Abstract
Males account for one third of global hip fracture patients, and their hip fracture-related mortality rate is higher than that of females. Scholars have primarily investigated self-evaluation indicators for elderly Caucasians and other ethnicities, and have rarely conducted large-scale cutoff point studies on Asian males. In this study, a large-scale database on bone mineral density (BMD) examinations was used to conduct a cutoff point study on males with osteoporosis. This study involved a retrospective research design. Males who accepted BMD examinations from 2009 to 2012 at a large teaching hospital in Taiwan were sampled in this study. This study used the database for analyzing effects with using osteoporosis self-assessment indicator, age, body weight, and body mass index (BMI) to discriminate osteoporosis in males, specifically focusing on accuracy, cutoff point, sensitivity, specificity, and positive and negative predictive values. Overall, receiver operating characteristic curve for the samples reached 70%. These cutoff points included: a body weight is less than 58.8 kg, BMI is 23 kg/m2, and the osteoporosis self-assessment score is −1.86, to identify osteoporosis. Involving body weight and BMI as optimal assessing indicators for assessing osteoporosis among males younger than 65 years, and the osteoporosis self-assessment tool was ideal for measuring males older than 65 years. Professional health staff should apply convenient, low-cost, and accurate evaluation tools that can assist Asian males in planning preventive osteoporosis strategies to avoid osteoporotic fractures and death.
Introduction
The World Health Organization (WHO) identified osteoporosis as a syndrome that results from losing bone mass with degradation of fine bone structure, increasing bone brittleness, or causing osteoporotic fractures (World Health Organization, 1994). Wright et al. (2014) estimated that 1 to 2 million American persons have osteoporosis and 8 to 13 million people have low bone density. Osteoporosis is potentially fatal. Unlike diabetes, which causes more than three clinical symptoms, early stage osteoporosis does not exhibit symptoms, and the first sign of osteoporosis is typically a fracture, which commonly occurs in the wrist, spinal column, and hip (Skorupski & Alexander, 2013). In the United States, approximately 13% of Caucasian males older than 50 years were at risk of osteoporotic fracture (Sidlauskas et al., 2014). Meanwhile, Zhang et al. (2014) mentioned age standardized prevalence of osteoporosis at the lumbar spine and femur neck in Chinese adults was 3.2% and 5.3 % in men aged ≥ 50 years, respectively. The most severe complication of osteoporosis was hip fracture, and males were having higher mortality rates than do females (Omsland et al., 2014). Compared with females, males exhibit twofold or greater mortality rates after a hip fracture (Pludowski et al., 2013). The United States expended US$1.7 billion treating osteoporosis, 25% of which are spent on males (Sidlauskas et al., 2014). Osteoporotic fractures cause higher mortality and morbidity rates among males than they do among females (Bliuc et al., 2009), thus, preventing osteoporosis among males is a critical public health concern.
Background
The osteoporosis diagnostic standard proposed by WHO (National Osteoporosis Foundation (NOF), 2008; World Health Organization, 1994) is as follows: normal bone mineral density (BMD) is over or equal to 1 SD of the average BMD for young people, osteopenia is identified if the BMD value ranges between 1 and 2.5 SD under the average BMD for young people, and osteoporosis is identified if the BMD is 2.5 SD or more under the average BMD for young people. Scholars have indicated that BMD examinations can be used to characterize bone condition and predict osteoporosis. The importance of using BMD in diagnosing osteoporosis is tantamount to that of using blood pressure monitoring and resulting values to predict strokes (Chang & Yang, 2013). Dual energy X-ray absorptiometry (DEXA) is the most accurate technique to test osteoporosis (NOF, 2008). However, BMD measurements are not available in all communities. In Taiwan, such examinations are limited by cost constraints associated with Bureau of National Health Insurance (BNHI); thus, not everyone can receive free BMD examinations. People with one of the three criteria can accept BMD examination without costs in Taiwan): increasing bone loss because of hormone diseases, having nontraumatic fractures, and females older than 50 years receiving treatments for osteoporosis. In addition, patients who have to take extra BMD examination cannot accept these tests in 1 year and cannot have over three times. People who do not qualify for free testing must pay the expenses required for BMD examinations. Currently, a clinical BMD radiological examination costs approximately US$30 to US$50, which is considerably high. Therefore, people may not be aware of their BMD conditions early, preventing them from taking preventive measures against fractures.
Because patients having low BMD are at high risk of fragility fracture, they should actively seek early screening to start prevention strategy. Following hip fragility fractures, males exhibit a 37.5% higher mortality rate than do females (Jiang et al., 2005). A simple self-assessment questionnaire must be developed to identify groups at risk of osteoporosis. In recent years, scholars (Koh et al., 2001; Morin, Tsang, & Leslie, 2009) have developed the simple osteoporosis self-assessment tool (OST), which originally used the equation 0.2 × (body weight [kg] − age) to evaluate the risk of osteoporosis among females (Koh et al., 2001). The aforementioned risk factors with simple evaluation indicators have been tested for postmenopausal, elderly Caucasian females or young Asian females. Gourlay et al. (2005) indicated that the sensitivity and specificity of OST for postmenopausal Caucasian females between the ages of 45 and 64 years were 89.2% and 45.0%, respectively (cutoff point ≤ 1). Osteoporotic Asian females between the ages of 30 and 49 years yielded sensitivity and specificity values of 76% and 49% (cutoff point ≤ 2; Chang & Yang 2013). The only study on Asian males was that of Lynn et al. (2008), which was focused on males older than 65 years in Hong Kong. However, few studies have compared the discriminate accuracy of osteoporosis for OST with males younger and older than 65 years.
Thus, simple self-assessments can be employed prior to BMD loss to elucidate the risk of osteoporosis. Meanwhile, facilitate implementing osteoporosis prevention strategies among Asian males could avoid deaths resulting from osteoporotic fractures.
Aim
This study employed a large-scale BMD medical history database (with chart review) to conduct a cutoff point study on males (older and younger than 65 years) with osteoporosis. Meanwhile, this research was using OST, body mass index (BMI), age, and body weight as indicators to identify osteoporosis.
Methods
Participants
The participants in this study were males who required BMD examinations. All of the cases were recruited from a large teaching hospital in Northern Taiwan.
Data Collection Methods
This study involved retrospective research methods. The T-score of the BMD at the femoral neck was defined as ≤−2.5. Males (N = 834) who received DXA BMD examination in the previous 3 years (2009-2011) were recruited as the study participants.
Ethical Considerations
This study was approved by the Research Ethics Office of National Taiwan University (2001204056RIC). All personal information was anonymous and rigorously safeguarded by the program executor.
Sample Size Determination
For calculating sample size, this research set that a 21% difference in the area under the receiver operating characteristic curve (AUROC) with the null hypothesis of 60% (i.e., the minimal level of clinical discrimination) was clinically significant based on Morin et al. (2009). With the Type I error rate (α level) of 0.05, power (1 − β) of 0.90, and ratio with samples of 5.85 (Morin et al., 2009), 590 was the minimum number of participants needed (including at least 86 patients exhibiting osteoporotic BMD values). The sample size was calculated using PASS 2008 (NCSS, LLC., Kaysville, UT, USA).
Statistical Analysis
The continuous variables were reported as mean and standard deviation, and categorical variables were demonstrated as individual numbers and percentages. Independent sample t test was carried out to explore the differences in the means of the continuous variables regarding the value of osteoporosis (based on T-score ≤ −2.5). In addition, a chi-square test was conducted to explore the differences in the categorical variable of osteoporosis.
This study divided body weight, BMI, and OST scores into four groups and listed the ratio of osteoporosis patients in each group. This investigation used logistic regression to calculate the odds ratio and confidence interval (CI) of osteoporosis, and a Cochrane–Armitage chi-square analysis was conducted to decide whether a linear trend in osteoporosis prevalence existed between each group. To determine the discriminate or diagnostic power of BMI, body weight, and OST scores for predicting osteoporosis, this research demonstrated a receiver operating characteristic (ROC) curve to analyze the overall sample and the potential optimal cutoff point, as well as the ROC results of each BMI and age level. Data analysis were reported using the Statistics Package for the Social Sciences version 17.0 (SPSS Inc., Chicago, IL, USA).
Results
Of the 834 valid samples (Taiwanese males) collected in this study, 321 were defined as having osteoporosis (38.5%). The mean age of participant was 71.9 years (SD = 13.3 years) and most participants (74.9%) were older than 65 years. The mean body weight and height was 63.9 kg (SD = 10.7 kg) and 165.9 cm (SD = 6.5 cm), respectively. The mean BMI was 23.2 kg/m2 (SD = 3.4 kg/m2), and most participants had a normal body weight (52.9%). Overall, 39.2% of the participants were overweight or obese. The mean OST score was −1.59 (SD = 3.78). In summary, osteoporosis patients were typically old, lightweight, short, and have low BMI and OST score (p < .05; see Table 1).
Baseline Characteristics of Study Sample (N = 834).

Note. BMD = bone mineral density; BMI = body mass index; OST = osteoporosis self-assessment tool. Categorical variables were presented as number (percentage) and continuous variables were presented as mean ± standard deviation.
The results of Cochrane–Armitage test reported the prevalence of osteoporosis increased as weight, BMI, and the OST quartiles decreased (p < .001). In the logistic regression analysis, using Group 4 as the reference group (reference category), this study identified that the odds ratio of the body weight quartiles Q1, Q2, and Q3 were significantly higher than that of Q4. Comparing the BMI with OST quartiles indicated that the odds ratio of Q1 and Q2 were significantly higher than those of Q4, but those of Q3 and Q4 did not differ significantly (see Table 2 and Figure 1).
Prevalence (%) of Osteoporosis by BMD Criteria in Relation to Weight, BMI, and OST Quartiles.

Note. BMD = bone mineral density; BMI = body mass index; OST = osteoporosis self-assessment tool; OR = odds ratio; CI = confidence interval.
Defined as T-score ≤ −2.5.

The association of quartiles of anthropometric variable with osteoporosis BMD.
The ROC results revealed that weight, BMI, or OST score exerted significant discrimination effect on osteoporosis because the 95% CI of the AUROC did not include 0.50. Of the three physical parameters, the OST demonstrated superior discrimination, attaining an AUROC of 70% (95% CI [0.66, 0.74]).
The findings indicated that the discriminate effects of body weight and BMI for predicting osteoporosis were superior among the nonelderly age group (<65 years) compared with elderly age group (≥65 years). However, the OST provided superior osteoporosis diagnostic effects for both the elderly and nonelderly age groups. Body weight was divided into two groups based on the BMI values. The effects of body weight and BMI on diagnosing osteoporosis were considerably high in the lightweight group; however, the osteoporosis diagnostic effects of the OST were identical for the lightweight, normal weight, overweight, and obese groups. When body weight was divided into four groups, the results identified that body weight yielded superior osteoporosis diagnostic effects on participants with low body weights (AUROC = 0.77). The OST exerted superior diagnostic effects on lightweight and overweight patients (AUROC was 0.71 and 0.73, respectively). Furthermore, the weight, BMI, and OST scores of the obese group could not effectively discriminate osteoporosis (see Table 3).
Association of Anthropometric Variables With Osteoporotic BMD.

Note. BMD = bone mineral density; BMI = body mass index; OST = osteoporosis self-assessment tool; AUROC = area under receiver operating characteristic curve; CI = confidence interval. BMD (defined as T-score ≤ −2.5).
Table 4 reported the outcome of each cutoff point of every physiological indicator. Overall, the ideal cutoff points were 58.8 kg for body weight (sensitivity 43.9%, specificity 78.2%, positive predictive values [PPV] 56.0%, and negative predictive values [NPV] 69.0%), 23.0 kg/m2 for BMI (sensitivity 60.4%, specificity 61.6%, PPV 50.0%, and NPV 71.0%), and −1.86 for OST score (sensitivity 69.2%, specificity 63.0%, PPV 54.0%, and NPV 77.0%; see Figure 2).
Selected Cutoff Point, Sensitivity and Specificity for the Association of Anthropometric Variables With Osteoporotic BMD.
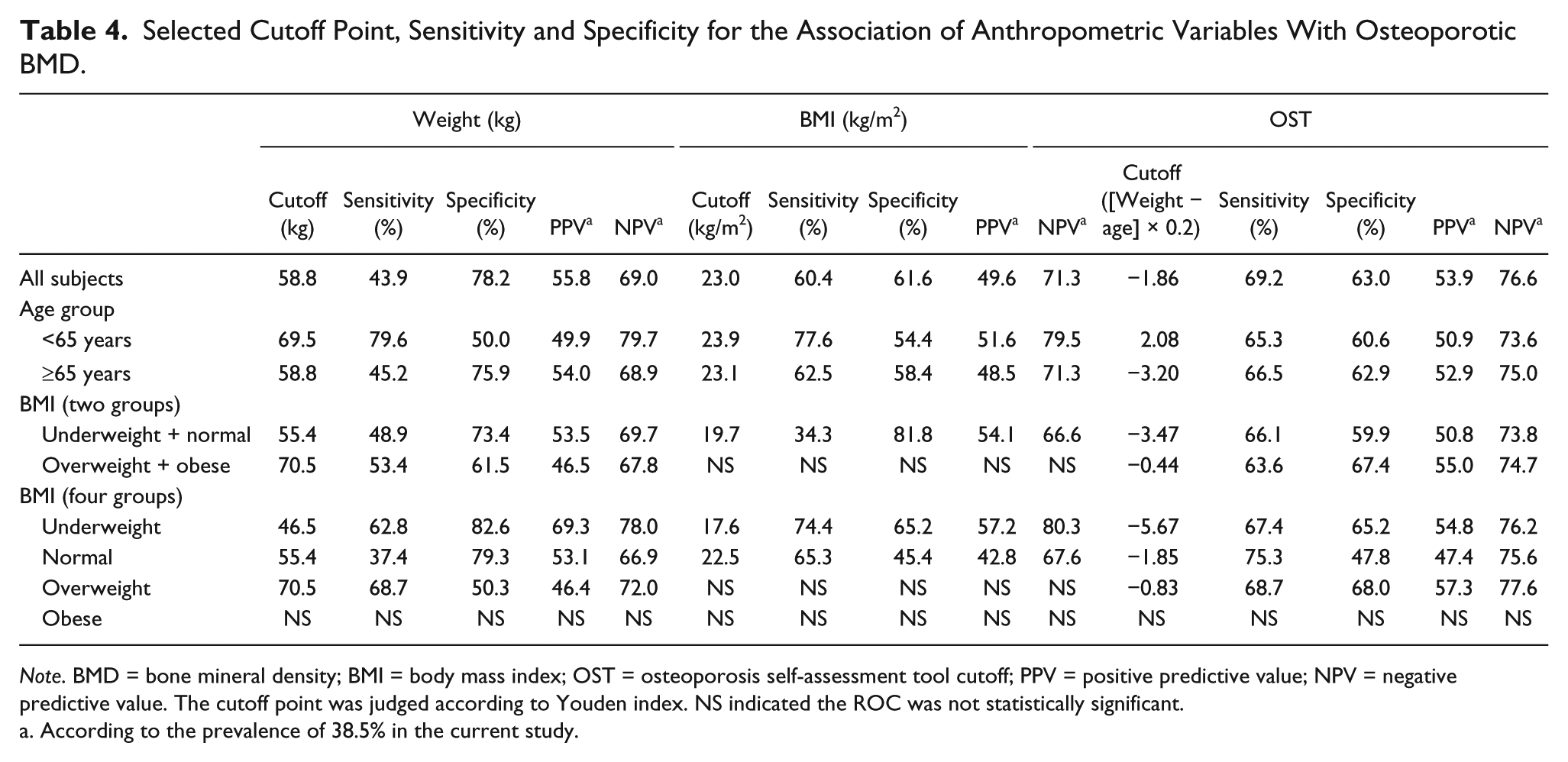
Note. BMD = bone mineral density; BMI = body mass index; OST = osteoporosis self-assessment tool cutoff; PPV = positive predictive value; NPV = negative predictive value. The cutoff point was judged according to Youden index. NS indicated the ROC was not statistically significant.
According to the prevalence of 38.5% in the current study.

The association of each anthropometric variable with osteoporosis BMD.
Discussion
The database indicated that most participants older than 65 years exhibited a relatively higher prevalence of osteoporosis compared with those aged ≥50 years by sampling Taiwan’s National Health Insurance (NHI) database between 1999 and 2001. The averaged prevalence of osteoporosis in NHI was 1.63% for men (Yang et al., 2006). The major reason was that most elderly people were evaluated by orthopedic surgeons, who perceived these males as being at risk of osteoporosis and required them to undergo BMD examinations. However, most studies have maintained that osteoporosis commonly occurs in elderly males (Costa, Cusano, Silva, & Bilezikian, 2014; Sibley, Voth, Munce, Straus, & Jaglal, 2014). Thus, this high-risk group requires specific care to avoid osteoporotic fractures, which can cause disability and death (Skorupski & Alexander, 2013).
By comparing with males with a normal bone status, this study identified that males with osteoporosis are old, lightweight, and short and have low BMI. Further analysis indicated that the group with the lightest weight and lowest BMI and OST score demonstrated a 2 to 3 times higher odds ratio of developing osteoporosis than that of the group with the heaviest weight and highest BMI and OST score. The results of this large research support associations of previous studies among age, weight, height, BMI, and BMD in males (Cheng et al., 2012). Felson, Zhang, Hannan, and Anderson (1993), Malabanan et al. (2013), and Tamaki et al. (2011) indicated that low body weight was a major predictor of peak bone mass in males. Adler, Tran, and Petkov (2003) and Lynn et al. (2008) have demonstrated that osteoporosis (i.e., T-score ≤ −2.5) was related to low weight, BMI, and OST score for male populations in the United States. Thus, males should be provided with osteoporosis prevention information early to reduce the occurrence of osteoporosis. Chang and Yang (2013) reported that patients should be encouraged to maintain an appropriate weight because weight loss has been associated with losing in strength of bone and further fractures. Thus, patients require behavioral changes and diverse intervention methods for maintaining the appropriate weight (Vescovi, Jamal, & De Souza, 2008).
The ROC demonstrated that weight, BMI, and OST score exerted significant diagnostic effects on osteoporosis; specifically, the OST diagnostic accuracy was satisfactory (70%). In this study, compared with older than 65 years, optimal results were obtained using body weight and BMI to discriminate osteoporosis in males younger than 65 years. Thus, young males can effectively prevent osteoporosis by paying attention to change in their body weight and BMI. This result was consistent with that of Sinnott, Kukreja, and Barengolts (2006), who analyzed how body weight and BMI could be used to forecast BMD and fractures among 128 African Americans aged 40 to 59 years. The results demonstrated that among young age groups, the body weight and BMI could be used to classify males with T-scores ≤−2.5. Notably, body weights and BMIs of males with light or normal weights yielded superior osteoporosis diagnostic effects compared with those of overweight and obese people. Therefore, underweight males could use the cutoff points of body weight and BMI to discriminate their risk of osteoporosis. De Laet et al. (2005) identified the BMI of 25 kg/m2 was the cutoff point to develop hip and osteoporotic fracture in the future. This research determined that applying the low BMI cutoff point of 23.0 kg/m2 yielded higher diagnostic accuracy for classifying osteoporosis in Taiwanese males than did using a weight criterion. WHO put forward a prediction tool to predict 10-year fracture probability, which was verified among cohorts studies of males and females older than 40 years old; this tool integrates various physiological indicators (e.g., BMI and BMD) (Kanis, Johnell, Oden, Johansson, & McCloskey, 2008). Prediction tables were produced to predict the risk of osteoporosis with BMI when BMD measurements are unavailable. This could be a valuable method in areas where DXA is inconvenient.
Previous studies, using the OST to discriminate osteoporosis in males, have primarily focused on Caucasian males (Adler et al., 2003; Lynn et al., 2008; Sinnott et al., 2006). Ghazi et al. (2007) analyzed 229 Moroccan males with an OST score of <2 to discriminate osteoporosis; the sensitivity ranged from 63% to 87% at the total hip, yielding BMD T-scores of −2.5 and a specificity range of 58% to 59%. Adler et al. (2003) studied 181 American males (mean age = 64.3 years) and determined that with an OST cutoff score of 3 could predict osteoporosis at sensitivity and specificity were 93% and 66%, respectively. Research on OST suitability has demonstrated that the OST is appropriate for diagnosing osteoporosis in males older than 65 years, who are overweight or obese. This result was similar to that of Lynn et al. (2008) and Nielson et al. (2011), who researched the accuracy of using the OST to discriminate Caucasian and Asian males older than 65 years. Both studies have discovered that the OST is appropriate for use among males of advanced age. This study demonstrated the cutoff of the OST was negative potentially because the OST equation compares differences in body weight and age. Thus, the weights of males tend to decrease with age, and males with osteoporosis demonstrate larger weight decreases than do those with normal bones. Salamat, Salamat, Abedi, and Janghorbani (2013) mentioned that decreased body weight was a critical risk indicator influencing BMD. As people age, they may become frail and develop sarcopenia because of malnutrition. These factors may be related to decreases in body weight among elderly males (Hubbard, Lang, Llewellyn, & Rockwood, 2010; Kelaiditi, van Kan, & Cesari, 2014; Morley, von Haehling, Anker, & Vellas, 2014), which may accompany the occurrence of osteoporosis.
Notably, the specificity values of weight and BMI exceeded the sensitivity values, indicating that these measures can be employed to eliminate the possibility of osteoporosis among males. The diagnostic accuracy of this tool is higher than mean; however, the low PPV hints that males with low BMD values are likely to be misclassified (Adler et al., 2003; Lynn et al., 2008). In this study, a high NPV was desirable because the primary purpose of an osteoporosis screening tool is to minimize failure in identifying those at risk. Thus, the PPV is compromised, indicating that elderly people who are not at risk of osteoporosis are likely to be identified.
Conclusions
Measuring easy physiological indicators, for example age, BMI, and OST scores, where BMD testing is limited or not available can facilitate identifying males at risk of osteoporosis. This study demonstrated that males with osteoporosis are typically older and lightweight and exhibit low BMIs and OST scores. Studies have indicated that using body weight and BMI as cutoff point indicators is appropriate for Taiwanese males younger than 65 years; however, the OST is appropriate for males who are older than 65 years and overweight.
Limitations
This study involved certain limitations. First, the participants were males who received BMD examinations as evaluated and referred by orthopedic surgeons; thus, the osteoporosis prevalence in males may be overestimated. Second, the analysis data revealed that although the body weight and BMI cutoff points were approximately 70% accurate in diagnosing osteoporosis in males younger than 65 years, this age group only accounted for 15.3% of the samples because of the limited NHI payment scheme. Patients referred for a BMD examination primarily comprised elderly males, which may have influenced the results of this study; therefore, this study recommend that large-scale studies be conducted on representative middle-aged groups in the future. Finally, body weight, BMI, and OST scores cannot be used as osteoporosis diagnostic tools for obese males; thus, other critical cutoff point factors that influence the BMD of obese Taiwanese males should be explored.
Footnotes
Acknowledgements
The authors are grateful to thank to Mr. Lin for his helpful comments on statistics of this article. The authors thank the National Science Council of the Republic of China, Taiwan, for financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. The National Science Council, Taiwan, Republic of China, supported the research by grant, NSC101-2314-B-227-001.
