Abstract
Despite a growing understanding that the social determinants of health have an impact on body mass index (BMI), the role of fatherhood on young men’s BMI is understudied. This longitudinal study examines BMI in young men over time as they transition from adolescence into fatherhood in a nationally representative sample. Data from all four waves of the National Longitudinal Study of Adolescent Health supported a 20-year longitudinal analysis of 10,253 men beginning in 1994. A “fatherhood-year” data set was created and changes in BMI were examined based on fatherhood status (nonfather, nonresident father, resident father), fatherhood years, and covariates. Though age is positively associated with BMI over all years for all men, comparing nonresident and resident fathers with nonfathers reveals different trajectories based on fatherhood status. Entrance into fatherhood is associated with an increase in BMI trajectory for both nonresident and resident fathers, while nonfathers exhibit a decrease over the same period. In this longitudinal, population-based study, fatherhood and residence status play a role in men’s BMI. Designing obesity prevention interventions for young men that begin in adolescence and carry through young adulthood should target the distinctive needs of these populations, potentially improving their health outcomes.
Introduction
As obesity continues to be a major public health priority for American families, investigating the contribution of the social determinants to changes in body mass index (BMI) is essential for prevention. With an estimated 70.1 million U.S. fathers (U.S. Department of Commerce, 2014), fatherhood is a key social determinant of men’s overall health (Durette, Marrs, & Gray, 2011). However, the relationship between BMI and fatherhood remains to be explored. Males report higher BMI compared with females (Ogden, Carroll, Kit, & Flegal, 2012) with fathers being more likely than mothers to be classified as overweight or obese (Magarey, Daniels, Boulton, & Cockington, 2003), and for their weight to be a better predictor of later child overweight and obesity than the mother’s (Brophy, Rees, Knox, Baker, & Thomas, 2012; Freeman et al., 2012). Men’s weight in adolescence and young adulthood, when they may be transitioning to fatherhood, is an important risk factor for diabetes (de Mutsert, Sun, Willett, Hu, & van Dam, 2014; Twig et al., 2014), accelerated coronary atherosclerosis (Flegal, Kit, Orpana, & Graubard, 2013; Jeffreys, McCarron, Gunnell, McEwen, & Smith, 2003; McGill et al., 2002; Schmidt, Bøtker, Pedersen, & Sørensen, 2014), future cardiovascular disease (Baker, Olsen, & Sorensen, 2007; Jeffreys et al., 2003; McGill et al., 2002), cancer (Han et al., 2014), and premature death (Schmidt et al., 2013; Yarnell, Patterson, Thomas, & Sweetnam, 2000).
The transition to fatherhood is a time of change for men. Evidence is accumulating that suggests this transition may affect the health behaviors of men which in turn may affect BMI, especially among at-risk populations. First, many men state becoming a father motivated them to change and improve their health behaviors such as eating a more nutritious diet, exercising more often, and drinking less alcohol (Garfield, Clark-Kauffman, & Davis, 2006). Yet little empiric work exists examining the role of becoming a father on BMI and whether the transition to fatherhood alone might affect the changes in BMI. Second, as our understanding of fatherhood and its different types expands, such as resident fathers who live with their children or nonresident fathers who live away from their children, an appreciation of the interactions between health risks and father type is beginning to emerge. For example, resident fathers are typically older, more educated, and employed with higher incomes compared with nonresident fathers (Castillo, Welch, & Sarver, 2011; Sorensen & Zibman, 2001). These characteristics have all been reported to be protective against increased BMI. In addition, being married, which many resident fathers are, also offers health benefits for men and may be protective of weight changes (Waite, 1995). Third, race/ethnicity and socioeconomic status have been implicated in terms of who becomes a father and when as well as in BMI changes (Berry et al., 2010; Hanson & Chen, 2007; Sobal & Stunkard, 1989; Wang, 2001). A systematic review of obesity rates reported that ethnic minorities and low-socioeconomic groups were disproportionally affected at all ages (Flegal, Carroll, Kit, & Ogden, 2012; U.S. Department of Health & Human Services, 2010; Wang & Zhang, 2006). Finally, lower education, which is associated with earlier and nonresident fatherhood, predicts obesity presumably because higher education enhances general nutrition knowledge, improved access to nutritional food, and increased access to information and tools for weight control (Galobardes, Morabia, & Bernstein, 2000; Jeffery, 1996). To our knowledge, no prior studies have accounted collectively for these factors when examining BMI trajectories over the life course and during the key developmental milestone of the transition to fatherhood.
This exploratory study examines associations between fatherhood and BMI. Specifically, the primary goal of this study is to assess whether the transition to fatherhood was associated with changes in BMI among men over time, whether this association varied by fathers’ residence status, and whether it differed when controlling for key social and demographic factors. Studies of the life course trajectory of paternal BMI have been hampered by a lack of data sets that are longitudinal, community-based, temporally correct, and represent a large enough population to have stable estimates. To address these limitations, data from the National Longitudinal Study of Adolescent Health (Add Health), which followed a representative sample of 20,745 American youth (10,253 males) from adolescence through early adulthood, were assessed. Add Health provides an exceptional opportunity to investigate the determinants of BMI over time, enabling comparisons of pre-fatherhood and post-fatherhood BMI while controlling for covariates, and to determine their relevance to population health and prevention. Understanding the progression of paternal BMI to delineate key points of intervention for prevention and treatment may benefit both fathers and their offspring.
Method
Study Sample
Add Health (Chen & Chantala, 2014) is a longitudinal study of a nationally representative sample of youth focusing on social, behavioral, and biomedical health as they progress from adolescence into adulthood. Wave I (n = 10,263 males; ages = 12-21 years) was conducted in 1994 to 1995; Wave II (n = 7,192, ages = 13-21 years) in 1996; Wave III (n = 7,192, ages = 18-28 years), 6 years later in 2001 to 2002, and Wave IV (n = 7,347, ages = 25-34 years), 6 years after that in 2007 to 2008. The institutional review board approved this study.
BMI
Height and weight measurement were collected in all four waves of the study. Height and weight data were self-reported in Wave I and measured by the interviewer in all subsequent waves. BMI was calculated from these height and weight values for all four waves. Clarke et al. (2014) demonstrated that men’s self-reported BMI and measured BMI were consistent from adolescence through adulthood. Add Health intentionally oversampled adolescents from various economic and racial/ethnic subgroups (Chen & Chantala, 2014); therefore, study-supplied weights, which also account for nonresponse and attrition and allowed for the sample to be representative of the adolescent population at baseline, were included.
Fatherhood Status
Fatherhood status was coded into three categories: resident fathers, nonresident fathers, and nonfathers. Each participant’s fatherhood status was defined using the household roster (reports of each member of their household) and live child data set (reports of each child they have fathered) from each wave; fathers were those who had listed a biological child. Residence status was defined by whether a child was listed in the household roster at entrance into fatherhood while controlling for interview year. Validity checks confirmed that the index child was indeed the first biological child for that respondent. Father classification was held constant through all future waves; nonfathers were the referent group for analyses.
Covariates
Covariates included respondents’ age, education, physical health, income, and number of children (all assessed at each wave) as well as race/ethnicity, own mother’s education (Wave I assessment). Race/ethnicity was categorized as White (referent), Black, Asian, Hispanic, and Other. Marital status was measured at entrance into fatherhood. Parents’ household income was used for each wave in which the respondent was still an adolescent. Health behaviors that may affect BMI were assessed at Waves I to IV and included screen hours (hours of television and computer summed/week) and daily physical activities (sum of times/week).
Statistical Analysis
Creating a Fatherhood-Years Data Set
A person-year file comprising data collected from adolescence through adulthood for young men was created using separate data records for each male encompassing study identification number, demographic, covariate, and BMI data to analyze BMI preentrance and postentrance into fatherhood. These “person-year” records used fatherhood status in each of the possible years of the data collection period and calculated time in fatherhood (i.e., “fatherhood years”) on the basis of wave-specific information regarding the age of the firstborn child. For example, a participant who first mentioned a 4-year old in Wave IV was coded as having +4 fatherhood years in Wave IV and −2 fatherhood years in Wave III (which was conducted 6 years prior to Wave IV), −8 fatherhood years in Wave II (conducted 12 years prior), and −9 fatherhood years in Wave I (conducted 13 years prior). Data from males who were never observed to be fathers were centered on the (weighted/winsorized) mean age of entrance into fatherhood for all fathers in the sample, age 23. Thus, “fatherhood years” for a nonfather observed at age 26 was coded as +3. Once complete, all participants had four rows of data corresponding to four waves of data collection. This allows for comparisons of fatherhood years over the 19-year range between nonfathers and fathers and resembles other approaches to defining person-years surrounding events of interest (Duncan, Wilkerson, & England, 2006; Garfield et al., 2014; Huang & Warner, 2005). For simplicity and comparisons, the term “fatherhood years” is used for fathers and nonfathers.
Multiple Imputations
Multiple imputations have been recommended from numerous sources to minimize the bias of data missing at random (Harel & Zhou, 2007; Rubin, 1987; White, Royston, & Wood, 2011). The primary advantage of using multiple imputations to handle missing data revolves around its use of random components incorporated into the estimates of missing values drawn from a distribution which allows inferences to be made from a stochastic method rather than through a method derived from strong (and often incorrect) assumptions, such as listwise deletion or hot-deck imputation. As recommended, 20 cycles were completed for five imputed data sets using the “ice” command in Stata (Royston, 2011).
Standardized BMI
Data were combined from each wave to support a continuous time analysis of respondents’ differing life stages before fatherhood and after fatherhood. BMI was standardized for all men at each wave. The mean estimate of each covariate on this standardized BMI variable was calculated for each fatherhood group over fatherhood years using a generalized estimating equation specifying an exchangeable correlation structure, normal distribution, and identity link, an approach commonly used (Chen & Chantala, 2014; Duncan et al., 2006; Garfield et al., 2014). A spline model was estimated with one knot modeling two mutually exclusive time category intervals: pre-fatherhood (−9 to 0 fatherhood years) and fatherhood (0 to +10 fatherhood years). Add Health intentionally oversampled adolescents from various economic and racial/ethnic subgroups (Chen & Chantala, 2014); therefore, study-supplied weights, which also account for nonresponse and attrition and allowed for the sample to be representative of the adolescent population at baseline, were included. Analyses were conducted in Stata/SE 12.1 (StataCorp, 2012).
Results
Descriptive Statistics and BMI Proportional Representation
Of 10,253 men in the sample, 6,828 (66.5%) were nonfathers and 3,425 (33.5%) were fathers by Wave IV (Table 1). Of those fathers, 686 (20.03%) were nonresident and 2,739 (79.97%) were resident at the time of the child’s birth. Most resident fathers were married (78%) compared with nonresident fathers (8%) and nonfathers (13%). Additional demographic and health characteristics are reported in Table 1.
Sample Characteristics of Nonfathers, Nonresident Fathers, and Resident Fathers.

Note. BMI = body mass index; FH = fatherhood; HS = high school.
p Values derived from F test for continuous and χ2 for categorical variables. bMean (SD). cn (%). dAs of 0 fatherhood years. eMean (Range).
BMI Over Fatherhood Years
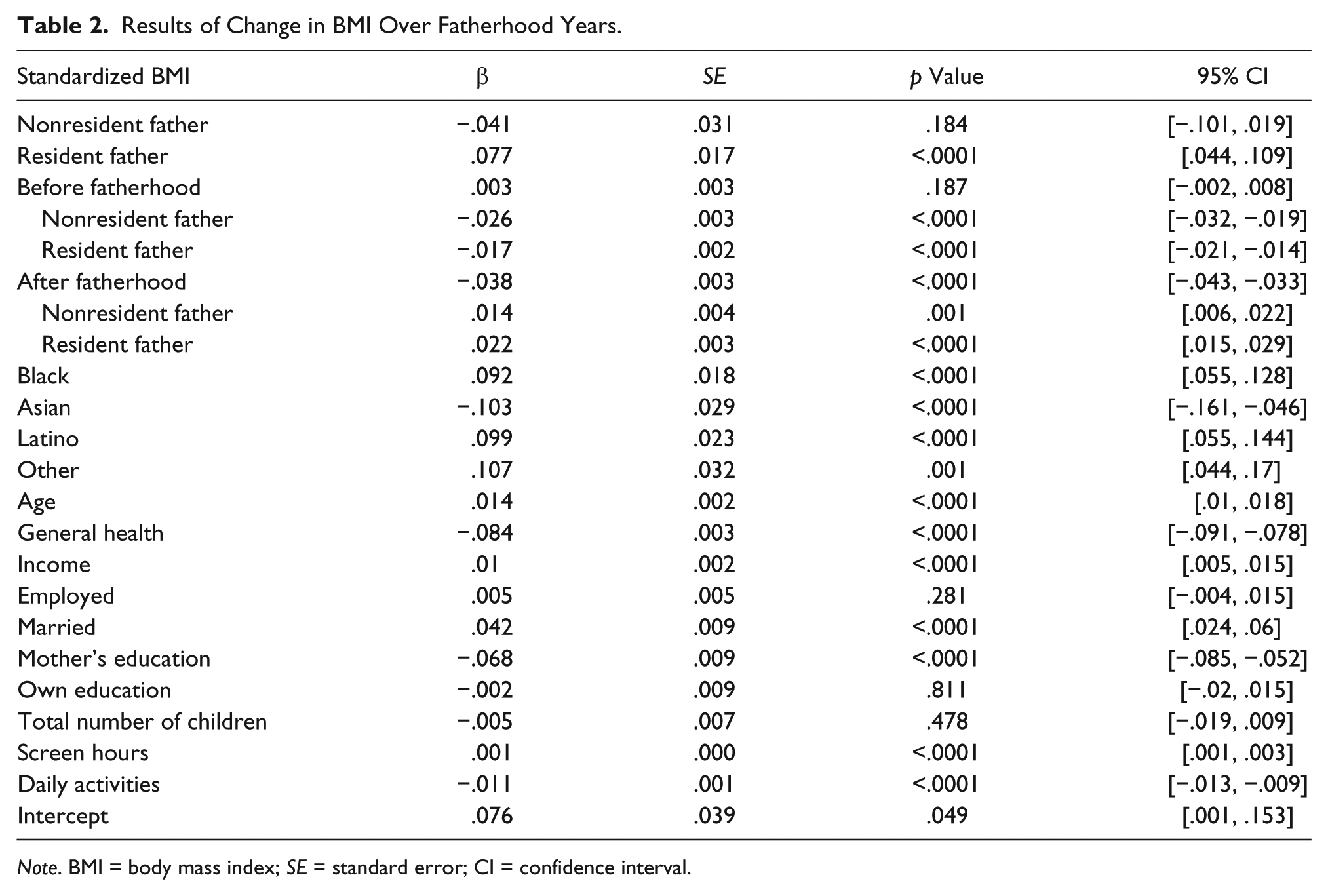
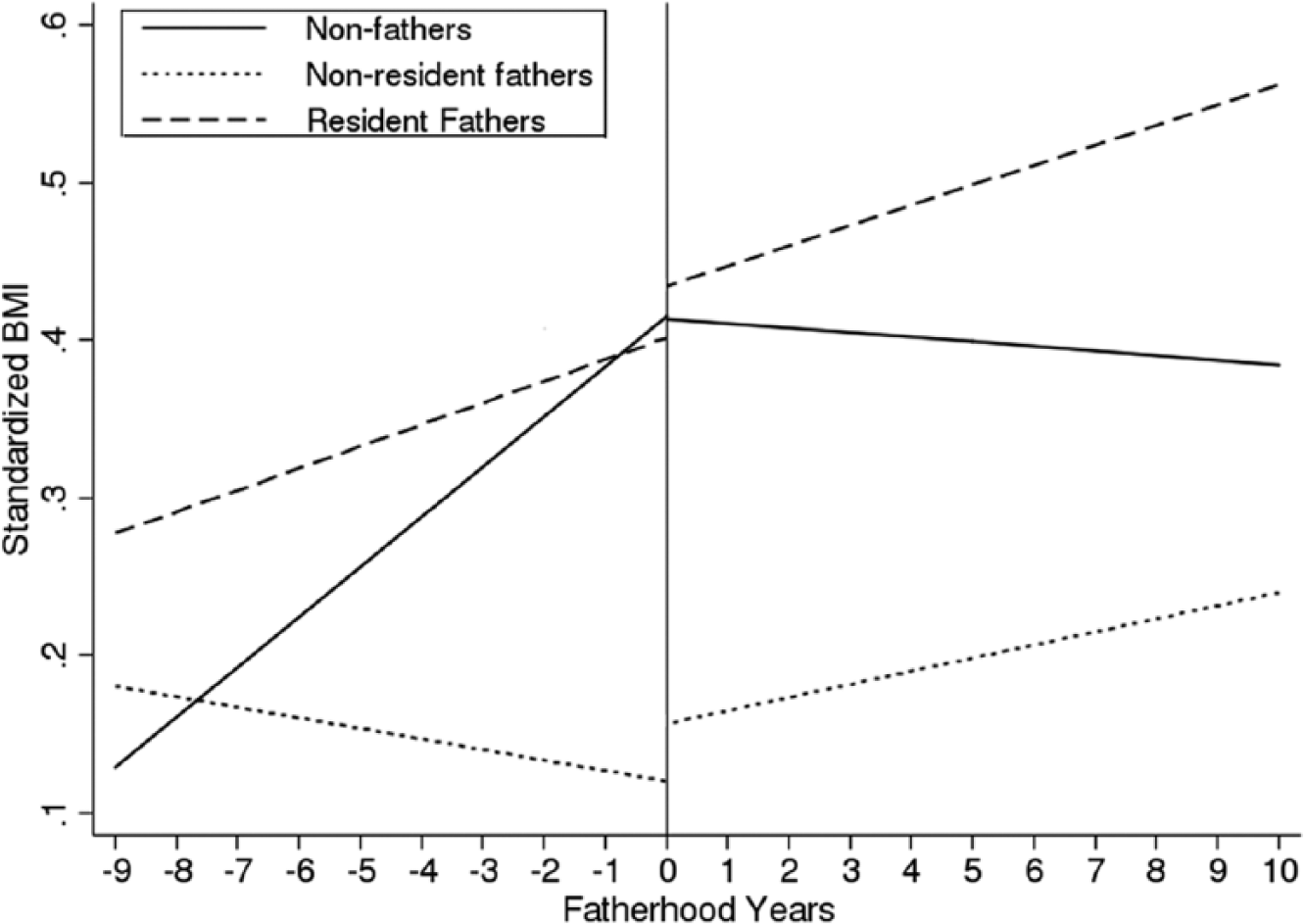
Age plays a major factor in BMI trajectory and was positively associated with BMI over all fatherhood years for all young men (β = .014; 95% confidence interval, CI [.010, .018]; Table 2). Change in BMI for nonfathers is due solely to age; comparing nonresident and resident fathers with nonfathers reveals different trajectories based on fatherhood status (Figure 1). While all young men’s BMI changes over time, the way it changes is contingent on fatherhood status and whether they become nonresident or resident fathers.
Results of Change in BMI Over Fatherhood Years.
Note. BMI = body mass index; SE = standard error; CI = confidence interval.
Predicted standardized body mass index (BMI) levels before and after entrance into fatherhood.
For young men who were nonfathers during the entire study period, BMI trends reveal that BMI slope did not statistically change before the age of 23 years and had a 0.04 SD decrease each year from age 23 until the end of the study (β = −.038; 95% CI [−.043, −.033]). The change in this time period is solely due to age. The mean BMI level for nonfathers conditioned on all of the independent variables increased by 0.29 SDs from the beginning of the study until age 23 and decreased by 0.02 SDs from age 23 until the end of the study. Since an SD of BMI represented roughly 4.5 units throughout the study, nonfathers therefore increased on average by 1.31 units from the entrance of the study to age 23—an increase of 6% in BMI—and decreased on average by 0.1 unit (less than 1%) from age 23 until the end of the study.
Young men who would become nonresident fathers had a decrease in BMI when compared with nonfathers in the pre-fatherhood period. Nonresident fathers had a 0.03 SD lower BMI compared with nonfathers in each year (β = −.026; 95% CI [−.031, −.020]). Once entering fatherhood, nonresident fathers had a slope that was 0.01 SDs larger than that of nonfathers (β = .014; 95% CI [.006, .022]). Therefore, nonresident fathers decreased on average by 0.07 SDs (0.32 BMI units) from the beginning of the study until entrance into fatherhood, a 1.5% decrease, and increased on average by 0.1 SDs (0.45 BMI units or 2%) from entrance into fatherhood until the end of the study.
Young men who would become resident fathers start with a BMI 8% higher than the comparison group of nonfathers (β = .076). The BMI slope for this group in the pre-fatherhood period was 0.02 SDs lower compared with nonfathers each year (β = −.017; 95% CI [−.021, −.014]) in the pre-fatherhood period. Once entering fatherhood, resident fathers had a slope that was 0.02 SDs higher than that of nonfathers (β = .022; 95% CI [.015, .029]). Therefore, resident fathers increased on average by 0.12 SDs (0.54 BMI units or 2%) from the beginning of the study until entrance into fatherhood and had a larger increase on average by 0.13 SDs (0.59 BMI units or 2.6%) from entrance into fatherhood until the end of the study.
In addition to these main findings, men of different races were all statistically significantly different from Whites with Asians having lower BMI and Blacks, Latinos, and other races having higher BMI (Table 2). Self-reported general health, mother’s education, and daily activities had negative associations with BMI. Income, marriage, and screen hours were all positively associated with BMI.
Discussion
In this first study of its kind examining young men’s BMI and fatherhood status in a nationally representative sample examined over a 20-year span, young men follow different weight gain paths based on their fatherhood status. While the BMI for all young men increases with age, there is a clear difference between the BMI trajectories of young men who enter fatherhood compared with those who do not. The entrance into fatherhood is associated with an increase in BMI trajectory regardless of fatherhood residency status, while those men who do not become fathers actually see an absolute decrease after age 23 (i.e., the post-fatherhood period of the study). Despite initial differences in BMI trajectories among the two fatherhood groups—wherein nonresident fathers exhibit increases and resident fathers exhibit decreases in BMI—following the transition to fatherhood, both groups exhibit increases in BMI of similar magnitudes.
Adolescent and early adulthood BMI often sets a course for men from which it is hard to deviate. While prior research examining men’s health and BMI trajectories point to increased heart, metabolic, cancer, and death risk (Baker et al., 2007; Flegal et al., 2013; Han et al., 2014; Jeffreys et al., 2003; McGill et al., 2002; Schmidt et al., 2013; Schmidt et al., 2014; Yarnell et al., 2000), these studies have not accounted for the influence of a major social and developmental milestone, the transition to fatherhood. Including fatherhood status and time in fatherhood is essential to understand how social characteristics may alter young men’s health, ultimately leading to innovative interventions and prevention.
As evidenced by this study, how young men’s BMI changes is influenced by when they become fathers and the type of father they become, either nonresident or resident. Prior to fatherhood, the BMI changes are in opposite directions for the two fatherhood groups, but after fatherhood both fatherhood groups have increasing BMI trajectories while nonfathers have decreasing BMIs. Though this change may appear small, it is important to remember the study examines mean estimates of BMI on a population level and therefore may provide conservative estimates. Nevertheless, becoming a father increases the BMI trajectories of young men.
Nonresident fathers achieve the lowest BMI immediately prior to entering fatherhood, suggesting an ideal time to focus clinically on health behaviors in this population. Nonresident fathers are already known to be at risk for lower education and income, which is associated with poor health; in this study, when controlling for these variables, this population is further at risk for increased BMI in early fatherhood. Prior research reports these men see becoming a father as motivation for positive change (Bozett, 1985; Cowan, 1988; Daniels & Weingarten, 1988; Palkovitz, 2002; Palkovitz, Copes, & Woolfolk, 2001; Strauss & Goldberg, 1999). For these men, being present in the health care system as pediatric chaperones may afford an opportunity for health education and counseling and has implications for family-centered care in pediatrics and family medicine.
Resident fathers in this study are the only group to have BMI increases before and after the transition to fatherhood. Examined by groups, resident fathers have the biggest increase overall and see the greatest increase in early fatherhood. Prior research reports becoming a father is a time when men attempt to improve their diets, increase physical activity, decrease alcohol use, and lower risk-taking behavior (Garfield, Isacco, & Bartlo, 2010; Guralnik, Butterworth, Patel, Mishra, & Kuh, 2009; Ringbäck Weitoft, Burström, & Rosén, 2004; Umberson, 1992; Umberson, Liu, & Powers, 2009; Weitoft, Haglund, Hjern, & Rosén, 2002); yet the increase in this study is evidence that these changes in health behaviors may have little impact. The majority of resident fathers in this study were married, an association in other research that has been reported to be advantageous in terms of education, income, and physical and mental health (Waite, 1995). In this study, marriage is associated with increased BMI, findings supported by other work (albeit without regard for fatherhood status; Brown, 2011; Jeffery & Rick, 2002). This may be due to unaccounted for changes in eating habits associated with marriage and parenting with potentially different kinds of foods, portions, and leftovers available; anecdotal evidence does exist of fathers cleaning their children’s dinner plates.
For nonfathers, early adolescence sees the highest increase in BMI followed by a lower increase in late adolescence and relative decrease in early adulthood. Though this study followed men until an average age of 29 years, it is important to recognize that many of these men will become fathers later in life. Thus, from a preventive and public health perspective, the implications are that by identifying these young men when they are at highest risk (i.e., during early adolescence) and implementing weight-related interventions and education at this time may offer the best potential to improve their own health and decrease negative influences on future BMI.
In addition to focusing on fatherhood as a social determinant, this study included a number of other social and contextual characteristics that were significantly associated with men’s BMI. Compared with Hispanic and Black men, those reporting poorer health and those with lower income had higher BMI, results supported by prior research (Bronte-Tinkew, Zaslow, Capps, Horowitz, & McNamara, 2007; Lawlor et al., 2003; Månsdotter, Lindholm, & Winkvist, 2007; Nomaguchi & Milkie, 2003). As would be expected, young men with more daily activities and fewer sedentary hours in front of screens (e.g., television, computer) had improved BMIs. This study adds to the debate around whether or not marriage is protective for men’s health, as married young men in this study have increased BMIs. Future study of the interrelationship between marriage, father’s residential status, and BMI is warranted.
From a clinical perspective as different subpopulations of men see fluctuations in BMI at different points over time, there is no one-size-fits-all approach to address these increases in weight. From a life course theory perspective, however, appropriately timed preventive efforts such as nutritional counseling and health promotion education that take into account developmentally significant stages may have enhanced potential for improving long-term outcomes and decreasing disparities (Cabrera, Fagan, & Farrie, 2008; Elder, 1998; Heckman, 2006; Robertson, Douglas, Ludbrook, Reid, & Teijlingen, 2008). The transition to fatherhood is one such time period where behavioral interventions (e.g., smoking cessation) for fathers may be especially welcome and effective (Robertson et al., 2008). There is mounting anecdotal evidence that expectant fathers are accompanying their partners to obstetric visits, yet another venue to consider finding ways to engage young men in the health care system. Health policy initiatives such as the Affordable Care Act (Patient Protection and Affordable Care Act of 2010) may also hold promise for improving access for young men who are less likely to have a primary care physician, to have seen a provider in the past year, and to have insurance compared with women (Garfield et al., 2006; Garfield, Isacco, & Rogers, 2012). Early results from the Affordable Care Act report a decrease in uninsured adults of child-rearing ages potentially alleviating insurance as a barrier to health care (Sommers et al., 2014 ).
The influence of fathers’ weight on children’s outcomes is just one more reason to focus preventive efforts on young men. In families with an overweight father and a normal weight mother, the odds of having an obese child 4 years later were 4.18 times greater compared with two normal weight parents, and, if the father was obese, the odds rose to 14.88 times greater (Freeman et al., 2012). Notably, having a normal weight father and an overweight/obese mother was not a significant predictor of child obesity (Freeman et al., 2012). Paternal behaviors have also been reported to increase the risk of obesity in preschoolers more than maternal behaviors (Wake, Nicholson, Hardy, & Smith, 2007). With the Centers for Disease Control and Prevention reporting 96% of resident fathers and 30% of nonresident fathers ate a meal with their <5-year-old child every day or several times a week (Jones & Mosher, 2013), there is ample opportunity for influence. The Healthy Dads, Healthy Kids program in Australia proved the feasibility and efficacy of targeting fathers to lose weight and be a role model for their children (Morgan et al., 2014). The Healthy Dads, Healthy Kids program resulted in significant weight loss and improved health-related outcomes in fathers and improved eating and physical activity among children (Morgan et al., 2011). Thus, targeting fathers during this developmental stage may be an especially efficacious approach to improving men’s and children’s health.
Limitations
The first wave of Add Health calculated BMI based on self-report; all later waves were interviewer collected and may be more accurate. Inferences for this study are limited to the average change in BMI during a certain period given certain characteristics. The model predicts at a population level and is unable to predict individual trajectories or estimate how changes in a particular covariate can affect the change of an individual’s BMI. Another limitation is the restriction of fatherhood status to one category over the study period despite recognizing that resident status change may potentially affect BMI and health. Future research with longer post-fatherhood transition study windows might better shed light on these shifts and BMI. Finally, the Add Health sample follows men through their 20s and into the 30s and is unable as of yet to examine the transition to parenthood for older fathers. Nevertheless, resident fathers’ age of fatherhood entry in Add Health, 25 years, is the same as the National Survey of Family Growth’s age at first birth for men from 2006 to 2010 (Martinez, Daniels, & Chandra, 2012).
Conclusion
Understanding the influence social characteristics have on young men’s BMI over time is a first step toward addressing unmet health needs for young men as they get older. These findings support the need for obesity prevention interventions specifically designed for and targeted toward young males in general and those transitioning to fatherhood in particular. Such interventions could begin in adolescence and carry through young adulthood, targeting the distinctive needs and concerns of young men over the life course.
Footnotes
Acknowledgements
Craig F. Garfield acknowledges the support of grant K23HD060664 from the Eunice Kennedy Shriver National Institute of Child Health and Human Development in conducting this study.
Authors’ Note
The study sponsor had no role in study design; collection, analysis, and interpretation of data; writing the report; and the decision to submit the report for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Craig F. Garfield’s effort was partially supported by grant K23HD060664 from the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
