Abstract
This study aims to determine main health-related anthropometric and metabolic parameters in young physically active male students in their first year of study. Sixty one male students from a Polish university of physical education were studied twice–at the beginning of their first and second academic years. Body weight, height, waist circumference, and body composition were measured. Plasma levels of thyroid-stimulating hormone (TSH), leptin, cortisol, and insulin, as well as insulin resistance (IR) using the homeostatic model assessment (HOMA) were determined. In 64% of the participants body weight and body mass index (BMI) significantly increased during the study. An average students’ weight gain amounted to 1.2 kg. In most of the subjects it resulted from the increase of fat-free mass (r = .886, p < .001). TSH and leptin levels did not change, but a significant increase of cortisol levels and a tendency toward decreased insulin levels and also HOMA-IR values were observed. However, there were no significant correlations identified between the changes in anthropometric and biochemical parameters. The study revealed that body weight gain seen in male students during their first year at the university of physical education was mainly due to the increase in fat-free mass. Apart from anthropometrics, changes in their insulin and cortisol levels were also detected, but further studies are required to evaluate these findings. The documented insufficient energy and carbohydrates consumption in physically active male students proves the need for nutritional education among them.
Obesity and obesity-related diseases constitute a major public health problem in many countries and therefore it is important to establish risk periods of weight gain and development of unhealthy behaviors. According to the recent data, the greatest increases in obesity rates have been observed among people aged 18–29 years and the first year of university could be seen as a critical period for the risk of weight gain among young adults (Crombie, Ilich, Dutton, Panton, & Abood, 2009; Gropper, Simmons, Connell, & Ulrich, 2012). Some studies report that almost one-quarter of first-year students gained about 1.3–3.1 kg of their weight (Pullman, et al., 2009; Wengreen & Moncur, 2009).
The phenomenon of weight gain during a student’s first year at university can be explained by lifestyle-related changes that may lead to weight increase. Students start living independently, away from their family homes. This transition is frequently accompanied by unhealthy behaviors including decreased physical activity, improper nutrition, increased alcohol consumption, and decreased sleep duration (Keller, Maddock, Hannöver, Thyrian, & Basler, 2008; Poortinga, 2007). Previous studies report high frequency of such behaviors among university students (Deforche, Van Dyck, Deliens, & De Bourdeaudhuij, 2015; Racette, Deusinger, Strube, Highstein, & Deusinger, 2008). It still has not been fully elucidated which of these factors are associated with weight gain in young adults. Several studies present insufficient amounts of physical activity as the most important contributor to weight increase in students (Jung, Bray, & Martin-Ginis, 2008; Pribis, Burtnack, McKenzie, & Thayer, 2010). In contrast, other researches demonstrate that weight gain is mainly a consequence of the increase in sitting time while studying (Salmon, Tremblay, Marshall, & Hume, 2011). It has also been observed that a reduced fruit and vegetables intake and increased consumption of unhealthy foods like high-fat foods, sweet snacks, alcohol drinks, and others are associated with students’ weight gain (Economos, Hildebrandt, & Hyatt, 2008). Previous studies have also documented that the amount of weight gain in young adults depends on their sex. Being a male is the strongest predictor of weight gain (Cluskey & Grobe, 2009; Girz et al., 2013).
It is worth noting that most of the existing studies related to weight gain in young people were conducted in the United States and Canada and focused only on total body mass and BMI changes (Gropper et al., 2011; Racette, Deusinger, Strube, Highstein, & Deusinger, 2005; Vella-Zarb & Elgar, 2010). Very little is known about body weight and body composition (e.g., fat and fat-free mass) changes in the population of European students (Deliens, Clarys, Van Hecke, De Bourdeaudhuij, & Deforche, 2013; Serlachius, Hamer, & Wardle, 2007). Metabolic consequences of changes in students’ weight and body composition have not been widely studied so far. Previous research points to the fact that changes in weight and body composition cause a number of metabolic effects. The most important for health and body fitness are changes in the metabolism of carbohydrates and lipids related to the altered secretion of such hormones as TSH, leptin, cortisol, and insulin (Bouchard, Tchernof, & Tremblay, 2014; Kitahara et al., 2012; Liao, et al., 2015).
Limited data are available on the changes in body weight and composition in individuals who commenced their studies at a university of physical education. The program of such studies, in addition to theoretical classes, contains many practical classes compelling students to major physical effort. Young people who choose to study physical education become future professional teachers of physical education, sport coaches, instructors of recreational and sport activities, or sports and recreation organizers. It could be expected that subjects who are regularly engaged in regular physical activities would be at a low risk of weight gain during their studies. Various weight gain risk factors linked with the transition to higher education such as changes in the place of residence and new company still apply to this group of students. Some previous studies showed that unhealthy eating behaviors were often observed among physically active students (Vadeboncoeur, Townsend, & Foster, 2015; Wierniuk & Włodarek, 2014). Due to the risk factors that still affect students despite their regular physical activity, the purpose of this study was to assess the changes in main health-related anthropometric and metabolic parameters in young physically active male students during their first year at university.
Materials and Methods
Study Design and Participants
The study was conducted between 2008 and 2009 at the University of Physical Education in Warsaw, Poland. There were 260 students (220 males, 40 females) enrolled in the Faculty of Physical Education in 2008. Recruitment for the study was conducted in the form of advertisements in student dormitories and by word of mouth. Only male students who were not engaged in competitive sport were eligible to participate in the research. Each subject was informed about the purpose and procedures of the study and provided written consent to it. The research protocol was accepted by the ethics committee of the Józef Piłsudski University of Physical Education in Warsaw.
The first assessment was completed by 87 out of the 100 students who agreed to take part in the study and 61 subjects completed the second one. This was the number which was considered in the final statistical analysis. The participants aged 18–23 years were healthy, nonsmoking, and on no medication. All the measurements were taken twice–at the beginning of the first (baseline) and second (follow-up) years of study. Students’ weekly number of obligatory physical activity hours (including swimming, running, games, and martial arts) varied from 5 to 7 hr.
Anthropometric Measurements
Basic anthropometric parameters were taken on both occasions. Students’ weight was measured with the accuracy of 0.1 kg and height with the accuracy of 0.1 cm according to international standards. BMI was calculated as weight (kg)/height (m2). Body components, that is, body fat (FM) and fat-free mass (FFM), were assessed by Bioelectrical Impedance Analysis (BIA) using Body Composition Analyzer TANITA BC-418 MA (Tanita Co., Japan). Inter- and intra-assay coefficients of variability for body fat measurements did not exceed 5%. Waist circumference was measured to the nearest 0.1 cm using a nonstretchable tape. Anthropometric measurements were taken in all the study participants by the same person in the morning with the subjects wearing light sports clothes without shoes.
Biochemical Analyses
Blood samples were taken from an antecubital vein between 7:30 and 9:00 a.m. in fasting conditions and were collected into lithium heparin tubes using disposable syringes and needles. The samples were centrifuged (15 min, 4,000 rpm, 4 °C) and the plasma was stored at −70 °C until analysis.
Plasma levels of TSH, cortisol, leptin, and insulin were determined by standard radioimmunoassay methods using BioSource commercial kits (Belgium). Inter- and intra-assay coefficients of variation for hormones did not exceed 7%. Blood glucose levels were determined by the oxidase method, using Randox commercial kits (Randox Laboratories, UK). Inter- and intra-assay coefficients of variability for glucose determination did not exceed 5%.
Insulin resistance (IR) was evaluated using the homeostasis assessment model (HOMA-IR) and calculated from fasting insulin and glucose concentration according to the formula: insulin (µIU/mL) x glucose (mmol/L)/22.5 (Matthews et al., 1985).
Energy Expenditure
Physical activity was briefly determined at both time points by an experienced interviewer using the Seven-Day Physical Activity Recall Questionnaire (SDPAR) (Sallis, 1990). The SDPAR is a detailed quantitative questionnaire which was found to be reliable for the assessment of different dimensions of physical activity and to be a useful tool in lifestyle-dependent diseases preventive screening (Czeczelewska, Czeczelewski, Wasiluk, & Saczuk, 2016).
Data concerning the duration, frequency, and intensity of various activities (occupational and leisure, sport participation, active transportation) within a week were collected. Light, moderate, hard, and very hard physical activity was defined as MET 1.5, 4, 6, and 10, respectively, and finally expressed in kcal/day. Because, apart from the data concerning physical activity, the SDPAR takes into account energy expenditure during sleep, it was possible to estimate the total daily energy expenditure (DEE, kcal/day) of the participants.
Energy Intake
The assessment of energy intake (EI) was done using a 24-hr food record provided by each appropriately instructed participant. The dietary information contained all foods and drinks consumed in 4 days (2 weekdays and the weekend) preceding blood collection and anthropometric measurements. The information was validated by a trained interviewer who established the exact size of meals using “Album of Photographs of Food Products and Meals” (Szponar, Wolnicka, & Rychlik, 2000). Subsequently, the household measures of food intake were converted into grams and the diet composition was analyzed using the computer program Dieta 5.0 designed by the National Food and Nutrition Institute in Warsaw (Poland).
Statistical Analyses
The results were analyzed using “STATISTICA” for Windows, v.12.0. In order to assess the changes in anthropometric and biochemical data between the first (baseline) and the second year of the study (follow-up), Repeated Measures ANOVA analyses were conducted. The magnitude of changes was presented by standardization of the variables from the follow-up measurement against mean and SD values of the baseline measurement. This procedure gives normalized mean value equal to zero and standard deviation equal to one. As a result, for each variable the same normalized values were obtained, regardless of the scale in which they were expressed without normalization. The normalized values were also used to analyze the correlation of each anthropometric and biochemical variable changes. Correlation analysis was carried out using Pearson test.
Results
Mean values ± SD of the analyzed data from the first and second years of the study are reported in Table 1, while the direction and magnitude of the changes in variables are shown in Figure 1. It was observed that body weight and BMI of the participants slightly but significantly increased during the study (F(1.60) = 5.96, p = .018, and F(1.60) = 6.21, p = .016, respectively). There were no statistically significant changes in body fat content and in waist circumference, however a significant increase in fat-free mass (F(1.60) = 15.7, p < .001) was observed. On the basis of standardization, it was determined that body weight and BMI increase occurred in 39 (64 %) participants (Figure 1). Correlation analysis proved that in 19 subjects (31.2 %) the increase of body mass was significantly related to the rise of fat mass (Figure 2A), while for a much larger number of students (30 subjects, 49.2%) it was a consequence of the increase in fat free mass (Figure 2B).
Characteristics of Participants in the First and Second Year of the Study (Mean ± SD).

Note. BMI = body mass index; %F = body fat percentage; FM = fat mass; FFM = fat-free mass; TSH = thyroid-stimulating hormone; DEE = daily energy expenditure; EI = energy intake; HOMA-IR = homeostatic model assessment for insulin resistance.
Trend toward significance (p < .1).
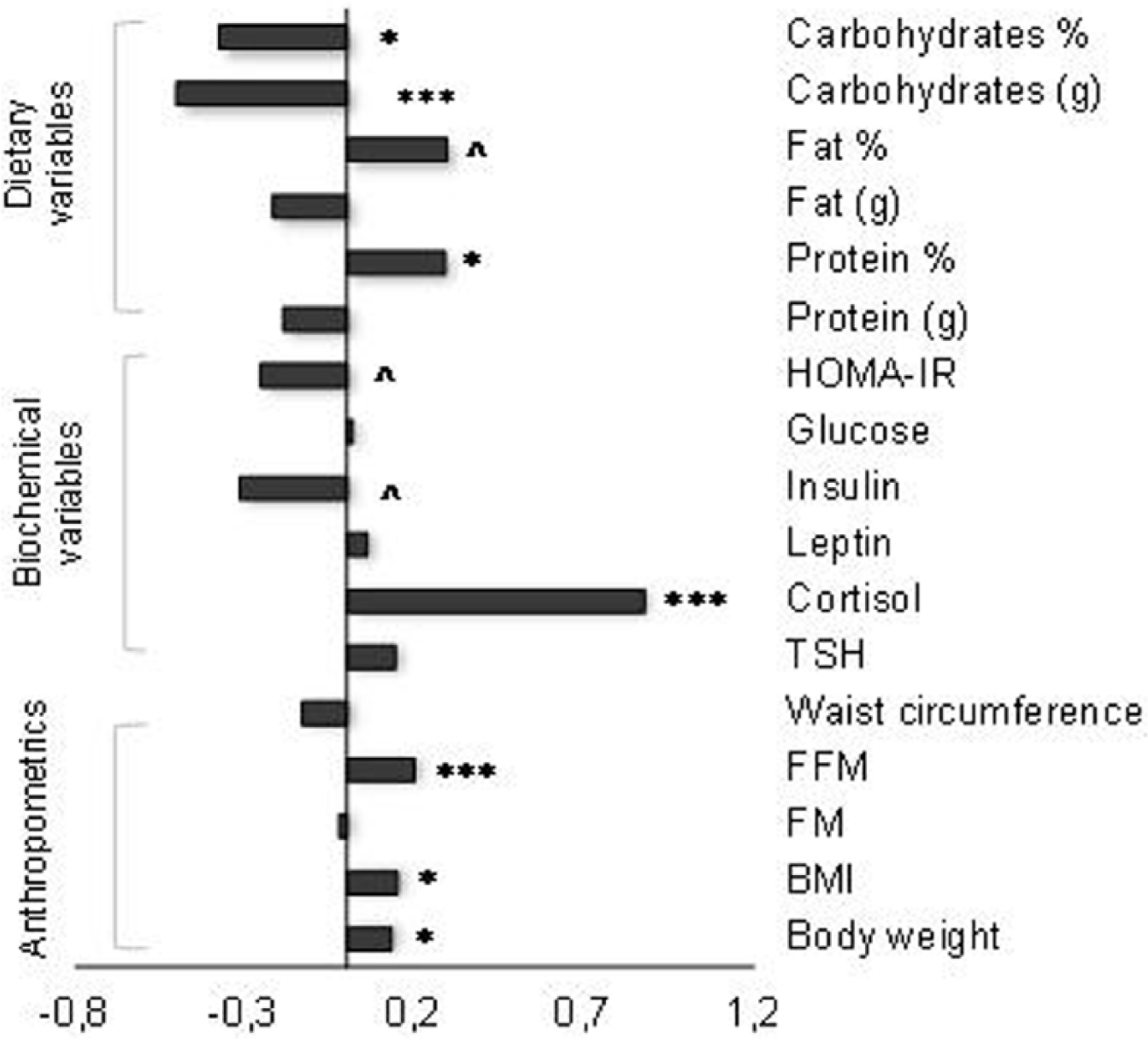
Direction of the changes in anthropometric, dietary and biochemical variables. *-p < .05; **-p < .01; ***-p < .001; ^-trend toward significance (p < .1).

Correlation between changes of body weight (BW) and fat mass (FM) (A) and fat-free mass (FFM) (B). ***-p < .001.
The biggest change of biochemical variables concerned the increase in cortisol level by 36.8% versus baseline values (Table 1). This increase affected 46 (75%) students. TSH and leptin levels did not change significantly during the study while mean insulin levels lowered from 6.8 µIU/ml to 6.0 µIU/ml, but it was only a trend toward significance (p < .1) (Table 1). Lower insulin levels in follow-up assessment were found in 38 (62%) participants (Figure 1). Also HOMA-IR values decreased by 12% in the second year of study (p < .105) and were observed in 39 (64%) students.
Although total daily energy expenditure did not change during the study, a significant decrease in students’ EI in the follow-up assessment was noted (F(1.60) = 10.08; p = .002). In addition, it was observed that within a year protein content in students’ diet significantly increased by 5.5%, while carbohydrates intake (in % and g) significantly decreased (by 14.5% and 6.4%, respectively). An increase in protein content in the diet was observed in 41 (67%) students, whereas a fall in carbohydrates consumption in grams in 37 (61%), and their share in the diet in 35 (57%) respondents (Figure 1).
There were no significant correlations found between the changes in anthropometric and biochemical parameters. Also, no interdependences were detected between dietary alterations and the studied metabolic indices.
Discussion
Prior research has demonstrated that regular physical activity is health enhancing, whereas physical inactivity is a major risk factor of chronic diseases related to obesity (Sacheck, Kuder, & Economos, 2010; Zaccagni, Barbieri, & Gualdi-Russo, 2014). Physical activity minimizes the risk of the aforementioned diseases by reducing body weight and changing its composition to decrease body fat content and increase fat free mass. Exercise-induced changes in body components in turn provide a number of significant metabolic benefits mainly due to the reduced amount of fuel stored in adiposities, as well as modification of the hormonal regulation of lipids and carbohydrates metabolism (Bishop-Bailey, 2013).
In this prospective study, 61 physically active male students were followed from the first until the second year of their university studies in order to assess changes in body composition and their influence on selected metabolic parameters. It was observed that, on average, students gained 1.2 kg of body weight during their first year of study. Comparable weight gain was demonstrated by Deliens et al. (2013) who examined weight and also body composition changes among first-year university students. They reported that after the first semester at university, Belgian students gained an average of 1.0 kg. However, the weight gain in their research mainly referred to the increase in fat mass rather than fat-free mass. In contrast, it was observed that in physical education students the first-year weight gain in most cases resulted from the increase in fat-free mass. The current study demonstrated that students’ participation in obligatory physical activity classes included in the university program protects them against the increase of body fat observed in their peers commencing studies in other fields. This positive change in body composition also means that higher weight and BMI values observed in the participants after one year of studying do not necessarily indicate any bigger health risks associated with overweight.
Previous research data highlight the fact that big waist circumference is a stronger than BMI causative factor of obesity-related diseases (i.e., cardiovascular disease, diabetes mellitus) (Balkau, et al., 2007). There were no significant changes observed in the waist circumference in participants of the present study. This result also confirms the observation that most of their weight gain was due to the increase in fat-free mass. Hence, these findings are in line with the studies reporting that BMI is not an appropriate risk predictor of obesity-related diseases in physically active individuals because it does not give any information about body fat content or fat distribution, which are usually different in active subjects in comparison to general population (De Lorenzo, et al., 2013).
Apart from the body mass and composition changes, several interesting changes in selected biochemical variables were also detected. Undoubtedly, the observed tendency of decreasing insulin levels and HOMA-IR values in most of the participants was a health-positive change. Insulin is the most important hormone in regulating plasma glucose level and HOMA-IR is a marker of IR (i.e., the responsiveness of insulin-sensitive tissues to the metabolic actions of this hormone) (Wallace & Matthews, 2002). It was proved that resistance to insulin-stimulated glucose uptake in adipose tissue, liver, and muscle preceded type 2 diabetes (Bano, 2013). Moreover, IR also interferes with lipid metabolism leading to atherosclerosis, hypertension, and cardiovascular disease (CVD) (Voulgari et al., 2011). So, the tendency of declining HOMA-IR values noted in participants of the current study can indicate a lower risk of aforementioned disturbances in the studied population. These findings are in agreement with other studies indicating that physical activity improves insulin sensitivity (Conn et al., 2015; O’Gorman & Krook, 2008). There are studies demonstrating that exercise-induced increase in insulin sensitivity has been linked with increased fat-free mass (Srikanthan & Karlamangla, 2011; Van Der Heijden et al., 2010). However, this observation cannot be clearly confirmed since there were no significant correlations between HOMA-IR values and anthropometric parameters established in the present research.
Another interesting result of this study is the observation that in 75% of young men plasma cortisol levels increased by almost 37% during their first year at the university of physical education. It has been documented that long-term, even mildly elevated cortisol levels can have detrimental effects on insulin production in pancreas and may significantly increase fat accumulation, diminish skeletal muscle mass, and alter carbohydrate and lipid metabolism (Pervanidou & Chrousos, 2012). Given that, the increased cortisol levels discovered in the second assessment of the study must be regarded as unfavorable.
Several factors could be responsible for this change. Data reporting increased cortisol levels in obese people and those with anorexia nervosa confirm the fact that physiological stress such as disruption in energy balance could alter cortisol secretion (Douyon & Schteingart, 2002). It is worth noting that despite no changes in energy expenditure, a significant decrease in EI was noted in students of the present study. The percentage of subjects with energy deficit increased from 16.4% at baseline to 34.4% at follow-up. Moreover, a significant decrease in their carbohydrates intake was observed, what according to some authors could also affect cortisol levels (Stimson et al., 2007). However, there were no significant correlations between energy deficit, macronutrients intake and cortisol levels. Perhaps the observed change in subjects’ diet composition was insufficient. Lack of significant changes in body fat and other analyzed metabolic parameters, that is, leptin and TSH, seems to confirm this suggestion. It has been proved that disruption in energy balance and changes in body composition are usually associated with altered secretion of cortisol and also leptin and TSH (Roef et al., 2012). Nevertheless, it is worth noting that insufficient caloric and carbohydrates consumption in young physically active men could have adverse health and fitness consequences. Low energy availability may result in muscle mass loss and lower bone density. It may also lead to an increased risk of injury, fatigue or other health problems (Mountjoy et al., 2014). Additionally, low or moderate reduction in carbohydrates consumption increases fat and protein intakes. Although carbohydrates reduction is actually a popular dietary trend, especially among people reducing their weight, many researches suggest that its potential health effects are unknown (Nilsson et al., 2013).
Lastly, the research concerning patients with anorexia nervosa demonstrated that their raised cortisol levels were partly associated with depression (Misra & Klibanski, 2014). It means that not only physiological but also psychological stressors have impact on plasma cortisol levels. The first year of university is certainly a period of increased stress for students. However, mental stress level was not measured in the current study and therefore its effect on plasma cortisol levels cannot be assessed.
A possible limitation of the current study could be the self-report of diet by the study participants. There are examples in literature pointing to the underestimation of food intake by physically active persons (Jonnalagadda, Benardot, & Dill, 2000; Rasmussen, Matthiessen, Biltoft-Jensen, & Tetens, 2007). In addition, due to the complexity of the study protocol, the size of the study group was modest. However, considering the obtained results further research regarding anthropometric and metabolic changes in young male students on larger sample groups is recommended. Finally, this study was limited to a student population from a single university and therefore it would be worth checking if these findings apply to a larger and more varied student population.
In summary, this study documents that body weight gain in male students during their first year at a university of physical education is mainly due to the increase in fat-free mass. Apart from anthropometrics, changes in insulin and cortisol secretion were also observed but further studies are required to evaluate these findings. The noted inadequate energy and carbohydrates consumption in physical activity students points to the need for nutritional education among them. Physically active people should be aware that proper energy supply in diet, coming mainly from carbohydrates, is necessary to maintain health and high physical fitness.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was jointly supported by the grants DS-132 and DS-147 from the Józef Piłsudski University of Physical Education, Warsaw, Poland.
