Abstract
Prostate cancer remains the most common and second most deadly cancer diagnosed amongst U.S. men. External beam radiotherapy is a standard-of-care definitive treatment option for localized prostate cancer and historically constituted an 8–9-week treatment course comprised of 39–45 doses of 1.8–2.0 Gy each (conventional fractionation, CF). Based on the notion that prostate cancer may respond favorably to a higher dose per day, considerable research efforts have been focused on characterizing the safety and efficacy profile of shorter and shorter radiation courses. Ultrahypofractionation (UHF) involves condensing the radiation course into just 5–7 treatments of 6–8 Gy each. When utilizing modern techniques that allow the precise sculpting of a dose distribution that delivers high doses to the prostate and lower doses to surrounding normal tissues over five or fewer treatments, this treatment is called stereotactic body radiotherapy (SBRT). Two randomized trials (HYPO-RT-PC and PACE-B) have compared UHF to longer radiation courses. The former demonstrated that UHF and CF have similar long-term toxicity and efficacy, while the latter demonstrated that modern SBRT has equivalent short-term toxicity as well. A separate report from a consortium of studies data provides prospective, albeit nonrandomized, data supporting the longer-term safety and efficacy of SBRT specifically. Thus, mounting high-level evidence suggests that SBRT is an acceptable standard care of option for men with localized prostate cancer.
Prostate cancer currently remains the most common and second most deadly cancer diagnosed amongst U.S. men—with an incidence of 118.2 cases per 100,000 men and 19.5 deaths per 100,000 men—both figures age-standardized to the 2000 U.S. standard population (Cronin et al., 2018). For localized prostate cancer—defined as disease confined to prostate and has not spread to regional lymph nodes or distant organs—treatment options generally center around surgery or radiation therapy to treat the prostate cancer locally.
In accordance with risk stratification as defined by the National Comprehensive Cancer Network (NCCN), the definitive treatment options available for localized prostate cancer generally depend on a patient’s risk, as determined by prostate specific antigen (PSA) level, clinical T-stage, Gleason pattern, and number cores positive (National Comprehensive Cancer Network). Treatment options generally consist of surgery, brachytherapy, external beam radiotherapy (EBRT), active surveillance, or a combination of these treatments.
Surgery for prostate cancer generally consists of radical prostatectomy possibly with a pelvic lymph node dissection dependent on risk of lymph node metastasis. Brachytherapy, which involves placing radioactive sources placed inside the body to treat tumor, may be performed via low-dose rate (LDR) techniques involving placement of permanent radioactive seeds, or high-dose rate (HDR) techniques involving placement of temporary catheters which transport high-dose radioactive sources in and subsequently out of the prostate. EBRT, the focus of this article, involves beams of ionizing radiation externally directed toward the tumor.
Although historically, EBRT for prostate had involved a treatment course spanning approximately 8 to 9 weeks with five 1.8–2.0 Gy fractions per week (conventional fractionation CF), there has been a recent trend toward hypofractionation (HF) with modern regimens clustered around ~60–72 Gy delivered in fractions of ~2.4–3.4 Gy/fraction each (Morgan et al., 2018), and even more recently, toward ultrahypofractionation (UHF) with total ~36–43 Gy treatment condensed in five to seven treatments with ~6–8 Gy delivered per fraction. When UHF is employed in five or fewer treatments with sophisticated modern radiotherapy delivery platforms, it is called stereotactic body radiotherapy (SBRT).
Radiobiologic Basis for Hypofractionation
The rationale for hypofractionation in prostate cancer arises from favorable radiobiology to high doses of radiation per fraction. This phenomenon is best described by the α/β ratio, which is a loose proxy describing the curvature of a curve plotting cell survival as a function of dose of radiation per fraction (Figure 1). Cells with a low α/β ratio are more likely to survive small doses per fraction than cells with a high α/β ratio; the converse is true as well, with cells with low α/β ratios preferentially being killed at high doses per fraction. Prostate cancer cells are thought to have particularly low α/β ratios compared to other tumors (Brenner & Hall, 1999), thereby providing the basis for treating prostate cancer with large doses per fraction (thus in significantly fewer fractions).

Example of Cell-Survival Curve With Varying Response to Varying α/β Ratio.
Historical Perspective Toward Hypofractionation
Although there is literature dating back several decades of patients successfully receiving dosing regimens similar to modern “hypofractionation” schedules (Preston et al., 1986) and even “ultrahypofractionation” schedules (Collins et al., 1991), randomized data regarding hypofractionation compared to conventional fractionation have only been available for the last 15 years. Numerous trials have now demonstrated comparable efficacy of hypofractionated regimens to conventionally fractionated regimens including the large RTOG 0415 (Lee et al., 2016), CHHiP (Dearnaley et al., 2016), and PROFIT trials (Catton et al., 2017), with one trial even reporting superior efficacy (Hoffman et al., 2018). Late toxicity in the aforementioned trials was overall comparable, except in PROFIT (lower late Grade 2 GI toxicity in hypofractionated regimen) and in RTOG 0415 (higher late GI grade 2 and higher late GU grade 2 toxicity in hypofractionated regimen, though this trial had biologically unequal doses of radiation in its two arms). As of 2018, the American Society of Radiation Oncology, the American Society of Clinical Oncology, and the American Urological Association have endorsed hypofractionation as a standard of care option for all patients with localized prostate cancer (Morgan et al., 2018).
Background on Prostate SBRT
The ability to further increase dose per fraction into the “ultrahypofractionated” range (i.e., ≥5 Gy per fraction) has coincided with important advances in precision related to simulation, treatment planning, and treatment delivery.
First, when treating with radiation, there are uncertainties in targeting related to patient positioning, target visualization, and, importantly, target motion. The prostate in fact exhibits a significant amount of motion both between and during treatments (Dang et al., 2018). This requires that radiation oncologists must place a “margin” around the actual prostate to ensure that, even if there is some uncertainty in position at any given time, the prostate requires adequate dose. This increases the area of tissue receiving radiation, potentially worsening toxicity. Modern advances in image-guided radiotherapy (IGRT), including the use of implanted fiducial markers inside the prostate to help direct radiation treatment both between treatments and potentially during treatment, allow physicians to feel increasingly confident treating patients with smaller margins.
Second, given the proximity of bladder and rectum to the prostate, it is also crucial to note the technological innovations that have allowed for sparing of dose in these surrounding tissues. The utilization of intensity-modulated radiotherapy (IMRT) has allowed for improved dose conformity—that is, the radiation dose can be more adequately sculpted such that the intended target receives a high dose of radiation while the dose to nearby critical structures is minimized. Together, the advent of modern IGRT and IMRT have greatly increased physician confidence in delivering SBRT.
Clinical Utility of SBRT in Localized Prostate Cancer
Definitive Treatment for Low-and Intermediate-Risk Prostate Cancer: Review of Clinical Series
An earlier review (Kishan & King, 2017) compiled 21 studies representing 1812 patients which included regimens ranging from 27.4 Gy to 50 Gy in 4–5 fractions. While several of the studies only studied low-risk or intermediate-risk patients, 13 studies contained some portion of high-risk patients (ranging from 7.4% to 65.9%). Irrespective of risk groups, biochemical recurrence-free survival (BCRFS) rates at 5 years (where reported) ranged from 81% to 100%. Overall, considerable variation in toxicity was seen—with acute grade 2+ GU/GI toxicity ranging from 4% to 45% and from 0% to 30.8%, respectively, and late grade 2+ GU/GI toxicity ranging from 2.2% to 26.4% and from 1% to 19.8%, respectively.
Several multi-institutional cohorts have also documented long-term efficacy and toxicity outcomes in SBRT patients. As displayed in Table 1, patients within these cohorts received 33.5–40 Gy in 4–5 fractions. Freedom from biochemical recurrence at 5 years varied by risk profile: 95%–100% (low risk), 90.7%–100% (favorable intermediate risk), and 81.0%–93.1% (unfavorable intermediate risk). Regarding acute toxicity, patients were reported to have acute grade 2+ GU toxicity ranging from 4% to 26%, and acute grade 2+ GI toxicity ranging from 1% to 8.1%, respectively. Regarding late toxicity, patients were reported to have late grade 2+ GU toxicity ranging from 8% to 14.7% and late grade 2+ GI toxicity ranging from 2% to 3.4%, respectively. As of early 2020, the largest multi-institutional series with long-term follow-up included 2142 patients with a median follow-up of 6.9 years and reported 7-year biochemical recurrence cumulative incidences of 4.5%, 8.6%, and 14.9% for low-, favorable intermediate-, and unfavorable intermediate-risk disease (Kishan et al., 2019). Seven-year cumulative incidences of grade 3+ GU and GI toxicity were 2.4% and 0.4%, respectively. A comparison of long-term toxicity between SBRT and other fractionation schemes is shown in Figure 2.
Long-Term Efficacy and Toxicity From Prostate SBRT as Seen in Five Pooled Analyses and in One Meta-Analysis.

BCRFS = biochemical recurrence-free survival; HDR = high-dose rate.

A recent meta-analysis compiled data from 38 prospective studies totaling 6116 patients who received SBRT for localized prostate cancer (Jackson et al., 2019). This aggregate cohort was made up of patients with low risk (45%), intermediate risk (47%), and high risk (8%). Median follow-up was 39 months. Median dose per fraction and number of fractions were 7.4 Gy (range 5–10 Gy) and 5 (range 4–9), respectively. Fifteen percent of patients received ADT with SBRT. In studies where available, 5-year BCRFS was reported to be 95.3% (95% CI: 91.3%–97.5%)—further broken down by risk group as 96.7% for low risk (95% CI: 95.2%–97.8%) and 92.1% for intermediate risk (95% CI: 89.2%–94.3%). Notably, meta-regression revealed dose, % of cohort receiving ADT, and NCCN risk group all as significant factors in BCRFS. Serious adverse events were uncommon, with acute grade 3/4 GU toxicity reported in 0.5/0% patients and grade 3/4 GI toxicity reported in 0.06/0.03% of patients. Late grade 3+ GU toxicity was reported in 2.0% of patients with late grade 3+ GI toxicity in 1.1% of patients. Dose was reported to be associated with grade 3+ GU toxicity but not for grade 3+ GI toxicity; this may be biased by the inclusion of studies that delivered >8 Gy per day of radiation, which is considered nonstandard off protocol.
Patient-Reported Outcomes Comparing SBRT With Other Modalities
In a pooled multi-institutional analysis of patients undergoing moderate hypofractionation (<5 Gy/fraction, n = 378) or SBRT (5–10 Gy/fraction, n = 534) for prostate cancer, patient-reported outcomes were collected at 1 and 2 years of follow-up (Johnson et al., 2016). After adjusting for age and cancer characteristics, patients receiving hypofractionation were more likely to experience worsening urinary symptoms at 2 years.
A population-based cohort of prospectively enrolled, newly diagnosed prostate cancer patients pursuing SBRT (n = 104), EBRT (n = 189), or active surveillance (n = 387) has studied patient-reported outcomes in patients at 3, 12, and 24 months after treatment (Moon et al., 2019). Although SBRT had favorable quality-of-life (QoL) results when compared to EBRT and comparable results when compared to active surveillance at 2 years of follow-up, the study’s small sample size is noted as a potential limiting factor.
Randomized Trials Comparing SBRT With Other Modalities
In 2019, data were published from two randomized trials comparing patients receiving ultrahypofractionation (SBRT) compared to conventional fractionation.
PACE-B was a Phase 3, noninferiority trial which randomized men with low-risk or favorable intermediate-risk prostate adenocarcinoma to conventionally fractionated (78 Gy in 39 fractions over 7–8 weeks) or moderately hypofractionated radiotherapy (62 Gy in 20 fractions over 4 weeks) [n = 441], or ultrahypofractionated SBRT (36.25 Gy in five fractions over 1–2 weeks) [n = 433]. On report of acute toxicity findings, grade 2+ acute GU toxicity was seen in 27% of patients receiving CF/HF vs. 23% of patients receiving SBRT (p = .16) and grade 2+ acute GI toxicity was seen in 12% of patients receiving CF/HF vs. 10% of patients receiving SBRT (p = .38) (Brand et al., 2019). Importantly, patient-reported outcomes were not different between the two arms. Results regarding late toxicity and biochemical control are expected to be reported in the upcoming 3–4 years.
HYPO-RT-PC was a phase 3, noninferiority which randomized men up to 75 years of age with intermediate-to-high-risk prostate cancer to conventional fractionated radiotherapy (78 Gy in 39 fractions, 5 days per week for 8 weeks) [n = 591 in per-protocol population] or ultra-hypofractionation (42.7 Gy in seven fractions, 3 days per week for 2.5 weeks) [n = 598 in per-protocol population] (Widmark et al., 2019). The estimated failure-free survival at 5 years was 84% (95% CI: 80%–87%) in both treatment groups (HR = 1.002, 95% CI: 0.758−1.325; log-rank p = ·99). Regarding acute toxicity, Grade 2+ GU toxicity was reported in 28% of patients receiving UHF vs. 23% of patients receiving CF (p = .057), while Grade 2+ GI toxicity was reported in 8.5% of patients receiving UHF vs. 6% of patients receiving CF. Regarding late toxicity, Grade 2+ GU toxicity was reported in 18% of patients receiving UHF vs. 17% of patients receiving CF, while Grade 2+ GI toxicity was reported in 10% of patients receiving UHF vs. 10% of patients receiving CF. The ability to achieve an erection for intercourse decreased from approximately 70% at start of radiotherapy to 35% at 5 years, with no significant difference between the two treatment groups—a conclusion also drawn by earlier studies (Dess et al., 2018; Wiegner & King, 2010). Patient-reported outcomes analysis indicated that urinary and bowel toxicity was slightly more pronounced in the seven-fraction arm at the end of treatment, and that the difference in urinary toxicity might persist for up to 12 months. Importantly, however, this trial used older radiation techniques—only 20% of patients received IMRT plans, and the planning margins used were very large by modern standards. Indeed, the acute rates of acute grade 2+ GU toxicity, for instance, are higher in HYPO-RT-PC than PACE-B (28% vs. 23%), and the late grade 3+ toxicity rates at 5-years are nearly 2–3 times higher in HYPO-RT-PC than in the Kishan et al. consortium study (4.2% vs. 1.8% for GU, and 1.5% vs. 0.4% for GI). Thus, the HYPO-RT-PC trial is in many ways a test of biology more than a test of modern SBRT.
Value of SBRT
Given the rapid rise in expected expenditures on prostate cancer care in the United States (Mariotto et al., 2011)—$11.85 billion in 2010 to $16.34 billion by 2020 per base-case estimates from 2011 NCI study—it has become increasingly important for the U.S. healthcare system to identify opportunities to preserve high-level care while improving value in oncology. In this setting, over the last decade, there has been growing interest in turning to SBRT as a source of value compared to conventionally fractionated techniques.
Using a time-driven activity-based costing approach, SBRT in low-risk prostate cancer has been reported to reduce costs to a provider by 50% when compared with a course of IMRT (Laviana et al., 2016). These costs account for personnel, space/equipment, and materials utilized during each phase of care for both treatment modalities. From the perspective of a health system, several Markov-based cost-effectiveness analyses have identified SBRT to be cost-effective to IMRT when treating prostate cancer (Hodges et al., 2012; Parthan et al., 2012; Sher et al., 2014).
Future Directions
Given its efficacy, toxicity profile, value, and convenience, SBRT has unsurprisingly continued to increase in utilization (Halpern et al., 2016). Although NCCN guidelines now list SBRT as a treatment option for very low, low, intermediate, high, and very high-risk disease, clinical trials are currently exploring multiple other indications (Table 2), including recurrent disease after prior RT and salvage after prior surgery. Additional trials are also studying the use of SBRT in combination with other modalities, comparison versus other modalities, alternate dosing/fractionation schema, and novel technological approaches to further limit toxicity.
Ongoing Clinical Trials Currently Studying SBRT in Prostate Cancer.
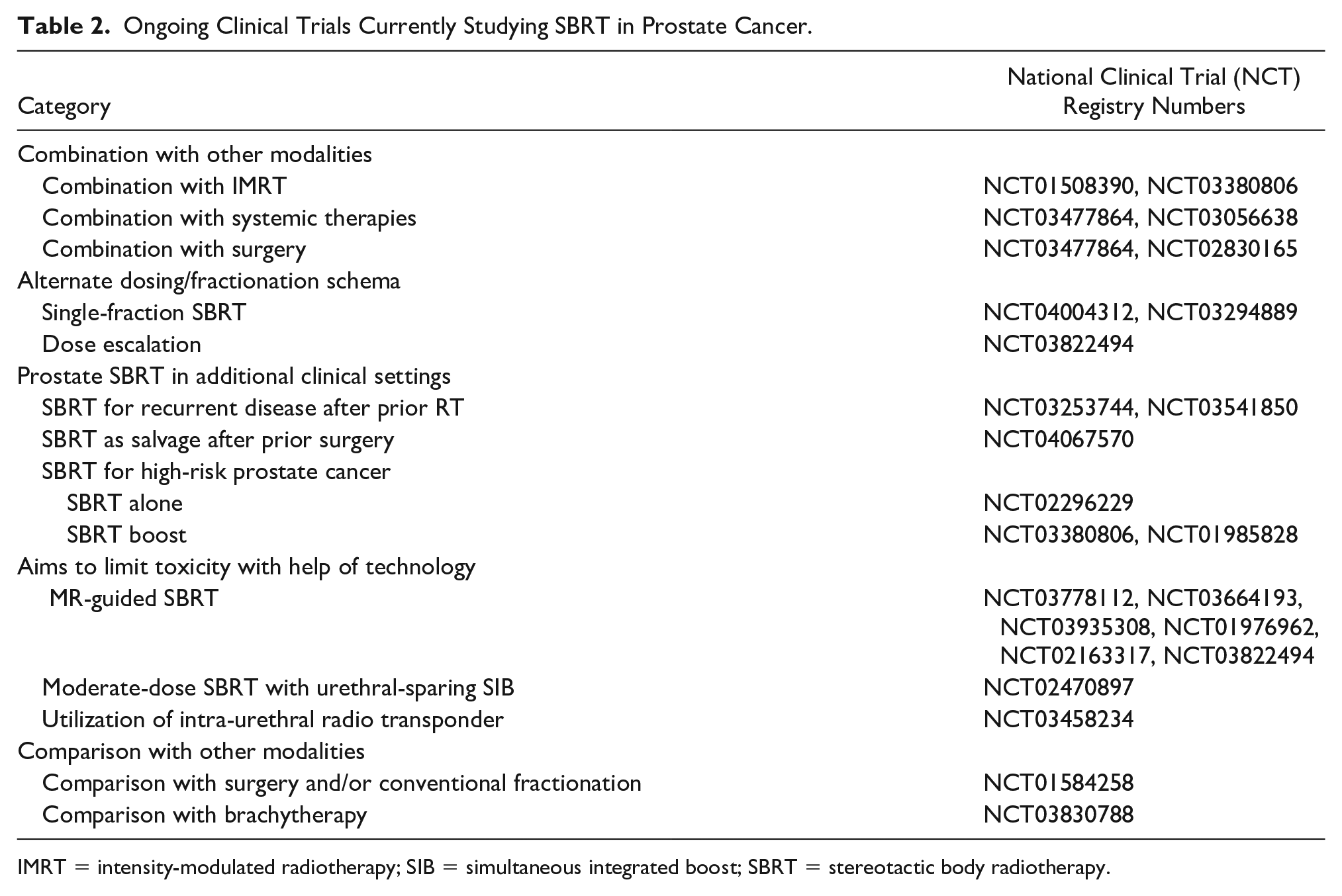
IMRT = intensity-modulated radiotherapy; SIB = simultaneous integrated boost; SBRT = stereotactic body radiotherapy.
Footnotes
Authors’ Contributions
N.R.P. and A.U.K. contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Kishan has received honoraria and consulting fees from Varian Medical Systems. He has received honoraria and research funding from ViewRay. He has received consulting fees from Intelligent Automation, Inc. and has served on an advisory board for Janssen.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
