Abstract
During adolescence, androgens are responsible for the development of secondary sexual characteristics, pubertal growth, and the anabolic effects on bone and muscle mass. Testosterone is the most abundant testicular androgen, but some effects are mediated by its conversion to the more potent androgen dihydrotestosterone (DHT) or to estradiol. Androgen deficiency, requiring replacement therapy, may occur due to a primary testicular failure or secondary to a hypothalamic–pituitary disorder. A very frequent condition characterized by a late activation of the gonadal axis that may also need androgen treatment is constitutional delay of puberty. Of the several testosterone or DHT formulations commercially available, very few are employed, and none is marketed for its use in adolescents. The most frequently used androgen therapy is based on the intramuscular administration of testosterone enanthate or cypionate every 3 to 4 weeks, with initially low doses. These are progressively increased during several months or years, in order to mimic the physiology of puberty, until adult doses are attained. Scarce experience exists with oral or transdermal formulations. Preparations containing DHT, which are not widely available, are preferred in specific conditions. Oxandrolone, a non-aromatizable drug with higher anabolic than androgenic effects, has been used in adolescents with preserved testosterone production, like Klinefelter syndrome, with positive effects on cardiometabolic health and visual, motor, and psychosocial functions. The usual protocols applied for androgen therapy in boys and adolescents are discussed.
Keywords
Developmental Physiology: An Essential Knowledge for Androgen Therapy
The testis has two main roles: the secretion of male hormones and the production of the male gamete. The relevance of these testicular functions varies along life.
Fetal Period
As soon as the testes differentiate in the first trimester of intrauterine development, Sertoli cells and primordial germ cells aggregate to form the seminiferous cords, which are embedded in the interstitial tissue containing Leydig cells. Sertoli and Leydig cells are the sources of testicular hormones, whereas primordial germ cells proliferate and subsequently differentiate in the process of spermatogenesis. During early fetal life, Sertoli cell production of anti-Müllerian hormone (AMH) plays a major role, together with Leydig cell androgen secretion, in the masculinization of the fetus. This early endocrine function of the testis occurs independently of pituitary gonadotropins (Figure 1; Grinspon et al., 2014; Makela et al., 2019). From the second trimester of fetal life, the endocrine function of the testis is regulated by luteinizing hormone (LH) and follicle-stimulating hormone (FSH), produced by the fetal pituitary in response to the hypothalamic gonadotropin-releasing hormone (GnRH). LH induces Leydig cells to secrete androgens and the insulin-like factor 3 (INSL3): Both are involved in testicular descent to the scrotum (Mamoulakis et al., 2015), whereas androgens are also responsible for the enlargement of the penis and scrotum. FSH promotes Sertoli cell proliferation and secretion of AMH and inhibin B (Grinspon et al., 2018).
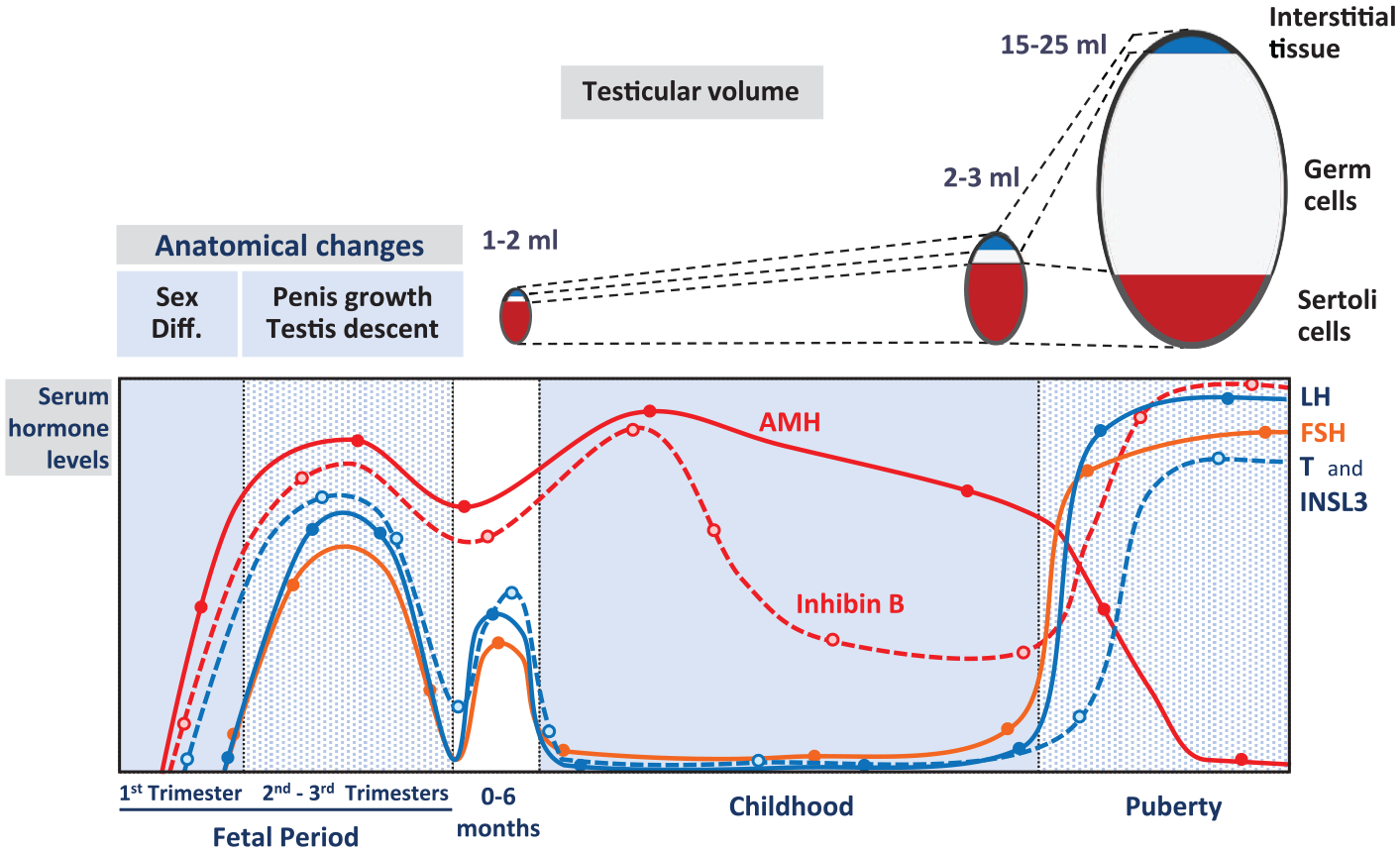
Changes in the anatomical aspects and circulating levels of sex hormones in males from fetal life through adulthood. In early fetal life, testicular hormones, which are produced independently of fetal LH and FSH, drive masculinization of the genitalia. In the second part of intrauterine life, luteinizing hormone (LH) and follicle-stimulating hormone (FSH) are the main regulators of gonadal endocrine activity resulting in testicular growth and descent and in enlargement of the external genitalia. During the first 3–6 months after birth (also called “mini-puberty”), gonadotropin and testicular endocrine function remains active. During the rest of infancy and childhood, gonadotropins, testosterone (T), and insulin-like factor 3 (INSL3) decline, but Sertoli cell secretion of anti-Müllerian hormone (AMH) and inhibin B persist at high levels. During pubertal development, the size of the testes increases notoriously due to spermatogenesis, driven by gonadotropins and testosterone. AMH is inhibited by testosterone, while inhibin B secretion is boosted by the action of FSH and germ cells. Reprinted, with permission, from Salonia et al. (2019). ©2019 Springer Nature Limited.
Infancy and Childhood
The hypothalamic–pituitary–testicular axis remains active after birth until the 3rd to 6th months (Bergadá et al., 2006; Kuiri-Hänninen et al., 2014; Makela et al., 2019). The lack of androgen receptor expression in Sertoli cells is responsible for the absence of full spermatogenesis in spite of high intratesticular androgen levels (Boukari et al., 2009; Chemes et al., 2008). Then, gonadotropin levels decline resulting in a dramatic fall of serum testosterone and INSL3 to very low or undetectable levels (Figure 1). However, basal AMH and inhibin B production remains active throughout childhood (Valeri et al., 2013). Germ cells proliferate by mitosis but do not enter meiosis; thus, spermatozoa are not produced (Edelsztein & Rey, 2019; Makela et al., 2019), owing to the low levels of intratesticular testosterone during childhood (Rey et al., 2009).
Adolescence and Adulthood
Between 9 and 14 years of age, the hypothalamic–pituitary–testicular axis is reactivated resulting in pubertal development. In the earliest stages of puberty, FSH promotes Sertoli cell proliferation resulting in a moderate testicular volume increase and LH induces Leydig cell androgen and INSL3 secretion (Figure 1). The increase in intratesticular testosterone concentration prompts Sertoli cell maturation—now expressing the androgen receptor—characterized by an arrest in its proliferation and a decrease in AMH secretion on one hand and by the acquisition of the capacity of sustaining meiosis and postmeiotic adult spermatogenesis leading to sperm production (Makela et al., 2019; Rey, 2014). Clinically, the onset of puberty is defined by the initial increase in testicular size to 4 ml associated with scrotal enlargement and change in the texture of the scrotal skin. Marshall and Tanner (1970) have thus characterized pubertal development of the genitalia from the preadolescent stage 1 to the adult stage 5. Interestingly, testosterone levels increase progressively in the circulation, and low or undetectable values are usually found during Stages 1 and 2, with a notorious increase during Stages 3 and 4 to reach adult values in Stage 5 (Grinspon et al., 2011; Knorr et al., 1974).
During adolescence and adulthood, androgens exert their actions on many organs, other than the testes themselves as already discussed. The most conspicuous effects of androgens in adolescents is the development of secondary sexual characteristics: enlargement of the penis and scrotum together with the development of sexual hair. Androgens also have a crucial role on pubertal growth, both by stimulating the secretion of growth hormone (GH) and insulin-like growth factor 1 (IGF1), through their conversion to estrogens, and by directly acting on the growth plate cartilage (Stancampiano et al., 2019). Strong evidence exists that androgens have anabolic effects on bone, both directly and after aromatization to estrogens (Mohamad et al., 2016), and on muscle mass (Welle et al., 1992) while reducing central adiposity (Chasland et al., 2019). Testosterone stimulates erythropoiesis although the underlying mechanism is not completely understood (Coviello et al., 2008). Finally, androgens have long been known for their positive effect on libido and behavior.
Testosterone is the most abundant testicular androgen, with a production rate of 2–11 mg/day in adults (Wu & Kovac, 2016). Most (40%–60%) circulates bind to sex hormone binding globulin (SHBG), 33% to albumin, and 1%–4% is unbound. Approximately 7% is converted to the more potent androgen dihydrotestosterone (DHT) by the enzyme 5α-reductase and less than 1% is transformed into estradiol by the enzyme aromatase (Stancampiano et al., 2019). Most testosterone actions are direct, through its binding to the androgen receptor. However, conversion to DHT is essential in the end organ for prostate development and virilization of the external genitalia during fetal life.
Hypoandrogenism as a Component of Hypogonadism
Androgen deficiency may occur during the fetal and neonatal periods and from adolescence through adulthood (Figure 2). Some essential concepts need to be considered in order to avoid misdiagnoses and inadequate management of hypogonadism in the different periods of life (Grinspon et al., 2019). Male hypogonadism is typically characterized by insufficient androgen and sperm production in the adult (Salonia et al., 2019), but it cannot be defined by those parameters during childhood since all normal boys have low or undetectable testosterone and do not produce sperm. Indeed, before puberty, impaired testicular function or hypogonadism is better ascertained by low Sertoli cell markers like AMH or inhibin B (Rey et al., 2013). Thus, the concept of male hypogonadism should not be equated just to hypoandrogenism but extended to all conditions with decreased function of any testicular compartment (Leydig, Sertoli, and/or germ cells), as compared to what is expected for the age.

Clinical presentation of male hypogonadism according to the period of life when it is established. The testes differentiate in the first trimester of fetal life, independently of LH and FSH, the pituitary gonadotropins. Testicular hormones drive the internal and external genitalia through the male differentiation pathway. Hypogonadism established in early fetal life leads to the existence of ambiguous or female genitalia in XY newborns. In later fetal life, androgens induce testicular descent and has a trophic effect on the external genitalia. Primary and central hypogonadisms established in this period result in a male newborn with micropenis, micro-orchidism, and/or cryptorchidism. During childhood, gonadotropins and androgens are very low or undetectable; thus, hypoandrogenism is physiological. At pubertal age, the gonadal axis is reactivated, and secondary sex characteristics develop. Hypogonadism may result in absent or incomplete pubertal development, infertility, and/or sexual dysfunction. Modified, with permission, from Grinspon et al. (2019). © 2019 Elsevier Limited. This figure was created using BioRender (https://biorender.com/).
Primary Hypogonadism
In primary hypogonadism, usually referred to as hypergonadotropic hypogonadism, the defect is primarily testicular. It may be congenital and exist from early fetal life or occur in later fetal life or after birth at any stage of development (Ladjouze & Donaldson, 2019). It may present with whole gonadal dysfunction, that is, Leydig, Sertoli, and germ cells are concomitantly affected, or with an initial cell-specific dysfunction (Table 1).
Male Hypogonadism Presenting With Hypoandrogenism at Different Periods of Life, Needing Androgen Therapy.

Note. DHT = dihydrotestosterone; IM = intramuscular.
An example of fetal-onset primary hypogonadism with whole gonadal dysfunction is testicular dysgenesis, while fetal primary hypogonadism with Sertoli cell–specific dysfunction occurs, for instance, in patients with mutations of the FSH receptor (Siegel et al., 2013) or of the AMH gene (Picard et al., 2017). In the latter, testosterone production by Leydig cells is initially preserved. Conversely, in patients with mutations of the LH receptor or steroidogenic enzymes (Auchus & Miller, 2012; Mendonça et al., 2010; Wisniewski et al., 2019), there is a fetal primary hypogonadism with Leydig cell–specific dysfunction.
Klinefelter syndrome is the most frequent disorder characterized by primary hypogonadism (Bojesen & Gravholt, 2007). Although the condition is of genetic origin, only the germ cell compartment seems to be defective from fetal life (Aksglæde et al., 2006). Sertoli and Leydig cell function remains normal until mid-puberty, as revealed by normal AMH, testosterone, and gonadotropin levels (Bastida et al., 2007). Thereafter, testicular hormone levels fall, and gonadotropins increase.
Primary hypogonadism can also be acquired. It presents with whole gonadal dysfunction, for example, in testicular regression or conditions that require gonadectomy leading to bilateral anorchia, or with cell-specific dysfunction, for example, in chemotherapy-induced testicular failure where germ cells are the primarily affected compartment. It is noteworthy that primary hypogonadism may present with normal gonadotropin levels, that is, it is not always hypergonadotropic during childhood (Grinspon et al., 2012).
Central Hypogonadism
In central hypogonadism, usually called hypogonadotropic hypogonadism, the primary defect resides in the hypothalamic–pituitary axis (Table 1). It can be congenital and affect the function of all testicular cell populations, like in Kallmann syndrome or in hypogonadotropic hypogonadism without anosmia (Young et al., 2019), or initially affect one specific compartment, like FSH synthesis defects impairing Sertoli cell function (Siegel et al., 2013) or LH synthesis defects affecting Leydig cell testosterone production (Valdes-Socin et al., 2004).
Central hypogonadism may also be acquired, like in patients with multiple pituitary hormone deficiency resulting from brain tumors in the sellar/suprasellar region or their surgical removal or from high-dosage cranial radiotherapy.
Clinical Consequences of Hypoandrogenism
As reviewed in detail elsewhere (Grinspon et al., 2019; Salonia et al., 2019), hypogonadism elicits different clinical consequences according to the type of hypogonadism (central/primary, whole gonadal/cell-specific) and to the period of life when it is established (Figure 2). In this review, only the clinical consequences of hypoandrogenism, that is, those resulting from impaired androgen production by the testes in pediatric ages, with a special focus on adolescence will be addressed. The consequences of primary Sertoli or gem cell defects are not within the scope of this article.
In Newborns
The clinical consequences of insufficient androgen action observed in the newborn vary according to the timing of the establishment of androgen impairment.
Hypoandrogenism in the First Trimester of Fetal Life: Disorders of Sex Development
Since androgens are essential for the development of the internal male genitalia from Wolffian ducts and the urogenital sinus and for the masculinization of the external genitalia, which take place between the 8th and 13th fetal weeks (Makela et al., 2019; Rey & Grinspon, 2011), androgen insufficiency established in early intrauterine life results in insufficient or complete lack of virilization of the fetus. These congenital conditions are most often of genetic origin and include complete or partial defects of testicular development (pure or partial gonadal dysgenesis, respectively) and specific disorders of Leydig cell differentiation (Leydig cell aplasia/hypoplasia due to mutations in the LH receptor) or steroid production (mutations in the genes coding for steroidogenic proteins). These are forms of primary hypogonadism, which may be subject to androgen treatment (Ladjouze & Donaldson, 2019). Androgen insensitivity is another form of Disorders of Sex Development (DSD) with undervirilization, but the response to androgen supplementation is dependent on the remaining activity of the mutated androgen receptor (Hughes et al., 2012).
In the most severe forms of DSD, androgen production or action is null, and the newborn has completely female external genitalia. These patients are raised as girls and will not require androgen treatment. Patients with partial forms of DSD (partial gonadal dysgenesis, mild forms of Leydig cell hypoplasia, or steroidogenic defects) present with various degrees of genital ambiguity and may be raised as males, requiring androgen therapy.
As can be deduced from the knowledge of developmental physiology, patients with impaired hypothalamic–pituitary function do not present as DSDs, since placental human chorionic gonadotropin (hCG) drives Leydig cell differentiation and steroid production during the first trimester of fetal life, acting through the LH receptor.
Hypoandrogenism in the Second and Third Trimesters of Fetal Life
Insufficient androgen production may be the consequence of a primary defect of the testes established after the critical period of sex differentiation: An example is the condition known as vanishing testes or testicular regression syndrome (Ladjouze & Donaldson, 2019). Hypoandrogenism in this period may also result from central defects of the hypothalamic–pituitary axis, that is, central or hypogonadotropic hypogonadism (Young et al., 2019). The lack of fetal LH stimulus on Leydig cells cannot be replaced by the declining hCG levels. These patients have male external genitalia, but the insufficient androgen levels result in micropenis. Testes, when present, are usually incompletely descended and smaller than normal.
In Childhood
As already mentioned, androgen production by the testes is extremely low or undetectable between the 6th month of postnatal life and the beginning of puberty. Therefore, conditions leading to primary or central hypogonadism established in this period are not expected to have evident clinical consequences before the age of adolescence. However, recent studies of early exposure to steroids in boys with Klinefelter syndrome has challenged this paradigm (Chan et al., 2019), as discussed later.
In Adolescents
The main clinical consequences of hypoandrogenism in adolescents is the lack of development of secondary sexual characteristics and of the pubertal growth spurt. These boys keep a childlike body aspect and their stature becomes progressively shorter than that of their peers. Boys with delayed or absent puberty usually present with distress, anxiety, and a generally impaired quality of life in dimensions related to psychological well-being (Stancampiano et al., 2019).
Absent puberty may be due to agonadism: Testosterone and all gonadal hormones are undetectable, while gonadotropins are extremely elevated usually after the age of 12–13 years (Grinspon et al., 2012). Together with a clinical history of empty scrotum and gonadectomy or bilateral testicular torsion, the diagnosis is easily made. Otherwise, undetectable AMH or absence of testosterone rise in response to an hCG test may be needed (Grinspon et al., 2018; Lee et al., 1997). Pubertal delay in primary hypogonadism is rare, since Leydig cells are quite resistant to diverse noxae like radio- or chemotherapy, orchitis, and cryptorchidism, and their androgen production is sufficient for pubertal onset in syndromic forms of primary hypogonadism, for example, Klinefelter, Prader–Willi, and Noonan syndromes (Ladjouze & Donaldson, 2019).
Constitutional delay of puberty is a relatively frequent cause of persistent hypoandrogenism in boys and may be difficult to distinguish from central (hypogonadotropic) hypogonadism, especially at the younger adolescent ages. In these boys, testosterone and gonadotropin levels remain in the prepubertal range, and androgen treatment may need to be established to induce pubertal changes in body aspect before a conclusive diagnosis can be made (Palmert & Dunkel, 2012; Salonia et al., 2019).
Androgen Treatment
Androgen effects on target tissues may be schematically classified into androgenic and anabolic. Androgenic effects include fetal virilization, development of male secondary sexual characteristics and of the reproductive tract (epididymis, seminal vesicles, prostate, etc.), male hair growth pattern, and typical sebaceous gland activity. Anabolic effects include enhanced protein synthesis leading to increased skeletal muscle mass and bone metabolism and growth (Orr & Fiatarone Singh, 2004; Wu & Kovac, 2016). Androgenic effects occur directly through binding to the androgen receptor present in target tissues: DHT is the most potent, whereas oxandrolone and nandrolone have the lowest androgenic potential. Anabolic actions involve both direct and indirect mechanisms: The direct effects are mediated by the androgen receptor, whereas the indirect actions occur by the compound’s capacity to bind the glucocorticoid receptor and antagonize the catabolic effects of glucocorticoids (Orr & Fiatarone Singh, 2004). Both androgenic and anabolic effects are generally sought when androgen therapy is indicated in male patients; however, specific effects may be pursued in certain cases. It should, however, be kept in mind that in practice it is not easy to accurately quantitate the anabolic versus the androgenic activities of these compounds.
Selective androgen receptor modulators (SARMs), chemical compounds with variable binding capacity to the androgen receptor and tissue specificity, are also being investigated in order to develop drugs showing anabolic activity while avoiding other side effects, but none is yet on the market (Solomon et al., 2019). Aromatase inhibitors, which repress the transformation of androgen to estrogens, have been used to provoke an increase in androgen levels, but limited to conditions where there is an unaffected androgen synthesis capacity (Varimo et al., 2019). Finally, it should be stressed that the beneficial effects of androgen treatment on most organs contrast with their negative effects on the testes: In fact, the administered androgens usually inhibit the gonadotrope, thus resulting in lowering (or completely blocking) LH and FSH secretion. As a consequence of the low intratesticular testosterone concentration, spermatogenesis is arrested at the premeiotic stage and testicular volume remains small (Coviello et al., 2004; Rey, 2014). In fact, modified testosterone compounds with androgenic and progestational activities, like dimethandrolone undecanoate (DMAU) and 11β-MNT dodecylcarbonate (11β-MNTDC), are being tested as hormonal male contraceptives (Ayoub et al., 2017; Yuen et al., 2020). Therefore, gonadotropin rather than androgen therapy is indicated when fertility is pursued in patients with central hypogonadism (Young et al., 2019). This review will briefly go through the general aspects of androgen formulations, with a more detailed description of those medications and treatment protocols used in adolescents.
Methods
A scoping review was performed to address the questions “What is known about conditions requiring androgen treatment in boys and adolescents and about the medications and treatment protocols available for those conditions?” The objectives of the review were to examine the extent, range, and nature of research activity on the subject, to summarize and disseminate the findings, and to identify research gaps in the existing literature (Arksey & O’Malley, 2005; Daudt et al., 2013). Therefore, this is a review of all the studies found after performing a search in PubMed (https://www.ncbi.nlm.nih.gov/pubmed/) using “androgen replacement,” “anabolic/androgenic steroids,” “constitutional delay of puberty,” disorders of sex development,” and “hypogonadism” as keywords, and “Species: Human,” “Languages: English,” “Ages: Child: birth-18 years,” and “Sex: Male” as filters.
Formulations Used for Androgen Therapy
Anabolic/androgenic steroids are derivatives of the testosterone or DHT (Table 2). The androgenic:anabolic ratio of testosterone, the prototypical anabolic/androgenic steroid, is 1:1 (Wu & Kovac, 2016). By modifying the basic structure of testosterone, different formulations aim at separating the androgenic from the anabolic activities. Thus, oxandrolone and nandrolone have special indication when anabolic effects are favored and androgenic effects need to be avoided. However, the androgenic effect is present in all anabolic/androgenic steroids when used at high dosage (Orr & Fiatarone Singh, 2004). Side effects for all androgenic agents when used for long periods are erythrocytosis, nervousness and irritability, inhibition of gonadotropin secretion resulting in reduced intratesticular testosterone concentration and spermatogenic arrest leading to reduced testis volume, persistent erections, gynecomastia due to aromatization to estrogens (except for those non-aromatizable), and, particularly in prepubertal or early pubertal boys, accelerated bone maturation and early pubic hair development and acne. Since androgen therapy is usually used indicated for terms that do not go beyond 3–6 weeks in boys and 6–12 months in adolescents with constitutional delay of puberty, monitoring hematocrit and liver function does not seem essential (Stancampiano et al., 2019).Conversely, for longer treatments, like in adolescents with congenital or acquired central hypogonadism, standardized monitoring as recommended for adults should be followed.
Androgenic Anabolic Steroids Commercially Available.

Testosterone
Following the scientific saga initiated in the late 19th century by Brown-Séquard in France and resumed by a few European scientists and pharmaceutic companies at the beginning of the 20th century, which led Ružička and Butenandt to be awarded the Nobel prize, testosterone has been available for androgenic therapy since 1935 (Nieschlag & Nieschlag, 2019). Different pharmaceutical formulations of testosterone exist, which are administered by various routes.
Intramuscular
The 17β-hydroxyl esters of testosterone, enanthate, cypionate, propionate, and undecanoate, are the most frequently used formulations in androgen therapy in adolescents. These compounds are prepared with oil-based vehicles for slow-release intramuscular (IM) injections.
Testosterone enanthate is commercially available in ampoules or ready-to-use syringes containing 200 or 250 mg (140 or 180 mg of testosterone, respectively). In young adults, testosterone levels reach supraphysiological levels in the 1st week after IM injection (Figure 3; Di Luigi et al., 2009; Schurmeyer & Nieschlag, 1984), with significant interindividual variations according to the genomic background of metabolizing enzymes (Ekström et al., 2011; Zitzmann, 2007). In young adults with hypogonadism, a single injection of 250-mg testosterone enanthate induced a rapid increase in serum testosterone, attaining 700–1,800 ng/dl (~ 24–62 nmol/l) by Day 7, then decreasing to 260–700 ng/dl (~ 9–24 nmol/l) by Day 14, and to mostly subphysiological levels for adults, roughly 100–400 ng/dl (~ 3.5–14 nmol/l), by Day 21. A similar pattern is observed in DHT levels (Di Luigi et al., 2009). The half-life is approximately 4.5 days, and elimination occurs through the kidneys (90%) and bile (10%). In adults, it is usually administered in dosages of 250 mg every 2–4 weeks. Although pharmacokinetic studies are not available in children or adolescents, this is the only IM preparation and the most commonly indicated testosterone formulation used in boys and adolescents.

Schematic of serum testosterone levels in hypogonadal adult males before (time 0) and after having received intramuscular injections of testosterone undecanoate (1,000 mg) or enanthate (250 mg). The graded area represents normal serum levels of testosterone for an adult male. Unfortunately, no data are available for boys or adolescents. Data obtained from Di Luigi et al. (2009), Morgentaler et al. (2008), Nieschlag and Behre (2010), and Zhang et al. (1998).
Testosterone cypionate is available in 100-mg or 200-mg vials for IM administration. Its half-life is approximately 4 days (Bi et al., 2018). Pharmacokinetics and pharmacodynamics are similar to those of testosterone enanthate. Testosterone propionate has a shorter half-life (Fujioka et al., 1986), and its use has been deemed less convenient from a clinical standpoint.
A mixture of very short and short-acting testosterone esters, namely, 30-mg propionate, 60-mg phenylpropionate, 60-mg isocaproate, and 100-mg testosterone decanoate, is available for IM administration in 250-mg ampoules. Its use is similar to that of testosterone enanthate in adolescents (Stancampiano et al., 2019; Vandewalle et al., 2018; Varimo et al., 2019) and adults (Valdes-Socin et al., 2004; Zitzmann & Nieschlag, 2000).
Testosterone undecanoate is commercialized in 1,000-mg ampoules (631.5 mg of testosterone) to be administered IM every 10–14 weeks or in 750-mg ampoules for IM injections every 8–10 weeks in adults (Aydogdu & Swerdloff, 2016). The pharmacokinetic profile is more physiological than that of enanthate or cypionate (Partsch et al., 1995), with less frequently observed supraphysiological levels in the 1st week and sustained physiological levels for at least 10 weeks in adults with hypogonadism (Figure 3; Morgentaler et al., 2008; Zhang et al., 1998). However, there is no experience in pediatric patients, and its appropriateness for those with pubertal delay raises concern owing to its long-acting period.
Transdermal
Transdermal formulations are the most frequently used form of androgenic/anabolic steroid therapy in adults with testosterone deficiency. These formulations are available as topical solutions or gels or as adhesive patches. The latter, originally designed for scrotal and then for non-scrotal application, have lost patients’ preference owing to the relatively high rate of skin irritation (McBride et al., 2015).
Liquids, ointments, and gels for topic administration produce less adverse effects locally in the skin but are more easily transferred to women and children by direct contact, which represents a special concern. A large variety of presentations exist in the market for adult use, but none of them has been approved for use in children or adolescents (Shoskes et al., 2016). Only one report exists on the use of a 5% testosterone ointment in boys with micropenis (Arisaka et al., 2001), and one preliminary study has compared testosterone gel with IM injections in adolescents with constitutional delay of puberty. This medication is available as a metered-dose pump that delivers 10 mg of testosterone (Chioma et al., 2018).
Subdermal Implants
Subdermal pellets, consisting of 12.5, 25, 37.5, 50, or 75 mg of crystalline testosterone, are intended for subdermal implant in the hip area or another fatty area, at a dosage of 150 to 450 mg every 3 to 6 months (Shoskes et al., 2016). Pellet extrusion, site infections, bleeding, and fibrosis are the main side effects. In addition, there is a requirement for skin incision and local anesthesia, which makes this formulation less favored.
Subcutaneous
Recently, formulations of testosterone cypionate or enanthate have been used for subcutaneous administration with success in female-to-male transgender patients (Spratt et al., 2017), and a 75-mg subcutaneous auto-injector for weekly self-administration of testosterone enanthate has been tested with satisfactory efficacy and safety in adults (Kaminetsky et al., 2019). Similarly, the 1,000-mg testosterone undecanoate formulation used for IM administration has recently been tested subcutaneously; while the pharmacokinetic features were similar to those observed with IM administration, pain after injection was greater when used subcutaneously, which lead to a lesser acceptability (Turner et al., 2019).
Oral and Buccal
Initially, 17α-alkylated derivatives of testosterone were synthesized to avoid hepatic first‑pass metabolism and proved efficacious; however, hepatotoxicity risk is high, and these compounds are no longer used (McBride et al., 2015; Shoskes et al., 2016). Instead, testosterone undecanoate for oral administration in 40-mg capsules is esterified at carbon 17β, which also partially avoids hepatic first‑pass metabolism by enhancing lymphatic transport and apparently is less hepatotoxic. Nonetheless, systemic absorption remains variable (McBride et al., 2015; Shoskes et al., 2016). A new formulation of testosterone undecanoate has recently been approved and is available in capsules of 158, 198, or 237 mg. An advantage of this new formulation is its better lipophilicity and more consistent absorption less dependent on a lipid meal (Ceponis et al., 2019). No experience has been reported yet in boys and adolescents. As compared to the injectable form, oral testosterone undecanoate shows lower risk for hypertension and polycythemia in adults with hypogonadism (Jick & Hagberg, 2013). A few studies have shown a good efficacy of oral testosterone undecanoate in the treatment of adolescent boys with constitutional delay of puberty (Albanese et al., 1994; Lawaetz et al., 2015), but no experience is published for patients with absent or delayed puberty due to hypogonadism.
Mucoadhesive tablets contain 30 mg of testosterone and are designed to adhere to the gum of the mouth or inner cheek. This buccal system provides sustained testosterone release as it hydrates. The usual dosage in adults is two tablets per day, one every 12 hr. The mucoadhesive tablets are well tolerated; the most common adverse effect is gingivitis (Shoskes et al., 2016).
Nasal
With a similar rationale to the buccal formulation, a nasal gel has been more recently developed. The advantages of intranasal testosterone administration include ease of delivery and decreased risk of transference. Peak levels in serum occur within 30–40 min and half-life is short, requiring two pumps of 5.5 mg of testosterone each per nostril three times a day, that is, a total dose of 33 mg daily in adults. One particular benefit of this formulation is that FSH and LH levels are not suppressed, thus resulting in maintained spermatogenesis (Masterson et al., 2018). Adverse effects include rhinorrhea and nasal discomfort or scabs (McBride et al., 2015; Shoskes et al., 2016). No experience exists in children and adolescents.
DHT
The natural and potent metabolite of testosterone via 5α-reductase enzymes type I or type II, DHT (also called androstanolone or stanolone), is found in 2.5% gels for skin application in very few countries. DHT has androgenic and anabolic effects, it is not converted to testosterone, and cannot be aromatized to estrogens. Therefore, DHT administration results in an increase in serum DHT and a decrease in testosterone and estradiol, following DHT inhibition at the hypothalamic level of the hypothalamic–pituitary–testicular axis (Cailleux-Bounacer et al., 2009; de Lignieres, 1993; Swerdloff et al., 2017). DHT is usually applied topically to the target organ, for example, the external genitalia in boys with micropenis (Becker et al., 2016), and is particularly useful in patients with defects of 5α-reductase (Aydogdu & Swerdloff, 2016; Xu et al., 2017).
Oxandrolone and Nandrolone
Oxandrolone and nandrolone are non-aromatizable drugs with an anabolic/androgenic ratio of approximately 10:1 to 11:1 (Orr & Fiatarone Singh, 2004; Wu & Kovac, 2016). Oxandrolone is available in 2.5- and 10-mg tablets for oral administration. It is well absorbed and relatively resistant to liver metabolization, with a half-life of approximately 9 hr. Its main side effects are related to those of 17α-alkylated steroids, namely, hepatotoxicity, though rarely serious or irreversible (Orr & Fiatarone Singh, 2004). Nandrolone decanoate exists for IM injections at 200 mg/ml in 1-ml vials. Hepatocellular neoplasia and hepatic peliosis have been reported in association with long-term therapy. These anabolic agents are used in patients with anemia or cachexia, but rarely for androgen replacement therapy. In boys and adolescents with Klinefelter syndrome, oxandrolone has been used with apparently positive effects on cardiometabolic health markers (Davis et al., 2017) and visual–motor and psychosocial functions (Ross et al., 2017). However, even if their androgenic action is low, these agents may impact on precocious genital development (Davis et al., 2018).
Treatment Protocols in Children and Adolescents
In pediatrics, IM testosterone is the prevailing therapeutic agent, with specific uses for topic DHT. Oral, transdermal, and subdermal testosterone preparations and oral oxandrolone have only been explored in a few clinical trials. It should be stressed that all commercially available androgenic/anabolic agents are designed for adults and none is marketed for use in patients below 18 years of age. Therefore, most treatment protocols used in children and adolescents are empiric.
In Newborns and During Infancy and Childhood
Newborns and untreated infants and children with signs of congenital hypoandrogenism due to primary hypogonadism leading to DSD or to primary or central hypogonadism leading to micropenis and cryptorchidism in the context of male genitalia may require transient androgen therapy (Hatipoğlu & Kurtoğlu, 2013; Ladjouze & Donaldson, 2019; A. D. Rogol, 2005a,b; Wisniewski et al., 2019). The main aims are penile enlargement and the enhancement of scrotal trophism.
The most usual practice is to give three IM doses of testosterone enanthate 25 mg every 3–4 weeks (Hatipoğlu & Kurtoğlu, 2013; Ladjouze & Donaldson, 2019; A. D. Rogol, 2005a,b; Wisniewski et al., 2019). It should be stressed that this may prove difficult, given that these oil preparations contain 200 or 250 mg in 1–2 ml. Side effects are pain and infections in the injection site, erections, and acne. These low doses of testosterone for short periods do not result in advancement of bone age or development of pubic hair. Whether testosterone replacement should be performed during the first 6 months of life to mimic the neonatal activation of the gonadal axis is unclear; in any case, replacement would better be done with gonadotropins to promote testicular effects (Bouvattier et al., 2012; Young et al., 2019).
Percutaneous treatment has been described in infants and children with micropenis: For 30 days, 10 mg (0.2 g of 5% testosterone cream) applied daily onto the phallus at night resulted in a significant increase of approximately 9 mm in penile length without changes in bone age (Arisaka et al., 2001).
In patients with DSD (Wisniewski et al., 2019), and particularly in those with 5α-reductase deficiency (Bertelloni et al., 2007; Odame et al., 1992; Xu et al., 2017), DHT gels may prove more efficacious. The 2.5% DHT gel is applied around the root of the penis or onto the penile shaft at a dose of 0.1–0.3 mg/kg/day, in two doses 12 hr apart, not exceeding 5 mg/day for a maximum of 3–6 months (Kaya et al., 2008; Xu et al., 2017).
Replacement therapy does not seem necessary for androgenic purposes during childhood since circulating testosterone is usually undetectable in this period of life. However, although hypoandrogenism has not been unequivocally observed in boys with Klinefelter, treatment with oxandrolone showed improvements in their behavior, cognition, and visual and motor capacities. Oxandrolone was administered orally at a daily dose of 0.05–0.06 mg/kg for 24 months to patients aged 4–12 years in placebo-controlled trials. Adverse events were mostly not clinically significant, for example, minor decline in serum HDL and advancement in bone age (Davis et al., 2017; Ross et al., 2017), except for the development of pubic hair and an increased risk of early gonadarche even if gonadal axis hormone levels were not affected (Davis et al., 2018).
In Early Adolescence
In adolescents >12–13 years old who do not show signs of pubertal onset, the goals of androgen therapy are to promote the development of secondary sexual characteristics and linear growth, which impact on the adolescent’s psychosocial well-being, together with the acquisition of muscle mass and bone mineral content. The timing and tempo of these androgenic and anabolic changes should mimic those of endogenous pubertal development, that is, the whole process from Tanner Stage 2 to Tanner Stage 5 should take approximately 3 years (Marshall & Tanner, 1970)). In this way, with a progression of approximately one stage per year, an adequate maturation of the growth cartilage is assured, avoiding a precocious early closure of the epiphyses.
Androgen therapy is used in patients with a known condition warranting permanent hypoandrogenism (anorchidism, congenital central hypogonadism, etc.) and in those with constitutional delay of puberty. The initial steps are similar, independently of the underlying condition. Usually referred to as “induction of puberty,” androgen therapy actually induces in these patients peripheral pubertal changes but not the reactivation of the hypothalamic–pituitary–testicular axis. Indeed, treatment with low doses of testosterone in boys with constitutional delay of puberty provokes a slower increase in testicular volume than in untreated boys (Bergadá & Bergadá, 1995; Chioma et al., 2018). In the most commonly used protocol, testosterone enanthate or cypionate is given IM at an initial dose of 50–100 mg every 4 weeks (Ladjouze & Donaldson, 2019; A.D. Rogol, 2005a,b; Salonia et al., 2019). Since the low doses used do not inhibit the reactivation of the hypothalamic–pituitary–testicular axis in boys’ constitutional delay of puberty, treatment can be discontinued at any time when a spontaneous progression of testis is observed. Alternatively, after a 6-month period, treatment can be discontinued for 3 months to see whether testis volume increases. In patients with a certified diagnosis of primary or central hypogonadism and in those with suspected constitutional delay of puberty and no testicular volume enlargement, the androgen dose is increased progressively in increments of 50 to 100 mg every 6 to 12 months. The aim is to mimic the increase of serum testosterone levels as observed during the progression of pubertal stages (Grinspon et al., 2011), that is, roughly 50 ng/dl in the first 6 months (equivalent to Tanner Stage 2), 150 ng/dl in the following 6–12 months (Tanner Stage 3), and above 250 ng/dl thereafter (Tanner Stages 4–5). Measuring serum testosterone in the 4th week after injection may be informative since, due to the pharmacokinetics of these formulations, there is almost no more drug present in circulation and the result will reflect endogenous testosterone production.
Little experience exists with transdermal, subdermal, and oral formulations in adolescents. A 2% testosterone gel has been tested in a small sample of boys with constitutional delay of puberty at a daily dosage of 10 mg, applied to the thighs once a day in the morning, showing satisfactory results in terms of secondary sexual characteristics development and linear growth, with no significant side effects (Chioma et al., 2018). One work was published more than 20 years ago with testosterone pellets administered at a dose of 8–10 mg/kg every 6 months for 18 months, with apparently good results (Zacharin & Warne, 1997); however, no further experiences seem to exist in this age group. A large cohort of boys with constitutional delay of puberty received testosterone undecanoate orally for 6 to 15 months. The initial dose was 40 mg/day in average (range 10–160) and was increased progressively to a maximal dose of 80 mg/day (20–160). Serum testosterone increased and remained within physiological levels, and a beneficial effect was noted on height velocity without any detrimental effect on bone age (Lawaetz et al., 2015).
In Late Adolescence and Adulthood
In patients with primary or central hypogonadism needing continued androgen therapy, the dosage of IM testosterone enanthate or cypionate is increased every 6 to 12 months until adult doses are attained, that is, 200 mg every 2–3 weeks or 250 mg every 3–4 weeks. Although clinical evaluation may be sufficient, treatment can be monitored by determining serum testosterone in the week preceding the next expected injection. Due to the pharmacokinetics of these formulations described earlier, a relatively subphysiological level should be expected. The IM formulation of testosterone undecanoate is approved for use in adults, showing pharmacokinetic advantages. Although no publication exists in adolescents, the use of testosterone undecanoate 1,000 mg IM every 10–14 weeks or 750 mg every 8–10 weeks could prove effective and safe in adolescents who have attained Tanner pubertal Stage 5 with the traditional testosterone therapy.
Some conditions are characterized by a milder hypoandrogenism and present with spontaneous puberty but a delayed tempo or a completely arrested progression. In these cases, treatment can be started with higher doses than those described for initial treatment in patients with absent puberty, depending on their bone age and predicted height.
The age at which patients with Klinefelter syndrome should start receiving treatment is controversial. Since testosterone production is within normal levels in most adolescents, replacement may be limited to those with clinical and biochemical signs of hypogonadism (Gravholt et al., 2018; Salonia et al., 2019). However, earlier exposure to steroid treatment seems to improve physical and neurocognitive outcomes (Chan et al., 2019). In adolescent patients with Klinefelter syndrome, administration of a 1% gel at 0.5 g once daily at bedtime, applied to the skin of the shoulders, upper arms, and/or abdomen, with a progressive increase up to 5 g/day resulted in doubling of serum testosterone and DHT with no clinically significant side effects (Rogol et al., 2014).
Other exceptions to the classical protocols of androgen therapy are patients with partial androgen insensitivity, who may need personalized dosage (Wisniewski et al., 2019), or patients with defects in 5α-reductase, who are deemed to respond better to DHT treatment (Becker et al., 2016; Xu et al., 2017).
None of the androgen preparations discussed in this review has an influence on liver function, even after long-term use. Hepatotoxicity occurred in the past when 17α-alkylated compounds were used.
Footnotes
Author Contributions
Both authors made a substantial contribution to the review and approved it for publication.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the review was written in the absence of any commercial or financial relationships that could be interpreted as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partially supported by grants PIP 11220130100687 of Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) and PICT 2014-2490, PICT 2016-0993, and PIDC 2017-0032 of Fondo para la Investigación Científica y Tecnológica (FONCYT), Argentina.
