Abstract
Erectile dysfunction (ED) is one of the main functional complications of surgical resections of the rectum due to rectal cancers or inflammatory bowel disease (IBD). The present systematic review aimed at revising ED management strategies applied after rectal resections and their efficacy in terms of improvement of the International Index of Erectile Function (IIEF) score. A literature search was conducted on Medline, EMBASE, Scopus, and Cochrane databases by two independent reviewers following the PRISMA guidelines. Randomized and nonrandomized controlled trials (RCTs, NRCTs), case-control studies, and case series evaluating medical or surgical therapies for ED diagnosed after rectal surgery for both benign and malignant pathologies were eligible for inclusion.
Out of 1028 articles initially identified, only five met the inclusion criteria: two RCTs comparing oral phosphodiesterase type-5 inhibitor (PDE-5i) versus placebo; one NRCT comparing PDE-5i versus PDE-5i + vacuum erection devices (VEDs) versus control; and two before-after studies on PDE-5i. A total of 253 (82.7%) rectal cancer patients and 53 (17.3%) IBD patients were included. Based on two RCTs, PDE-5i significantly improved IIEF compared to placebo at 3 months (SMD = 1.07; 95% CI [0.65, 1.48]; p < .00001; I2 = 39%). Improved IIEF was also reported with PDE-5i + VED at 12 months. There is a paucity of articles in the literature that specifically assess efficacy of ED treatments after rectal surgery. Many alternative treatment strategies to PDE-5is remain to be investigated. Future studies should implement standardized preoperative, postoperative, and follow-up sexual function assessment in patients undergoing rectal resections.
Introduction
Erectile dysfunction (ED) is defined as the persistent inability to attain and maintain an erection sufficient to permit satisfactory sexual performance (Hatzimouratidis et al., 2010). ED is one of the most frequent functional complications of radical pelvic surgery (Zippe et al., 2006), in particular rectal surgery, with an incidence rate ranging from 5% to 90% of patients according to the different studies (Celentano et al., 2017; Sorensson et al., 2019), and as high as 73% after low anterior rectal resections (Havenga et al., 1996; Keating, 2004; Schmidt, Bestmann, Kuchler, Longo, et al., 2005). This may be due to direct nerve injuries that occur during the mesorectal plane dissection, but also as a consequence of thermal injuries, stretching, inflammation, and ischemia that indirectly damage the pelvic nerves (Fang et al., 2019; Giglia & Stein, 2019). Several psychological factors may play a substantial role in the development of poor sexual function following rectal surgery, as changes in body perception related to wound scars or ostomies often contribute to lower sexual motivation and libido (Pucciarelli et al., 2011; Towe et al., 2019).
Multiple risk factors for ED after rectal surgery have been reported, including age, smoking, presence of diabetes and hypertension, specific surgical techniques, and surgeon’s experience (Adam et al., 2016; Giglia & Stein, 2019; Towe et al., 2019; Zippe et al., 2006). In case of oncologic surgery, tumor stage, tumor invasion, and administration of neoadjuvant and adjuvant chemoradiation therapies have been reported to increase the risk of ED (Bonnel et al., 2002; Giglia & Stein, 2019; Huang et al., 2016; Lange et al., 2009). Conversely, surgical rectal resections performed for benign pathologies, like inflammatory bowel disease (IBD), appear to be associated with a lower incidence of ED, but the risk is not completely eradicated even when nerve-sparing techniques are applied (Celentano, 2017; George et al., 2018).
Spontaneous erectile function recovery is rare and it may take 2 years or longer with only few patients returning to their preoperative erectile function (Sivarajan et al., 2014). The most common ED management strategies include psychological evaluation and support, pharmacological therapies, and mechanical treatments (e.g., vacuum erection devices [VEDs] and penile prosthesis implantation; Madiraju et al., 2019; Montague et al., 2005; Towe et al., 2019; Wespes, 2006). Medical treatments comprise the oral administration of phosphodiesterase type-5 inhibitors (PDE-5is [i.e., sildenafil, tadalafil, vardenafil, avanafil]; Lindsey et al., 2002), as well as the direct drug delivery of prostaglandins via topical creams (i.e., alprostadil; Anaissie & Hellstrom, 2016) and intra-urethral suppository or intracavernosal injections. Oral PDE-5is are currently considered the first-line therapy for ED. Their efficacy has been largely investigated after radical prostatectomy (Liu et al., 2017) but remains poorly studies in ED patients after rectal surgery.
The population of rectal surgery survivors is susceptible to steadily increase because of the raising global burden of both benign and malignant rectal pathologies (Collaborators, 2020; Ferlay et al., 2018; Siegel et al., 2017), and the progressive improvement of postoperative outcomes and survival after surgery. Thus, there is a compelling need for efficacious strategies to manage secondary morbidity and long-term functional complications, such as ED, which are known to drastically impact the patient’s quality of life (Downing et al., 2019).
The present systematic review aims to revise the available literature about ED management strategies applied after rectal resections and investigate ED treatment efficacy in terms of improvement of the International Index of Erectile Function (IIEF) score over time.
Methods
The present systematic review was conducted and reported according to the Preferred Reporting Items for Systematic reviews and Meta-Analysis (PRISMA) statements checklist (Moher et al., 2010) to answer the following research questions: (a) What are the treatment strategies applied to manage ED after rectal surgery? (b) What is the efficacy of ED treatments in patients developing it after rectal resections?
The study inclusion criteria were established before starting the literature search according to the following Population, Intervention, Comparisons, Outcomes, Study Design (PICOS) schema:
The following diseases were considered: rectal cancer, IBD, familial adenomatous polyposis syndrome (FAP). The following rectal surgeries were considered: total mesorectal excision (TME), partial mesorectal excision (PME), abdominal perineal resection (APR), Hartmann’s procedure, restorative proctocolectomy with intestinal pouch anal anastomosis (IPAA). All surgical approaches were considered, including laparotomy, laparoscopy, transanal TME (Ta-TME), and robotic surgery.
Literature Search
The following online databases were searched for relevant articles: Medline (through PubMed), EMBASE, Scopus, and Cochrane. Specific keywords and MeshTerm were used, including: erectile dysfunction, treatment, management, penile rehabilitation, penile implant, penile prosthesis, rectal cancer, rectal tumor, inflammatory bowel diseases, familial adenomatous polyposis syndrome, rectal resection, proctectomy, rectal surgery, total mesorectal excision, partial mesorectal excision, and neoadjuvant chemoradiation therapy. Research equations were adapted to each database to assure the exhaustiveness of the search. Reference lists of review articles, letters, and commentaries were cross-checked to identify potential additional records. The literature search was performed in February 2020 and considered articles written in English from 1960 onward.
Study Selection and Quality Assessment
Articles were searched and reviewed for inclusion by two independent authors (MN and BA). Direct exclusion was limited to those articles judged nonpertinent by both reviewers. All disagreements were solved by a third reviewer (NdeA). A first screening was conducted at the title and abstract level. For this step, the web application Rayyan QCRI (Ouzzani et al., 2016) was used. Then, the remaining articles underwent a full-text evaluation to finalize the selection process. Once retrieved the final pool of articles meeting the inclusion criteria, the two reviewers proceeded to data extraction and study quality assessment.
For RCTs, the RoB-2 Cochrane tool for the assessment of risk of bias was used (Higgins et al., 2011; Sterne et al., 2019). For NRCTs, the Risk of Bias in Nonrandomized Interventional Studies tool (ROBINS-I; Sterne et al., 2016) was used. The Axis tool (Downes et al., 2016) was adopted to assess the reliability of noncomparative cross-sectional studies.
Data Extraction and Analysis
A dedicated excel spreadsheet was used to extract the following data from each included study: authors’ name, year of publication, country, study time frame, study design, sample size, patient population, comorbidities, rectal pathology, type of surgery, surgical approach, presence of stoma, neo-adjuvant and adjuvant therapies, preoperative sexual function evaluation, immediate postoperative erectile evaluation, and postoperative sexual function evaluation at any reported follow-up time point (e.g., 3, 6, and 12 months).
Qualitative and quantitative analyses of the extracted data were performed, whenever possible by estimating mean differences (MD) or standardized mean difference (SMD) and 95% confidence interval (CI). If standard deviation (SD) was not reported, it was estimated from the CI and p value. The pooled estimates of the MD/SMD were calculated using random effects models to consider potential inter-study heterogeneity and to adopt a more conservative approach. Heterogeneity was assessed by I2 statistics and values of 25%, 50%, and 75% were considered as low, moderate, and high, respectively. Pooled effects were considered significant if p < .05. Meta-analyses were performed using RevMan software (version 5.3; Cochrane Collaboration).
Results
Selection of Studies
Out of the 1028 studies initially identified, 140 (13.6%) were excluded because they were written in languages other than English. The remaining 888 were retrieved for title and abstract assessment: of these, 352 were excluded because of wrong study population of patients (e.g., female, pediatric, or urological patients); 431 were excluded because of wrong study designs (e.g., review articles). Full-text evaluation was thus conducted for 105 articles. Finally, five studies met the inclusion criteria and were selected for the present systematic review (Figure 1).

PRISMA diagram.
Characteristics of Studies
Among the included studies, there were two RCTs (Lindsey et al., 2002; Park et al., 2015), one NRCT (Deng et al., 2017), one prospective (Nishizawa et al., 2011), and one retrospective before-after (noncomparative) study (Lindsey et al., 2003). Two studies were conducted by the same author group in the United Kingdom (Lindsey et al., 2002, 2003); the other studies were conducted in Korea (Park et al., 2015), China (Deng et al., 2017), and Japan (Nishizawa et al., 2011).
The first version of the IIEF score (based on 15 questions, score ranging from 0 to 75; Rosen et al., 1997) was adopted in one study published in 2002 (Lindsey et al., 2002). The fifth version of the IIEF (based on five questions, score ranging from 0 to 25) was used in the more recent studies (Deng et al., 2017; Lindsey et al., 2003; Nishizawa et al., 2011; Park et al., 2015). A total of 253 (82.7%) rectal cancer patients and 53 (17.3%) IBD patients were included.
All studies investigated the medical treatment of oral PDE-5i administration. One study also tested the efficacy of a combined treatment strategy, associating PDE-5i with a VED for ED (Deng et al., 2017). No study was found on surgical management strategies with implant placement, or on psychological interventions and pelvic floor rehabilitation for ED in patients receiving rectal surgery.
Study characteristics and outcomes are summarized in Tables 1 and 2.
Summary of Characteristics and Outcomes of RCTs and NRCTs.

Note. APR: abdomino-perineal resection; CD: Crohn disease; IIEF: International Index of Erectile Function; IPAA: intestinal pouch anal anastomosis; LAR: lower anterior resection; NRCT: non-randomized controlled trial; NR: not reported; PDE-5I: phosphodiesterase type 5 inhibitor; RCT: randomized controlled trial; TME: total mesorectal excision; UC: ulcerative colitis; VED: vacuum erection device.
Significant difference between the PDE-5i and placebo/control groups (p < .05).
Significant difference between the PDE-5i and PDE-5i + VED groups (p < .05).
IIEF-5 scale is used (0–25/25).
the first version of IIEF is used (0–75/75).
Summary of the Characteristics and Outcomes of the Before-After Studies.

Note. APR: abdomino-perineal resection; ED: erectile dysfunction; IBD: inflammatory bowel disease; IIEF: International Index of Erectile Function; ISR: Inter-sphincteric resection; LAR: lower anterior resection; NR: not reported; PDE-5i: phosphodiesterase type 5 inhibitor; RP: Restorative proctocolectomy; TME: total mesorectal excision.
Efficacy of PDE-5i Treatment for ED After Rectal Surgery
Both RCTs (Lindsey et al., 2002; Park et al., 2015) reported a statistically significant improvement in sexual function, as evaluated on IIEF scores, in the PDE-5i group compared to the placebo group at the 3-month postoperative follow-up. Lindsey et al. (2002) reported an IIEF score in the placebo group that was significantly lower than in the PDE-5i group (respectively 34.5 vs. 57.4, p = .005; Lindsey et al., 2002). Park et al. (2015) also observed a significantly lower IIEF-5 score in the placebo group compared to the PDE-5I one (respectively 10.8 vs. 14.3; 95% CI [1.3, 5.5]; p < .001; Park et al., 2015). However, these values were no more significantly different at the 6-month follow-up (Park et al., 2015). In the same study considering exclusively rectal cancer patients (Park et al., 2015), the authors also performed a subgroup analysis concerning the intervention arm; they considered the IIEF-5 increase after ED therapy and reported that age (cut-off 54 years), neo-adjuvant radiotherapy, surgical approach (i.e., open vs. laparoscopic vs. robotic surgery), and type of anastomosis (i.e., colorectal vs. coloanal) were not related to significant differences in the treatment response.
Deng et al. (2017) in 2016 conducted an NRCT comparing three treatment arms and reported a significant difference between the groups treated with PDE-5i or PDE-5i + VED and the group of ED patients receiving no treatment (control group) at 6- and 12-month follow-ups (IIEF-5 scores at 6 months for control vs. PDE-5i vs. PDE-5i + VED: 9.2 vs. 14.9 vs. 18, respectively; p = .01; IIEF-5 scores at 12 months: 10.9 vs. 15.1 vs. 18.7, respectively; p = .04; Deng et al., 2017). See Table 1.
A pooled data analysis was performed including only the two RCTs (Lindsey et al., 2002; Park et al., 2015) that compared the mean of IIEF score between the PDE-5i and placebo groups. Differences between the treatment arms were assessable only for the 3-month follow-up (ΔIIEF 3-baseline: IIEF score assessed after 3 months of treatment – IIEF score at baseline). The overall SMD was 1.07 in favor to PDE-5i treatment with a moderate heterogeneity (I2 = 39%; Figure 2).
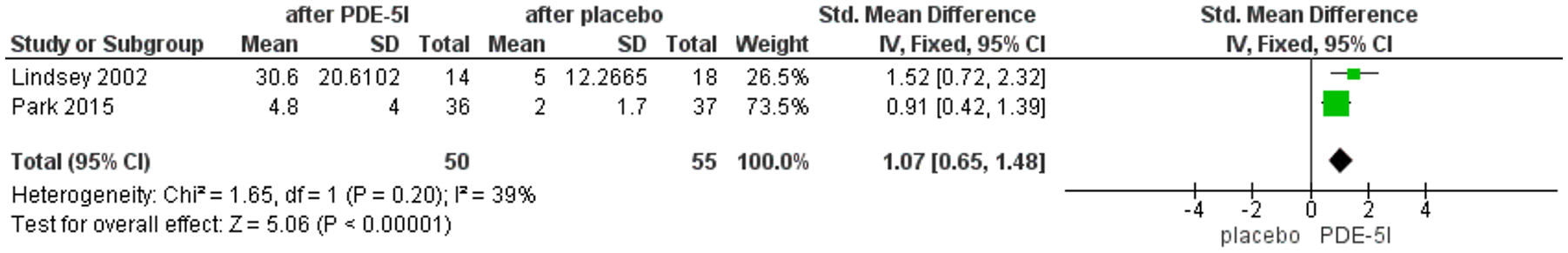
Forest plot of PDE-5i efficacy.
The two before-after studies by Lindsey et al. (2003) and Nishizawa et al. (2011) assessed the efficacy of PDE-5i treatment using a noncomparative study design and including patients with rectal cancer and IBD (Lindsey et al., 2003) or rectal cancer only (Nishizawa et al., 2011). Both studies observed an improvement in erectile function in, respectively, 69% (Lindsey et al., 2003) and 87% (Nishizawa et al., 2011) of patients who received oral PDE-5i (Table 2).
Adverse events were reported in only three out of five studies (Lindsey et al., 2002; Nishizawa et al., 2011; Park et al., 2015), with incidence rates varying between 0% to 50% of patients receiving PDE-5i. They mainly consist in minor symptoms such as facial flushing and headache.
Study Quality Assessment
Based on the RoB-2 tool, the two RCTs were judged at moderate (Park et al., 2015) and high (Lindsey et al., 2002) risk of bias (Supplemental Table 1). Based on the ROBINS-I tool, the NRCT (Deng et al., 2017) was judged at moderate risk of bias (Supplemental Table 2). The Axis tool used for two before-after studies (Supplemental Table 3) indicated that both articles (Lindsey et al., 2003; Nishizawa et al., 2011) have a poor score because of the lack of detailed inclusion criteria (Lindsey et al., 2003) and reported results (Nishizawa et al., 2011), the absence of characterization of nonresponders, and the unmotivated choice of not using IIEF score for ED after treatment (Lindsey et al., 2003; Nishizawa et al., 2011).
Discussion
This is the first systematic review, in our knowledge, to focus on ED treatment after rectal surgery. The literature review and critical appraisal reveal that, despite the high frequency of ED, there is a paucity of data concerning the efficacy of ED treatments in the specific subset of patients having undergone rectal resections. The existing evidence supports the short-term efficacy of medical therapy with PDE-5is also in the context of post-rectal surgery ED. Alternative treatments remain essentially unexplored.
The lack of studies evaluating the different treatment strategies for post-rectal surgery ED is even more evident when comparing it to the pertinent literature on urological pelvic surgery (Cui et al., 2016; Liu et al., 2017; Tian et al., 2017). This difference can be partially explained because post-surgical sexual impairment straddles the limit between the digestive domain and the urological one; as a consequence, its management could be hypothetically left to a specialist not directly involved in the index surgical procedure, impairing a full insight on its efficacy and impact on the patient’s quality of life (Kirby et al., 2014).
Several treatment options exist for the management of ED. However, when exploring the available literature about ED as a complication of rectal surgery, only the use of PDE-5is has been investigated, either as the stand-alone treatment or in combination with VED (Deng et al., 2017). Although direct comparisons and pool data analyses were not possible, except for 3-month IIFE outcome of the two RCTs, the literature is consistent observing that the nightly administration of PDE-5i is associated with ED improvements that are significantly higher than under placebo (Lindsey et al., 2002; Park et al., 2015), and may be even potentiated with a combined therapy PDE-5i + VED (Deng et al., 2017).
Several limitations have been noted in the selected studies; the treatment protocols applied were described in detail only in the NRCT of Deng et al. (2017); information about the nightly oral dose and/or on demand dose, the association with other devices/treatment, and the therapy duration was substantially missing (Liu et al., 2017). It is surprising to note that no study evaluated alternative ED management strategies, such as intracavernosal injections, penile implants, or psychological interventions, whose indications and efficacy cannot be directly supposed from the urological literature and applied to ED patients having undergone rectal surgery (Liu et al., 2017). This highlights a global lack of awareness on the extent of the ED problem and the need of multidisciplinary cares.
As suggested by Capogrosso et al. (2020) in a systematic review evaluating patients who underwent different pelvic surgical procedures including radical prostatectomy, radical cystectomy, and rectal surgery, sexual activity impairment requires a multi-step evaluation: preoperative assessment of sexual function using standardized questionnaires and taking into account the patient’s age (Schmidt, Bestmann, Kuchler, & Kremer, 2005) and comorbidities (Briganti et al., 2010); detailed report of the surgical procedure (e.g., approach, nerve sparing technique, surgeon’s experience); postoperative erectile evaluation (consistent with pre-surgical conditions); and penile rehabilitation. The evaluation of sexual function is challenging because of its multiple psychological implications. The most commonly used self-assessment tool is the IIEF (Cappelleri et al., 1999; Rosen, 1998; Rosen et al., 1997, 1999), which explores different domains, such as self-confidence, erection, penetration, ejaculation, and overall satisfaction. Its use was a selection criterion for the present systematic review, but it must be noted that the IIEF score was not specifically developed and validated for post-surgery ED (Capogrosso et al., 2020).
In the present study, we considered both benign and malignant indications for elective rectal resections: three studies enrolled patients with rectal cancer only (Deng et al., 2017; Lindsey et al., 2002, 2003; Nishizawa et al., 2011; Park et al., 2015), whereas two studies included both rectal cancer and IBD patients (Lindsey et al., 2002, 2003). Despite the different surgical procedures, both benign (17.3% of IBD cases [Lindsey et al., 2001]) and malignant (31%–76% of rectal cancers after TME [Celentano et al., 2017]) diseases are associated with postoperative sexual dysfunction in RCT-based meta-analyses. Differences related to the surgical indication and the surgical procedure are expected and future studies should investigate homogeneous sample of patients undergoing surgery for the same indication.
It has been demonstrated in a meta-analysis published in 2017 and based on retrospective and prospective studies (Ma et al., 2017) that there exists a significant association between chemoradiation therapy and postoperative nerve damage, with an ED relative risk of 2.25 (95% CI [1.51, 3.35] p < .01; Ma et al., 2017). In the present systematic review, four articles out of five (Deng et al., 2017; Lindsey et al., 2003; Nishizawa et al., 2011; Park et al., 2015) reported this information. Nevertheless, Park et al. (2015) did not identify a significant difference in IIEF-5 score after PDE-5i treatment in cancer patients who underwent radiotherapy compared to cancer patients who did not.
In case of rectal cancers, TME was also reported to be a potential risk factor for ED (Dulskas et al., 2016) regardless of the operative approach (Andersson et al., 2014). In this context, nerve-sparing surgery may play an important role to prevent sexual dysfunctions, as demonstrated in a recent neuro-histological comparative study published by Liu et al. (2019). However, there are still no conclusive data to support the superiority of laparoscopic surgery over open surgery in terms of postoperative functional outcomes (Celentano et al., 2017). Similarly, the ROLARR trial (Jayne et al., 2019) reported comparable sexual functions in patients with rectal cancer undergoing robotic and laparoscopic rectal resections. Concerning the present findings, none of the five included studies performed subgroup analyses to deem the impact of the surgical approach on ED occurrence and severity, or ED treatment efficacy, hampering any conclusion about the role of the surgical approach.
Consistent with the preoperative sexual function evaluation, postsurgical assessment must be prospectively recorded. Only those patients who are sexually active after surgery should be considered (Capogrosso et al., 2020). The ideal timing is at the end of the surgical recovery and before any eventual adjuvant therapy. Baseline sexual function evaluation was one of the main elements of heterogeneity among the selected articles; indeed, it was correctly reported in the two RCTs (Lindsey et al., 2002; Park et al., 2015), whereas it was reported with cut-off values (Lindsey et al., 2003), incompletely (Nishizawa et al., 2011), or not reported (Deng et al., 2017) in the other three studies. The literature lacks long-term studies (follow-up >12 months) that should monitor important treatment efficacy parameters, such as the response rate, dose adjustment, and patient’s satisfaction over time. The length of follow-up duration is indeed particularly relevant as post-surgical recovery can last up to 2 years (Lee et al., 2015). Spontaneous improvement is eventually possible without therapy, as two of the included studies reported (Deng et al., 2017; Lindsey et al., 2002; Park et al., 2015), but significantly better results are usually achieved with specific treatments.
The present systematic review has some limitations mainly related to the paucity of articles found in the literature on the specific topic of ED treatments after rectal surgery. The available studies deal with small samples of patients and investigate only PDE-5i treatment; they are heterogeneous in terms of study design, sexual function evaluation (different scores and timing), baseline patients’ characteristics, treatment protocols, and statistical reporting.
Future RCTs should be designed to better investigate the response rate and long-term efficacy of ED treatments in patients having undergone rectal surgery. These need to include homogeneous sample of patients as well as standardized sexual function evaluation performed preoperatively and consistently repeated postoperatively. Detailed description of the surgical procedure and the surgeon’s experience must be provided. Multiple arms of therapy should be implemented, including alternative treatments such as psychological interventions and pelvic floor rehabilitation. Ideally, the IIEF-5 score should be assessed during a 2-year lasting follow-up, and throughout subgroup analyses (e.g., benign/malignant diagnosis, TME/PME, adjuvant chemotherapy).
Conclusion
Despite the limited evidence available in the literature, the oral administration of PDE-5is appears to improve IIEF score, especially in the short term, in male patients diagnosed with ED after rectal surgery. Further studies are needed to explore long-term efficacy and alternative treatment strategies.
Supplemental Material
7_SUPPLEMENTAL_MATERIAL – Supplemental material for PDE-5i Management of Erectile Dysfunction After Rectal Surgery: A Systematic Review Focusing on Treatment Efficacy
Supplemental material, 7_SUPPLEMENTAL_MATERIAL for PDE-5i Management of Erectile Dysfunction After Rectal Surgery: A Systematic Review Focusing on Treatment Efficacy by Margherita Notarnicola, Valerio Celentano, Paschalis Gavriilidis, Bilal Abdi, Nassiba Beghdadi, Daniele Sommacale, Francesco Brunetti, Federico Coccolini and Nicola de’Angelis in American Journal of Men's Health
Footnotes
Statement of Authorship
Conception and Design: Nicola de’Angelis and Margherita Notarnicola
Acquisition of Data: Margherita Notarnicola, Bilal Abdi, and Nassiba Beghdadi
Analysis and Interpretation of Data: Nicola de’Angelis, Margherita Notarnicola, Valerio Celentano, and Pascal Gavriilidis
Drafting the Article: Nicola de’Angelis, Margherita Notarnicola, Federico Coccolini, Valerio Celentano, and Francesco Brunetti
Revising It for Intellectual Content: Nicola de’Angelis, Margherita Notarnicola, Federico Coccolini, Valerio Celentano, Pascal Gavriilidis, Francesco Brunetti, and Daniele Sommacale
Final Approval of the Completed Article: All authors
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
