Abstract
Research investigating the role of paternal age in adverse birth outcomes is limited. This population-based retrospective cohort study used the Missouri maternally linked data set from 1989 to 2005 to assess whether paternal age affects fetal birth outcomes: low birth weight (LBW), preterm birth (PTB), stillbirth, and small size for gestational age (SGA). We examined these outcomes among infants across seven paternal age-groups (<20, 20-24, 25-29, 30-34, 35-39, 40-45, and >45 years) using the generalized estimating equation framework. Compared with infants born to younger fathers (25-29 years), infants born to fathers aged 40 to 45 years had a 24% increased risk of stillbirth but a reduced risk of SGA. A 48% increased risk of late stillbirth was observed in infants born to advanced paternal age (>45 years). Moreover, advanced paternal age (>45 years) was observed to result in a 19%, 13%, and 29% greater risk for LBW, PTB, and VPTB (very preterm birth) infants, respectively. Infants born to fathers aged 30 to 39 years had a lower risk of LBW, PTB, and SGA, whereas those born to fathers aged 24 years or younger had an elevated likelihood of experiencing these same adverse outcomes. These findings demonstrate that paternal age influences birth outcomes and warrants further investigation.
Keywords
Introduction
In recent years, a growing body of literature has examined the influence of maternal age on adverse fetal birth outcomes (Abel, Kruger, & Burd, 2002; Astolfi & Zonta, 1999; Croen, Najjar, Fireman, & Grether, 2007; de la Rochebrochard & Thonneau, 2002; Salihu, Shumpert, Slay, Kirby, & Alexander, 2003; Salihu, Wilson, Alio, & Kirby, 2008). Such studies have produced a wealth of evidence of an association between advanced maternal age and increased risks of fetal loss, preterm delivery, and small size for gestational age (Astolfi, De Pasquale, & Zonta, 2006; Astolfi & Zonta, 1999; Fretts & Usher, 1997; Nahum & Stanislaw, 2002; Raymond, Cnattingius, & Kiely, 1994; Salihu et al., 2008). Conversely, studies have also found young maternal age to be a risk factor associated with preterm delivery (Adams, Elam-Evans, Wilson, & Gilbertz, 2000; Fraser, Brockert, & Ward, 1995), neonatal mortality (Gilbert, Jandial, Field, Bigelow, & Danielsen, 2004), and low Apgar score (Chen, Wen, Demissie, Rhoads, & Walker, 2007).
Although the relationship between maternal age and adverse fetal birth outcomes has been widely studied, less attention has been devoted to the role of paternal factors, particularly paternal age. Adverse birth outcomes because of advanced paternal age have some biologic plausibility. Placental growth and development is influenced by the expression of paternal genes, and harmful gene mutations are more frequent among older men (Frank et al., 2002; Miozzo & Simoni, 2002; Reichman & Teitler, 2006), which may affect fetal development.
Studies that examined paternal age as a risk factor for adverse birth outcomes have yielded mixed results. Although some studies found an association between advanced paternal age and increased risk of spontaneous abortion (de la Rochebrochard & Thonneau, 2002; Kleinhaus et al., 2006; Slama et al., 2005), preeclampsia (Harlap et al., 2002), stillbirth (Nybo Andersen, Hansen, Andersen, & Davey Smith, 2004; de la Rochebrochard & Thonneau, 2002), schizophrenia (Kühnert & Nieschlag, 2004; Malaspina et al., 2001), autism (Reichenberg et al., 2006), low birth weight (Tough, Faber, Svenson, & Johnston, 2003), and other birth defects (Kühnert & Nieschlag, 2004; Savitz, Schwingl, & Keels, 1991; Yang et al., 2007), other studies found no evidence of a relationship between advanced paternal age and adverse fetal birth outcomes (Abel et al., 2002; Chen et al., 2008; Nahum & Stanislaw, 2003; Parker & Schoendorf, 1992). However, there are a variety of important limitations affecting these findings, including the lack of information on the role of important sociodemographic factors, including maternal smoking status, prenatal care, and race/ethnicity. The effect of young paternal age on feto-birth outcomes has been less documented; however, a previous study found younger paternal age to be associated with an increased risk of preterm birth, low birth weight, small size for gestational age, low Apgar score, neonatal mortality, and postneonatal mortality (Chen et al., 2008).
To further elucidate the relationship between paternal age and adverse feto-infant birth outcomes, we examined the effect of paternal age on the risk for low birth weight, preterm birth, stillbirth (early and late), and small size for gestational age using a validated database with births records between 1998 and 2005, while controlling for maternal sociodemographic characteristics.
Method
For this analysis, we used the Missouri maternally linked vital statistics records for the period of 1989 to 2005 (inclusive). Siblings were linked to their biological mothers using a unique identifier. Previous reports have explained the methods and algorithms used to link infant data to maternal records in this data set (Herman et al., 1997). The Missouri vital record system is not only a reliable data source, but it is also considered a standard to validate national data sets that involve matching and linking procedures (Martin, Curtin, Saulnier, & Mousavi, 2003).
Of the 1,035,547 births in the Missouri maternally linked cohort data files, 755,334 were used for analysis, after applying inclusion and exclusion criteria for the study. Only singleton infants born during the study period (1989-2005) were eligible for this study, which resulted in 55,698 cases being excluded. From the remaining births (n = 979,849), we excluded births at less than 20 weeks of gestation or greater than 44 weeks of gestation (n = 55,807). Furthermore, those cases lacking information on maternal or paternal age (n = 168,391), small size for gestational age (n = 86), and birth weight (n = 231) were excluded. Detailed information on this process is provided in Figure 1.

Flow diagram of exclusion criteria (Missouri: 1989-2005)
The main exposure of interest in this study was paternal age. Information on paternal age and other demographics are routinely recorded on birth certificates after delivery. We classified paternal age into the following categories: <20 years, 20-24 years, 25-29 years, 30-34 years, 35-39 years, 40-45 years, and >45 years. Fathers in the age-group 25-29 years were used as the referent category in the analysis.
The main outcomes of interest were low birth weight, preterm birth, very preterm birth, stillbirth (early and late), and small size for gestational age. Low birth weight was defined as <2,500 grams at birth. Preterm birth was defined as birth between 33 and 37 weeks of gestation. Very preterm birth was defined as birth at ≤33 weeks of gestation. Stillbirth was defined as in utero fetal death at ≥20 weeks of gestation (early ≤28 weeks, late >28 weeks). Small size for gestational age was defined as less than the 10th percentile of birth weight for gestational age (Alexander, Kogan, Martin, & Papiernik, 1998). Gestational age was calculated from the date of last normal menses minus the date of birth, in weeks.
We compared the following sociodemographic characteristics across paternal age-groups: maternal age (<35 and ≥35 years), maternal race (White, Black, or Other), maternal educational status (<12 or ≥12 years), maternal marital status (married, single, or not stated), prenatal smoking (yes or no), prenatal alcohol consumption (yes or no), prenatal care (adequate or inadequate), and common obstetric complications. To describe the level of prenatal care utilization, we used the revised graduated index algorithm (R-GINDEX; Alexander & Cornely, 1987; Alexander & Kotelchuck, 1996). The index assesses the adequacy of care based on the trimester of prenatal care initiation, the number of visits, and the gestational age of the infant at birth. Inadequate prenatal care was defined as either missing prenatal care information, suboptimal prenatal care, or no prenatal care at all. A pregnancy was considered to have complications if any of the following obstetric conditions were present: preeclampsia, eclampsia, hypertension, placental abruption, placental previa, or anemia.
Statistical Analysis
Differences in sociodemographic characteristics and maternal pregnancy complications across paternal age-groups were analyzed using the chi-square test. Continuous variables (i.e., birth weight and gestational age) were analyzed using the analysis of variance test. The independent association between paternal age and the risk of adverse fetal outcomes was assessed with multivariable logistic regression, in which we controlled for maternal characteristics and common obstetric complications. Based on biologic plausibility, findings in the literature, and statistical significance testing, we retained the following covariates in the final model: maternal age, race, education, marital status, prenatal smoking, alcohol use, prenatal care, year of infant birth, and obstetric complications.
In our study, 565,336 (74.85%) of mothers had more than one pregnancy. Naturally, siblings from the same mother are more or less subjected to similar prevailing conditions and, thus, are equally affected by attributes that characterize the mother, such as young maternal age, adequacy of prenatal care, and smoking and alcohol use during pregnancies. As a result, adverse fetal outcomes in early pregnancies may increase adverse fetal outcome in subsequent pregnancies. Therefore, to avoid this bias and account of intraclass correlation to assess the independent effect of paternal age on fetal birth outcomes, we applied the generalized estimating equation (GEE). Since some women contributed to more than one birth outcome, observations on siblings will be more correlated than those for nonsiblings as a result of shared risk factors (e.g., same genes, same maternal behaviors, such as smoking, etc.). This would lead to increased intracluster correlations that need to be adjusted for in the analysis, hence, the need to use GEE framework. Tests of hypothesis were two tailed with a Type 1 error rate fixed at 5%. SAS version 9.2 (SAS Institute, Cary, NC) was used to perform all analyses. This study was approved by the institutional review board at the University of South Florida prior to initiation.
Results
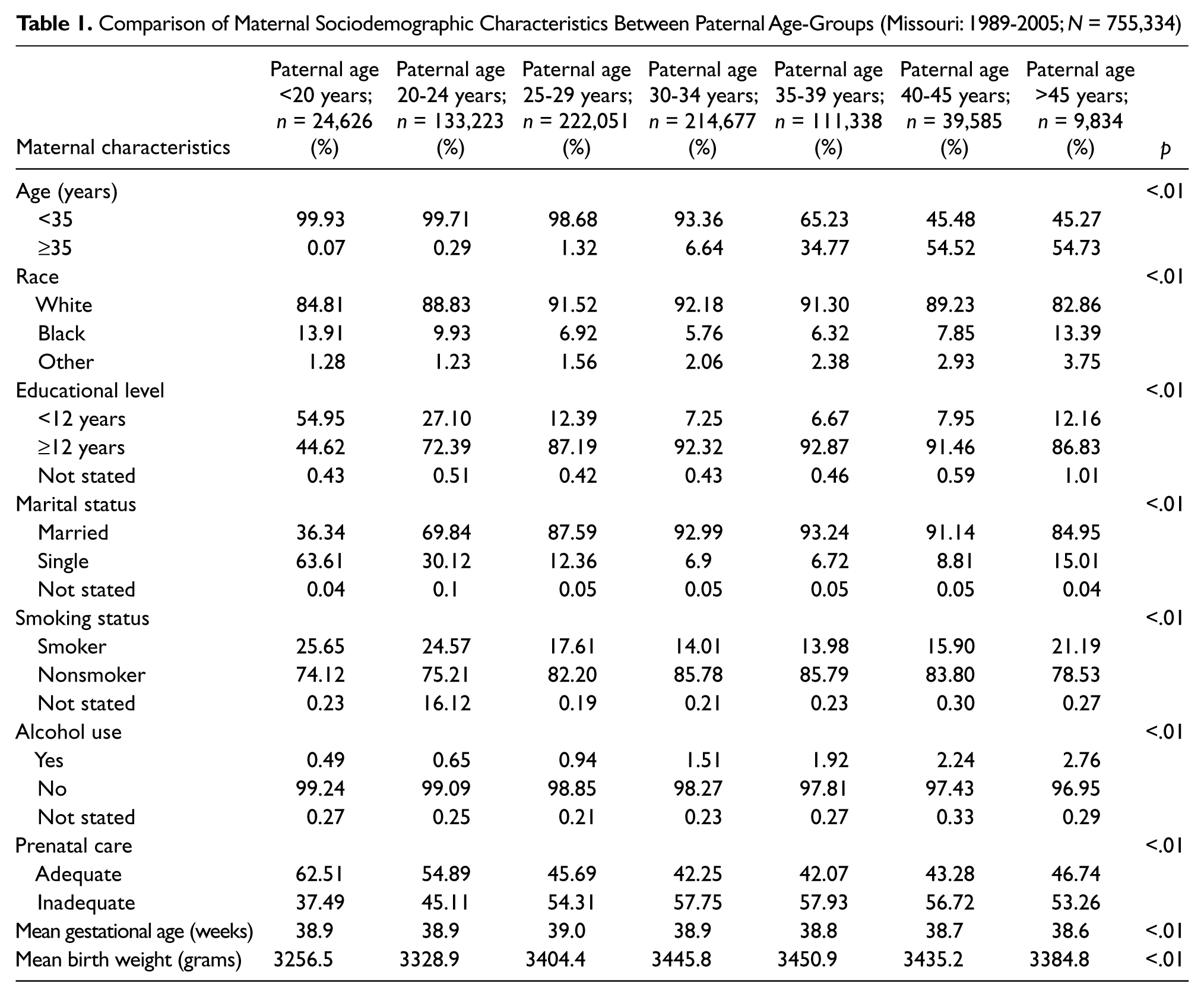
A comparison of selected sociodemographic characteristics between paternal age-groups is presented in Table 1. The majority of births were to fathers between the ages of 25-29 years (n = 222,051, 29.4%) and 30-34 years (n = 214,677, 28.4%), whereas the fewest births occurred among fathers older than 45 years of age (n = 9,834, 1.3%). Infants born to fathers who were younger than 20 years old were more likely to be born to a Black, single, younger mother with a lower level of education and a higher likelihood of adequate prenatal care and prenatal cigarette smoking. Infants born to fathers of advanced age (>45 years) were more likely to have an older mother with a higher educational level and a greater likelihood of smoking and alcohol use during pregnancy. The mean gestational age in our study population was 38.91weeks (standard deviation: 2.35), and the mean birth weight was 3,406 grams (standard deviation: 563.9). A significant difference in both mean gestational age (p < .01) and mean birth weight (p < .01) existed among the paternal age-groups.
Comparison of Maternal Sociodemographic Characteristics Between Paternal Age-Groups (Missouri: 1989-2005; N = 755,334)
The frequencies of common obstetric complications by paternal age are provided in Table 2. Female partners of the fathers with advanced age (>45 years) were the most likely to experience hypertension, placental abruption, and placental previa, whereas the highest proportions of anemia, preeclampsia, and eclampsia were observed in mothers who had male partners who were younger than 20 years (p < .01). A modest dose–response relationship was observed between paternal age and the maternal complications of placental previa and hypertension, with rates increasing with increasing paternal age (p for trend <.01).
Comparison of Maternal Complications Between Paternal Age-Groups (Missouri: 1989-2005; N = 755,334)

Table 3 depicts the crude frequencies of fetal birth outcomes by paternal age-group. Overall, the highest rates of stillbirth and preterm birth were observed among infants with fathers of advanced paternal age (>45 years), whereas small size for gestational age and low birth weight were most frequent in infants of young adult fathers (aged 20-24 years). In this analysis, there were a total of 72,058 preterm and very preterm births, and almost all of them (n = 72,025) had information on whether the preterm and very preterm delivery resulted from induced births or not. It was observed that 16.75% (n = 12,061) of the preterm and very preterm births were induced (data not shown).
Crude Frequency Comparison of Fetal Birth Outcomes Between Paternal Age-Groups (Missouri: 1989-2005; N = 755,334)

Table 4 summarizes the results of the analysis for the association between paternal age and fetal birth outcomes, accounting for sibling correlation. After adjustment for maternal age, paternal race, maternal education, smoking and alcohol use during pregnancy, adequacy of prenatal care, and year of infant birth, advanced paternal age (>45 years) was associated with a 48% increased risk of late stillbirth (adjusted odds ratio [AOR] = 1.48, 95% confidence interval [CI] = 1.04-2.10), a 19% increased risk of low birth weight (AOR = 1.19, 95% CI = 1.09-1.29), a 13% increased risk of preterm birth (AOR = 1.13, 95% CI = 1.05-1.22), and a 29% increased risk of very preterm birth (AOR = 1.29, 95% CI = 1.15-1.44), as compared with the referent category (fathers in the age-group 25-29 years). Among infants of fathers aged 40-45 years, there was a 24% increased risk for stillbirth (AOR = 1.24, 95% CI = 1.04-1.47) and a 5% lower risk of small size for gestational age (AOR = 0.95, 95% CI = 0.90-0.99). Infants born to fathers aged 30-39 years had a reduced risk for low birth weight, preterm birth, very preterm birth, and small size for gestational age, as compared with the referent group. Conversely, infants born to young fathers aged 24 years or less had an elevated risk of low birth weight, preterm birth, very preterm birth, and small size for gestational age, as compared with the referent group. Overall, for low birth weight, preterm birth, and very preterm birth, a J-shaped curve was observed with heightened risk among infants of young fathers (<20 years) and older fathers (>45 years) and reduced risk among those of fathers in their 30s.
Adjusted Estimates of Adverse Fetal Birth Outcomes by Paternal Age-Groups (Missouri: 1989-2005; N = 755,334)
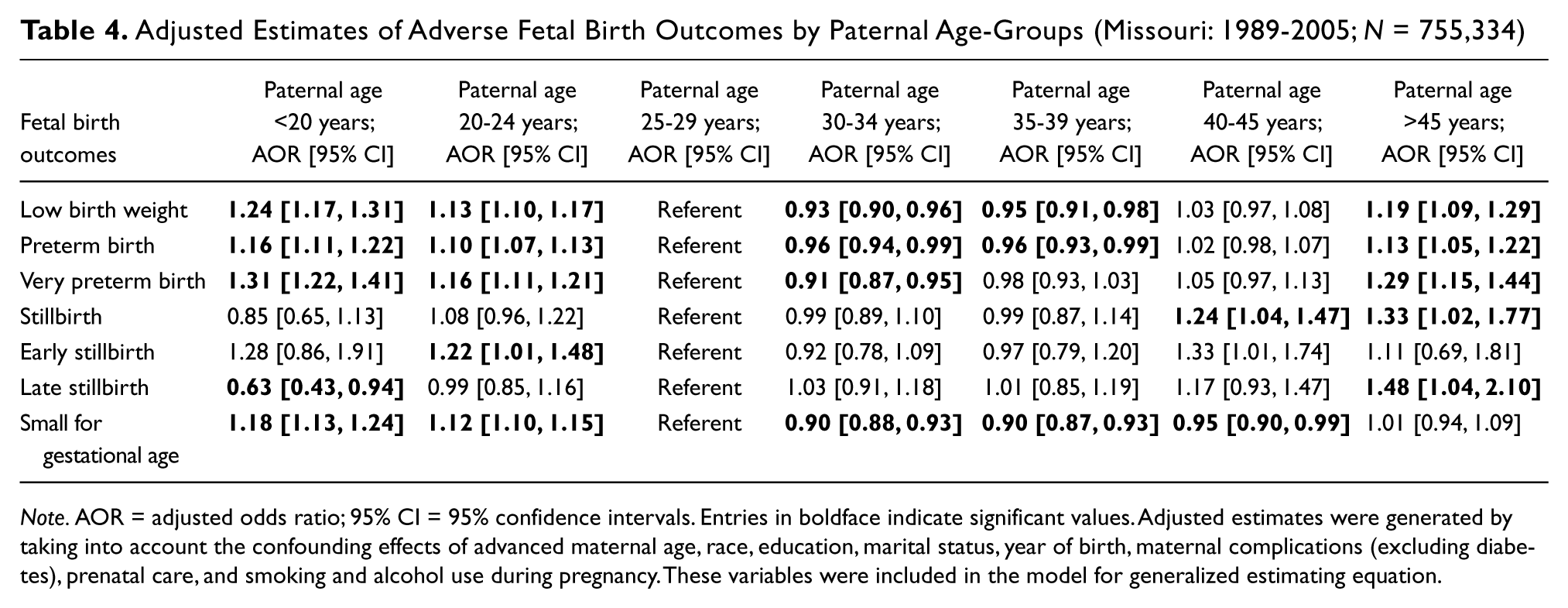
Note. AOR = adjusted odds ratio; 95% CI = 95% confidence intervals. Entries in boldface indicate significant values. Adjusted estimates were generated by taking into account the confounding effects of advanced maternal age, race, education, marital status, year of birth, maternal complications (excluding diabetes), prenatal care, and smoking and alcohol use during pregnancy. These variables were included in the model for generalized estimating equation.
Discussion
In this study, infants born to fathers aged 40 to 45 years had a 22% greater likelihood for stillbirth compared with fathers aged 25 to 29 years. Previous studies confirm the findings of higher risk of adverse birth outcomes among older fathers (Astolfi, De Pasquale, & Zonta, 2004; Reichman & Teitler, 2006; Zhu, Vestergaard, Madsen, & Olsen, 2008). However, few have investigated the association between paternal age and stillbirth (de la Rochebrochard & Thonneau, 2002; Nybo Andersen et al., 2004). Even though no risk difference was found between advanced paternal age (>45 years) and the risk of overall stillbirth, our results show a 48% elevated risk for stillbirth associated with older paternal age in the later period of gestation. Previous research has similarly noted that advanced paternal age is related to an increased risk of late fetal death (Nybo Andersen et al., 2004). As paternal genes contribute to placental growth and harmful gene mutations are more frequent with age (Frank et al., 2002; Miozzo & Simoni, 2002; Reichman & Teitler, 2006), this may potentially explain the increased risk of late stillbirth among infants born to older men. However, this remains purely speculative since we do not have genetic information in the data set that we analyzed.
Fathers of advanced age (>45 years) in our study also had a 13% to 29% increased risk of low birth weight, preterm birth, and very preterm birth. This is similar to previous findings of other investigators (Tough et al., 2003). Additionally, our study reinforced the findings of Chen et al. (2008), which reported that infants born to young adult and adolescent fathers have a heightened risk of low birth weight, preterm birth, and small size for gestational age. However, in both these quoted studies, the risk estimates are not comparable to the present study because of differences in age categorizations and data analysis approaches. While our study provides further evidence of the relationship between paternal age and these critical feto-infant morbidities, previous research yielded mixed results, with some revealing no evidence of an association (Abel et al., 2002; Chen et al., 2008; Nahum & Stanislaw, 2003; Parker & Schoendorf, 1992). Given this conflicting evidence, additional investigation is warranted to further elucidate the relationship between paternal age, particularly adolescence and older age, and fetal birth outcomes.
Our study has some limitations. The analysis we performed included a time range during which the study population would have been exposed to varying obstetric practices. However, the inclusion of the year of birth in the adjusted model minimized the influence of this source of bias. Without information on income and economic status, uncontrolled effects could have biased our findings because educational attainment captures only a narrow spectrum of socioeconomic status (Braveman et al., 2005). Furthermore, as the data set used in this study does not include various paternal sociodemographic characteristics, such as race and educational level, the findings are restricted in the comprehensive examination of factors that could potentially mediate the relationships between paternal age and adverse birth outcomes.
Despite these shortcomings, our study has some noteworthy strengths. The large sample size for this study allowed for the adjustment of several potential confounding factors. Another merit is the utilization of a population-based data set, which enhances the generalizability of these findings. Using an entire population data source minimizes the chances of selection bias influencing the results. Finally, we were also able to examine various feto-infant morbidity outcomes at birth, unlike other studies that focused only on preterm birth or low birth weight. We controlled for several potential confounders, although we cannot rule out residual confounding because of unmeasured variables.
The influence that paternal age has on fetal birth outcomes may have important implications. The results of this study raise the question of whether prenatal care is intrinsically related to adverse birth outcomes. The lack of prenatal care or inadequate prenatal care is considered a reliable predictor of fetal health (D’Ascoli, Alexander, Petersen, & Kogan, 1997; Wilcox & Mark, 1994). However, in our study, infants born to fathers aged 30-45 years were least likely to have adequate prenatal care, yet they had reduced risk for low birth weight, preterm birth, and small size for gestational age. These findings suggest that other critical paternal sociocultural, sociodemographic factors may be at play that could not be assessed because of the limitations of the data set used in this analysis. Previous studies have indicated that an association exists between paternal race/ethnicity (Krishnakumar et al., 2011; Palomar, DeFranco, Lee, Allsworth, & Muglia, 2007), education (Blumenshine, Egerter, Libet, & Braveman, 2011; Bray, Gunnell, & Smith, 2006; Chen et al., 2008; Kiernan, 1997; Saikh et al., 2011), support and involvement (Alio et al., 2011; Alio, Kornosky, Mbah, Marty, & Salihu, 2010; Ghosh, Wilhelm, Dunkel-Schetter, Lombardi, & Ritz, 2010), and alcohol, tobacco, and drug use (Chen et al., 2008; Little & Sing, 1986; Vine, 1996) and adverse feto-infant outcomes. Consequently, it may be beneficial for interconception care, which incorporates prenatal care, to include both mothers and fathers to prevent or minimize health problems for mother and child (Johnson et al., 2006; Mercer et al., 1999; Surkan, Stephansson, Dickman, & Cnattingius, 2004). Further investigation of the role of paternal lifestyle characteristics on fetal birth outcomes is needed to yield further understanding of this relationship that will aid in the development and implementation of appropriate interventions.
The aim of this study was to assess the effect of paternal age on adverse fetal birth outcomes. We found that paternal age was an independent risk factor for adverse fetal birth outcomes, in which elevated risks for stillbirth and late stillbirth were observed for the older paternal age categories. These findings support the current hypothesis that in utero fetal development and survival are influenced by not only maternal but also paternal characteristics and is supported by previous research on paternal aging and stillbirth risk (Astolfi et al., 2004). Furthermore, the findings of this study are congruent with the notion of including fathers within the current framework of prenatal care and decision making, as well as counseling during the preconception period, to optimize pregnancy outcomes. Future study direction should incorporate a comprehensive approach that encompasses physical, social, and biological influences contributed by the father.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
