA for men and may provide insight for clinicians to help men who have experienced CSA improve their cognitive functioning and overall well-being. Notably, due to the limited sample size included in this study, future research is needed with a large, diverse sample. Men who have experienced CSA could benefit from clinician support for processing their traumatic memories, for coping with distressing emotions and establishing a healthy sense of self. For clinicians, AM is an avenue that can be accessed to understand and provide support to update a client’s self-concept to establish a state of psychological well-being (Grilli & Ryan, 2020; Hitchcock et al., 2017). This study highlights the need for more research with men who have experienced CSA so we can fully understand their altered neural response to emotional memories and support their mental health and continued wellness.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a team grant from the Canadian Institutes of Health Research (610738-190799; Principal Investigator Dr. Christine Wekerle).
