Abstract
Objective:
Totally endoscopic coronary artery bypass (TECAB) procedures pose significant challenges, motivating the development of Octocon, an automated endoscopic connector designed for coronary anastomoses in off-pump and endoscopic settings. This feasibility study aimed to assess Octocon’s functionality and maneuverability in closed-chest conditions during robot-assisted TECAB simulations.
Methods:
The Octocon deployment comprises a 3-step procedure. Initially, delicate self-aligning microstapling technology is used to attach connector halves to individual blood vessels. Subsequently, the connector halves are joined to accomplish the anastomosis process. TECAB conditions were simulated using a dedicated box housing ex vivo porcine hearts. The study, conducted by 3 experienced surgeons, investigated the feasibility and standardization potential of a robot-assisted procedure employing Octocon. It evaluated maneuverability in closed-chest conditions and assessed the effectiveness of grafting internal mammary artery segments to different heart regions using single graft, jump graft, and Y-graft constructions.
Results:
The robot-assisted procedure, using 4 standard instruments, successfully completed all 3 steps in 18 anastomotic procedures. In 96% of cases, the procedural steps were accomplished on the first attempt. The feasibility of constructing jump graft and Y-graft geometries on both anterior and posterior heart walls was demonstrated. Furthermore, experiences affirmed the device’s endoscopic user-friendliness, ease of teachability, reproducibility, and potential to achieve expedient, leak-free anastomoses.
Conclusions:
This ex vivo study confirmed Octocon’s potential suitability and functionality for TECAB. The device can create diverse grafting strategies and achieve wide-open vascular connections on various heart regions, highlighting its potential in advancing minimally invasive, robot-assisted coronary procedures. These promising results justify further exploration for integration into clinical practice.

Keywords
Central Message
Octocon is an automated endoscopic connector for coronary anastomoses, which was evaluated in a feasibility study for totally endoscopic coronary artery bypass. Results indicate high efficiency when integrated onto the robot platform, successfully facilitating reliable vascular connections and warranting further clinical investigation.
Introduction
The challenges associated with performing reliable hand-sewn coronary anastomoses in constrained spaces are well established. 1 Despite the proven benefits of minimally invasive surgical techniques for myocardial revascularization, such as improved patient outcomes and faster recovery compared with conventional open sternum operations, the adoption of these techniques remains limited.2–5 This persistent challenge highlights the ongoing need for advancements in cardiac surgical procedures. In addition, the worldwide adoption of sternal-sparing techniques for coronary artery bypass grafting in robot-assisted minimally invasive direct coronary artery bypass and totally endoscopic coronary artery bypass (TECAB) procedures faces a slow uptake. 6 In this context, we set out at University Medical Center Utrecht to develop an innovative microconnector technology with the aim of improving the precision of coronary anastomoses through the use of an automated device. Our novel microconnector technology is designed to address the challenges associated with off-pump and endoscopic coronary anastomosis construction. By offering simplicity, versatility, and precision, it has the potential to open up broader adoption of minimally invasive approaches in coronary surgery.
Recognizing the importance of a real-world application, we have chosen to evaluate the performance of the Octocon™ Vascular Connection System (OctoVascular, Utrecht, The Netherlands) in the most demanding setting of TECAB. This study explored the feasibility and potential standardization of a robot-assisted procedure using the connector. The findings from this research are expected to provide valuable insights into the practical implementation and effectiveness of Octocon, paving the way for advancements in minimally invasive coronary revascularization procedures.
Methods
Procedure Setup
The procedure setup was tailored to emulate robot-assisted TECAB procedures. The study was performed using the da Vinci Si system (Intuitive Surgical, Sunnyvale, CA, USA). A range of instrumentation as often used for “conventional,” hand-sewn TECAB was available, including high-resolution 0^ and 30^ scopes. To simulate the chest, a closed box with preexisting holes was used, offering the option to choose and implement multiple port placements at appropriate sites. Three ports were placed at typical locations, mimicking the standard TECAB positions at the second, fourth, and sixth intercostal spaces (ICSs). The robotic arms were docked in a standard configuration on the left side of the surgical table. A service port, AirSeal® 12 mm (CONMED Corporation, Largo, FL, USA) was positioned in the fourth ICS at the parasternal line, to enable the introduction of the Octocon into the “chest.” Although not strictly necessary for our purpose, a fourth subcostal port was inserted to allow the introduction of a cardiac stabilizer to replicate a real-life setting as accurately as possible. Digital 720p recording equipment was connected to the da Vinci system to document the entire experiment for subsequent analysis and evaluation.
Endoscopic Octocon
For endoscopic applications, the Octocon device underwent a redesign, transforming it into a sleek version tailored explicitly for fully endoscopic platforms. This refined model is compatible with insertion through 12 mm ports and creates oval-shaped side-to-side anastomoses on target vessels with internal diameters ranging from 1.3 to 3.0 mm. The Octocon system comprises several components including a hydraulically driven expanding applier and 2 titanium connector halves for sequential delivery to the graft and coronary vessels. The expandable Octocon applier, introduced through a 3 mm arteriotomy, deploys 1 connector half at a time. Each connector half is designed with a very thin flange that resides almost entirely outside the bloodstream. Only the blunt inner surface of the connector orifice and the 12 pairs of microstaples are exposed to the bloodstream, totaling a mere 3 mm2 of metal surface area. The device offers an anastomotic orifice area exceeding 7 mm2. After first stapling the bypass and then the coronary target, the Octocon halves are moved to facing positions and can be joined together seamlessly with a clicking mechanism, establishing a complete vascular connection. Thus, the anastomosis construction involves a logical process of 3 controlled steps: deploying the graft connector half, deploying the coronary connector half, and joining the 2 halves to form a complete vascular connection (Fig. 1). No apex positioner was used.
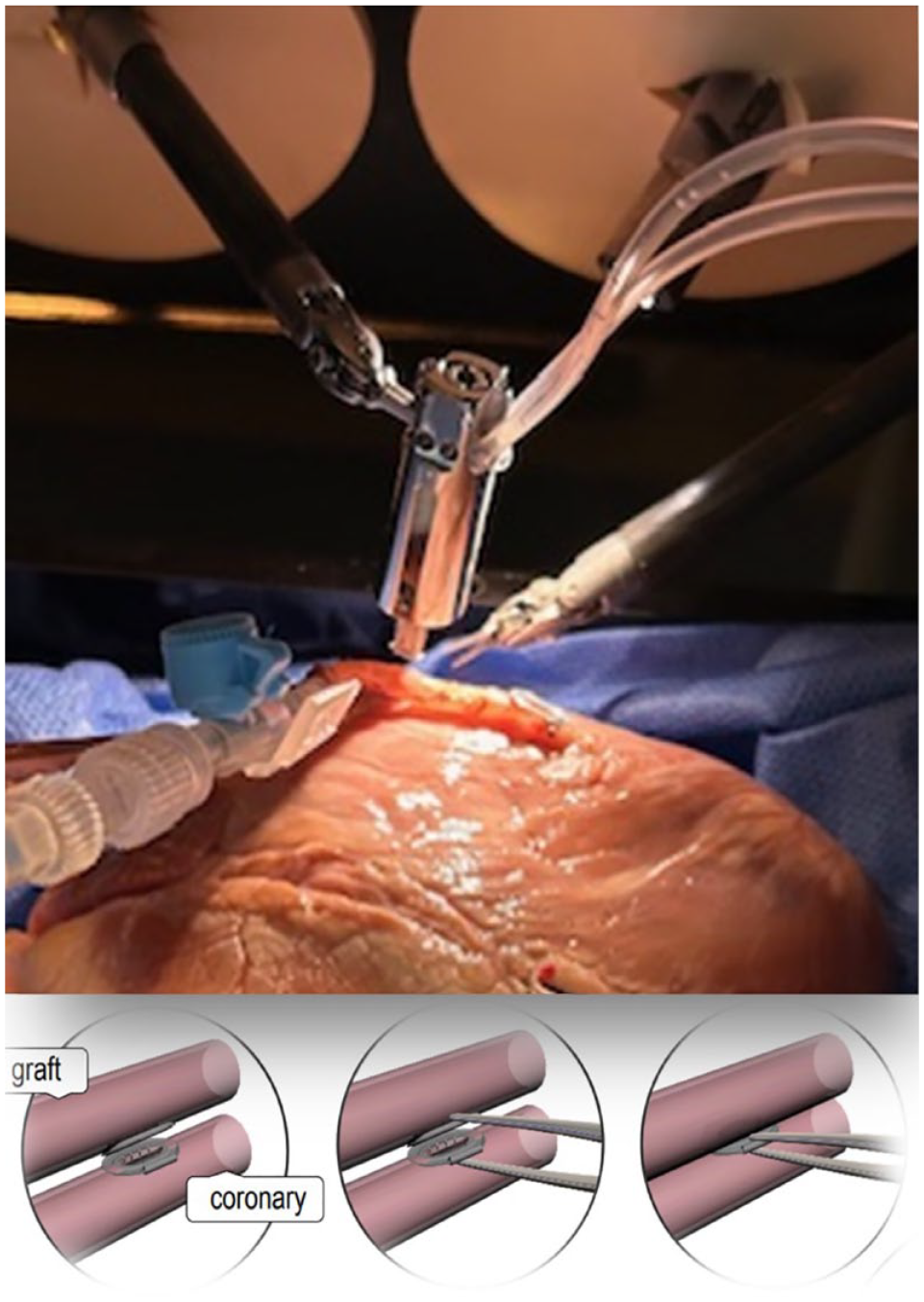
In the picture, the Octocon applier is positioned within the endoscopic box for deploying the graft connector half. The illustration demonstrates the process of joining the 2 halves to form the complete vascular connection.
Outcomes
First, we assessed the prerequisites for using Octocon on the robotic platform, followed by standardizing the procedure for constructing both single and jump grafts on the anterior and posterior heart walls. Both regions on the heart wall were feasible targets inside the endoscopic box, specifically the left anterior descending artery (LAD) and the obtuse marginal (OM) branches. The procedural standardization involved a detailed examination of the necessary instruments for Octocon manipulation and the spatial requirements for its maneuverability within the simulated chest wall. Subsequently, our attention pivoted toward appraising the efficacy of Octocon.
Primary outcome
The primary objective was to assess the effectiveness of Octocon, measured through the success rates of executing all 3 crucial steps required to create an anastomosis. These steps involved accurately deploying the first connector half in the graft and the second connector half in the coronary vessels, followed by achieving precise alignment of the 2 halves, essential for their final permanent fusion. Each step was deemed equally vital in the creation of the anastomoses, thus prompting individual evaluation in this feasibility study. An unsuccessful attempt in executing a step was defined as an inability to complete correctly that specific step, regardless of the number of attempts made.
Secondary outcomes
Secondary outcomes included the number of attempts needed to successfully execute a step, proficiency in conducting a fully camera-guided procedure, and the tally of unintentional drops of the device applicator. We evaluated the feasibility of performing anastomoses on both the anterior and lateral walls of the heart as well as the feasibility of executing various graft geometries such as jump graft and Y-graft constructions. The time required to complete a robot-assisted Octocon anastomosis was recorded. The quality of the completed anastomoses was evaluated visually after opening the anastomoses by longitudinally cutting the graft or hydrostatically with a pressurized water test. The pressure reached by the system during the test was recorded in mm Hg. Finally, we evaluated the ease of use, teachability, and reproducibility of the Octocon’s deployment procedure by introducing 2 additional surgeons with no previous experience with this system and recorded their results.
Statistical Analysis
Given the limited sample size, formal statistical analysis was not performed. Descriptive statistics such as percentages, means, or frequencies were calculated.
Results
Over a 5-day period, 3 cardiac surgeons, each with different levels of experience in robotic surgery, assessed the endoscopic Octocon connector device on a robotic platform. Among them, only 1 surgeon had prior experience in performing anastomoses with the Octocon, albeit not in a robotic setting. A second surgeon conducted a single manual deployment before attempting the procedure robotically for the first time. The third surgeon, an expert in TECAB, directly attempted robotic procedures without prior manual experience but had observed a manual procedure before transitioning to the robotic approach.
Surgical Procedure
Setup and device maneuverability
The endoscopic box simulating the chest anatomy measured approximately 45 cm in length, 35 cm in depth, and stood at a height of 30 cm. A swine heart was positioned inside the box, positioned to simulate a standard setting for TECAB. The graft material used in various sessions included segments of cadaveric internal mammary artery (IMA) graft and a dissected right coronary artery from a swine heart, with dimensions comparable to the human IMA. The Octocon applicator could be inserted smoothly into the box without resistance through the AirSeal 12 mm working port, positioned on the parasternal line at the fourth ICS. To manipulate the device safely, a 5 cm clearance between the thoracic wall and the mediastinal structures proved sufficient. The experiment setup and endoscopic box are shown in Figure 2. Various types of forceps were assessed during the standardization process of the instruments to optimize the Octocon’s delivery process, including the Resano and DeBakey forceps and the large needle driver. The most optimal maneuverability and grip stability for the Octocon were attained with the large needle driver on the right robotic arm. Atraumatic tissue forceps, such as the DeBakey forceps, did not perform as well due to a less stable grip resulting from their distinct coarsely ribbed grip panel. Resano forceps, albeit relatively large, were a viable alternative option. The Octocon device was easily manipulated by gripping it at its upper side, which was equipped with gripping features on 4 sides. Throughout all experiments, no involuntary falling occurred.

Experiment setup and endoscopic box.
Step 1: graft incision and Octocon’s graft half deployment
The approximately 3 mm vessel incision, required for the device insertion, was performed using standard Potts scissors having a millimeter scale to facilitate length eyeballing. Next, the device’s front end was gently inserted into this incision with a tilting motion, similar to the action of putting on a shoe. As mentioned, the applicator was manipulated with the large needle driver in the right hand to maintain a stable grip. In the left hand, the small Black Diamond forceps proved to be very suitable for delicately manipulating the anatomical structures. The introduction and deployment of the Octocon in the graft are illustrated in Figure 3. Subsequently, the applicator was retrieved and reloaded for the next step.
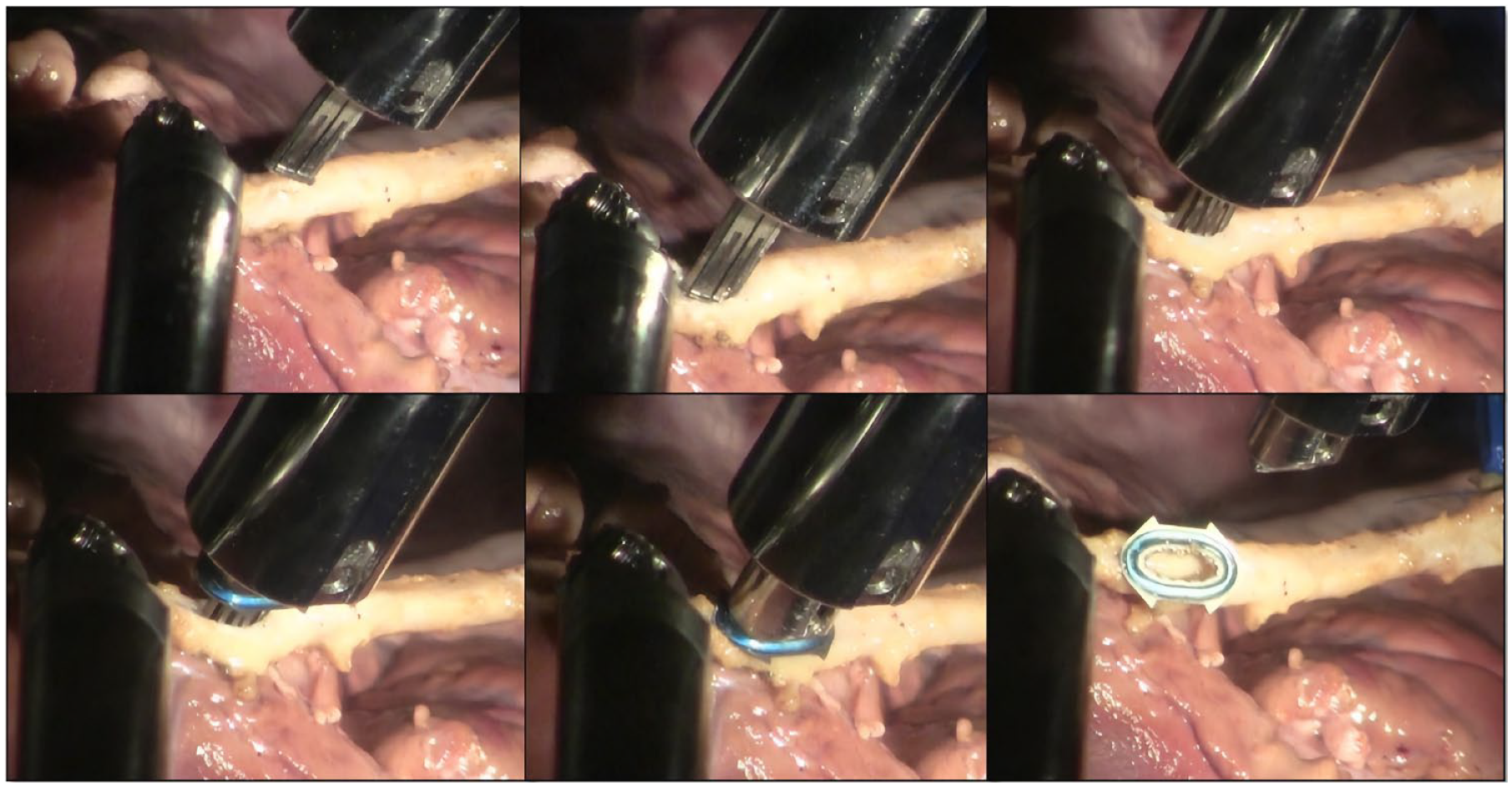
Stepwise introduction and deployment of the Octocon connector’s half in the graft.
Step 2: coronary incision and Octocon’s coronary half deployment
This step repeated the previous step, now focused on deploying the Octocon half to the coronary artery. After making a similar 3 mm incision with the Potts scissors, the Octocon applicator was reintroduced through the working port for insertion into the coronary vessel, mimicking the earlier process. Following this deployment, the applier was retrieved out of the working port.
Step 3: fusion of the Octocon’s halves and anastomoses completion
Having both the graft and coronary Octocon halves positioned, the execution of the third step benefitted from a right instrument change into a Black Diamond forceps. The critical task here was the alignment for fusion of the 2 Octocon halves. The fine Black Diamond forceps in both robotic arms proved very suitable for the precise alignment and subsequent clicking fusion of the graft and coronary Octocon halves. Correct alignment was visually checked, optionally zooming the camera in. Visualization appeared optimal with the 30° scope, angled 10° to 30° downward (Fig. 4).
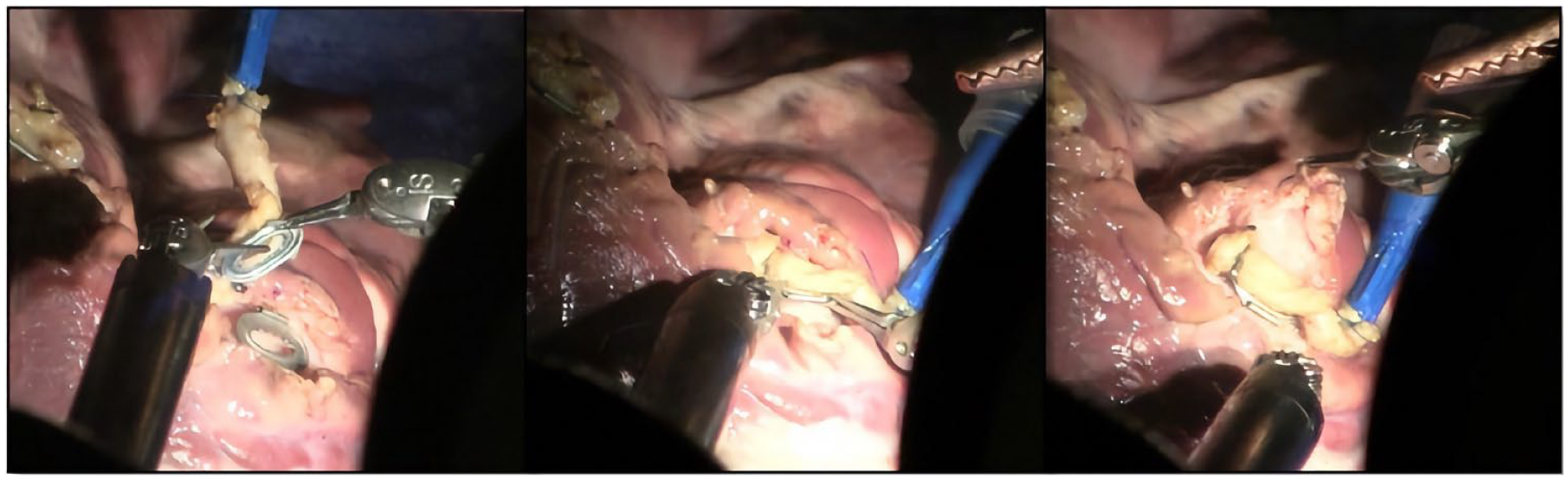
Correct alignment of the 2 connector’s halves before final fusion.
Efficiency of Robot-Assisted Octocon
A total of 18 anastomoses were attempted on the robotic platform, with 17 performed in the simulated closed-box environment described above. However, the very first anastomosis was performed in an open box. Each anastomosis procedure comprised the 3 essential steps, amounting to 54 consecutive steps individually evaluated in this feasibility study. The success rate for completing the entire 3-step procedure robotically was 100% (Table 1, Supplemental Video).
Results of the 3-Step Procedure and Consecutive Octocon Graft Connections Across 18 Cases.

Attempts Required for a Robot-Assisted Octocon Anastomoses
The number of attempts required to successfully complete an Octocon anastomosis on the robotic platform was evaluated. In 96% of the cases, the procedural steps were successful on the first attempt. Overall, a second attempt was required in 4% of cases, in both cases for achieving the optimal deployment of the Octocon’s graft half (i.e., in the conduit). The first instance was related to poor, thick-walled graft quality. The initial deployment attempt resulted in a partially obstructed anastomotic orifice. This was likely the result of the applicator not being fully tilted into the lumen on 1 side. Thus, upon expansion, the applicator anvil caused a local vessel wall dissection (Supplemental Fig. 1). Awareness of this failure mode helped to avoid it by a visual check. Next, the affected graft section was surgically removed, and a subsequent deployment was successfully performed. In the second instance, a graft incision that was too small resulted in a similar improper full insertion of the applicator head, also leading to a partial obstruction of the anastomosis orifice. In this case, the connector half was cautiously detached from the graft with the intention of redeploying the Octocon in the same location. However, after connector removal, the graft’s incision length, as measured with Potts scissors, exceeded the standard 3 mm. This issue was resolved by shortening the effective incision length with a crossed polypropylene 7-0 stitch at the downstream side. This adjustment enabled the successful redeployment of a new connector at the original location. No need for a second attempt in coronary graft deployment was observed.
Coronary Target and Graft Geometry
Among the procedures, 10 (55.6%) targeted the anterior heart wall (with LAD), 1 (5.6%) the anterolateral wall, and 5 (27.8%) the posterior heart wall (with OM). In 1 OM case, the graft was successfully deployed using a top-down approach (Supplemental Fig. 2). Eleven procedures (61.1%) were single distal anastomoses, 5 (27.8%) were jump grafts, and 2 (11.1%) realized the proximal connections of Y-graft constructions.
Procedural Time
The time required to perform the 3-step anastomoses varied throughout the experiment, primarily due to graft preparation, incision, and measuring the incision length. The surgeon with TECAB experience was the fastest in performing the graft and coronary incision with the required length (min). After the third procedure, all surgeons reached a comparable result. After completing both the graft and coronary incisions, all surgeons consistently completed the entire sequence of the 3 Octocon steps (from introducing the applier in the chest box simulator to closing the 2 plates) in less than 5 min. Reloading the applier outside the endoscopic box consistently took less than 1 min.
Ability to Perform a Completely Camera-Guided Procedure
The standard camera port position in the TECAB setting enabled a fully camera-guided procedure. All procedural steps were carried out under direct camera visualization. Zooming in and out facilitated navigation of the Octocon within the simulated chest wall: zooming out for overall orientation and zooming in for precise introduction of the Octocon into the vessel and alignment of its halves. During coronary deployment and alignment on the posterior heart wall for grafting the OM coronary, the camera angle was shifted from 30° upward to 30° downward.
Procedure’s Ease of Use
All 3 surgeons were able to successfully perform all of the steps necessary for creating an anastomosis with the Octocon on the first attempt. The time needed to perform a vascular connection was similar between the surgeons.
Quality Assessment
In the first 2 cases, quality assessment was conducted through visual inspection only. The graft was longitudinally cut open above the anastomoses, allowing observation of the anastomotic orifice area. Subsequent anastomoses underwent leak testing using pressurized water. The maximum pressures reached depended on the target vessel runoff. No significant leakage was observed in the Octocon anastomoses at pressures up to 250 mm Hg. Table 2 presents the quality assessment alongside the coronary target, graft geometry, and surgeon performance.
Assessment of Quality and Characteristics in 18 Consecutive Octocon Anastomoses by 3 Surgeons.

Abbreviations: AL, anterolateral wall; LAD, left anterior descending artery; OM, obtuse marginal.
Inspection or pressure test (mm Hg).
Unintentional Octocon Applier Drop
No unintentional applier drops were observed. On one occasion, the Octocon applier was intentionally released from the large needle driver after the deployment of the coronary half while still inside the coronary. Subsequently, it was carefully removed without visible damage, and the joining of the 2 halves was successfully completed.
Discussion
In this ex vivo feasibility experiment, the endoscopic procedure-adapted Octocon version demonstrated a 100% success rate in performing all necessary steps for completing an anastomosis with robotic manipulation. The procedure succeeded on the first attempt in 96% of cases; only 2 instances (4%) required a second attempt for completion. Notably, both instances were associated with graft (conduit) deployments. The issues could be traced back to a single failure mode, which was incomplete insertion of the Octocon applicator head into the vessel lumen, either due to increased wall thickness or a short vessel incision. This highlights a learning point that turned out to be relatively easily addressable. Specific visual inspection combined with gentle graft manipulation proved quite effective to verify full insertion to entirely avoid this issue. Both issues arose with the surgeon who had the least experience in robotic surgery. Corrective measures following such inadvertent misfires include either removing a graft segment if the remaining length is sufficient or, potentially, removing the connector half and shortening the vessel incision length with a stitch, if necessary.
In each instance, precise alignment of the 2 plates could be achieved, allowing the fusion of the 2 half connectors. This delicate alignment was accomplished using fine forceps such as the Black Diamonds, coupled with camera zooming for increased precision. However, the clicking mechanism was not evaluated here due to known manufacturing inconsistencies in some sets, which could result in connectors becoming loose too easily.
The Octocon device demonstrated very good maneuverability within the simulated chest, with the use of just 4 instruments—standard in TECAB procedures—being sufficient for optimal device performance. All maneuvers were camera guided, with camera views verifying each of the 3 procedural steps. There were no accidental drops, and the applier’s insertion into the 12 mm working port was smooth and trouble free. Both anterior and lateral wall anastomoses were successfully completed. For the best visualization of the OM branches, a 30° upward camera angle was usually employed, while a top-down approach was useful on more posterior branches, ensuring clear visibility during connector half introduction and application. Various graft geometries, such as jump grafts and Y-grafts, were also achievable under continuous camera guidance. Despite differing levels of robotic experience, all 3 surgeons completed a full anastomosis successfully on the first attempt. By the end of the first day, each surgeon consistently completed key procedural steps in less than 5 min.
This study has several limitations. First, our analysis was limited to an ex vivo porcine model, which prevented assessment of the Octocon device’s suitability for beating-heart techniques. Second, our study solely focused on applying the device on a robotic platform, using visual and semiquantitative result evaluations. This feasibility study did not include precise assessments of patency, measurements of leakage, or histological examinations. These aspects are crucial for establishing safety and efficacy and necessitate further comprehensive studies.
Conclusions
This ex vivo feasibility study highlighted the successful application of the endoscopic Octocon coronary connector within a closed-chest robotic platform in performing distal coronary anastomoses on both the anterior and lateral heart walls. The ease of use and teachability of the procedure are promising. Different graft geometries were successfully tested using standard robotic instruments and camera guidance, demonstrating feasible and intuitive maneuvering of the Octocon. Despite varying levels of robotic experience, all 3 surgeons achieved successful results with the Octocon. With limited experience, the time required for an anastomosis was less than 5 min. We believe the endoscopic Octocon coronary connector has the potential to promote broader adoption of limited access and TECAB procedures.
Supplemental Material
sj-docx-1-inv-10.1177_15569845241288540 – Supplemental material for Coronary Connector Facilitated Total Endoscopic Coronary Artery Bypass: An Ex Vivo Feasibility Study
Supplemental material, sj-docx-1-inv-10.1177_15569845241288540 for Coronary Connector Facilitated Total Endoscopic Coronary Artery Bypass: An Ex Vivo Feasibility Study by Monica Gianoli, Anne Roos de Jong, Harmen Matthijs Wassink, Paul F. Gründeman, Bob Kiaii, Husam H. Balkhy and Willem J. L. Suyker in Innovations
Footnotes
Acknowledgements
We acknowledge Robert Burgin for his contributions in the skills lab at University of Chicago Medicine. Furthermore, we acknowledge the substantial contributions of Paul Suyker and the colleagues at the Medical Technology and Clinical Physics (MTKF) department in Utrecht, whose expertise and dedicated efforts were pivotal in the development of the Octocon.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors M.G., H.M.W., P.F.G., and W.J.L.S. are coinventors and declare a financial interest in the Octocon technology (patent pending), including W.J.L.S. as the CMO with stock options for OctoVascular BV. M.G. is a member of EACTS-endorsed Robotic Cardiothoracic Surgery Taskforce. B.K. is a consultant with Medtronic, Johnson & Johnson, and Abbott and receives lecture honoraria from Corcym. H.H.B. is a proctor for Intuitive, receives lecture honoraria from Medtronic and Edwards Lifesciences, and participates on the DSMB for Edwards Lifesciences. A.R.d.J. declares no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
