Abstract
Myofibroblastoma is a rare mesenchymal tumor located not only in breast but also in extramammary sites. This is the 79th case of breast myofibroblastoma reported in the literature. This tumor presents a great variety of morphological features, which increase the difficulty of differential diagnosis. The authors report a breast myofibroblastoma diagnosed in a 73-year-old male. The article discusses a bilateral gynecomastia and a palpable right breast well-defined tumor, without calcifications on mammography, which was surgically removed. Macroscopically, a well-circumscribed uncapsulated nodule was seen with lobular arrangement on cut section, and microscopically, a nodule with pushing borders and a connective pseudocapsule was seen. The spindle cells were arranged in fascicular clusters, with focally collagen bundles and a rich reticulinic network stained black with Gömöri impregnation. Immunohistochemically, the tumor cells were marked by vimentin, CD34, desmin, and smooth muscle antigen and did not express cytokeratin, S-100 protein, CD99, CD10, and factor VIII–related antigen. More than 90% of the cells expressed estrogen receptor. No recurrences were reported 2 years after surgical excision. This case is a variant of cellular myofibroblastoma, with a rich reticulinic network and scanty collagen bands.
Myofibroblastoma is an uncommon benign mesenchymal tumor that may arise in several organs and tissues, including soft tissues of retroperitoneum (Sagar, Vargiamidou, & Manikkapurath, 2011) or inguinal area (McManamin & Fletcher, 2001), abdominal wall, meninges, suprasellar area (Shinojima et al., 2002), vagina, and either vulva (Wei & Zhu, 2011). Although most of the reported cases were located in breast (Magro, Gurrera, & Bisceglia, 2003), it is extremely rare, representing less than 1% of breast tumors (Tavassoli & Soares, 2003).
Breast myofibroblastoma occurs mainly in men aged 25 to 87 years, with a median age of 64 years (Mele, Jensen, Wronecki, & Lelkaitis, 2011; Qureshi & Kayani, 2008), but females in the age range of 25 to 80 years (mainly postmenopausal) may also be involved (Qureshi & Kayani, 2008; Uchôa, Cruz, Schaefer, Pegas, & Cambruzzi, 2010; Yoon-Ki et al., 2010). A recent study mentioned that, despite the predilection for elderly men, in the recent past, the gender distribution seems to have become insignificant (Yoon-Ki et al., 2010). Only one case was diagnosed in the breast of an adolescent Omani boy (Alam, Samarasinghe, & Pillai, 2002).
This breast tumor is solitary, usually unilateral, and less than 4 cm (Qureshi & Kayani, 2008). Only one case has been reported as bilateral breast myofibroblastoma, in a 25-year-old woman (Yoon-Ki et al., 2010). It may occur in the right (Lee, Gilcrease, Wu, & Yang, 2010) or left breast (Landeyro, Diaz, Raventos, Vadillo, & Martinez, 2011), usually near the nipple (Desrosiers, Rezk, Larkin, Khan, & Li, 2007), but no predilection for a specific quadrant has been reported.
The first case of breast myofibroblastoma was described in 1987 by Wargotz, Weiss, and Norris (1987). From 1987 to 1997, the number increased to 40 reported cases, and according to Lee et al. (2010), approximately 70 cases were reported between 1987 and 2009. Since then, between 2010 and 2011, 8 other cases have been reported (Landeyro et al., 2011; Mele et al., 2011; Roman-Diaz et al., 2010; Uchôa et al., 2010; Wahbah, Gilcrease, & Wu, 2010; Yang, Chang, Jan, & Wang, 2010; Yoo, Shin, Ko, Han, & Oh, 2010; Yoon-Ki et al., 2010), which confirms its increasing frequency (Desrosiers et al., 2007). Our case should be the 79th case of breast myofibroblastoma reported between 1987 and February 2012, in the articles cited in PubMed and KoreaMed.
In this article, along with the rarity of this case we have also presented its histological characteristics based on the review of pertinent literature. A special focus was on the understanding of its histogenesis based on the immunohistochemical (IHC) stains.
Case Presentation
A 73-year-old man presented with a 2-month history of bilateral gynecomastia. Any palpable breast mass, nipple discharge, or visible changes of the breast skin was noted by the patient himself. Cirrhosis and other hormone-related systemic disorders were excluded.
On physical examination, a well-defined nodule, near the nipple, was detected on palpation of the right breast. No axillary lymph nodes were palpable. Right breast echography revealed a well-demarcated nodule in the breast parenchyma, 0.5 cm from the nipple. No associated microcalcifications were observed at mammography.
The patient underwent surgical excision of the tumor mass. Macroscopically, the surgical specimen was a well-circumscribed round-grey nodule, which was nonencapsulated, hard in consistency, 10 mm in diameter, and with a lobular cut surface appearance. No necrosis or hemorrhage was observed.
The histological examination showed a sharply defined nodular tumor with pushing borders and connective pseudocapsule. The tumor cells were arranged in a highly cellular fascicular cluster with focally collagen bundles (Figure 1) stained red with Van Gieson staining and smooth muscle component. At high-power view, we noticed spindle cells with eosinophilic scanty cytoplasm and elongated monomorphic nuclei, with small nucleoli and occasional grooves. No atypical cells, mitotic figures, epithelial components, necrosis, or hemorrhages were observed.
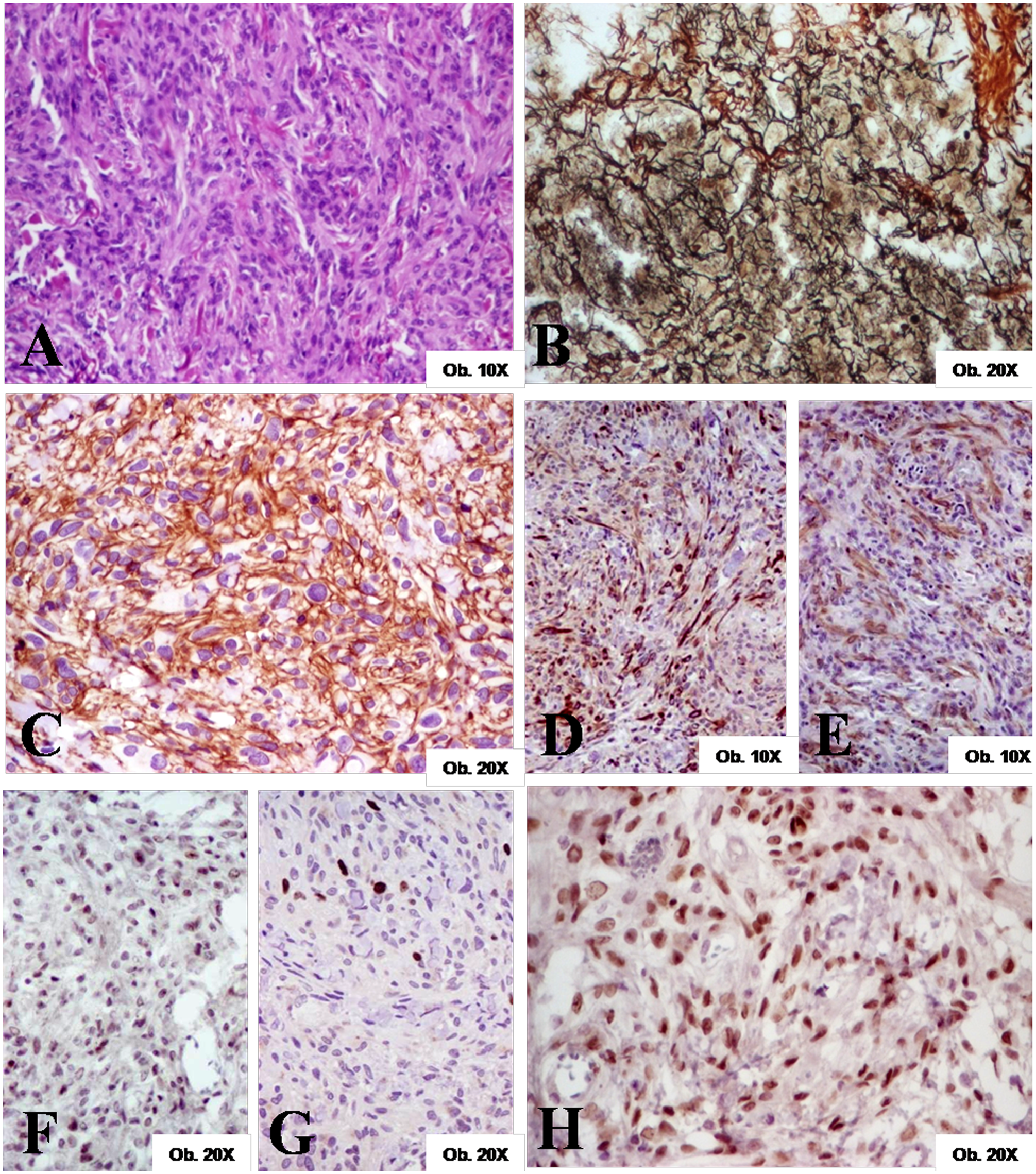
Breast myofibroblastoma
A particular feature of this case was the unusual stroma-rich reticulinic network, stained black with Gömöri impregnation (Figure 1).
Immunohistochemically, the tumor cells were diffusely positive for vimentin and CD34 and presented focal positivity for desmin and smooth muscle antigen (SMA). They did not express cytokeratins (CAM 5.2 and AE1/AE3), bcl-2, CD10, CD99, factor VIII–related antigen, or S-100 protein. Estrogen receptors marked 90% of the spindle cells tumor. There was no expression of progesterone receptors. For p53 and Ki67 the nuclear positivity index was less than 5% (Figure 1).
Our patient did not present recurrence 2 years after surgical intervention. He did not receive other additional therapies, such as radiotherapy or chemotherapy.
Discussion
Myofibroblastoma is a rare solitary benign tumor that may exhibit several morphological features. The fascicular structure was first reported in 1987 by Wargotz et al. First described tumors were characterized by fascicles of spindle cells having features of myofibroblasts, with intervening large hyalinized collagenous stroma, without necrotic or hemorrhagic areas, and having a surrounding pseudocapsule composed of compressed breast tissue (Wargotz et al., 1987).
The histological variants of this tumor include cellular (Schmitt & Ac Mera, 1998), collagenized, epithelioid, palisaded, lipomatous (Wahbah et al., 2010), hemangiopericytoma-like, and infiltrant features. Association of cartilaginous or smooth muscle areas and giant cells components were also reported (Miller, Levine, & Simmons, 1997). Sometimes, myofibroblastoma may arise in a mammary hamartoma and may present interspersed epithelial structures, which may increase the difficulty of diagnosis (Uchôa et al., 2010). Also described were cases with infiltrating patterns and calcifications seen on mammography (Kobayashi et al., 1996) or collision tumors (Wei & Zhu, 2011), which may lead to an incorrect diagnosis.
Our case presented a particular morphological aspect. It was about the classical cellular variant associated with a rich reticulinic network and scanty collagen bands. Other similar cases have been reported (Desrosiers et al., 2007), without mentioning the rich reticulinic network.
Regarding the final diagnosis of myofibroblastoma, all authors admit the crucial importance of IHC in establishing it. All the reported cases revealed a characteristic expression of vimentin, CD34, and desmin (Desrosiers et al., 2007; Lee et al., 2010; Sagar et al., 2011; Uchôa et al., 2010). Some tumor cells expressed SMA and calponin, and caldesmon is present in 50% of the cases reported (Magro et al., 2003). At the same time, they are negative for cytokeratin, factor VIII–related antigen, and S-100 protein (Lee et al., 2010; Uchôa et al., 2010) and may be negative or positive for bcl-2 (Desrosiers et al., 2007; Uchôa et al., 2010). Although in most of the cases tumor cells are negative for CD10 and CD99 (Uchôa et al., 2010), in some cases they can be positive (Magro, Caltabiano, Di Cataldo, & Puzzo, 2007; Tavassoli & Soares, 2003). Sometimes, myofibroblastoma may express androgen, estrogen, or progesterone receptors (Desrosiers et al., 2007; Magro et al., 2007) and may associate gynecomastia, as in our case.
The expression of these markers is independently by the histological type, except palisaded myofibroblastoma, which is a particular entity involving the lymph nodes, with a different histological origin and behavior. In the palisaded nodal myofibroblastoma, the tumor cells are positive for vimentin and SMA but do not express CD34 or desmin (Sagar et al., 2011). Compared with the other variants that are solitary tumors with pure benign behavior, the nodal palisaded myofibroblastoma may be multicentric and recurrent and may present bone metastases (Ciralik, Ezberci, Bulbuloglu, & Aydin, 2005).
The IHC pattern of myofibroblastoma can provide information about its histogenesis, which is incompletely understood. On one hand, it usually arises from pluripotent stroma fibroblasts, which are also positive for CD34. During maturation, these fibroblasts may be differentiated in several types of cells: myofibroblasts, smooth muscle cells, adipocytes, multinucleated cells, or condrocytes (Magro et al., 2003). It may explain the probable relationship of myofibroblastoma to spindle cell lipoma and solitary fibrous tumor, as mentioned by Desrosiers et al. (2007) in a recent article.
The cell origin of myofibroblastoma seems to be the modified fibroblasts between fibroblasts and smooth muscle cells (Ciralik et al., 2005). This differentiation may partly explain the smooth muscle component of myofibroblastomas, which was also seen in our case.
The peculiar stroma of our case, which is rich in reticulin fibers, could sustain this hypothesis of pluripotent stroma fibroblasts. It seems that, in the case of myofibroblastoma, the tumor cells retained the capacity for synthesis of reticulin fibers.
On the other hand, based on the strong positivity of tumor cells for estrogen receptors and associated gynecomastia (Wargotz et al., 1987), it is tempting to speculate that hormonal status might also play a role in tumorigenesis. This statement is sustained by the occurrence of myofibroblastoma in patients following androgen ablation therapy for prostate cancer (McManamin & Fletcher, 2001) and also the prevalence of breast myofibroblastoma in elderly males and postmenopausal females (Lee et al., 2010).
Depending on the type of cells derived from pluripotent fibroblasts, several tumors with similar features may develop in breast, including myofibroblastoma. This is the reason why the differential diagnosis is very difficult and may be based on the clinical, imagistic, histological, and IHC features. It should take into consideration the following breast lesions: reactive conditions (nodular fasciitis and fibromatosis), benign tumors (solitary fibrous tumor, leiomyoma, neuroma, neurofibroma, spindle cell lipoma, and vascular tumors), and their malignant variants, including dermatofibrosarcoma protuberans, phyllodes tumor, and malignant fibrous histiocytoma.
No recurrences have been reported in the literature after surgical local excision of breast myofibroblastomas, except in one case diagnosed in a 25-year-old woman, which is also the only reported bilateral breast myofibroblastoma (Yoon-Ki et al., 2010).
In conclusion, we underline that the diagnosis of breast male myofibroblastoma may sometimes be difficult and should be based on the clinical history and imagistic and pathological exams, including immunohistochemistry. Further studies are necessary to elucidate the molecular mechanism of controlling growth and proliferation of myofibroblasts using growth factors such as tumor growth factor-β, tumor necrosis factor, or fibroblast growth factor and their receptors.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the Romanian National University Research Council, Ministry of Education and Research, Projects Frame: PN II-PD, No. 504/2010, and by the Sectoral Operational Programme Human Resources Development, financed from the European Social Fund and by the Romanian Government under Contract Number POSDRU/89/1.5/S/60782.
