Abstract
Minimally invasive coronary revascularization techniques aim to avoid median sternotomy with its associated complications, while facilitating recovery and maintaining the benefits of surgical revascularization. The 3 most common procedures are minimally invasive coronary artery bypass grafting, totally endoscopic coronary artery bypass, and hybrid coronary revascularization. For a variety of reasons, including cost and technical difficulty, not many centers are routinely performing minimally invasive coronary revascularization. Nevertheless, many studies have assessed the safety and efficacy of each of these procedures in different clinical contexts. Thus far results have been promising, and with the evolution of procedural techniques, these approaches have the potential to redefine coronary revascularization in the future. This review highlights the current state of minimally invasive coronary revascularization techniques by exploring their benefits, identifying barriers to their adoption, and discussing future potential paradigms.
Keywords
Central Message
To ensure patients receive optimal intervention, high-volume centers should consider establishing a minimally invasive coronary program. Large, multicenter, randomized trials can then be designed to assess long-term outcomes for each intervention.
Introduction
Current guidelines for the treatment of coronary artery disease (CAD) suggest coronary artery bypass grafting (CABG) as the gold standard revascularization strategy for multivessel and left main CAD. 1 -3 The guidelines have largely been influenced by large, important clinical trials, such as SYNTAX and FREEDOM, 4 , 5 which have firmly established the advantage of CABG over percutaneous coronary intervention (PCI), particularly in an environment of increasingly complex CAD, obesity, and diabetes. Recent trials, such as Everolimus-Eluting Stents or Bypass Surgery for Left Main Coronary Artery Disease (EXCEL Trial) 6 and Nordic-Baltic-British left main revascularization study (NOBLE Trial) 7 have reconfirmed CABG superiority over PCI in survival and freedom from adverse cardiovascular outcomes, resulting in a resurgence of CABG in North America. 8,9 However, CABG performed via sternotomy remains a highly invasive procedure and, when given the choice, many patients still choose PCI for multivessel CAD, despite the fact that 1-year risk of death and risk of repeat procedures for PCI far exceeds that of CABG. 10 Therefore, minimally invasive coronary revascularization techniques have been introduced to redefine surgical revascularization and address patient preference for a less invasive procedure, while maintaining the clinical outcome advantage of CABG over PCI.
Today, several minimally invasive CABG techniques have emerged with various degrees of robotic and video assistance, myocardial protection, graft usage, incision placement, anastomotic techniques, endoballoon usage, and combination with PCI. Among these approaches, 3 minimally invasive coronary revascularization techniques are of note. Minimally invasive (MICS) CABG performed via a mini left anterior thoracotomy, totally endoscopic coronary artery bypass (TECAB) using robots, and hybrid coronary revascularization (HCR) combining the anastomosis of the left internal mammary artery (LIMA) to the left anterior descending artery (LAD) with PCI to non-LAD lesions. These approaches confer the benefits of MICS: avoidance of sternotomy, smaller incisions, shorter hospital stay, earlier recovery, decreased bleeding, less transfusion, and reduced risk of sternal infection. 11 -13 However, the uptake of minimally invasive coronary revascularization has been limited by barriers such as increased cost, lack of randomized controlled trials, technical difficulty, and, in some cases, lack of optimal surgical instruments. In this manuscript, we provide a thorough overview of the current state of minimally invasive coronary revascularization techniques, address challenges to their adoption, and discuss future outlooks.
Minimally Invasive Coronary Artery Bypass Grafting
Overview
MICS CABG was first introduced in 2005 by McGinn et al. as a modified version of minimally invasive direct vision coronary artery bypass (MIDCAB) from 1990. 14 It still uses the left thoracotomy incision and a direct vision to perform the anastomoses, however, it is a more lateral and a smaller (4 to 6 cm) incision in the fifth intercostal space, which allows the LIMA to be harvested with less risk of costochondral or rib injury, although chest wall trauma may occur secondary to the heavy retractors needed to lift the anterior ribs in order to facilitate exposure of the ascending aorta. 14 Its detailed operative technique is outlined in the literature. 14,15 MICS CABG also allows for a multivessel coronary operation, unlike MIDCAB where the incision does not allow for a proximal anastomosis of the graft. 16 A detailed approach for MIDCAB can be found here. 17 Nonetheless, the term MIDCAB is still interchangeably used in the literature to describe a multivessel CABG procedure using left thoracotomy under direct vision.
Outcomes
Long-term clinical data on MICS CABG is limited, but several observational studies for MICS CABG have demonstrated promising clinical results. Studies have reported perioperative mortality rate at a range of 0%-1.3%, 13,14,18 -20 perioperative stroke rate from 0%-0.4%, 13,14,19 and conversion rate from 0%-6.7%. 13,19,21 Studies have also found decreased transfusion, and lower surgical site infection rates with MICS CABG, and decreased hospital length of stay (LOS) and earlier return to full physical function in comparison to a sternotomy off-pump coronary artery bypass (OPCAB). 13,14,22 -24
The graft patency in MICS CABG was studied by Ruel et al. in 2014 where they showed complete revascularization in all 89 patients with 52.5% receiving >3 conduits and 92% patency for all grafts and 100% patency for LIMA at 6 months using computed tomography angiography. 21 Nambiar et al. have also demonstrated an excellent outcome for MICS CABG using bilateral internal mammary arteries (BIMA) in 940 patients with 97.9% complete revascularization, 0.9% mortality, and 99.3% freedom from major adverse cardiac and cerebrovascular events (MACCE) at average follow-up of 33 months. From the group, 10 (1.06%) patients required reintervention, with 2 of them due to LIMA-RIMA Y anastomosis blockage. Although the reduced risk of sternal wound infection was mentioned in the paper, the number was not reported in their data. 20
The clinical benefits of MICS CABG shown in observational studies were challenged in the only randomized controlled trial (STET trial) conducted to date with 184 total patients. 25 Although the patients in MICS CABG group in comparison to OPCAB group had reduced proinflammatory cytokines, reduced intubation time (256 vs 321 min), and fewer postoperative arrhythmias (23% vs 35%), MICS CABG had longer median hospital LOS (6 vs 5 days), and median operative time (4.1 vs 3.3 h). The 2 groups also had a similar pain score and quality of life scores at 3 months and 12 months. Others have postulated that the steep learning curve of the multivessel MICS CABG might have impacted the outcome of the trial. 16 Another randomized trial comparing MICS CABG to conventional CABG called Minimally Invasive coronary surgery compared to STernotomy coronary artery bypass grafting (MIST Trial) is currently recruiting to assess quality of life and MACCE. 26 A summary of MICS CABG studies is highlighted in Table 1.
Summary of Major MICS CABG Studies

Abbreviations: LOS, length of stay; MICS CABG, minimally invasive coronary artery bypass grafting.
Patient Selection
MICS CABG has been previously described to be performed on stable patients with preserved ventricular function unless cardiopulmonary bypass (CPB) is used. 14,27 Absolute contraindications were listed as emergency surgery with hemodynamic compromise, severe chest deformities, and pulmonary comorbidities. A relative contraindication is patients with increased body mass index, as exposure through the thoracotomy may be restricted. The limitation in heart manipulation through a small left thoracotomy incision required preoperative assessment of distal coronary artery target quality to ensure adequate anastomosis. Diffuse and intramyocardial vessels were deemed unsuitable for MICS CABG. 16 MICS CABG showed potential in providing a favorable long-term outcome in patients over 75 in a study by Barsoum et al. where 5-year all-cause mortality for MICS CABG was reported to be lower than conventional sternotomy CABG at 19.7% and 47.7%, respectively. Similar results were found for patients younger than 75 years. 28
Barriers to Adoption
The technical difficulty, especially achieving multivessel revascularization with a MICS CABG, presents the greatest barrier to the widespread adoption of this technique. This approach is also challenging when attempting to expose the distal segment of the LIMA through the thoracotomy incision. Formalized initiation steps to an off-pump multivessel MICS CABG are described by Une et al. where surgeons sequentially increase vessel number with or without CPB assistance. 16 Rodriguez et al. have also emphasized that surgeons should be a very experienced OPCAB surgeon via sternotomy before performing an off-pump MICS CABG. 27 Therefore, MICS CABG uptake and training will likely be limited to the institutions that routinely perform OPCAB procedures. With the CABG Off or On Pump Revascularization Study (CORONARY trial) showing no difference in clinical endpoints and identical survival at 5 years for both on-pump and off-pump CABG, 29 centers that utilize the OPCAB technique will likely remain limited thereby restricting the number of surgeons to be trained in MICS CABG.
The learning curve of MICS CABG has been analyzed by Une et al. who showed no difference in clinical outcomes between the first 25 and the remainder for off-pump multivessel MICS CABG. However, there was a 14.5% conversion rate associated with poor exposure, hemodynamic instability, LIMA injury, and intolerance to single lung ventilation. Operative time reached an acceptable level at the 66th case for single vessel MICS CABG and the 40th case for multivessel MICS CABG. 22 Rodriguez et al. have examined rates of reoperation in 306 patients who underwent MICS CABG procedure where 7% received repeat revascularization at 1.7 ± 1.6 years postoperatively with the rate decreasing from 11% in the first half of the series to 2% for the latter half. 30
The STET trial showed a 10% increased hospital cost with MICS CABG versus OPCAB (£5,079 vs £4,566), which included £869 of reoperation cost for 4 patients in the MICS CABG group versus £0 in the OPCAB group. 25 The addition of a robot in the LIMA takedown changes the cost of the approach and Pasrija et al. have shown that a robot-assisted (RA) MIDCAB is less costly in comparison to a totally endoscopic coronary artery bypass (TECAB; $22,679 vs $33,769). 31 Finally, Merkle et al. reported higher costs associated with the MIDCAB group versus the PCI group in their analysis (10,879€ vs 4,009€). 32
Future Outlook
The greatest advantage of MICS CABG over other techniques is its ability to achieve multiarterial vessel grafting with BIMA without the associated risk of sternal wound infection, while keeping the operative cost relatively low. Studies exploring multiarterial grating with MICS CABG are being conducted. Nambiar et al. have shown excellent clinical results with BIMA use, 20 Nishigawa et al. demonstrated V composite grating using right internal mammary artery (RIMA) with MICS CABG, 33 and Kikuchi et al. have shown the possibility of in situ RIMA harvesting and anastomosis with MICS CABG. 34 However, to increase the number of cases, subsequent larger randomized trials need to demonstrate clear clinical benefit over conventional CABG that would justify the technical difficulty, which is the main disadvantage associated with multivessel MICS CABG. Such trials should also pay close attention to the difficulty in ensuring excellent outcomes when this operation is performed in obese patients. Establishing a structured training program may be required to train surgeons on this challenging surgery to overcome its technical barriers.
Totally Endoscopic Coronary Artery Bypass
Overview
TECAB is a procedure performed entirely through trocar accesses without a surgical incision. It was first performed in Hôpital Broussai in Paris by Loulmet and colleagues in 1998. 35 Since the initial enthusiasm to use robotics in cardiac surgery, its adoption has been limited to few dedicated centers around the world due to its barriers such as high cost, steep learning curve, evolving instrumentation, few training opportunities, and long procedural time. In the United States, 1,439 RA CABG procedures were performed in 2010 and the number dwindled to only 590 procedures in 2015. Similarly, in Europe, 173 RA CABG in 2001 declined to 80 procedures in 2015. 36 While TECAB has been undergoing significant developments in robots and anastomotic techniques, it is still evolving where there is great potential for further major advances. Widespread adoption of this technique will depend on a few factors, including achieving economic and training feasibility and establishing clear clinical superiority over conventional CABG as demonstrated by large, multicenter, randomized controlled trials. Also, if there is going to be further and more widespread adoption of this technique, companies dedicated to TECAB should continue the innovative research and development process and ensure newer generation of devices receive approval from the appropriate regulatory bodies to facilitate their transition into clinical settings. Finally, TECAB is likely at least as technically difficult as MICS CABG. However, it may facilitate a less invasive procedure for a few reasons, including technological components can be present in the operative field instead of a keyhole view, the dexterity of the instruments, and ergonomics.
Outcomes
The majority of TECAB studies in the literature reflect single-institution observational experiences with limitations in sample size and generalizability. The studies are also variable in their number of target vessels treated, anastomotic techniques (EndoWrist, Flex-A, U-clip), generation of da Vinci® robot used (da Vinci, da Vinci S, da Vinci Si; Intuitive Surgical, Sunnyvale, CA, USA) as well as CPB and endoballoon cardioplegia use as a stepping stone for overcoming learning curves. With newer technical advancements, there are improvements in conversion rates, operative time, and the impact of the learning curve on the outcome. 37
The first meta-analysis on TECAB studies was conducted by Leonard et al. in 2018 with a total of 3,721 patients. 38 Of the studies, 62.1% were arrested heart TECAB and 69.1% were single-vessel treatments. The meta-analysis showed a low operative mortality of 0.8%, and acceptable perioperative stroke and myocardial infarction (MI) rate of 1.5% and 2.3%, respectively. Long cross-clamp time, suboptimal myocardial protection, and increased cardiac biomarker checks could have caused the reported perioperative MI rates to rise in this metanalysis. 38 Similar clinical outcomes were noted in recent systematic reviews by Göbölös and Cao. 37,39 Göbölös and colleagues analyzed a total of 2,397 TECAB cases with less than half being multivessel procedures. That study demonstrated an acceptable rate of perioperative mortality (0.8%) and perioperative major events such as stroke (1%), renal failure (1.6%), de novo atrial fibrillation (13.3%), and operative revision rate due to postoperative hemorrhage (4.2%). However, this cohort had both beating heart and arrested heart TECAB patients who underwent the subsequent hybrid procedure (16.8% and 30.3%, respectively), thus the data cannot be extrapolated as being purely a TECAB study. 37 Cao and colleagues analyzed 9 beating heart TECAB studies with an average of fewer than 2 vessels bypassed. The results were similar in perioperative mortality rate (1.7%), perioperative MI rate (1.1%), stroke rate (1.1%), acute kidney injury rate (3.4%), and reoperation for bleeding rate (3.2%). 39 The reported mortality rates of the above studies are lower than the FREEDOM trial with 1.7% and the SYNTAX trial with 3.5% at 1 year. Göbölös suspected this to be due to lower risk patients being chosen for this technically challenging procedure, despite only a small number of studies having performed formal risk evaluation. Summary of major TECAB studies is represented in Table 2.
Summary of Major TECAB Studies
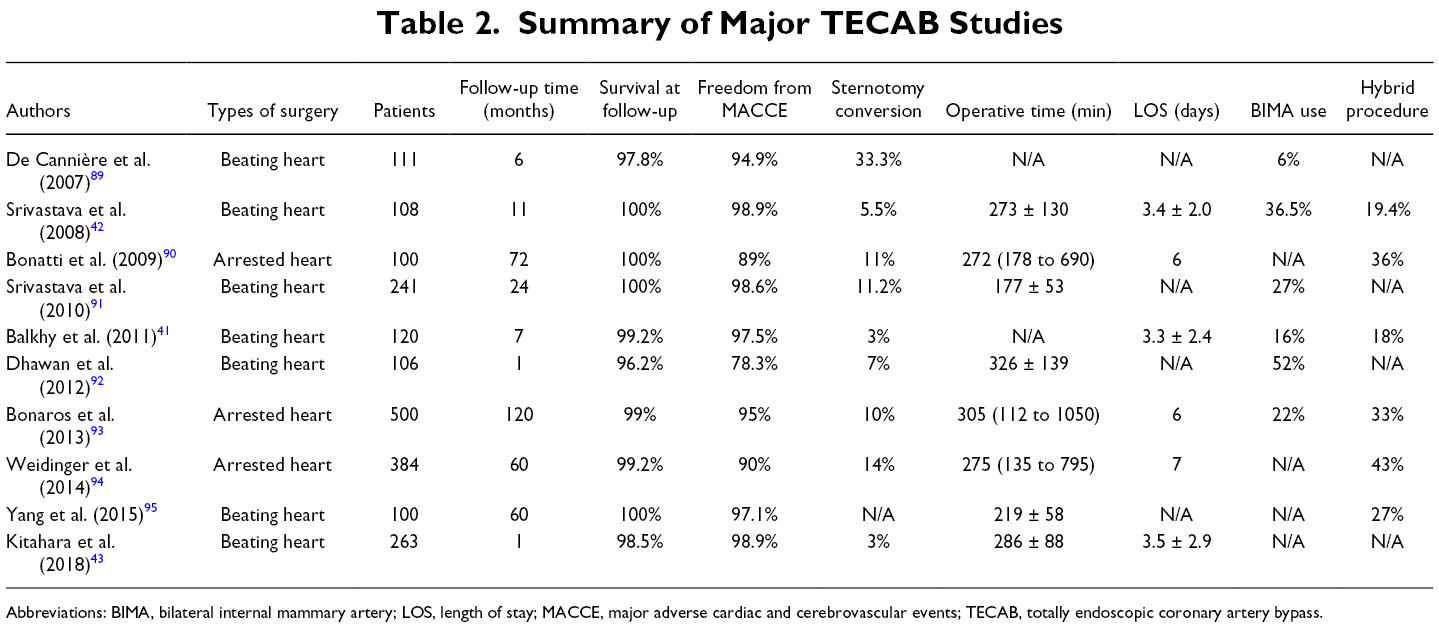
Abbreviations: BIMA, bilateral internal mammary artery; LOS, length of stay; MACCE, major adverse cardiac and cerebrovascular events; TECAB, totally endoscopic coronary artery bypass.
Graft patency in TECAB was studied by Kitahara et al. in 2019 with 1,220 IMA grafts. They showed comparable patency rates of IMA grafts as in conventional CABG with 98.8%, 95.8%, and 93.6% at early (<1 month), midterm (<5 years), and long-term (>5 years) follow-up, respectively. 40 A meta-analysis by Leonard et al. also revealed good short-term graft patency of 94.8% at mean follow-up of 10.1 months. 38 Mean hospital LOS with TECAB in the 2 recent systematic reviews by Göbölös and Cao were reported as 5.6 and 5.8 days, which is not a drastic improvement compared to the conventional CABG. 37,39 Göbölös also reported a shorter length of hospital stay of 4.7 days for beating heart TECAB in comparison to 6.6 days for arrested heart TECAB. Within the beating heart TECAB groups, studies with over 100 reported cases consistently showed short hospital LOS of 3.3 to 3.5 days. 41 -43
RA CABG is not only bound to TECAB but to other techniques with various degree of robot utilization such as the minimally invasive RA direct coronary artery bypass (RADCAB) technique. RADCAB uses the robot to harvest the LITA and is followed by a manual anastomosis the graft to LAD using off pump techniques. Notably, Giambruno et al. have demonstrated excellent results with RADCAB with mortality rate of 0.3% with a graft patency of 92.7% at 95.8 months of follow-up duration. 44
Patient Selection
TECAB can target LAD, diagonal, ramus, and obtuse marginal branch, and the right coronary artery (RCA). The contraindications for TECAB are similar to that of MICS CABG: patients with hemodynamic instability, cardiogenic shock, severe chronic obstructive pulmonary disease (COPD), acute MI, and prior radiation to the chest. 42 The clinical benefits of TECAB are best realized in treating high-risk patients for conventional CABG. Twenty-eight octogenarians (9.1%) who underwent TECAB were compared with younger patients in a study by Kitahara et al. where 60.7% were multivessel TECAB. They showed mean hospital LOS of 3.9 days with 78.6% being discharged home and no in-hospital mortality. 40,45 These studies built on top of the previous work by Lee et al. which identified old age, hemodialysis, cerebrovascular accident, and higher risk scores as factors prolonging LOS following TECAB. 46 Kitahara et al. also showed the beneficial outcome of TECAB for patients with body mass index >35, 45 female patients, 43 redo surgery, 47 and high STS score. 48
In a study by Bonaros, TECAB also showed earlier functional recovery with the restoration of daily activities 2 to 3 weeks faster than conventional CABG, and significantly better quality of life scoring in physical health and bodily pain. 49 Bonatti and colleagues also showed that patients with TECAB were able to return to household activities in 14 days, drive in 21 days, and perform sports in 42 days. 50
Barriers to Adoption
The cost of robotic cardiac surgery presents a major barrier to the wide adoption of TECAB and robotic cardiac surgery as a whole. The initial purchase price of a da Vinci robot is over 2 million USD and over 100,000 USD is required for maintenance fee. 51,52 Cost analysis done by Pasrija found the median cost per TECAB patients to be $33,769 when compared to the RA-MIDCAB group of $22,679. 31 This was significantly more expensive to Levyi’s cost analysis of conventional CABG at $18,601. 53 Pasrija noted that $11,701 of the TECAB cost came from the stabilizer equipment while the operative cost was not significantly different when compared to RA-MIDCAB. 31 The notion that the cost can be offset by the clinical benefits of TECAB remains speculative as no formal cost analysis has been conducted to date. Even if those clinical benefits are realized, TECAB still needs to justify its cost against other minimally invasive coronary revascularization techniques that carry many of the same clinical benefits that TECAB offers. Moreover, as noted above, newer generation, TECAB-specific instruments, which further simplify the operation, must also be developed and introduced to the clinical setting after receiving approval from the appropriate health regulatory bodies.
The conversion rate for TECAB has been trending down since the Göbölös publication 37 with many recent studies now suggesting rates <1%. 54 Reported operative times have also been decreasing with 20 to 30 cases to overcome the learning curve. 37,55 Cross-clamp time and CPB time of 67.9 min and 100.4 min, respectively, were reported in a meta-analysis by Leonard. 38
Future Outlook
Robotic cardiac surgery is still in the early development stages, with rapid innovations in equipment and techniques. Improvements in structured training and robotic technology, including stabilizers, connectors, and anastomotic devices that can eliminate endoscopic suturing will lessen the technical difficulty, the learning curve, and operative times. Unfortunately, with the introduction of new and expensive devices and robots, cost is likely to be the biggest barrier against its wide adoption. There needs to be a greater number of thorough cost analysis studies, and a reasonable financial incentive structure for new institutions to adopt and maintain their robotic cardiac surgery programs. Additionally, new technological improvements would mean more variability in the way that TECAB is practiced and also in the training programs that are established across institutions. Standardization of the practice will be an important factor in the development of TECAB. Randomized controlled trials in TECAB in addition to observational studies will need to establish excellent long-term patency, morbidity, and mortality rates. Unless these barriers are addressed, the numbers of RA CABG may continue to decrease, as seen in current usage trends.
Hybrid Coronary Revascularization
Overview
HCR, first described by Angelini et al. in 1996, 56 combines the surgical anastomosis of LIMA to LAD with PCI to non-LAD lesions. LIMA to LAD anastomosis is currently being done with either a MIDCAB, RA-MIDCAB, or a TECAB thereby avoiding a sternotomy. PCI to non-LAD lesions further reduces the invasiveness and technical complexity of graft usage in minimally invasive coronary revascularization techniques. HCR only composes 0.48% of the total isolated CABG volume in the United States, 57 and with 358 (50.4%) hospitals having performed HCR between 2009 and 2017, 97.3% of the hospitals performs <1 procedure per year. 58 However, its safety and feasibility are well demonstrated in the literature and appropriately reflected in the current guidelines (Supplemental Table). 1 -3 The consensus regarding HCR today is that the procedure will remain limited to few dedicated centers and specific patients until multicenter, randomized controlled trials demonstrate its efficacy with superior clinical outcomes, and that its barriers to adoption such as learning curve, cost, logistics are addressed.
Multivessel Grafting
Determination of the optimal multivessel coronary revascularization strategy rests on multiple factors such as clinical setting, heart function, patient characteristics and preference, comorbidities, graft availability, and most importantly, anatomic factors. 11 CABG has shown to be especially superior to PCI in complex multivessel lesions by large, multicenter, randomized trials such as SYNTAX and FREEDOM. However, compared to well-established therapeutic excellence of LIMA to LAD graft with 10-year patency of greater 90%, 52,59 -64 venous grafts show inferior results that are being challenged with improvements in drug-eluting stent (DES) replacements. PCI with DES against venous grafts has shown lower rates of failure and re-stenosis, as well as having lower stroke rate, less invasiveness, and shorter recovery when compared with CABG. 65 -67 PCI can now provide a comparable short and midterm outcome to CABG in low-risk patients (low SYNTAX score) and in those with single-vessel disease, which are now reflected in the guidelines. 1,2,66,68
With respect to whether PCI to a non-LAD artery will be appropriate against multiarterial grafting of multivessel CAD, Royse et al. and Lytle et al. have shown excellent arterial conduit outcomes leading to multiarterial grafts becoming the gold standard in multivessel revascularization. 69,70 However, confusion around conflicting evidence has led to poor uptake of multiarterial grafts in the United States. Only 10% of CABG are done with >2 arterial grafts and <1% with 3 grafts, whereas over 50% of patients receive >2 arterial grafts in Australia. 71 Thus, the comparison of clinical outcomes between HCR against conventional CABG in the United States should always distinguish the type of graft utilized in conventional CABG. Moreover, the broader adoption of HCR should be considered in the context of lower rates of multiarterial grafts used in the United States.
Sequence
The sequence in which HCR is performed today is currently divided into (1) CABG then PCI, (2) PCI then CABG, and (3) simultaneous. Presently, two-thirds of the HCR volume is performed where it is CABG first, followed by PCI. 72 Each approach has its advantages and disadvantages and the choice remains up to the institution’s preference or logistics on a per-patient basis.
CABG then PCI has the advantage of protected LAD territory during the PCI and angiographic confirmation of LIMA-LAD patency during PCI. Its disadvantages include the risk of nephropathy due to surgical intravenous contrast and the potential need for a reoperation due to failure of PCI. In this scenario, aspirin is given before CABG is performed and a second antiplatelet agent is added before PCI for anticoagulation. 27,73
PCI then CABG has the advantage of being able to address any stent complications with CABG in addition to managing acute coronary syndrome with PCI to non-LAD lesions. 73 Its disadvantage includes a high risk of ischemia to the unprotected LAD territory, risk of stent thrombosis in the perioperative period due to the hypercoagulable state during subsequent surgery, 74 and risk of bleed with the use of dual antiplatelet in the perioperative period. 75,76
Finally, the cases where CABG and PCI are done simultaneously have the advantage of shorter intensive care unit (ICU) and hospital LOS, angiographic confirmation of patency of LIMA-LAD graft, protection of LAD territory, and the ability to repair unsuccessful stents. Their disadvantages include a higher risk of bleeding after surgery due to overlap of anticoagulation and antiplatelet administration, risk of nephropathy due to surgery and intravenous contrast, increased risk of stent thrombosis due to inflammation, and high cost of a hybrid operating room. 27,73
Outcomes
The variability present in the HCR techniques in the limited number of procedures performed compromise the comparability of clinical outcomes. They include different types of stents, revascularization type, techniques, sequence, and heterogeneity of the study population. Particular attention should be paid to the supposed advantage of HCR, which includes reduced stroke rate due to the elimination of aortic manipulation and CPB, decreased hospital LOS, and earlier recovery. Complete revascularization and reintervention rate should also be considered in the context of replacing venous grafts with DES.
A Society of Thoracic Surgeons database analysis of 950 HCR cases with 33% RA and 30.5% simultaneous HCR revealed no differences in the composite in-hospital mortality and major morbidity between HCR and conventional CABG. This was despite the HCR group having higher cardiovascular risk profiles. 57 Similar results were found in a meta-analysis of observational studies of HCR where there were no significant differences in individual components and composite of death, MI, stroke, or repeat revascularization during hospitalization and at 1-year follow-up. 77,78 A summarized report of major studies of HCR is shown in Table 3.
Summary of Major Hybrid Coronary Revascularization Studies
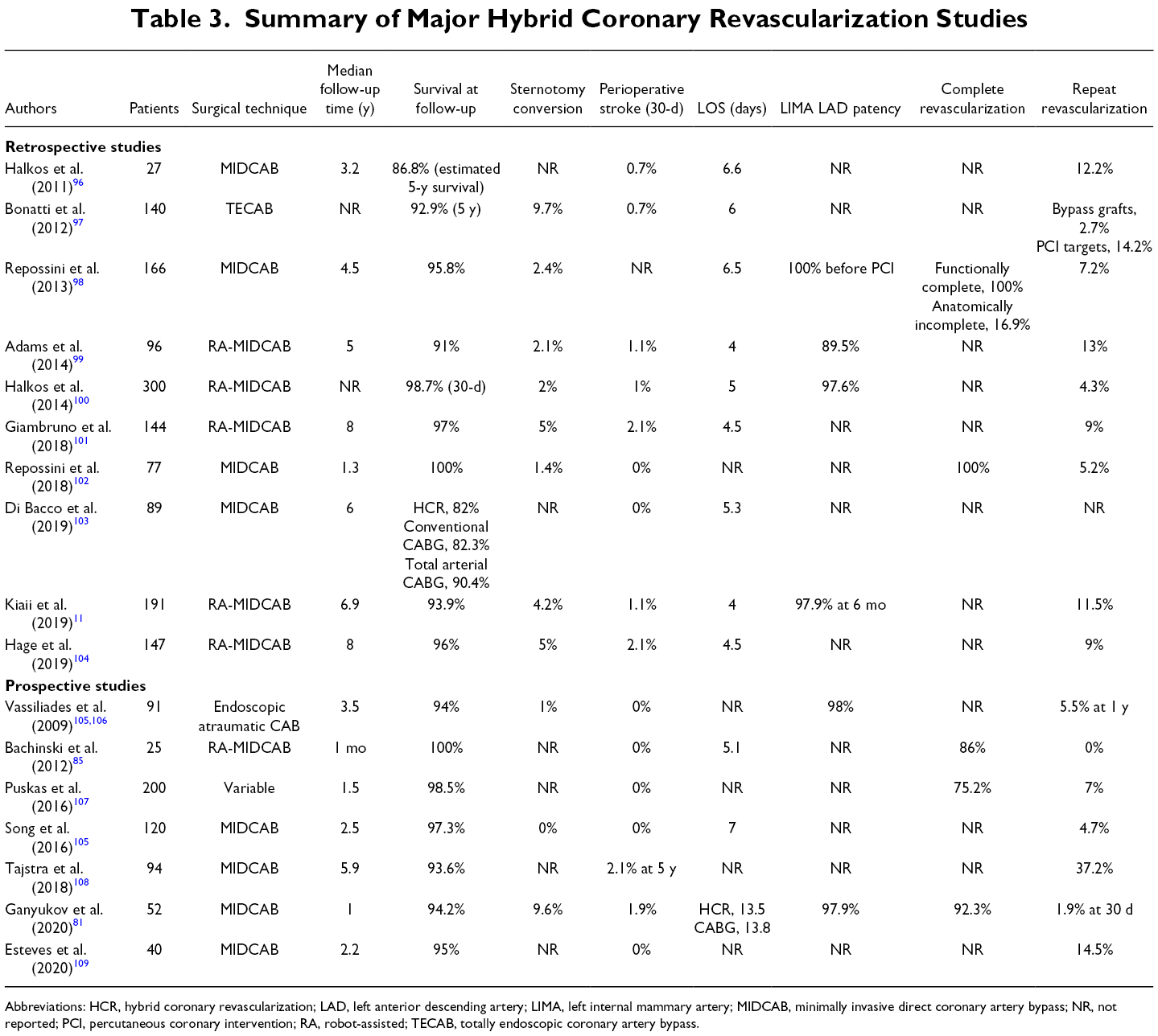
Abbreviations: HCR, hybrid coronary revascularization; LAD, left anterior descending artery; LIMA, left internal mammary artery; MIDCAB, minimally invasive direct coronary artery bypass; NR, not reported; PCI, percutaneous coronary intervention; RA, robot-assisted; TECAB, totally endoscopic coronary artery bypass.
There are 2 small randomized controlled trials for the hybrid procedures to date. The Hybrid Revascularization for Multivessel Coronary Artery Disease (HYBRID) trial compared the hybrid procedure against conventional CABG. 79 At a mean follow-up time of 5.89 years, 9 patients were lost to follow-up with 94 in the HCR arm and 97 in the conventional CABG arm. MIDCAB and Everolimus eluting stents were used for HCR with a mean SYNTAX score of 23. At 5 years, all-cause mortality was similar for HCR and conventional CABG at 6.4% and 9.2%, respectively. Also, no significant difference was observed in the rates of MI, repeat revascularization, stroke, and MACCE. The relatively high repeat revascularization rates of 37.2% for HCR and 45.4% for conventional CABG remain unexplained in the study. Although not statistically significant, the stroke rate was reported to be lower for HCR than conventional CABG at 2.1% and 4.1%. The major bleeding rate was identical at 2.0% and complete revascularization was reported at 77.6% for HCR and 78.2% for conventional CABG. These are higher rates than previously reported for PCI and complete revascularization rate of 50%-60%. 4,80
On the other hand, the Hybrid coronary REvascularization Versus Stenting or Surgery (HREVS) trial had 155 patients who were randomized 1:1:1 to hybrid, conventional CABG, and PCI. 81 SYNTAX scores for the groups were between 19.3 and 19.5. At 12 months, mortality was 5.8%, 2.0%, and 3.8%, respectively. Interestingly, the hybrid arm was the only group with the incidence of stroke at 3.8% and repeat revascularization at 1.9%. Major bleeding rates were reported at 9.6% for the hybrid procedure, 20.0% for conventional CABG, and 0% for PCI. The complete revascularization rate was reported higher than previous observational studies and the HYBRID trial at 92.3%, 92%, and 94.3%. The conversion rate was reported at 6.1% for the HYBRID trial and 9.6% for the HREVS trial.
Hospital LOS, ICU LOS, and transfusion rates were reported to be significantly shorter for HCR versus conventional CABG in a meta-analysis by Zhu et al. 78,82 However, hospital LOS was similar between the hybrid and conventional CABG arms at 8.6 ± 4.1 and 8.5 ± 5.2, respectively. It should be noted that hospital LOS for the 9.6% of hybrid procedures that were converted to conventional CABG were still included in the hybrid group.
Patient Selection
The current value proposition of HCR lies with a specific set of patients where neither CABG nor PCI alone can effectively provide the best revascularization option for the patient. They include patients unsuitable for CABG due to severe preexisting comorbidities, lack of suitable graft conduits, calcified aorta, tortuous vessels, or chronic total occlusion of distal left main or LAD arteries. 3 The patients would, in turn, need to have non-LAD vessels that are amenable to PCI and distal LAD anatomy suitable for LIMA-LAD graft. Contraindications for HCR include emergency surgery and hemodynamic instability, previous chest surgery, significant obesity, and moderate to severe COPD. 83 When the barriers to its adoption are addressed and excellent long-term outcomes are demonstrated in large randomized trials, HCR may be offered not only as an alternative but also as a primary surgical option for patients who meet the indications.
Barriers to Adoption
Technical difficulty is less than that of multivessel MICS CABG or TECAB, as HCR only requires a LIMA-LAD anastomosis. However, off-pump MIDCAB remains a technical challenge for surgeons with results being case-load and surgeon dependent with 50 to 100 cases required to cross 95% reassurance boundary. 59 HCR also carries a logistical challenge of coordinating the revascularization, training the care team, and the planning of hybrid operating rooms in the case of simultaneous HCR.
The cost of HCR was compared to OPCAB in a meta-analysis by Dong et al. that demonstrated increased cost with HCR despite shorter ICU and hospital stay and decreased transfusion rates. 84 Bachinsky et al. also showed higher cost for HCR compared to OPCAB despite lower postoperative cost ($33,984 ± $4,806 vs $27,816 ± $11,172). 85 However, Halkos et al. showed that HCR results in a greater contribution margin for hospitals in comparison to OPCAB possibly due to higher reimbursement in the United States and improved postoperative resource utilization. 86 Adjusted for LOS and predicted risk of mortality score, the study still showed a total contribution margin of $8,771 to be greater for HCR versus OPCAB.
Future Outlook
Retrospective observational studies of HCR have demonstrated safety and feasibility, which are reflected in the current guidelines. Larger multicenter trials that can effectively remove the bias associated with smaller trials and observational studies are needed to determine some of the contradicting results seen in the trials to date. 87 Current trials that are in recruiting and active stages are highlighted in Table 4. Within this context, it is important to highlight the CTSN HYBRID trial (NCT03089398), which aimed to randomize patients to either HCR or multivessel PCI with metallic DES in patients with multivessel CAD involving the LAD or left main territories. The trial was stopped given poor patient enrollment. Therefore, the theoretical benefit of decreased stroke rate, lower reintervention rates, and earlier recovery need to be studied further, given the aforementioned barriers to this procedure. Clinical equipoise will not suffice for HCR to gain wide adoption among the surgeons. Finally, HCR also provides an opportunity for advanced hybrid coronary interventions, where both internal mammary arteries can be harvested and grafted minimally invasively and combined with PCI. In the future, this strategy may be indicated for a growing patient population that would benefit from double internal mammary bypass grafts.
Summary of Current Clinical Trials on HCR
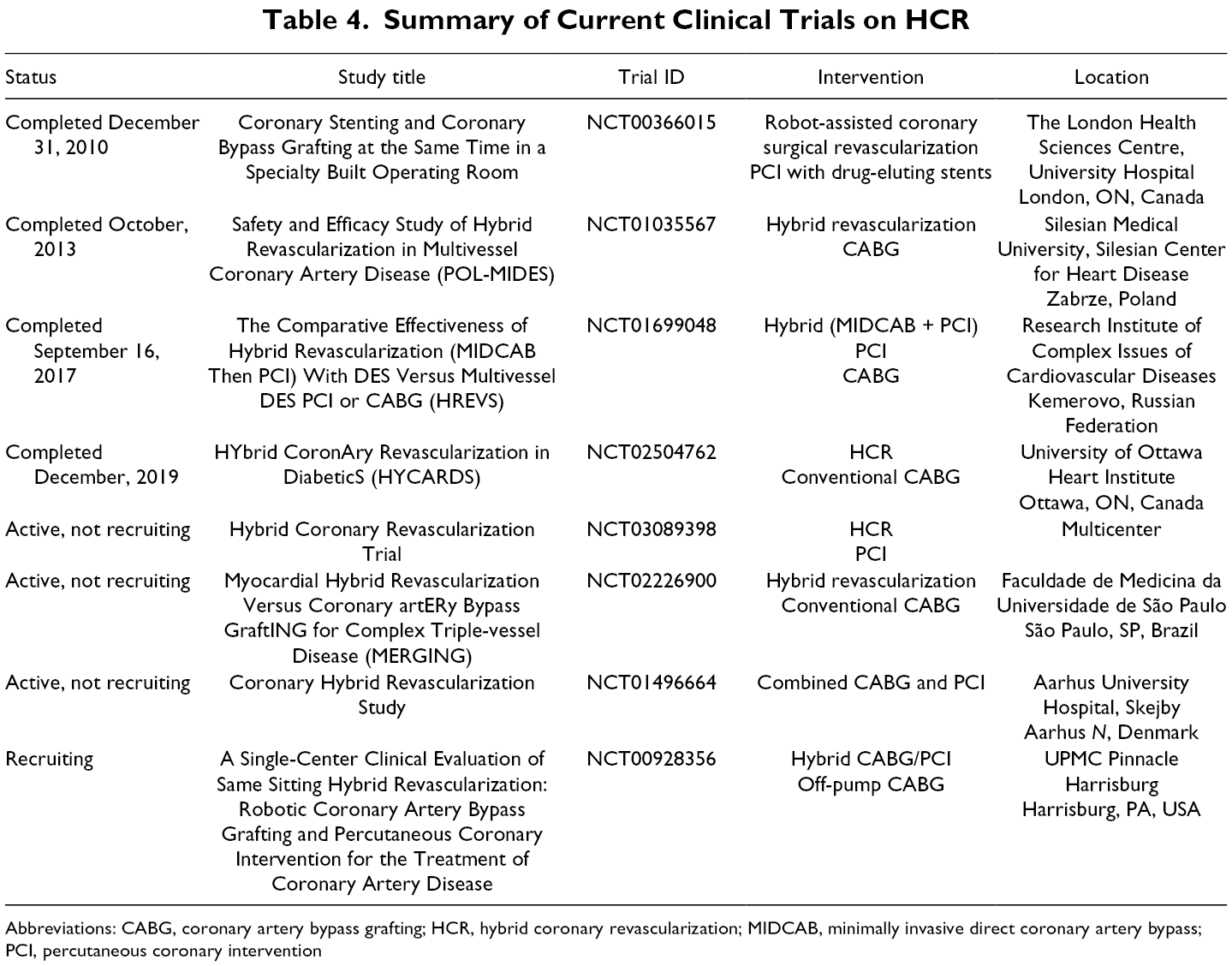
Abbreviations: CABG, coronary artery bypass grafting; HCR, hybrid coronary revascularization; MIDCAB, minimally invasive direct coronary artery bypass; PCI, percutaneous coronary intervention
Conclusions
Since the introduction of CABG, midline sternotomy has been the steadfast approach for coronary revascularization, with excellent clinical outcome and reproducibility. With limitations of stent technology, manifested by inferior long-term results for multivessel disease, CABG will continue to be the gold standard treatment of CAD for the foreseeable future. However, cardiac surgery as a specialty is becoming less invasive with evolving surgical technique supported by innovations in robotic technology and surgical instruments. Recently, there has also been an increase in catheter-based interventions and in some cases, conventional heart operations are being replaced by catheter-based technology. This has resulted in an increase in the demand for minimally invasive interventions. Even an operation with excellent outcome and reproducibility may be perceived by patients and referring physicians as extremely invasive, to a point where an increasing number of patients may accept inferior long-term results over surgical revascularization.
To date, 3 main approaches have been adopted for minimally invasive coronary revascularization techniques: MICS CABG, TECAB, and HCR. All of these strategies are technically challenging and require special logistical considerations, which has resulted in slow and modest uptake among cardiac surgeons around the world. To expand the application of MIS CABG, improvements are required to continue to deliver optimal, precise, and durable results with revascularization. Predicting which minimally invasive technique will gain the greatest traction depends on how each of those approaches overcome their barriers. For MICS CABG, it is the technical difficulty of its multivessel procedure; for TECAB, it is the cost, technical complexity, and logistics of owning and operating a robot; and for HCR, it is the lack of large well-designed trials that show the comparable results of HCR over conventional CABG and potentially superior results to PCI.
Despite the early stage of these techniques, minimally invasive surgical coronary revascularization is a rapidly evolving area that has the potential to reshape the future treatment and management of coronary artery disease. In addition to larger and well-designed trials that can assess and objectively determine clinical outcomes, future studies should further explore patient recovery status, patient quality of life, cost-benefit analysis, and training strategies. Moreover, to encourage better adoption rates for any of these techniques, it is imperative to not only show superior benefits and outcomes but to also continue to improve instruments that can facilitate training. Finally, there is a real opportunity for large centers to develop specialized programs that focus on minimally invasive coronary surgery. The onus is on the surgical community to spearhead the institution and training of future surgeons who are comfortable and competent in ensuring optimal clinical outcomes for minimally invasive coronary surgery. There is already a precedent in cardiac surgery, with the success of minimally invasive valve surgery, so it is important for surgeons to seize this opportunity and lay the foundations for successful minimally invasive coronary surgery programs.
Supplemental Material
Table S1 - Supplemental material for Review of Contemporary Techniques for Minimally Invasive Coronary Revascularization
Supplemental material, Table S1, for Review of Contemporary Techniques for Minimally Invasive Coronary Revascularization by Ali Fatehi Hassanabad, Jimmy Kang, Andrew Maitland, Corey Adams and William D. T. Kent in Innovations: Technology and Techniques in Cardiothoracic and Vascular Surgery
Supplemental Material
Presentation S1 - Supplemental material for Review of Contemporary Techniques for Minimally Invasive Coronary Revascularization
Supplemental material, Presentation S1, for Review of Contemporary Techniques for Minimally Invasive Coronary Revascularization by Ali Fatehi Hassanabad, Jimmy Kang, Andrew Maitland, Corey Adams and William D. T. Kent in Innovations: Technology and Techniques in Cardiothoracic and Vascular Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
