Abstract
Previous studies have reported the clinical efficacy of avanafil for erectile dysfunction (ED), but these findings are controversial. This study aims to investigate the safety and efficacy of avanafil for ED. EMBASE, PubMed, and Cochrane Library were searched extensively to obtain eligible studies. Clinical outcomes including successful vaginal penetration (SVP), successful intercourse (SI), International Index of Erectile Function-Erectile Function domain (IIEF-EF) score and treatment adverse events (TAEs) were compared using RevMan v.5.3. Eight RCTs involving 3,709 patients were included. The analysis demonstrated that compared with placebo, the SVP (RR = 3.20, 95% CI [2.60, 3.95], p < .001), SI (RR = 2.53, 95% CI [2.19, 2.92], p < .001), change in IIEF-EF score (MD = 4.57, 95% CI [3.68, 5.46], p < .001) and TAEs (RR = 1.78, 95% CI [1.38, 2.31], p < .0001) were significantly higher in the avanafil. In addition, avanafil 200 mg were higher than avanafil 100 mg in SI (RR = 0.86, 95% CI [0.75, 0.99], p = .03) and change in IIEF-EF score (MD = −1.34, 95% CI [−1.67, −1.01], p < .001), but there were no obvious differences in SVP (RR = 0.89; 95% CI [0.74, 1.08], p = .23) and TAEs (RR = 0.97, 95% CI [0.83, 1.14], p = .74) between the two doses. The present evidence suggests that avanafil (especially 200 mg) has the potential to be the drug of choice for ED, but more strict and larger sample size RCTs are need to validate the findings.
Erectile dysfunction (ED) is defined as the inability of men to obtain or maintain enough erection to complete a satisfactory sexual activity (Hatzimouratidis et al., 2010). As a public health issue, ED has caused a serious negative psychological impact on patients and may affect the quality of life and the marital relationship of patients. The prevalence of ED increases with age, and it is necessary to find an effective and safe way to treat male ED (Laumann et al., 2005).
Oral phosphodiesterase type 5 inhibitors (PDE5-Is) are considered to be an effective method for the treatment of ED. Three of these drugs (such as sildenafil, vardenafil, and tadalafil) are recommended by the European Medicines Agency (EMA) and the Food and Drug Administration (FDA) for the treatment of ED (Bruzziches, Francomano, Gareri, Lenzi, & Aversa, 2013; Smith et al., 2013). In the past 10 years, due to the occasional failure and adverse events, many patients are not satisfied with those drugs. As a new generation of PDE5-Is, avanafil is gradually accepted by patients due to its high selectivity and low adverse event response rate (Burke & Evans, 2012).
Although Wang performed a meta-analysis of the effectiveness of avanafil in the treatment of ED, the results are still controversial (Wang et al., 2014). The study was designed to include more relevant randomized controlled trials (RCTs) and analyze more parameters (such as the International Index of Erectile Function-Erectile Function domain [IIEF-EF] score) to assess the safety and efficacy of avanafil for the treatment of ED and provide updated clinical evidence.
Methods
Search Strategy
This study was conducted under the Preferred Reporting Items for Systematic Reviews and Meta-analysis statement. As of April 2019, EMBASE, PubMed, and Cochrane Library were searched by the computer to distinguish all RCTs about the treatment of ED with avanafil. The search language was limited to English, and the following search terms were used: “avanafil,” “erectile dysfunction,” “randomized controlled trial.” Besides, the author manually searched and applied Google scholarly literature to avoid the omission of studies.
Inclusion/Exclusion Criteria
If all correlative RCTs suffice the following criteria, they were included in the analysis: (a) all the studies on the therapy of ED with avanafil; (b) all patients were 18 years or older, and were clinically diagnosed with ED; (c) the control group in this study was either a placebo or a different dose of avanafil; (d) this study provided at least one indicator of outcomes that can be analyzed. On the contrary, studies were excluded if (a) the research data were based on the results of animal experiments; (b) the study data could not be obtained; or (c) all studies in non-RCTs. Figure 1 presents a flow chart of the study selection process.

Flow diagram of bibliographic retrievals and results.
Data Extraction
After examining the title, abstract, and full text, two authors selected the literature in strict accordance with the inclusion criteria and then extracted the data according to the predesigned table for cross-checking. Differences can be dealt with by discussion. Data including first author, year of publication, type of study design, interventions, total number and age of subjects, characteristics of the patient populations, and outcome indicators were extracted. Data were collected independently by two investigators, and the missing data were acquired by contacting the author. The following outcomes data were extracted: successful vaginal penetration (SVP), successful intercourse (SI), change in IIEF-EF score, and treatment adverse events (TAEs). When continuous variables appeared in the studies in the form of the median (range), the mean values (standard deviation) was calculated by us (Luo, Wan, Liu, & Tong, 2018).
Quality Assessment
According to the new five-level evidence grading standard established by the Oxford Center for Evidence-Based Medicine (OCEBM), the level of evidence for all RCTs was 2b (Table 1). The quality assessment was based on methodological quality assessment criteria recommended by the Cochrane Handbook for Systematic Reviews of Interventions version 5.1.0 (Higgins et al., 2011). Where “high risk” stands for the high risk of bias, “low risk” stands for the low risk of bias and “unclear risk” stands for the absence of adequate information in the research to conduct the bias evaluation (Figure 2). All differences were resolved through discussions between the two commentators.
Basic Information and Characteristics of Studies for Meta-Analysis.
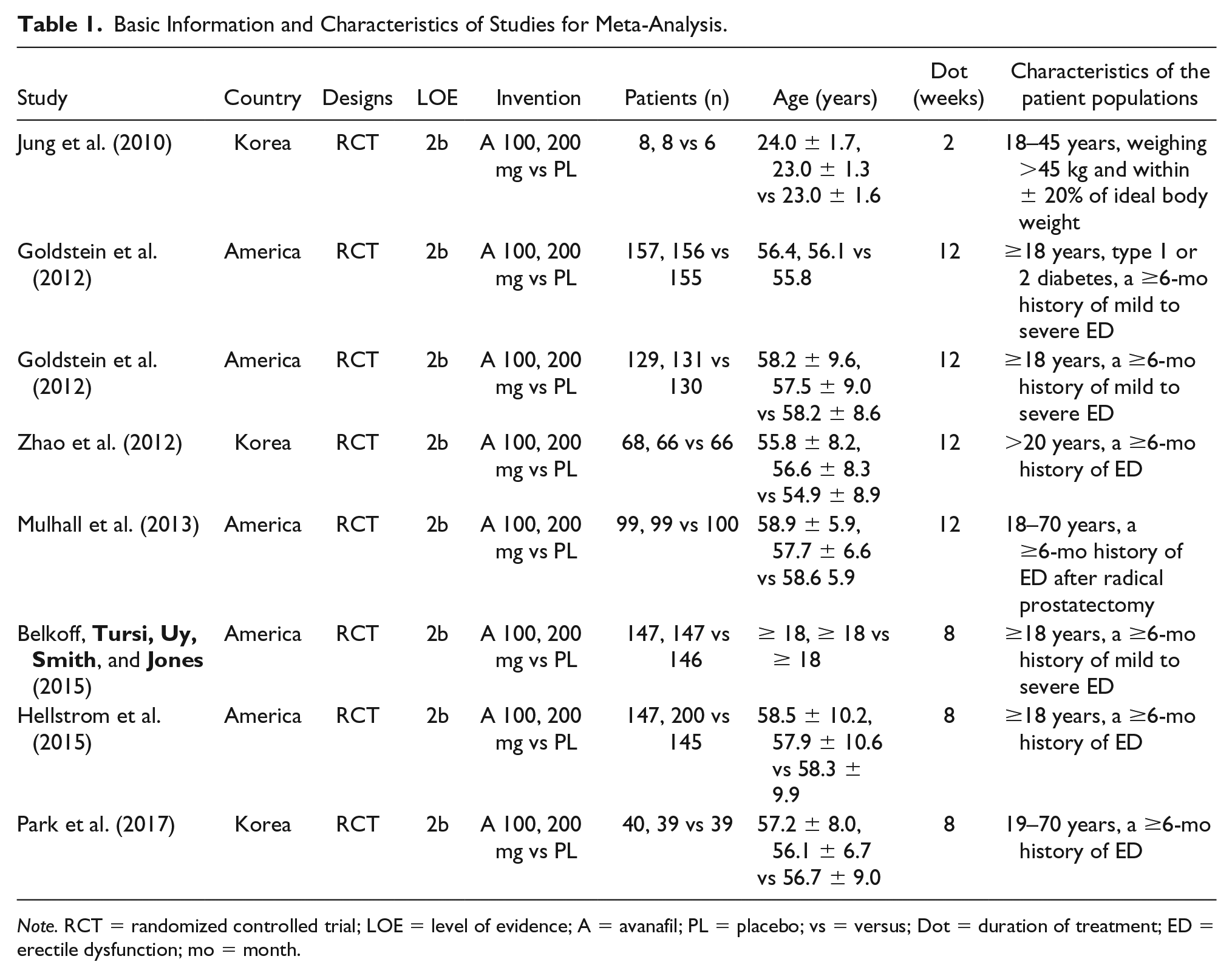
Note. RCT = randomized controlled trial; LOE = level of evidence; A = avanafil; PL = placebo; vs = versus; Dot = duration of treatment; ED = erectile dysfunction; mo = month.
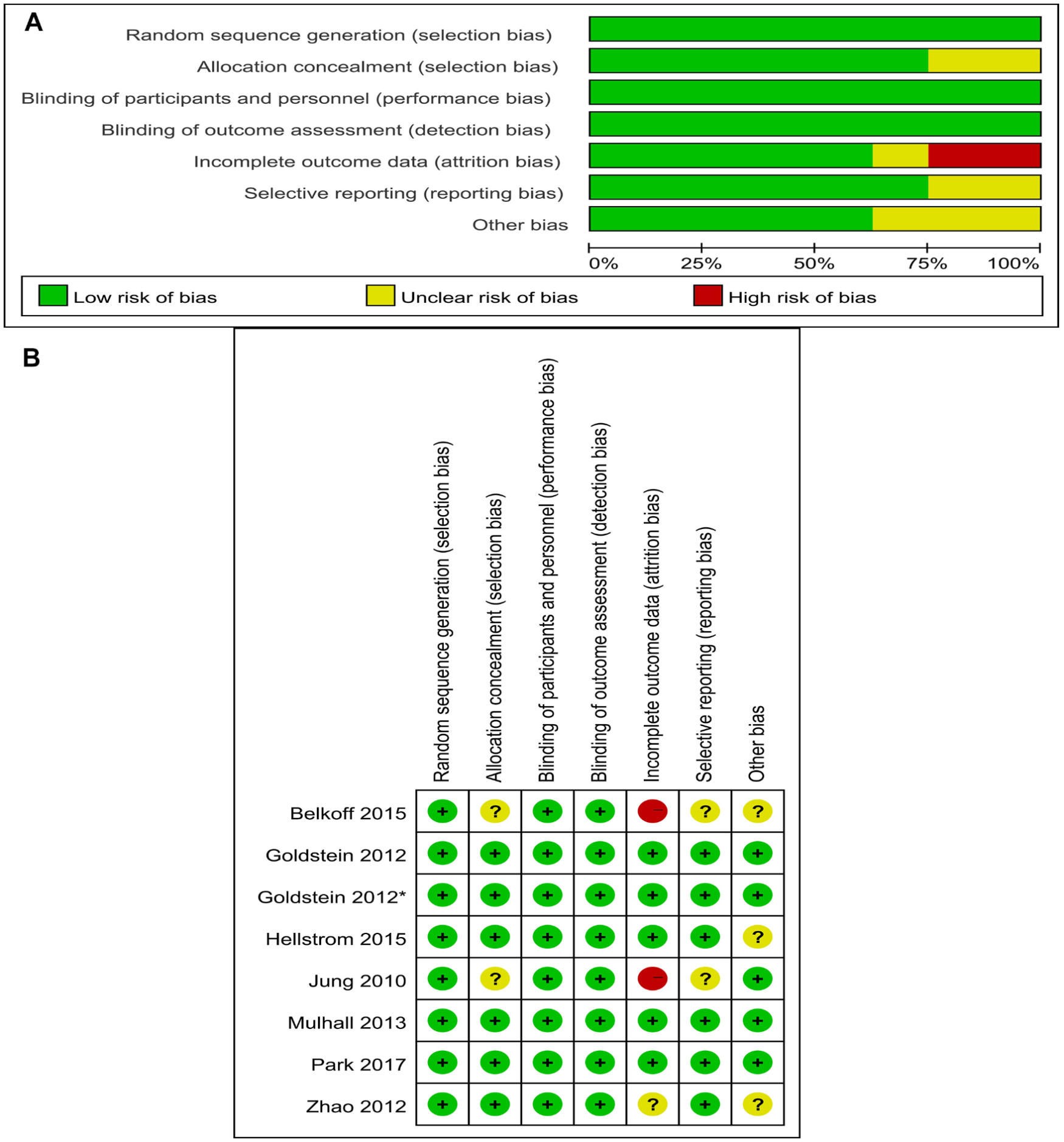
Quality of studies was assessed with the Cochrane Collaboration’s tool (
The funnel plot contributed a qualitative appraisal of the bias of published studies, and no evidence of bias was observed (Figure 3).

Funnel plot of the studies represented in the meta-analysis. RR = risk ratio; SE = standard error.
Statistical Analysis
The RevMan 5.3 and Stata 14.0 were applied for data analysis. Mean difference (MD) and risk ratio (RR) were used as the effect indexes for continuous and dichotomous data respectively, and p value and 95% confidence interval (CI) were given for both. Heterogeneity between studies was judged by Cochran’s Q and I2 statistics. When there was statistical homogeneity between studies (p > .1, I2 < 50%), a fixed-effects model was chosen for meta-analysis. Otherwise, a random-effects model was utilized. About all statistical consequences, p < .05 was regarded as statistically significant.
Results
Study Characteristics
A total of 426 related literature were obtained by a preliminary examination. Three hundred and ninety duplicate and unrelated studies were removed, and 36 studies were selected. After further reading the full text, eight RCTs (Belkoff, Tursi, Uy, Smith, & Jones, 2015; Goldstein, Jones, et al., 2012; Goldstein, McCullough, et al., 2012; Hellstrom et al., 2015; Jung et al., 2010; Mulhall et al., 2013; Park et al., 2017; Zhao et al., 2012) involving 2,398 patients were included in the meta-analysis (Figure 1). In addition, the sample size was estimated according to the methodology introduced in the study (Belkoff et al., 2015), and some data were obtained from the Cochrane Central Register of Controlled Trials (Goldstein, Jones, et al., 2012; Goldstein, McCullough, et al., 2012; Mulhall et al., 2013). The basic information and baseline characteristics of the incorporated studies are reported in Table 1, and the methodological quality evaluation of RCTs is presented in Figure 3.
Successful Vaginal Penetration
In the included studies, the data of SVP were reported in six RCTs involving 1,865 patients (Goldstein, Jones, et al., 2012; Goldstein, McCullough, et al., 2012; Mulhall et al., 2013; Park et al., 2017; Zhao et al., 2012). The combined results displayed a significant improvement in SVP of patients in the avanafil group compared with the placebo group (RR = 3.20, 95% CI [2.60, 3.95], p < .001). In the study, a subgroup analysis of patients treated with 100 mg and 200 mg of avanafil for ED was performed. The pooled analysis demonstrated that compared to the placebo group, the 100 mg group had obvious statistical significance (RR = 3.02, 95% CI [2.24, 4.07], p < .001), and similar results were identified in the 200 mg group (RR = 3.39, 95% CI [2.60, 3.95], p < .001; Figure 4A).
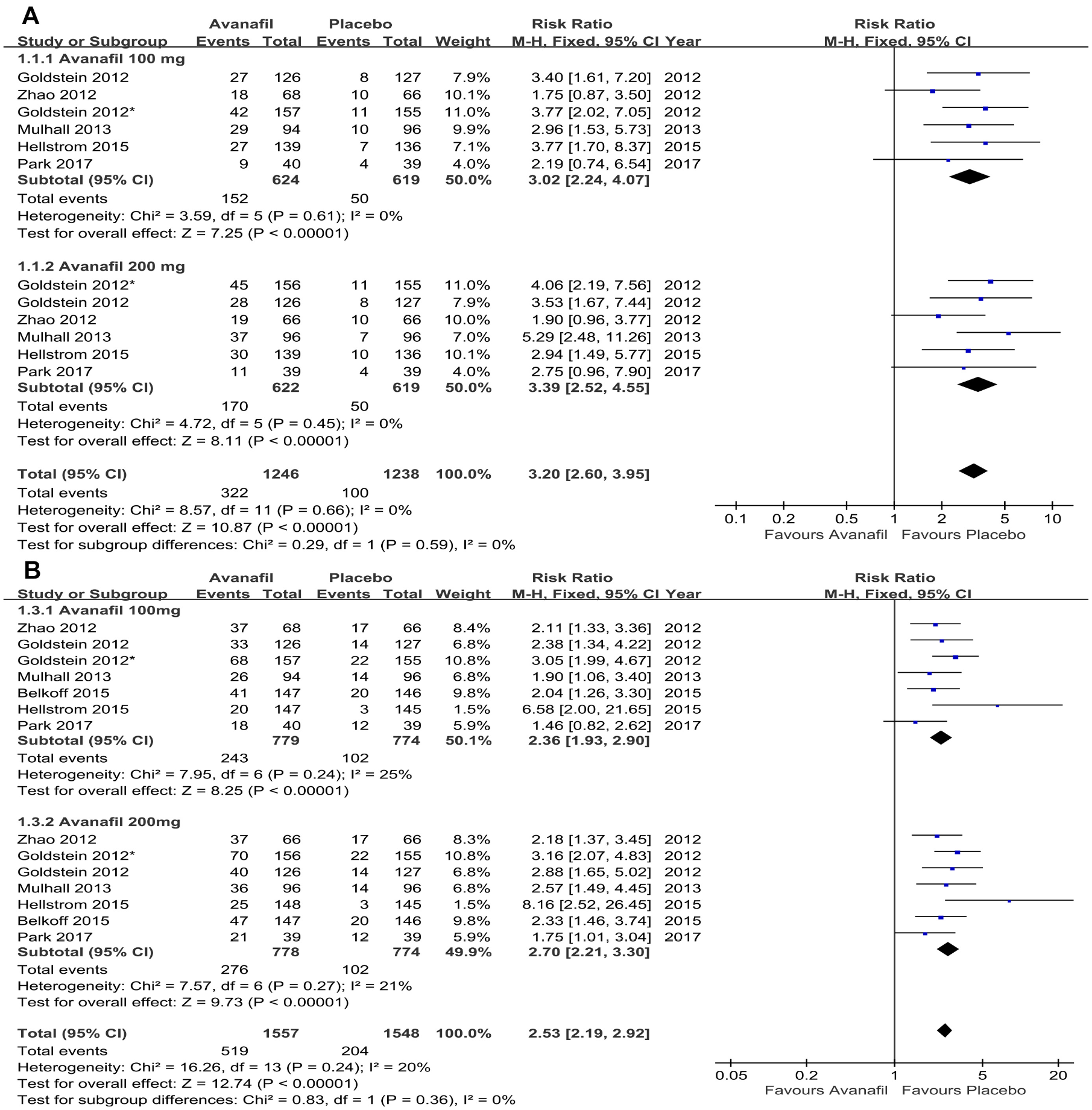
Forest plot for the comparison of the avanafil (100 mg and 200 mg subgroup) and placebo group (
Successful Intercourse
The SI data were extracted from seven RCTs (Belkoff et al., 2015; Goldstein, Jones, et al., 2012; Goldstein, McCullough, et al., 2012; Hellstrom et al., 2015; Mulhall et al., 2013; Park et al., 2017; Zhao et al., 2012); the comprehensive analysis demonstrated that the proportion of SI in the avanafil group was significantly greater than that in the placebo group (RR = 2.53, 95% CI [2.19, 2.92], p < .001). The subgroup analysis results showed that both the avanafil 100 mg and 200 mg groups had significantly higher SI ratios than the placebo group (100 mg: RR = 2.36, 95% CI [1.93, 2.90], p < .001; 200 mg: RR = 2.70, 95% CI [2.21, 3.30], p < .001; Figure 4B).
IIEF-EF Score
Five RCTs recorded the change in IIEF-EF score data (Goldstein, Jones, et al., 2012; Goldstein, McCullough, et al., 2012; Hellstrom et al., 2015; Mulhall et al., 2013; Park et al., 2017). Results indicated that the change in the IIEF-EF score of the avanafil group was significantly higher than that of the placebo group (MD = 4.57, 95% CI [3.68, 5.46], p < .001). In addition, similar results were found in the subgroup analysis. The MD was 3.88 (95% CI [2.69, 5.07], p < .001) for avanafil 100 mg group and 5.26 (95% CI [3.68, 5.46], p < .001) for avanafil 200 mg group (Figure 5A).
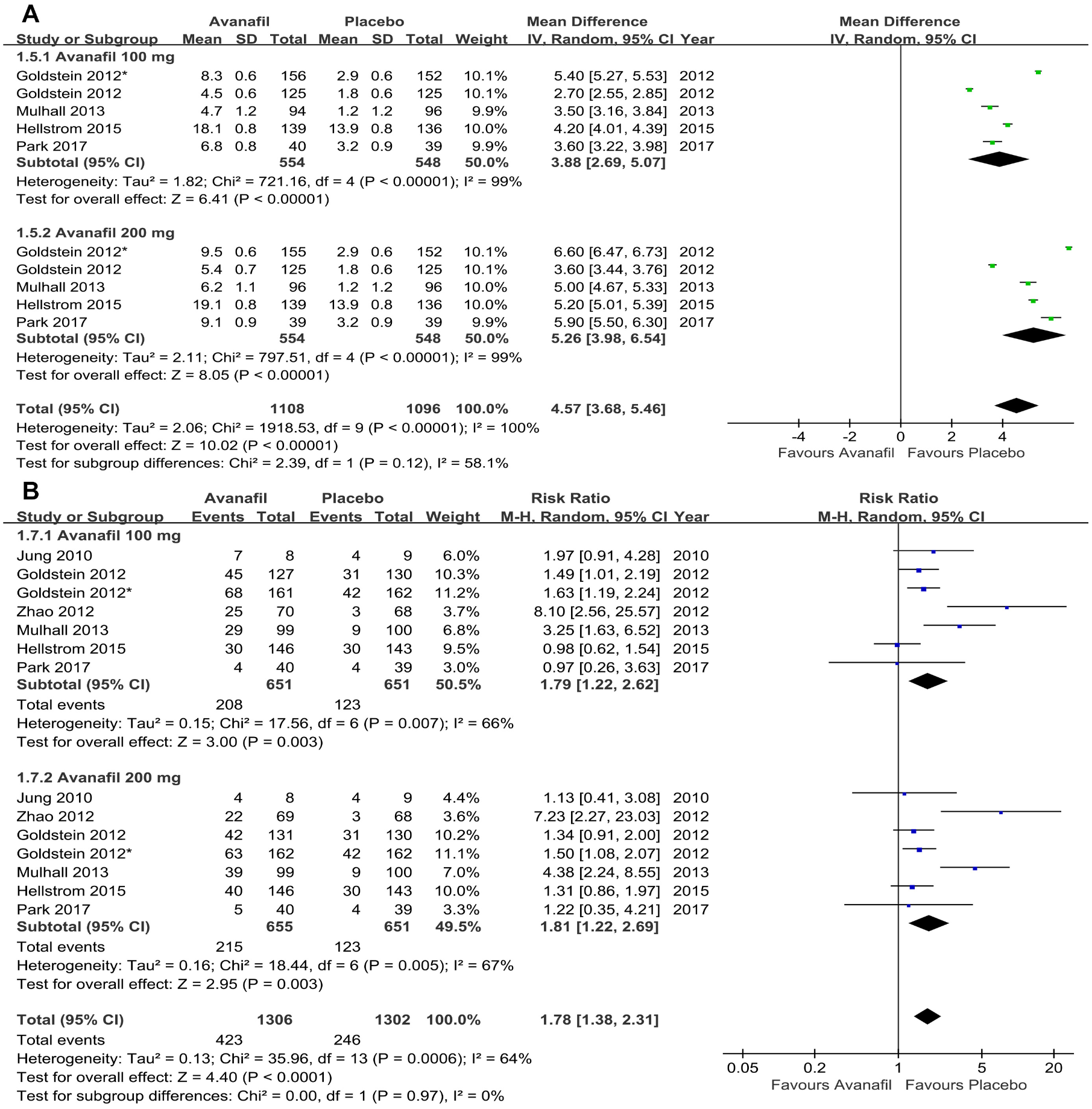
Forest plot for the comparison of the avanafil (100 mg and 200 mg subgroup) and placebo group (
Treatment Adverse Events
In the final statistical analysis, seven RCTs reported the TAEs (Goldstein, Jones, et al., 2012; Goldstein, McCullough, et al., 2012; Hellstrom et al., 2015; Jung et al., 2010; Mulhall et al., 2013; Park et al., 2017; Zhao et al., 2012). The number of TAEs increased significantly in the avanafil group compared with the placebo group (RR = 1.78, 95% CI [1.38, 2.31], p < .0001; Figure 5B). The subgroup analysis suggested that the number of TAEs increased significantly in the 100 mg group compared with the placebo group (RR = 1.79, 95% CI [1.22, 2.62], p = .003). The same results were observed in the 200 mg group (RR = 1.81, 95% CI [1.22, 2.69], p = .003).
Avanafil 100 mg Versus Avanafil 200 mg
This analysis evaluated whether there were differences in efficacy and safety for ED treatment using either 100 mg or 200 mg avanafil (Figure 6). The pooled analysis revealed that avanafil 200 mg were higher than avanafil 100 mg in SI (RR = 0.86, 95% CI [0.75, 0.99], p = .03) and change in IIEF-EF score (MD = −1.34, 95% CI [−1.67, −1.01], p < .001), but there were no differences in SVP (RR = 0.89; 95% CI [0.74, 1.08], p = .27) and TAEs (RR = 0.97, 95% CI [0.83, 1.14], p = .74) between the two doses.
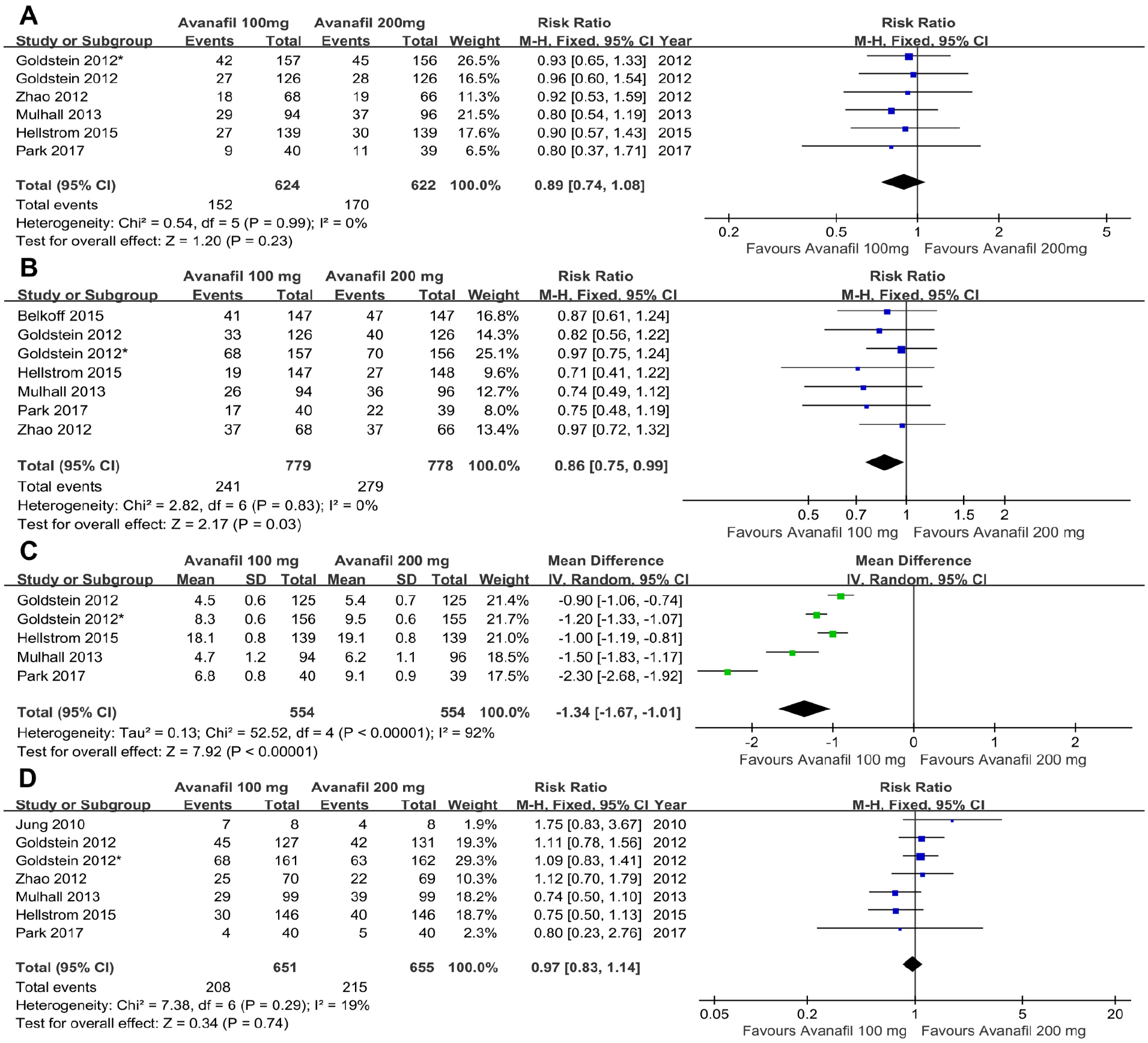
Forest plot for the comparison of the 100 mg and 200 mg avanafil group (
Sensitivity Analysis
Sensitivity analysis was performed to determine the impact of each study data on the final outcomes. There were no significant differences in overall pooled RRs and MDs, regardless of which study was deleted (Figure 7).

Sensitivity analysis. (
Discussion
Although ED is a benign disease, it affects the patient’s body and mental health and is closely related to the patient’s quality of life, sexual relationship, and family stability (Laumann et al., 2005). The treatment of ED can only improve the degree of an erection and alleviate the pain of patients, but it cannot be completely cured. Avanafil is a new type of PDE5-Is. Compared with others, it has good pharmacodynamic and pharmacokinetic effects and has high selectivity for PDE5 isozyme, so there are fewer adverse reactions (Burke & Evans, 2012).
The present study showed that improvement of SVP in ED patients with avanafil at different doses (100 mg and 200 mg) was better than that in placebo patients, which was consistent with the analysis results of Wang et al. (2014) and Corona, Rastrelli, Burri, Jannini, and Maggi (2016). PDE5 is the major reactive enzyme for cGMP catabolism in cells and mediates the signal of erection disappearance. And the PDE5-Is (especially avanafil) are analogs of the cGMP structure, they competitively bind to the catalytic group of PDE5 and inhibit the hydrolysis of cGMP, contributing to the increase of cGMP level, thereby increasing penile blood flow and amplifying the neural signal of erection (Andersson, 2003; Dean & Lue, 2005). It suggested that oral avanafil was an effective method for the treatment of ED. In addition, both Goldstein, McCullough, et al. (2012) and our study proved that there was no statistically significant difference in SVP between avanafil at 100 mg and 200 mg. Consequently, it is debatable whether high doses of avanafil have an advantage in improving SVP in ED patients.
This statistical analysis demonstrated that compared with placebo, avanafil (100 mg and 200 mg) significantly increased the proportion of SI in ED patients. A higher proportion of SI in patients receiving 200 mg of avanafil than in the lower-dose (100 mg) group. A previous meta-analysis incorporating four RCTs concluded that there was no difference in SI between the avanafil 100 mg and 200 mg groups (Cui, Li, Zong, Yan, & Zhang, 2014). There are a variety of reasons for this difference, for example, the study population’s region, race, number, and age. Data from an epidemiological survey in China identified that higher prevalence of ED was observed in adult men with severe smoking, diabetes, and benign prostatic hyperplasia (Zhang, Yang, Li, & Li, 2017), and a recent study suggested that diabetic men with depressive symptoms may have a higher risk of developing ED (Wang, Yang, Cai, Wang, & Weng, 2018). These studies suggested that the treatment of ED patients may be affected by a combination of factors. The study conclusions are reasonable and warrant further objective evaluation of the impact of avanafil on improving SI at different doses (100 mg and 200 mg).
Since its introduction in 1997 (Rosen et al., 1997), the International Index of Erectile Function score (IIEF) has been widely accepted for its sensitivity and specificity in detecting changes in the treatment of ED patients. Hence, the IIEF-EF score has become one of the principal methods to estimate the quality of male ED. Our meta-analysis of change in IIEF-EF score demonstrated that different doses (100 mg or 200 mg) of avanafil were better than the placebo, which was consistent with the results of previous RCTs. Also, subgroup analyses indicated that higher doses (200 mg) of avanafil significantly increased change in IIEF-EF score in ED patients compared with avanafil 100 mg. There is no suspicion that avanafil can increase the IIEF-EF score significantly in patients with ED, and 200 mg of avanafil is better than 100 mg.
This meta-analysis showed a higher incidence of total TAEs in avanafil compared with placebo, and no difference was identified between avanafil 200 mg and 100 mg. According to the results of previous clinical trials, the total TAEs of avanafil were acceptable. In a 12-week phase III clinical study (Goldstein, McCullough, et al., 2012), the overall TAE rates for the avanafil 100 mg group, avanafil 200 mg group, and the placebo group were 42.9%, 38.9%, and 26.1%, respectively. The overall incidence of TAEs in patients receiving avanafil and placebo was 32.4% (423/1036) and 18.9% (246/1302), respectively. Further, the overall occurrence of TAEs was 18.2% (253/1389) and 22.5% (269/1197) in patients taking 100 mg and 200 mg avanafil, respectively. The most frequently reported adverse events included headache, flushing, nasal congestion, nasopharyngitis, and back pain (Belkoff et al., 2013; Goldstein, McCullough, et al., 2012; Hellstrom et al., 2012). It is gratifying to note that the extent of adverse events is relatively mild, patients can tolerate them, and there are few reports of serious complications in all studies.
Swearingen and his colleagues reported that compared with sildenafil, sublingual nitroglycerin had a lesser effect on blood pressure and heart rate after oral administration of avanafil for 1 h (p < .05); the adverse events associated with a clinically significant reduction in systolic blood pressure (≥30 mmHg) induced by avanafil were less common than sildenafil (15% vs 29%, p < .05; Swearingen, Nehra, Morelos, & Peterson, 2013). Another study also reported that avanafil is 120-fold and 10,000-fold more selective for PDE5 than PDE6 and PDE1, while vardenafil is 21-fold and 1,000-fold and sildenafil is 16-fold and 380-fold (Kedia, Uckert, Assadi-Pour, Kuczyk, & Albrecht, 2013). There is a reason to believe that avanafil is a wise choice for patients who cannot tolerate TAEs to sildenafil and vardenafil.
There are certain limitations in the present analysis: (a) part of the study has a small sample size; (b) because the etiology of ED in some studies differs in the severity of ED and the expected response to the drug, the potential heterogeneity of the subjects used in the meta-analysis may be greater; (c) the follow-up period in the eight studies was shorter. Besides, most of the RCTs included did not explicitly describe allocation concealment.
Conclusion
The current evidence suggests that the SVP, SI, and IIEF-EF score in men who received avanafil improved significantly. Although some TAEs exist, they are within the tolerable range of patients. In summary, avanafil (especially 200 mg) has the potential to be the drug of choice for the treatment of ED, but more strict and larger sample size RCTs are needed to verify the findings of this meta-analysis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
