Abstract
Metabolic syndrome (MetS), a cluster of cardiometabolic risk factors, is a challenging public health issue. The aim of current study was to test the hypothesis that concentrations of plasma adropin and leptin differ between patients with MetS and comparable age- and sex-matched control groups. This case-control study involved 153 subjects (51 per group). The study group included obese subjects with MetS and the two control groups included weight-matched subjects without MetS (“healthy”: obese) and normal weight subjects without MetS. Body composition parameters were measured using bioelectrical impedance analysis. Plasma levels of adropin, leptin, and their ratio were measured. Leptin was significantly different between obese patients with/without MetS groups and normal weight subjects. Patients with MetS had higher levels of leptin (14 ± 12.4) compared with those without MetS (11.2 ± 9.3 vs. 7 ± 7.1 obese and normal weight without MetS, respectively; p = .002). Compared with healthy obese and normal weight subjects, MetS subjects had lower levels of plasma adropin (p < .001) and a lower plasma adropin to leptin ratio (p < .001), which remained significant when adjusted for body fat mass by analysis of covariance (p < .001). This study demonstrates low levels of adropin are correlated with MetS and hence identify it as a potentially protective agent against MetS development. Variation in adropin levels may partly explain the “healthy obese” phenomenon.
Introduction
Metabolic syndrome (MetS) or insulin resistance syndrome is characterized by the concurrent presence of several cardiometabolic risk factors including central obesity, dyslipidemia, insulin resistance, glucose intolerance, and hypertension (Grundy, Hansen, Smith, Cleeman, & Kahn, 2004; Hanson, Imperatore, Bennett, & Knowler, 2002). This combination of risk factors dramatically increases the risk of type 2 diabetes mellitus and coronary heart disease (Lloyd-Jones et al., 2010; Sarti & Gallagher, 2006). The prevalence of MetS is rising rapidly worldwide (Expert Panel on Detection, 2001). Globally, approximately 20% to 25% of adults already have MetS (Cameron, Shaw, & Zimmet, 2004; Ford, Giles, & Mokdad, 2004). According to the Tehran Lipid and Glucose Study, the prevalence of MetS is 42% in adult Iranian women and 24% in Iranian men (Azizi et al., 2003). These prevalence rates are among the highest in the world (Azizi et al., 2010). Due to unclear underlying pathophysiologic processes that underpin its development and confusion between the conceptual definitions, MetS continues to be a source of medical controversy (Kocaoglu, Buyukinan, Erdem, & Ozel, 2015).
Obesity is known as the most important characteristic of the MetS and often precedes MetS. In fact, MetS is sometimes considered as a consequence of obesity. Although the prevalence of MetS increases with obesity, MetS can be present in lean individuals (Hanley et al., 2002). A proportion of obese patients do not develop MetS (Hanley et al., 2002). Such subjects have been referred to as the “healthy obese.” Although there has been a large research effort directed at understanding the molecular mechanisms that underpin MetS, such mechanisms remain unclear. Hormonal signals related to body fat, such as leptin, may underpin the development of MetS (Butler et al., 2012; Chiu et al., 2012; Kocaoglu et al., 2015; Mirza et al., 2011).
Adropin is a hormone that was first identified by Kumar et al. in 2008. It is encoded by the energy homeostasis–associated gene that is expressed in the liver and the brain (Kumar et al., 2008). Low adropin levels may associate with a higher risk of obesity, insulin resistance, and probably MetS (Kumar et al., 2008). In human subjects, a previous study has reported low levels of plasma adropin in diabetic patients (Wu et al., 2014). It has also been identified that adropin uniquely affects the regulation of endothelial function; impaired endothelial function contributes to the development and progression of diverse metabolic diseases (Lovren et al., 2010). Aydin (2014) reported evidence of the relationship of adropin with energy regulation.
Leptin, an adipocytokine, plays critical roles in regulating food intake (Mantzoros et al., 1996), energy expenditure (Stehling, Doring, Ertl, Preibisch, & Schmidt, 1996), and body weight (Mantzoros et al., 2011). Increases of leptin levels in plasma, suggesting leptin resistance, is an early manifestation of MetS (Zimmet, Boyko, Collier, & Courten, 1999). Serum leptin levels in patients with MetS are higher than those in healthy subjects (Özçelik et al., 2010). An increased leptin level predicts MetS development independent of body mass index (BMI; Franks et al., 2005).
Adropin and leptin have not been studied together in obese patients with MetS compared with obese patients without MetS and normal subjects, and consequently, their possible combined roles in relation to MetS are unknown. The aim of current study was to test the hypothesis that concentrations of plasma adropin and leptin differ between patients with MetS and comparable age- and sex-matched control groups.
Materials and Method
Subjects
The current study was a case-control study comprising 51 cases and 102 controls. The cases, patients newly diagnosed with MetS and having never received medications, were recruited from patients referred to the Endocrinology Center of Tehran University of Medical Sciences. The control groups were age and gender matched to the cases, made up of 51 weight-matched overweight/obese subjects without MetS the “healthy obese” and 51 normal weight subjects (BMI 18-25) without MetS selected from those attending the center for routine medical care. All subjects provided written informed consent to participate in the study. Subjects were informed in detail about the study purpose before completing their written informed consent. The research protocol of the study was reviewed and approved by the scientific research committee of Tehran University of Medical Science.
MetS was defined using the National Cholesterol Education Program–Adult Treatment Panel III (NCEP-ATP III) criteria. The NCEP-ATP III definition requires the presence of three or more of the following: (a) waist circumference (WC) greater than or equal to 102 cm for men and greater than or equal to 88 cm for women, (b) triglyceride (TG) level greater than or equal to 150 mg/dL, (c) high-density lipoprotein–cholesterol (HDL-c) cholesterol less than 40 mg/dL for men and less than 50 mg/dL for women, (d) systolic blood pressure (SBP) greater than or equal to 130 and/or diastolic blood pressure 85 mmHg, and (e) fasting blood sugar (FBS) greater than or equal to 100 mg/dL.
Exclusion criteria included having a family history of coronary artery disease, acute or chronic renal failure, acute infection within the previous 7 days, acute or chronic hepatic failure, hematological disorder, presence of any chronic inflammatory and autoimmune disease, and any known malignancy. Other nonpathological exclusion criteria included pregnancy, breastfeeding, postmenopause, smoking, professional athlete, thyroid disorder, use of medications for dyslipidemia or hypertension, hypnotics, sedatives and immunosuppressive, or having a special diet for any reason prescribed by the clinic dietitian.
Hormonal Assays
Blood samples were collected in the morning, after 8 to 12 hours of overnight fasting and 20 minutes of supine rest. Venous blood was drawn into ethylenediaminetetraacetic acid (EDTA) tubes and promptly centrifuged at 4 °C. Plasma was frozen at −80 °C until analyses of adropin and leptin were performed. FBS was measured by an automated glucose oxidase method (Automatic Analyzer 2700, Olympus, Japan). TG and HDL-c were measured by glycerol phosphate oxidase and phosphotungstic acid (Pars Azmoon Inc., Tehran, Iran), respectively.
Adropin and Leptin
The plasma adropin levels were measured using a commercial ELISA kit (Cusabio Biotech Co, Wuhan, CN) according to the manufacturer’s instructions. The assay recognizes recombinant and natural human adropin. No significant cross-reactivity or interference was observed. Test range was 1.56 pg/mL to 100 pg/mL. The sensitivity of the assay was 0.39 pg/mL, and interassay and intraassay coefficients of variation were less than 10% and 8%, respectively. Plasma samples required 500-fold dilution. A commercial ELISA kit for human leptin (Mediagnost, Germany) was used and the assay was conducted according to the manufacturer’s instructions. The sensitivity of the assay was 0.2 ng/mL, and interassay and intraassay coefficients of variation were less than 10% and 7%, respectively.
Statistical Analysis
All values were expressed as the mean ± SD. Continuous variables were analyzed by one-way analysis of variance (ANOVA) to compare the differences among the three groups. If the result of the ANOVA test was significant, a least significant difference test was used to locate which of the means differed. Multiple linear regression with backward method was used for assessing the association between adropin, leptin, and related factors. Also, the receiver operator characteristic curve (ROC curves) was used for determining the appropriate cutoff point of adropin in diagnosing MetS. For all analyses, a two-tailed p < .05 was considered statistically significant. All of the analyses were performed using SPSS for Windows (version 16.0; SPSS Inc., Chicago, IL, USA).
Results
Subjects
MetS characteristics and general characteristics of the study population are reported in Table 1. Most subjects in all groups were 44 (86.3%) men and 7 (13.7%) women in each group. In terms of age, sex, and marital status, there were no statistically significant differences between the three groups, indicating the matching was successful. MetS components, including blood pressure, TG, FBS, and WC were significantly higher among MetS patients compared with both weight-matched and nonweight-matched control subjects, while HDL-c was lowest in the case patients. No significant difference was observed for MetS components between the two control groups (Table 1).
General Characteristics and MetS Parameters of Subjects Based on Study Groups.*

Note. MetS = metabolic syndrome; WC = waist circumference; BMI = body mass index; SBP = systolic blood pressure; DBP = diastolic blood pressure; FBS = fasting blood sugar; TG = triglyceride; HDL = high-density lipoprotein. Dissimilar values (superscripts a, b, c) of each row are significantly different.
Values are analyzed by one-way analysis of variance; values are mean ± SD.
Body Composition
The mean of the anthropometric measurements including total and regional body composition of subjects for all three groups are displayed in Table 2. The obese groups with and without MetS had a significantly higher total and regional body fat and lower fat-free mass compared with the lean control group, while no significant differences were observed for body fat and fat-free mass between the MetS group and the overweight/obese matched controls without MetS.
Comparison of Total and Regional Body Composition Among Case and Control Groups.*
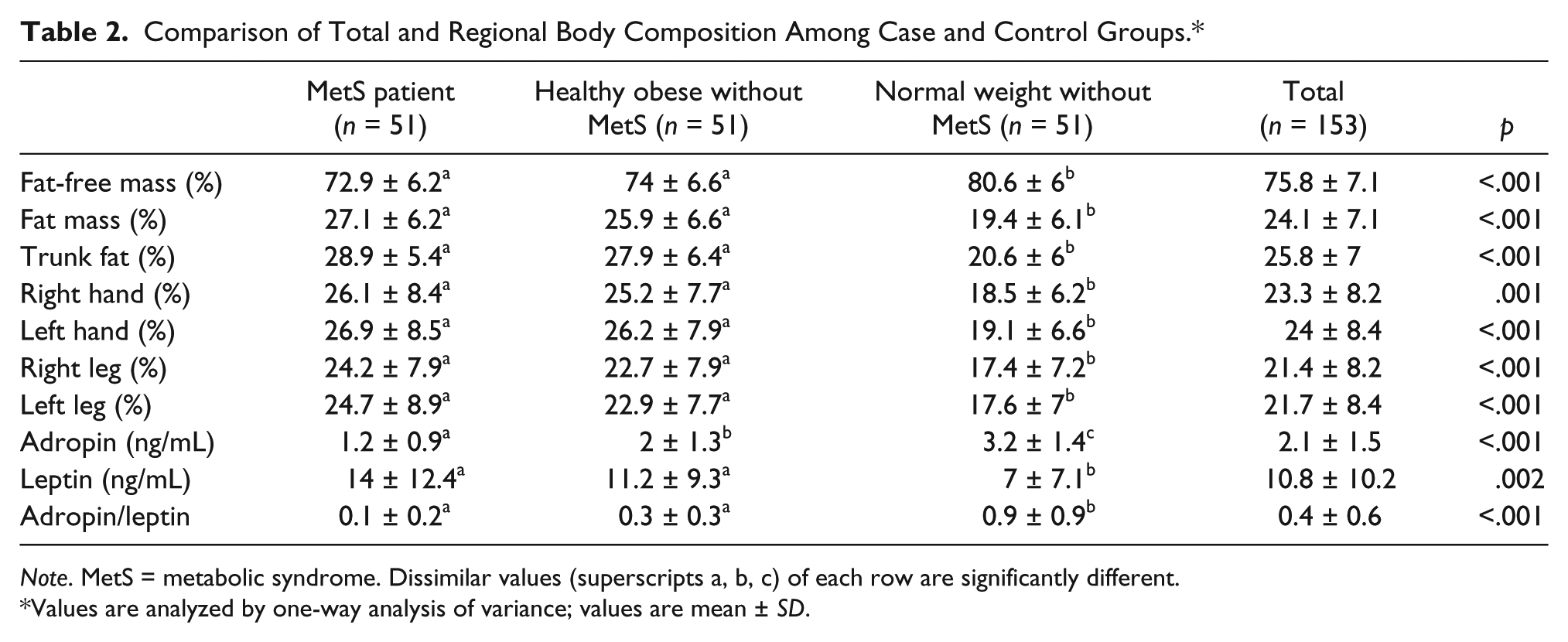
Note. MetS = metabolic syndrome. Dissimilar values (superscripts a, b, c) of each row are significantly different.
Values are analyzed by one-way analysis of variance; values are mean ± SD.
Adropin, Leptin, and Adropin/Leptin Ratio
MetS patients had an average adropin concentration of 1.2 ± 0.9 ng/mL which was significantly lower compared with the average concentrations of 2 ± 1.3 ng/mL in the obese group and 3.2 ± 1.4 ng/mL in the normal weight group (p < .05). Subjects with normal weight had the highest adropin levels, while the lowest level of adropin was seen in patients with MetS (p < .05). After adjustment for fat content, there was still a significant association between adropin and the study groups (analysis of covariance [ANCOVA]: p < .001) so that the adjusted mean of adropin in MetS patients was still significantly lower compared with average concentrations in the obese and normal weight groups (Tables 3 and 4).
Analysis of Covariance for Assessing Association Between Adropin and Leptin With Study Groups.*

Estimated Marginal Means of Adropin and Leptin Among Case and Control Groups.*

Note. MetS = metabolic syndrome. Dissimilar values (superscripts a, b, c) of each row are significantly different.
Values are analyzed by analysis of covariance; values are mean ± SD.
In one-way ANOVA, leptin was significantly different between obese patients with/without MetS groups and normal weight subjects. Patients with MetS had higher levels of leptin (14 ± 12.4) compared with those without MetS (11.2 ± 9.3 vs. 7 ± 7.1 obese and normal weight without MetS, respectively; p = .002; Table 2). After adjustment for fat content, there was still a significant association between leptin and study groups (ANCOVA: p = .015) so that the adjusted mean level of circulating leptin in the normal weight group was significantly higher compared with average concentrations in the obese and MetS patient groups (Tables 3 and 4).
The adropin/leptin ratio (ng/mL adropin:ng/mL leptin) was 0.1 ± 0.2, 0.3 ± 0.3, and 0.9 ± 0.9 in MetS patients, obese non-MetS and normal weight subjects without MetS, respectively. The obese groups with and without MetS had a significantly lower adropin/leptin ratio compared with the normal weight group (ANOVA: p < .0001; Table 2). After adjustment for fat content, there was still a significant association between study groups for adropin/leptin ratio (ANCOVA: p = .001) so that the obese groups with and without MetS still had a significantly lower adropin/leptin ratio compared with the normal weight group (Tables 3 and 4).
ROC curves and area under the curve for determining the ability of adropin in diagnosing of MetS are reported in Figure 1. The area under the ROC curve was 0.79 for adropin levels (p = .001). The sensitivity and specificity values of adropin levels were 83% and 76%, respectively (cutoff value 1.62).

The ROC curve for determining the adropin cutoff point in diagnosing of MetS.
Linear Regression Analysis of Adropin Levels Against Other Study Parameters
Linear regression analysis was used to assess the association between adropin and other study parameters (Table 5). There was a significant association between adropin and gender (β = 0.71, p = .04), study group (β = 0.98, p < .001), weight (β = −0.06, p < .001), WC (β = −0.06, p < .001), BMI (β = −0.15, p < .001), SBP (β = −0.04, p < .001), FBS (β = −0.076, p = .001), TG (β = −0.003, p < .001), MetS component (β = −0.49, p < .001), fat mass (β = −0.04, p = .013), fat-free mass (β = 0.04, p = .013), and trunk fat (β = −0.06, p < .001). In a second model including adjustment for demographic variables, there was a significant association between adropin and leptin (β = −0.05, p = .004), study groups (β = 0.96, p < .001), weight (β = −0.04, p < .001), WC (β = −0.06, p < .001), BMI (β= −0.14, p < .001), SBP (β= −0.04, p < .001), FBS (β = −0.02, p < .001), TG (β = −0.003, p < .001), number of MetS components (β = −0.48, p < .001), fat mass (β = −0.1, p < .001), fat-free mass (β = 0.1, p < .001), and trunk fat (β = −0.09, p < .001).
Multiple Linear Regression for Assessing Association Between Adropin and Related Factors.

Note. WC = waist circumference; BMI = body mass index; SBP = systolic blood pressure; FBS = fasting blood sugar; TG = triglyceride; HDL = high-density lipoprotein; MetS = metabolic syndrome. aCrude model. bAdjusted for demographic variables such as sex, age, marital status, and educational level. cAdjusted model (all variables with p < .2 entered the model such as sex, groups, weight, WC, BMI, SBP, TG, HDL, MetS component, BMR, fat mass, fat-free mass, trunk fat). dMultivariate backward linear regression model. eStudy groups including the following: 1 = MetS patient, 2 = healthy obese, 3 = normal weight without MetS.
In multiple linear regression analysis, with adjustment for all variables with p value less than .2, there was still a significant association between adropin and the study group (β = 0.70, p = .005). With increasing one unit in the study group, from MetS to healthy obese group and healthy obese to the normal weight group, the adjusted adropin levels were increased by on average 0.70 ng/mL. Fitting the backward linear regression analysis reported that there was a significant association between adropin and study groups (β = 0.54, p = .002) and weight (β = −0.023, p = .006).
The results of the linear regression report that there was a significant association between the adropin/leptin ratio with the number of MetS components (β = −0.378, p < .001) and study groups (β = −0.170, p < .001). With increasing one unit in number of MetS components, the adropin/leptin ratio was decreased by 0.378, so that there was a decreasing trend of the adropin/leptin ratio according to the number of MetS components and study groups. There was a significant association between leptin and the number of MetS component (β = 1.87, p = .003), study groups (β = 3.48, p = .001), weight (β = 0.14, p = .007), WC (β = 0.26, p < .001), BMI (β = 1.059, p < .001), HDL (β = 0.22, p = .031), fat mass (β = 1.133, p < .001) and fat-free mass (β = −1.133, p < .001). With increasing one unit in number of the MetS components, the leptin was increased by 1.87 ng/mL, so that there was an increasing trend of leptin according to number of MetS components and study groups (Figures 2 and 3).

Trend of adropin and leptin according to number of MetS components.

Trend of adropin and leptin according to study groups.
Discussion
The current study compared the concentrations of plasma adropin, leptin, their ratio, and body composition (fat mass, fat-free mass, and trunk fat) among patients with MetS and two control groups. This study provides evidence suggesting a negative correlation between circulating adropin levels and occurrence of MetS compared with healthy obese controls and lean controls.
Obese subjects without MetS in the present study, in spite of having an unhealthy body composition and serum leptin level in terms of MetS components (FBS, HDL-c, TG, and blood pressure), were in a healthy status (the “healthy obese” phenomenon). Differences in body composition and serum leptin may not be the key explanation for having a normal metabolic status in the healthy obese. Other factors, especially endocrine and metabolic factors, need to be considered as risk factors for developing this disorder. These data suggest that high levels of serum adropin in healthy obese individuals compared with MetS patients may be associated with the prevention of MetS in healthy obese subjects. Limited studies have investigated adropin levels in patients with metabolic disorders (Expert Panel on Detection, 2001; Wu et al., 2014). These findings remained consistent after adjusting for demographic variables such as age, sex, and marital status.
It seems that normal lean subjects have adropin at a basic level (in the present study: 3.2 ± 1.47 ng/mL). As weight, BMI, and WC increased with obesity, the level of this hormone decreased until a certain level was reached (1.62 ng/mL in the present study, calculated by ROC curve). Whether this reduction is causally linked to the development of cardiometabolic disorders is uncertain. These data suggest that if plasma adropin does not remain higher than 1.62 ng/mL along with developing obesity, then MetS may develop. The main difference of the present study compared with others is that the present study measured adropin in healthy obese subjects as well as those who were diagnosed from MetS. No previous studies have reported serum adropin levels in healthy obese subjects. One interesting finding of the current research was that while it confirmed the results of earlier research about lower levels of adropin in obese people (Kumar et al., 2008), the present study additionally identified that the simultaneous presence of obesity and MetS in a person, compared with the presence of obesity without MetS, was associated with reduced levels of adropin. In subjects of this study, high levels of serum adropin were correlated with normal metabolic function even if the person was obese, the so-called “healthy obese phenomenon.” Future studies with longitudinal design are needed to see the changes of adropin over the time coincide with the development of obesity to clarify the potential role of adropin in the prevention of MetS.
The current correlational study could not distinguish whether low adropin levels cause the increase in MetS or whether MetS reduces adropin levels. Previous studies in mice strongly suggest that the effects of low levels adropin are possibly causal for the development of MetS (Kumar et al., 2008). In diet-induced obese mice, transgenic overexpression or systemic administration of adropin resulted in markedly attenuated insulin resistance and glucose intolerance, a reduction in diabetes, and weight loss, key features of the metabolic stress response (Kumar et al., 2008). Furthermore, supporting this hypothesis, adropin knockout mice have increased adiposity and fasting TGs, hepatic steatosis, insulin resistance, and increased propensity for impaired glucose tolerance with diet-induced obesity (Ganesh et al., 2012). In human subjects, a previous study has reported that low levels of plasma adropin are present in diabetic patients (Wu et al., 2014).
Conclusion
Data from the present study suggest that in addition to the important metabolic effects of low adropin, predisposing to MetS, high levels of adropin may be involved in the “healthy obese phenomenon.” If these causal links are correct, then administration of adropin, or pharmaceuticals that promote its production, may be a novel approach for treatment or prevention of MetS development.
Footnotes
Acknowledgements
The research group would like to thank all subjects who took part in the current study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
