Abstract
While life expectancy continues to increase, aging can bring several distinct endocrine and psychosocial changes. The study aimed to investigate the interplay between biopsychosocial factors of healthy aging in specifically healthy aging men. Ninety-seven healthy aging men were investigated at two time points spanning 4 years. Participants completed questionnaires measuring several psychosocial dimensions and gave saliva samples for hormone quantification during a laboratory appointment. The study applied a random intercept mixed-model approach. Age-related changes were found in most endocrine markers (cortisol, testosterone, dehydroepiandrosterone-sulfate, and progesterone), except for estradiol. Psychosocial measures remained stable, except for increased social support. Further, changes in endocrine and psychosocial measures were independent of each other. The results suggest that in healthy aging men, age-related endocrine changes occur, but do not necessarily determine a change in psychosocial measures. Potentially, preventive interventions can be derived from these results.
Advancements in the prevention and treatment of diseases as well as novel technologies in biomedicine have led modern societies to experience an unprecedented increase in life expectancy (United Nations, 2009). As a consequence, people are spending a substantial part of their lives in older age. There are currently 962 million individuals aged 60 years and older worldwide, and global projections indicate that this figure will reach almost 2.1 billion by 2050 (United Nations, 2017). While a longer life is a precious gift, aging comes with distinct changes in biopsychosocial markers, leading to multifaceted processes of adaptation and increased morbidity and mortality. Biological aging is therefore considered as a major risk factor for chronic diseases and disabling conditions, causing tremendous societal and economic burden (Barzilai et al., 2018). Although some influential pioneering studies have indicated the relevance of psychosocial factors for age-related health changes (Hawkley et al., 2006; Johnson et al., 1989; Weng et al., 2000), the role played by psychosocial factors regarding the age-related increase in morbidity and mortality has not yet been sufficiently recognized. Prolonging the health span and reducing health-care costs are emerging as central goals for modern societies, which can only be achieved by investigating the underlying biopsychosocial aging processes and identifying novel options to improve healthy aging (Beard et al., 2016).
Healthy aging can be defined as “the process of developing and maintaining the functional ability that enables well-being in older age” (World Health Organization, 2015). Aging is associated with distinct changes in hormone secretion. Throughout a man’s lifespan, endocrine systems undergo specific changes: Activity of the hypothalamic-pituitary-gonadal (HPG) axis decreases with age, whereas the hypothalamic-pituitary-adrenal (HPA) axis becomes hyperactive with higher age (Ebner et al., 2014). At around the age of 40 years, the function of the Leydig cells in the testes begins to decrease, diminishing the levels of sex steroids (Fiacco et al., 2018). Most prominently, testosterone (T) decreases, which can potentially lead to a deterioration of physical and mental health (Ford et al., 2016). An age-related T decrease has also been observed in specifically healthy aging men (Harman et al., 2001). While Sartorius et al. (2012) assume that very good health may prevent aging men from sex steroid decline, there is increasing evidence to support a continuous but decelerated age-related decline in T in healthy men (Feldman et al., 2002; Walther et al., 2016). Dehydroepiandrosterone (DHEA) and its sulfate (DHEAs) have also been identified to decrease in aging men due to the attrition of DHEA(s)-producing cells in the zona reticularis of the adrenal cortex (Ohlsson et al., 2010; Parker, et al., 1997). Age trends regarding progesterone (P) and estradiol (E2), however, are conflicting, with studies reporting that these hormones remain stable (Oettel & Mukhopadhyay, 2004; Sartorius et al., 2012), increase (Jasuja et al., 2013), or slightly decline (Lewerin et al., 2014; Zitzmann et al., 2005). Similarly to the case for androgens, good self-reported health has been suggested to buffer the potential age-related changes in P and E2 (Sartorius et al., 2012; Walther et al., 2016). Contrary to the suggested decline in sex steroids, cortisol (C) increases with age (Karlamangla et al., 2013). This is due on the one hand to changes in the number of mineralocorticoid and glucocorticoid receptors and a change in sensitivity of the negative feedback mechanisms of the HPA axis (Gupta & Morley, 2014) and on the other hand to an age-related increased number of corticotropin-releasing hormone (CRH) neurons and subsequently in CRH secretion, further increasing cortisol output of the HPA axis (Zhou & Swaab, 1999). Age-related changes in the endocrine system also harbor the risk of physical or psychological pathologies, if mechanisms of adaptation fail to buffer these changes.
Parallel to endocrine changes, psychosocial adaptations can also be observed during aging. Subjective health seems to be an important indicator of healthy aging (Han et al., 2015). Symptoms of aging in males, such as decreased potency, muscular strength, or energy, are exhibited naturally with higher age (Jakiel et al., 2015). Large population-based cohort studies have also demonstrated a decline in general mental health with age (Norman et al., 2009; Payne et al., 2014). This association is potentially strongly influenced by physical illness and its consequences (Lorem et al., 2017). Sexual function, as part of overall quality of life, decreases in aging men (Giovanni Corona et al., 2013; Walther et al., 2017). For instance, 61% of 40–69-year-old males have experienced erectile dysfunction at least once in their life, whereas this rate increases to 77% among ≥ 70-year-olds (Wagle et al., 2012). Psychological factors such as self-esteem and social support also undergo changes with age. With regard to self-esteem, the findings are conflicting, with some studies reporting that males’ self-esteem increases until their sixties and subsequently declines (Orth et al., 2015; Robins et al., 2002) and other studies indicating a relative stability until close to death (Wagner et al., 2015). Social support is an important resource associated with health, with findings suggesting that age-related changes in social networks occur voluntarily and that social support increases with age (Charles & Carstensen, 2009). Aging can also foster loneliness (Hawkley & Kocherginsky, 2018), which is associated with several negative health outcomes (Shankar et al., 2017; Tian, 2016) and increased mortality (Harris et al., 2015). Taken together, subjective good health is associated with healthy aging, aging symptoms, such as decreased potency, increase and sexual function decreases with higher age. Results on self-esteem are conflicting, showing either a decline or stability, while social support can be beneficial for health, and loneliness—the opposite of social support—can foster negative effects on health.
Endocrine parameters are emerging as promising markers for the continuous monitoring of healthy aging since changes in androgens and estrogens were reported to be predictive for all-cause and cause-specific mortality in men (Hsu et al., 2016). Psychosocial measures, on the other hand, have also been identified to predict the aging process (e.g., Dainese et al., 2011). Isolated views on either psychosocial or endocrine factors alone neglect the complex interactions between the two domains and potentially impede the investigation of healthy aging as a whole. An integrative biopsychosocial approach as first described by George and Engel (1980) could be beneficial, especially within the employed multidimensional framework of healthy aging.
According to the biopsychosocial model, endocrine markers and psychosocial measures potentially influence each other. For symptoms of aging in men, the literature identified more symptoms of aging in the case of lower T secretion (Handelsman et al., 2015). Low T secretion is also associated with a higher risk of age-related illnesses such as dementia (Ford et al., 2018). A cross-sectional study with specifically healthy aging men failed to link subjective health to T secretion (Dunbar et al., 2001). Lower T secretion can further elevate symptoms of sexual dysfunction in men (Corona et al., 2009) or moderate the association between age and sexual dysfunction (Walther et al., 2017). Findings regarding endocrine associations with psychosocial measures such as self-esteem and social support are scarce. A lack of social support has previously been associated with flatter diurnal C slopes (Friedman et al., 2012; Gaffey et al., 2016), while self-esteem has either been associated with increased morning C (Zorrilla et al., 1995) or has shown no direct association with C (Pruessner et al., 1999), although the latter study investigated stress-related C secretion while the former investigated basal C secretion. An influential review revealed that various further studies did not identify links between endocrine secretion and psychosocial measures (Campbell & Ehlert, 2012), thus underlining the need to further investigate these associations and highlighting the importance of assessing changes in both psychosocial measures and endocrine markers.
To the knowledge of the authors, no previous study has investigated aging in specifically healthy men employing a wide range of biological, psychological, and social measures in a longitudinal design. The study aims to investigate how endocrine markers and psychosocial measures develop in the distinct population of healthy aging men over a time span of 4 years. The first hypothesis is that T and DHEAs would decrease with age, while C would increase, and E2 and P would remain stable. The second hypothesis is that health-related measures would decline, while self-esteem and social support would remain stable. The last hypothesis expects to find a buffering effect of self-esteem and social support on endocrine markers and that endocrine secretion would influence the development of all psychosocial measures since the literature suggests that endocrine development and psychosocial changes influence one another.
Materials and Methods
Participants
A total of 98 healthy men aged 40 years and over from the Men’s Health 40+ study (Walther et al., 2016) were investigated using a longitudinal design. At the beginning of the investigation, participants were aged between 40 and 75 years. An inclusion criterion for both time points of the study was a subjective health rating of “good”, “very good,” or “excellent” to enable a specific investigation of healthy individuals. This was assessed through the first question of the Short Form 36 Health Survey (SF-36, Bullinger et al., 1995), “How would you describe your current health condition?”, with response options ranging from “very bad” to “excellent”. Participants with self-reported acute and chronic medical conditions, psychiatric disorders, psychopharmacological treatment, or psychotherapy were screened out and all participants had to be fluent in German. The study was approved by the ethics committee of the Canton of Zurich in Switzerland. All participants accepted the terms of the study and gave their written informed consent.
Procedure
The study comprised a baseline measurement and a follow-up conducted 4 years after the initial investigation. At both time points, participants first underwent a screening to test whether all inclusion criteria were met. After being accepted for the study, participants subsequently completed three psychometric test batteries (in German), which consisted of various validated self-rating questionnaires. Each test battery required approximately 60 min to complete. Participants underwent a biological examination in the laboratory, during which various physiological (e.g., body composition) and endocrine parameters were obtained by trained study personnel between 8:00 and 8:15 am. Participants were instructed not to eat, drink, or consume cigarettes for 2 hr before the laboratory examination. They were asked to refrain from physical or sexual activity for 24 hr before the examination and alcohol or caffeine consumption for 48 hr before sampling (Stalder et al., 2016). Contaminated saliva samples (e.g., through prior coffee consumption or eating) produced missing values and could not be analyzed. In general, saliva samples, if collected under controlled conditions, are a reliable, noninvasive alternative to serum samples and have shown to be applicable for the analysis of steroid hormones (Gröschl, 2017). Detailed descriptions of the biological examination are described elsewhere (Walther et al., 2016).
Psychosocial Measures
Health-Related Quality of Life
To assess health-related quality of life, the Short Form 36 Health Survey (SF-36; Bullinger et al., 1995) and the more specific Aging Males’ Symptoms rating scale (AMS; Zimmermann et al., 1999) were used. The SF-36 is a 36-item self-report questionnaire which provides sum scores for eight subscales and two general scales. The general scale “physical health” was used in the present study. It has been used in multiple studies in a variety of fields over the last decades and is a well-established instrument for the assessment of health-related quality of life. Internal consistency for the present sample was α = .63.
The Aging Males’ Symptoms rating scale consists of a general sum score, which was used in the present study, and three distinct subscales. Participants rated the 17 symptom-related items in terms of severity from “no” to “very severe”. This questionnaire was specifically designed to assess male-specific symptoms of aging and thus to measure health-related quality of life for this population. It is a widely used and well-documented instrument within male-specific aging studies. Cronbach’s alpha for the current sample was α = .84.
Sexual Function
The German version of the International Index of Erectile Function (IIEF; Rosen, 1997; Wiltink et al., 2003) was used to measure erectile function with 15 items. The proposed German scoring was used for the present analyses. The IIEF consists of sum scores, including two distinct subscales: sexual function, which was used here, and sexual desire (Wiltink et al., 2003). The IIEF is a self-report measurement tool which is well-validated and widely used in clinical as well as epidemiological studies. Cronbach’s alpha for this sample was α = .64.
Mental Health
The Brief Symptom Inventory 18 (BSI-18) is a well-established 18-item self-report questionnaire measuring mental health on three subscales and a general sum scale, which was used in the present study (Spitzer et al., 2011). The BSI-18 is a time-efficient measurement tool which is widely used. The BSI-18 is a shortened version of the Brief Symptom Inventory (Franke & Derogatis, 2000). The questionnaire showed good internal consistency (α = .83) in the present sample.
Self-Esteem
The Multidimensional Self-Esteem Scale (MSWS; Schütz & Sellin, 2006) was used to assess self-esteem as a multidimensional construct. It investigates self-esteem as a trait. The questionnaire consists of 32 items which form a hierarchical structure of subscales, with two scales representing distinct factors for general self-esteem (used in the present study) and body-related self-esteem. Cronbach’s alpha for the current sample was α = .89.
Social Support
The Berlin Social Support Scale (BSS; Schwarzer & Schulz, 2003) is a well-established instrument to measure social support. The questionnaire consists of four subscales and a general score, which was used in this study. Internal consistency was α = .90 in the present sample.
Hormone Analyses
Saliva samples were obtained between 8:00 and 8:15 am under standardized laboratory conditions using SaliCaps (SaliCaps, IBL International GmbH, Hamburg, Germany). Samples were immediately stored at −20°C until required for biochemical analysis. Enzyme-linked immunoassays were used to analyze C, DHEAs, and P, while T and E2 were analyzed using luminescence immunoassay (Chiappin et al., 2007; Goncharov et al., 2006). All hormones were analyzed at the Biochemical Laboratory at the Institute of Psychology of the University of Zurich. The intra- and interassay variation was less than 10%, while sensitivity was 0.003 μg/dl for C, 1.8 pg/ml for T, 1.5pg/ml for DHEAs, 0.3 pg/ml for E2, and 2.6 pg/ml for P.
Statistical Analysis
Bootstrapped zero-order and partial Pearson correlations were conducted to examine direct cross-sectional and longitudinal associations between baseline and follow-up measures (detailed information is provided in the supplemental material). Partial correlations were controlled for physical activity, income, educational level, fat mass, and age to control for any potential bias. To examine changes in endocrine and psychosocial factors over time, a linear mixed-model approach with random intercepts was used, which enables a more specific analysis of the longitudinal effects than simple correlation and regression analyses. Visual inspection for normality of the residuals was performed for every single model; if normality of the residuals was violated, the respective variable was log-transformed (Sollberger & Ehlert, 2016). Linear mixed models were calculated in R (Version 3.4.3) using the package “lme4” (Bates et al., 2015). The analysis was conducted in two subsequent steps: first, a more general analysis of the change over time and second the specific moderation analyses. Within the first step, the dependent variables were the respective hormones or psychosocial measures, with the time variable as an independent fixed effect variable, displaying intraindividual changes between the two measurement time points. Within the current set of hypotheses, random effects (i.e., whether there are differences between the participants) are no priority in the current set of hypotheses and thus will not be reported or discussed in detail. All calculations were controlled for physical activity, educational level, and income. Endocrine and health-related measures were further controlled for fat mass. The aforementioned moderation analyses were conducted with not only the baseline values but the longitudinal data for both the dependent variables and the moderators, which enabled us to, additionally, investigate the changes in these variables. The model for the moderation analyses consisted of the respective dependent variable and as predictors, the time*moderator term as well as the covariates were included. For all mixed-model analyses, participants’ endocrine sampling adherence was included in the models, but none of the parameters were significant predictors and did not change the general results. To apply the principle of parsimony, they were therefore not included in the final analyses.
Moderation analysis was conducted for the influence of social support and self-esteem on endocrine markers and for the influence of all endocrine markers separately on the psychosocial markers. All calculations, again, were controlled for physical activity, educational level, and income. Endocrine and health-related measures were further controlled for fat mass. The Akaike information criterion (AIC) was used to assess model quality for the change over time, as well as the moderation analyses. For all calculations within this study, the Benjamini–Hochberg procedure was applied to the p values to control the false discovery rate (Benjamini & Hochberg, 1995).
Results
Descriptive Data
Out of 271 participants from the baseline assessment, 130 (48%) completed the screening and 98 (36%) attended the follow-up assessment (Figure 1). The participants were initially informed that they would be participating in a cross-sectional study. Three years after the baseline assessment, participants were asked whether they would like to participate in a follow-up assessment. Twenty-two (8%) of the 271 participants had an invalid e-mail address and 60 (22%) did not respond to repeated e-mails. Out of the 130 participants who completed the screening, 15 (12%) were no longer eligible to participate due to physical illnesses and 17 (13%) were no longer interested in participating. Finally, 98 (75%) participants participated in the follow-up assessment, 97 (99%) of whom completed the laboratory assessment.

Flow Chart of Participant Participation
Descriptive data of these 97 participants are presented in Table 1. Participants’ mean age was 61.26 years (SD = 10.02) at baseline and 65.18 (SD = 9.98) at follow-up. Most participants were in a relationship (N = 8, 8.25%) or married (N = 70, 72.16%) throughout the complete study. The largest proportion of participants had a university degree and an annual income between 50,000 and 150,000 Swiss Francs. A total of 84.54% (N = 82) reported being nonsmokers at baseline and 86.46% (N = 83) at follow-up. Around 60% (N = 59) of the participants did not take any medication at baseline, while 53% (N = 51) were medication-free at follow-up. Antihypertensives were the most commonly taken medication type (46% for both time points, N = 23/28), followed by the category “Other”, which mostly comprised cholesterol medication and bowel-regulating medications. The largest proportion of participants (Baseline: N = 40,41%; Follow-up: N = 51, 53%) reported undertaking 4–6 hr of physical activity per weeks.
Sample Characteristics.

Participants could indicate more than one medication.
Note. N = number of participants; M = mean; Freq. = frequency; SD = standard deviation; % = percent; C = cortisol; T = testosterone; DHEAs = dehydroepiandrosterone-sulfate; E2 = estradiol; P = progesterone; AMS = Aging Males’ Symptoms scale; SF-36 = Short-Form 36; BSI = brief symptom inventory; IIEF = International Index of Erectile Function; MSWS = Multidimensional Self-Esteem Scale; BSSS = Berlin Social Support Scale; CHF = Swiss Francs.
Analyses comparing differences between the participants who participated at both time points and those who only participated at the first time point showed that the former were older and healthier overall, thus supporting the aim to investigate participants who were able to maintain their health. There were no significant differences in endocrine measures between participants of T1+T2 and only T1, with the exception of P, which was significantly lower for participants who did not return. Further analyses comparing eligible and ineligible participants who returned at the second time point showed that the eligible participants were indeed healthier, although they reported higher values with regard to pain. Overall, the aim to investigate healthy participants was supported (detailed information in the supplemental materials). Correlations between all parameters can be found in the supplemental materials.
Changes Over Time
Changes over time for endocrine markers and psychosocial measures are displayed in Figure 2. Linear mixed-model analyses with random intercepts yielded significant changes over time (Table 2). Longitudinal effects for endocrine secretion showed significant decreases in T (b = −0.3, p < .001, −3.07% per year) and DHEAs (b = −0.47, p < .001, −12.18% per year). Furthermore, C (b = 0.44, p =.01, 7.36% per year) and P (b = 02, p = .001, 5.96% per year) increased, while E2 (b = 0.07, p = .623, 2.51% per year) did not change significantly. It was decided to interpret the change in C despite a slightly worse model fit because the covariates were entered in a theory-based manner. After excluding educational level, which was not a significant predictor of C, the model fit improved.

Changes Over Time for Endocrine Markers and Psychosocial Measures. (A) Shows the changes over time for endocrine measures. DHEAs = dehydroepiandrosterone-sulfate; E2 = estradiol. Changes on percent scale with higher values are shown as 100%. (B) Shows the change over time of psychosocial measures. Scale shows percent of maximum achievable value on respective questionnaire scale. For mean and SD see Table 2.
Linear Mixed Model Results Over Time.
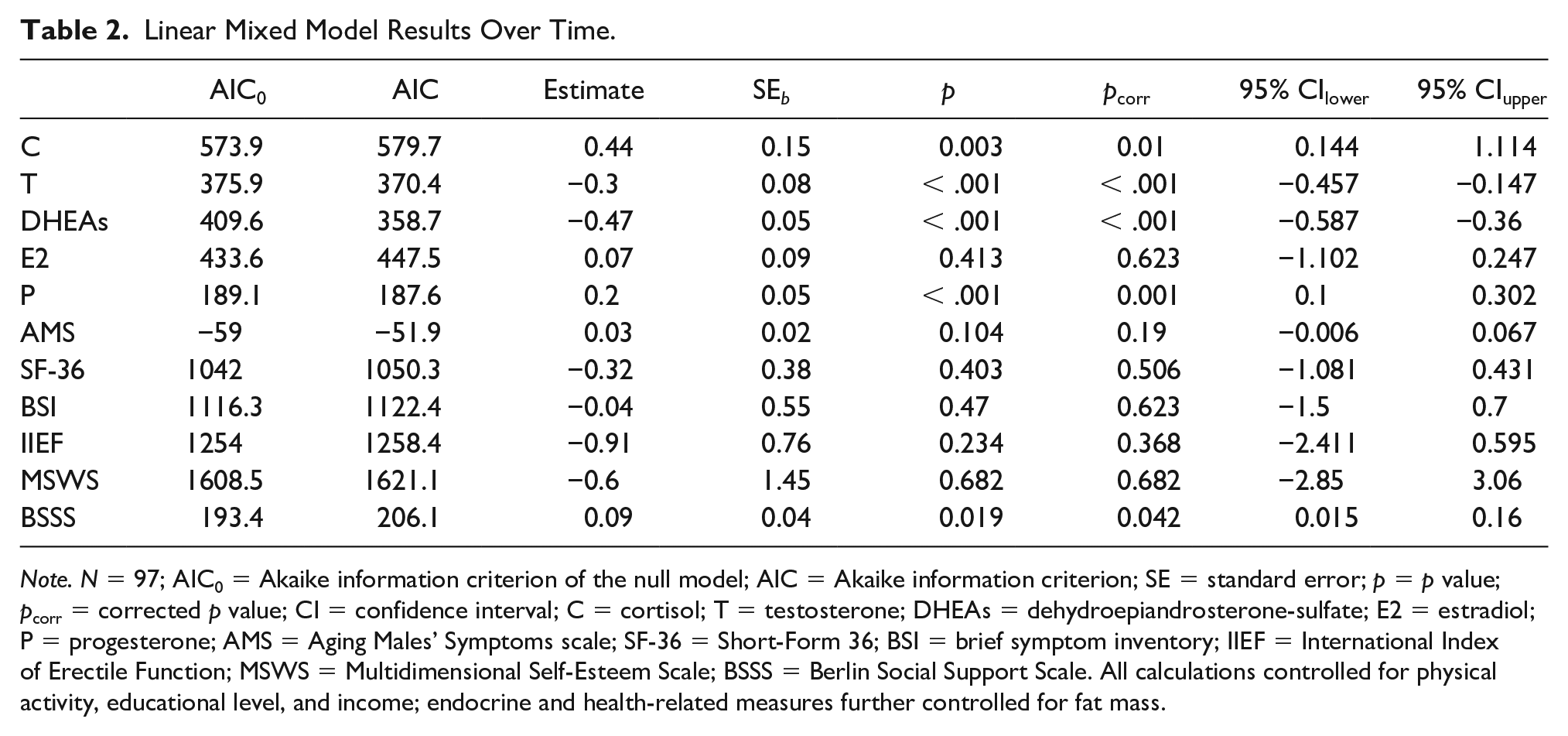
Note. N = 97; AIC0 = Akaike information criterion of the null model; AIC = Akaike information criterion; SE = standard error; p = p value; pcorr = corrected p value; CI = confidence interval; C = cortisol; T = testosterone; DHEAs = dehydroepiandrosterone-sulfate; E2 = estradiol; P = progesterone; AMS = Aging Males’ Symptoms scale; SF-36 = Short-Form 36; BSI = brief symptom inventory; IIEF = International Index of Erectile Function; MSWS = Multidimensional Self-Esteem Scale; BSSS = Berlin Social Support Scale. All calculations controlled for physical activity, educational level, and income; endocrine and health-related measures further controlled for fat mass.
The results for the psychosocial measures show no significant changes in male aging symptoms (b = 0.03, p = .190), physical health (b = −0.32, p = .506), mental health (b = −0.04, p = .623), sexual function (b = −0.91, p = .368), and self-esteem (b = −0.6, p = .682). Social support significantly increased over time (b = 0.09, p = .042). Similarly to C, the change in social support was interpreted despite slightly worse model fit, since covariates were selected in a theory-based manner.
Moderation Analyses
Results of the moderation analyses showed that by trend, social support moderated the age-related change in C (b = 0.544, p = .087) and in DHEAs (b = 0.010, p = .079). As a further trend, C (b = 0.198, p = .055) and T (b = 1.410, p = .054) moderated the change in sexual function over time. Social support was found to significantly moderate the change in C over time (b = 0.013, p = .012), insofar as higher social support was associated with lower C secretion. This finding did not remain significant after adjusting for the false discovery rate (Table 3). In conclusion, no significant moderation effects were identified.
Linear Mixed Model Results for All Moderation Analyses.

Note. N = 97; AIC0 = Akaike information criterion of the null model; AIC = Akaike information criterion; SE = standard error; p = p value; pcorr = corrected p value; CI = confidence interval; C = cortisol; T = testosterone; DHEAs = dehydroepiandrosterone-sulfate; E2 = estradiol; P = progesterone; SF36 = Short-Form 36; AMS = Aging Males’ Symptoms scale; BSI = brief symptom inventory; IIEF = International Index of Erectile Function; MSWS = Multidimensional Self-esteem Scale; BSSS = Berlin Social Support Scale. All calculations controlled for physical activity, educational level, and income; endocrine and health-related measures further controlled for fat mass.
Discussion
In the present study examining 97 healthy men at baseline and 4-year follow-up, an age-related decrease in T and DHEAs and an increase in P and C was identified. No significant change in E2 was detected within this period of time. Scores on all health-related psychosocial measures such as self-reported health, mental health, self-esteem, and sexual function remained stable, while social support increased. No moderation effects of longitudinal self-esteem scores and social support on endocrine changes were found, and there were no moderation effects of endocrine markers on psychosocial measures. Therefore, in the healthy aging men examined, age-related changes in endocrine markers did not match changes in psychosocial measures. These findings reveal that despite age-related changes in hormones, it is still possible to maintain or even increase a high quality of life, mental health, sexual function, self-esteem, and social support.
The results for endocrine measures are mostly in line with the literature. An increase in C and a decrease in T and DHEAs, as previously reported by other studies (Ford et al., 2016; Gupta & Morley, 2014) was identified. Findings for E2 are also in accordance with previous studies, which showed either stability or a slight decrease, as described in a detailed review by Fiacco et al. (2018). However, the increase in P in the present study is in contrast to previous research demonstrating either stability or a decline with age (Oettel & Mukhopadhyay, 2004). As P is synthesized not only in the Leydig cells, which decrease in function with age, but also in the adrenal glands, which are also crucial for male secretion of P (Oettel & Mukhopadhyay, 2004; Schumacher et al., 2014), the adrenal glands might attempt to compensate for the reduced Leydig cell function by producing more P. This could explain the increase in P despite the decline in T, which is also synthesized in the testes.
Interestingly, health-related measures remained stable in the present study, which is yet to be discussed. Various studies reported that different aspects of quality of life such as mental health (e.g., Payne et al., 2014) or sexual function (Corona et al., 2013) decreased with age while symptoms of aging increased (Jakiel et al., 2015). Previous studies predominantly investigated general or clinical populations rather than specifically healthy individuals as in the present study. The current findings therefore add valuable insights into the healthy aging process. Moreover, the present results give rise to the question of whether the decrease in psychosocial parameters is natural or whether it is due to physical illnesses. The latter was also proposed by Lorem et al. (2017), and is further supported by the stability of subjective physical health in this sample of healthy aging men.
The finding of maintained self-esteem in healthy aging men is partially in line with the literature, which reported that self-esteem increases until the age of around 60 and then shows a steady but slow decline until old age (Orth et al., 2010, 2012). Another recent study reported that self-esteem remains stable until very old age and only declines shortly before death (Wagner et al., 2015). An explanation for the current findings of this study might lie in the specific sampling of healthy participants, because as Wagner et al. (2015) stated, low subjective health and its potential constraints are important factors that negatively influence self-esteem. Furthermore, a high educational level increases self-esteem (Orth et al., 2012), which is relevant since 44.33% of this sample had a university degree.
This healthy aging sample reported increasing levels of social support over time. Due to the lack of studies specifically investigating subjective social support over the lifespan, and given the heterogeneity of methods used to assess social support, integrating the findings into the existing literature is challenging. As older individuals play an active role in designing their social networks (Charles & Carstensen, 2009), the significant increase found in the present study might indicate that healthy aging men indeed actively manage their social networks. It is crucial to mention that the size of the social networks themselves was not assessed, but rather measured subjective, self-rated social support using a validated questionnaire. It cannot reliably be concluded that the social network itself changed, although a change in the subjective perception of social support can be concluded. Scholz et al. (2012) further reported that for older individuals, social support was associated with higher (emotional) well-being.
The current study showed that age-related endocrine changes and changes in psychosocial measures are independent in healthy aging men. Three potential explanations for this can be suggested: First, most previous studies investigated general populations or participants with illnesses. In this respect, it is important to discuss by way of example that the decline in T did not have any effects on mental health in this study, in contrast to a previous study in a randomly selected sample and therefore also including individuals with disorders or potentially in a premorbid state (Ford et al., 2016). This premorbid state might have led to biological changes prior to the manifestation of the respective illness, which were not present in this healthy sample. Another study with subjectively healthy aging men further failed to link T to subjective health, although the analyses were cross-sectional (Dunbar et al., 2001). Second, there may be a dissociation between physiological and psychological measures in healthy aging men, as discussed for psychoendocrine research in general by Campbell and Ehlert (2012). Third, the associations might have been influenced by a potential confounding variable that was not assessed, as also proposed by Campbell and Ehlert (2012). Nevertheless, associations between endocrine markers and social support (Friedman et al., 2012) have previously been observed in the general population, whereas the literature on the associations between self-esteem and endocrine markers remains conflicting (Pruessner et al., 1999; Zorrilla et al., 1995).
Some limitations of the present study need to be noted, the strongest of which is the aforementioned high dropout rate. This was mostly due to the fact that information about the follow-up was only provided 3 years after the baseline. It is further important to note that compared to other European countries, residents of Switzerland have a rather high income and level of education (BfS, 2015), which limits the ability to generalize the findings to other countries. Income and educational level were added as covariates in all calculations. The study design did not include an unhealthy group, rendering direct comparisons impossible and thus limiting potential conclusions beyond healthy aging populations. To increase the validity and reliability, it can be beneficial to employ a design with more than one hormone sample for each time point. Reliable and valid results can also be obtained with single or two-time measurements while controlling for sampling time and presample behavior, as also applied within the current study (Barrett-Connor et al., 1999; Clifton et al., 2016; Feldman et al., 2002). Similarly, the assessment of more than two time points within the 4 years of the study duration, or even expanding the study with a study design of more than 4 years would increase the potential conclusions that could be derived. Finally, for the questionnaires on sexual function and subjective health-related quality of life, internal consistency was rather low, possibly because most questionnaires were predominantly developed to measure dysfunctions in nonhealthy populations. Accordingly, they may have been less applicable to healthy participants, therefore decreasing the reliability for this population.
Nevertheless, the present study also entailed several strengths. First, a sample which was able maintain their health between the two time points was investigated. Second, the use of longitudinal data enabled a more detailed investigation of age-related changes compared to cross-sectional data. Further strengths of the study include the use of validated questionnaires, strict inclusion criteria, and standardized procedures for endocrine measurements and for the overall study procedure.
Future studies should incorporate more data (e.g., experience sampling and more time points) and include a nonhealthy group within a larger overall sample. To enable reliable use in healthy aging participants, it would be beneficial to develop health-focused rather than illness-focused questionnaires.
Conclusion
In this sample of healthy aging men, levels of psychosocial measures were maintained despite age-related endocrine changes. These age-related endocrine changes were not associated with changes in psychosocial measures or vice versa. While C and P increased, DHEAs and T decreased, and E2 remained stable. In terms of psychosocial measures, levels of subjective health, sexual function, mental health, self-esteem, and social support were maintained or even increased, despite the age-related endocrine changes. These findings are of crucial interest for the public. Since more people are experiencing better health with higher age, the current finding that endocrine changes are independent of psychosocial changes is encouraging. It is therefore important to continue to integrate biological and psychosocial parameters into definitions of healthy aging and to take into account the potential independence of their developmental course in (healthy) aging. As a novel area of research, studies could aim to investigate the potential for maintaining high levels of protective psychosocial factors to help prolong general health into old age.
Supplemental Material
RevisedSupplemental_Material_Paper1_TL – Supplemental material for Age-Related Alterations in Endocrine Markers Do Not Match Changes in Psychosocial Measures: Findings From the Men’s Health 40+ Longitudinal Study
Supplemental material, RevisedSupplemental_Material_Paper1_TL for Age-Related Alterations in Endocrine Markers Do Not Match Changes in Psychosocial Measures: Findings From the Men’s Health 40+ Longitudinal Study by T.J. Lacker, A. Walther and U. Ehlert in American Journal of Men’s Health
Footnotes
Acknowledgements
The authors would like to thank the Men’s Health 40+ study team, namely Nina Chiastra, Rahel Furrer, Anja Vandersmissen, and all interns, without whom the study would not have been possible. They are also grateful to Dr Firouzeh Fahramand for her extraordinary work in performing all hormone analyses. The authors further thank all participants for their time and motivation to join the study.
Authors’ Contributions
T.J.L. and U.E. designed the concept of the study. T.J.L. and A.W. organized and conducted the study and collected the data. T.J.L. wrote the first draft of the manuscript. U.E. and A.W. contributed with important intellectual content and edited subsequent versions of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the University Research Priority Program (URPP) “Dynamics of Healthy Aging” of the University of Zurich awarded to UE. The funding organization played no role in the design, analysis, or interpretation of the current study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
