Abstract
Objective
To compare lesion size and depth between a 1-step, a sequential, and a delayed radio-frequency ablation in a hybrid setup.
Methods
Left atrium tissues obtained from fresh porcine hearts were mounted into the ABLABOX simulator. Based on the time differences between the index epicardial (epi) and consequent endocardial (endo) ablation, 3 study groups were compared: a 1-stage (SEQ- 0) group (0-minute delay), an SEQ 1 group (60-minute delay), and an SEQ 2 group (240-minute delay). During the experiment, a constant epicardial (300 gr) and endocardial (30 gr) force were applied. Per group, 20 samples were studied, and the resulting lesion size and depth were quantified with morphometric evaluation.
Results
Overall, no transmural lesion was obtained. Lesions in SEQ 0 had better maximum and minimum diameters (P < 0.001), a larger total area (P < 0.001), and volume (P < 0.001) than SEQ 1 and SEQ 2. There was no statistical difference in morphometric parameters (all, P > 0.05) between the delayed procedures (SEQ 1 and SEQ 2).
Conclusions
In our in vitro model, different time sequences of combined epi–endo ablation did not result in transmural lesions. However, simultaneous epi–endo ablation produced broader and deeper lesions. Our findings need to be confirmed by further research.
Central Message
Using fresh porcine heart tissue, morphometric results indicated that in hybrid ablation, the lesion transmurality is not dependent on the time frame between the epicardial and the endocardial procedure.
Introduction
Hybrid ablation of atrial fibrillation (AF) is a treatment approach that combines 2 separate steps: a thoracoscopic epicardial ablation with an endocardial catheter ablation. 1 The early report on hybrid AF ablation showed promising results, 2 and these initial results have been confirmed in other, more recent reports. 3,4 Nevertheless, the hybrid approach is based on a mutually reinforcing collaboration between the surgeon and the electrophysiologist and by concept inextricable linked to the continued search for improvements, resulting in a more effective treatment approach. 5 -7
Potential development is the optimization of the time lapse between the endocardial and epicardial stage, which can vary from minutes to months. 5,8 -10 To date, it is not known whether a 1-stage or a 2-stage procedure translates in better efficacy. 11 The objective of this study is to evaluate, in a vitro model, the size and depth of lesions made through simultaneous, sequential, or delayed epi–endocardial approach.
Methods
Samples
The left atrium tissue was obtained from freshly slaughtered porcine hearts, washed with NaCl 0.9% (B. Braun, Melsungen, Hessen, Germany) and put into a transport medium RPMI 1640 (Sigma-A Samples Aldrich, St Louis, MO, USA). This medium was also used for sample conservation.
The Simulator
The ABLA-BOX simulator (IDEE—Instrument Development Engineering and Evaluation, Maastricht University, The Netherlands) has been previously described. 12 Briefly, the chamber mimicking the internal part of the left atrium has an inlet and outlet access plugged into an external pump (COBE precision blood pump, COBE Cardiovascular Inc, Arvada, CO, USA) for blood circulation (Fig. 1a and b). A heating plate (IDEE—Instrument Development Engineering and Evaluation, Maastricht University, Maastricht, The Netherlands) and a heating exchange device (Bio Cal 370, Medtronic, Minneapolis, MN, USA) are employed to keep the temperature constant throughout the experiments. The stirring motor mimicking AF blood turbulence is connected with its dedicated controller (Cyclone 1Y100, HMCEurope, Tuessling, Germany). Two pressure transducers (AE sensors, Dordrecht, The Netherlands) allow accurate control of the applied forces.

(a) Ablabox: 1 = pump flow outlet, 2 = endocardial catheter holder, 3 = pump flow inlet, 4 = heating plate, 5 = epicardial catheter holder. (b) Ablabox System: 1,2 = digital screen for catheter force application, 3 = heating plate control console, 4 = magnetic stirrer motor control console, 5,6 = catheter contact force sensors.
The 2 cm × 2 cm specimens were mounted between 2 plastic plates magnetically held to keep the samples of tissue firmly (Fig. 2).

Specimen holder open and closed with its lid (a, b, c). Placement into the Ablabox (d, e, f).
Catheters
An epicardial pen (Isolator long-pen TT, AtriCure, Cincinnati, OH, USA) and an endocardial catheter (Celsius FLTR, Biosense Webster Inc, Diamond Bar, CA, USA) were employed. The epicardial catheter was plugged in a power console (Ablation Sensing Unit ASU, AtriCure, Cincinnati, OH, USA). In contrast, a Biosense Webster Stockert 70 RF Generator was used to power the endocardial catheter. The energy power was set up at 25 and 30 W (probe temperature, 65°C; impedance 100 Ω impedance; tissue temperature, 38°C) for the epicardial and the endocardial catheters, respectively. 13
Experimental Setup
Left atrial tissue samples were randomly assigned to 3 groups, based on the time delay between the epicardial and the endocardial ablation application: (1) SEQ 0 (0-minute delay), i.e., simultaneous procedure; (2) SEQ 1 (60-minute delay); and (3) SEQ 2 (240-minute delay). A total of 60 samples were analyzed/studied (20 for each group).
The thicknesses of the samples were 4.3 ± 0.2, 4.2 ± 0.2, and 4.3 ± 0.2 in SEQ 0, SEQ 1, and SEQ 2, respectively (P ≥ 0.09).
In the delayed procedures (SEQ 1 and SEQ 2), the epicardial approach was performed first, followed by the endocardial ablation, to mimic the sequence in the clinical setting. 1 In all samples, the epicardial ablation cycle was automatically terminated by the console, whereas the endocardial ablation time was 30 seconds.
Based on our previous experiences, 12,14 the endocardial contact force was set up at 30 gr. From the epicardial side was chosen a 300 gr force as currently employed in surgery.
Moreover, the flow rate was set up at 5 L/min, the temperature at 38°C, and the stirrer motor at 400 rpm.
Morphometric Evaluation of Myocardial Tissue Ablation
The samples were included in 10% neutral buffered formalin (NBF) for 48 hours, immerged in a sucrose solution (20%), and finally put on a metallic disc and embedded to Tissue-Tek® OCT ™ compound. The blocks obtained were put into a cryostat at −20°C and sliced in consecutive sections (100 µm) perpendicular to the direction of the catheter ablation. The slices were laid down on the acetate sheet and covered and dried at room temperature for 1 hour. Afterward, the slices were digitalized at 300 dpi using a flatbed scanner. Later, the images were imported into computer software for image processing (Image J version 1.48 software; National Institutes of Health, Bethesda, MD, USA). In total, we obtained 42.05 ± 4.33 layers from each sample, and for every single slice, total maximum diameter (D max), total minimum diameter (D min), and total ablated area (AA) were measured. Besides, for any slice, volume (V) was obtained multiplying the area by the thickness of a single slice, fixed to 100 µm by the setup of the cryostat. Likewise, maximum and minimum lesion’s diameter (D epi and D endo, d epi and d endo), area (a epi and a endo), and volume (v epi and v endo) were measured at both epicardial and endocardial sides (Fig. 3).
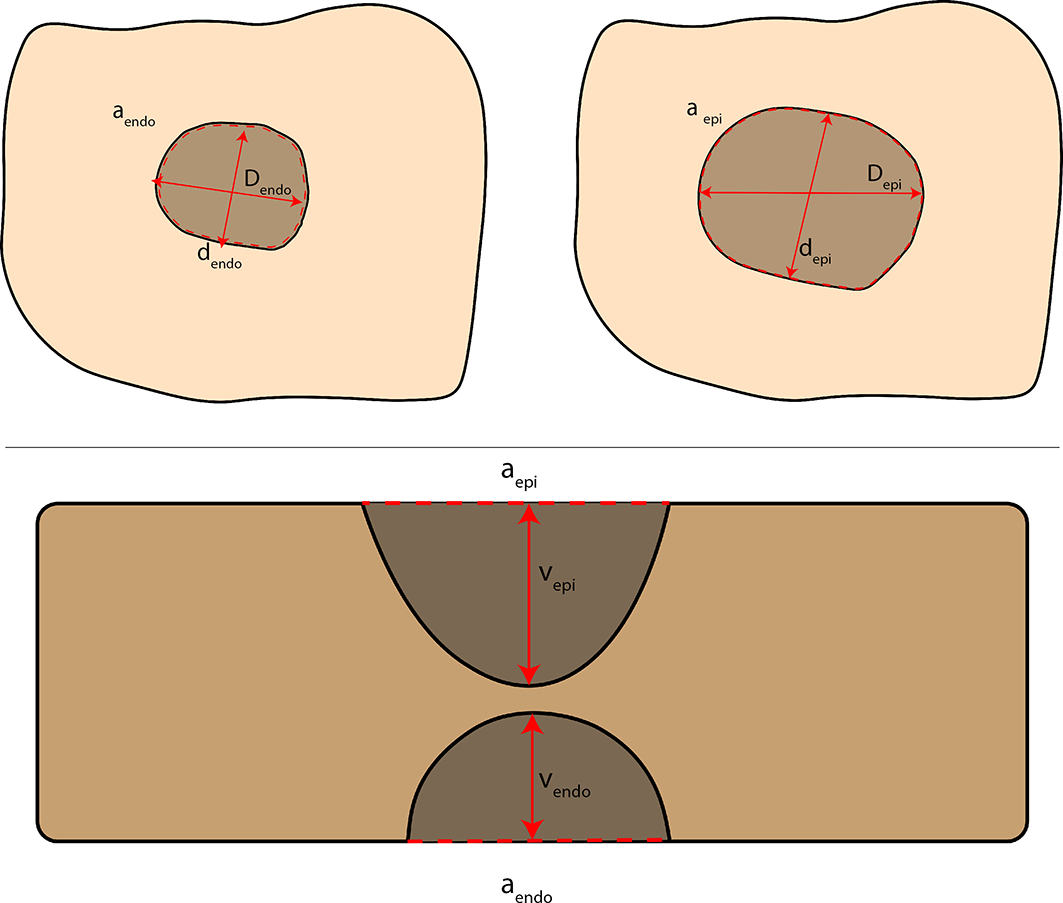
Scheme of the measurements acquired: maximum endocardial diameter (D endo), maximum epicardial diameter (D epi), minimum endocardial diameter (d endo), minimum epicardial diameter (d epi), endocardial area (a endo), epicardial area (a epi), epicardial penetration volume (v epi), endocardial penetration volume (v endo).
Statistical Analysis
The Shapiro–Wilk normality test was performed and variables were expressed as median and interquartile range. Lesion measurements were compared with the Kruskal–Wallis test. Intraobserver and interobserver variabilities were tested on 5 samples randomly chosen, and κ-statistics were used to determine the degree of intraobserver and interobserver agreement after correction for the agreement expected by chance. The variables tested were D max and AA. A κ-value has a maximum of 1.0 when the agreement is perfect. A value of 0 indicates no agreement better than chance agreement. All statistical analyses were conducted using SPSS Statistics Version 18.0 (SPSS Inc., Chicago, IL, USA).
Results
The intraobserver and interobserver variabilities were D max (k = 0.98), AA (k = 0.93), and D max (k = 0.88), AA (k = 0.86), respectively. During the experiment, no overheating steam pops occurred. Overall, no transmural lesion was identified (Fig. 4, Fig. 5). Morphometric data are shown in Table 1.
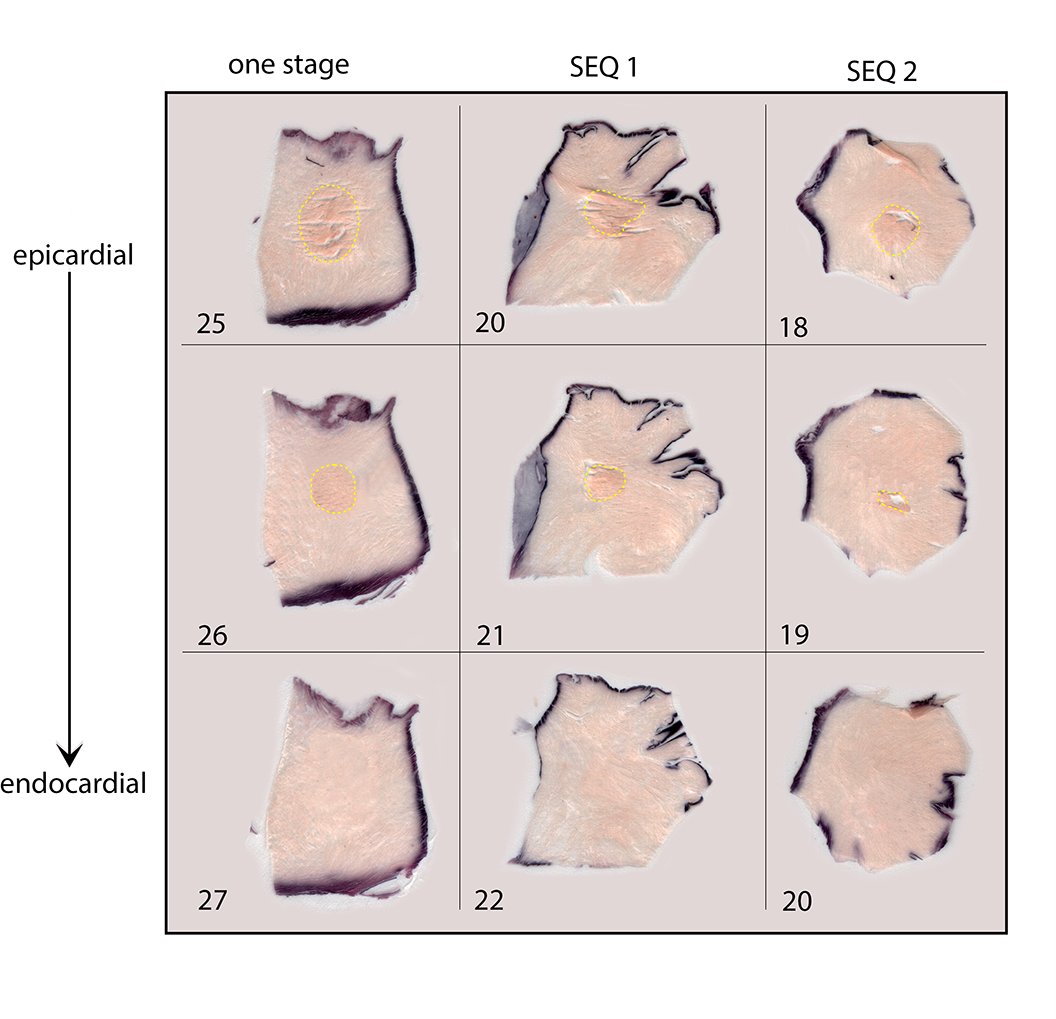
Progression of lesions within the groups and the number of corresponding slices. There is no transmurality evidence. In the 1-stage sample, the ablation lesion disappears at the 27th slice, whereas in the other 2, the lesion stops before, showing a lower penetration.
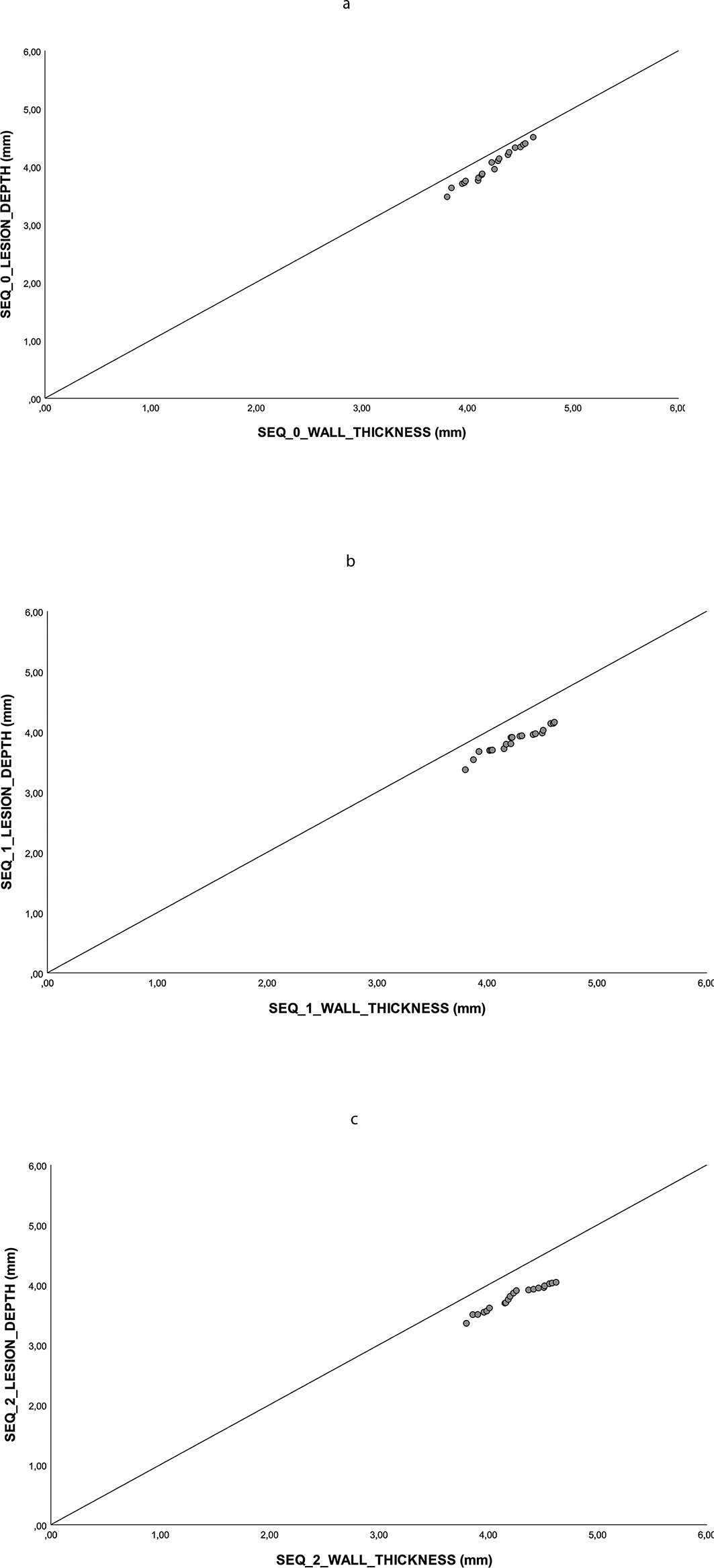
Lesion depth is plotted against tissue thickness. If the lesion is transmural, its point will fall on the line of identity. (a) SEQ 0 group, (b) SEQ 1 group, (c) SEQ 2 group.
Ablation Lesion Results.
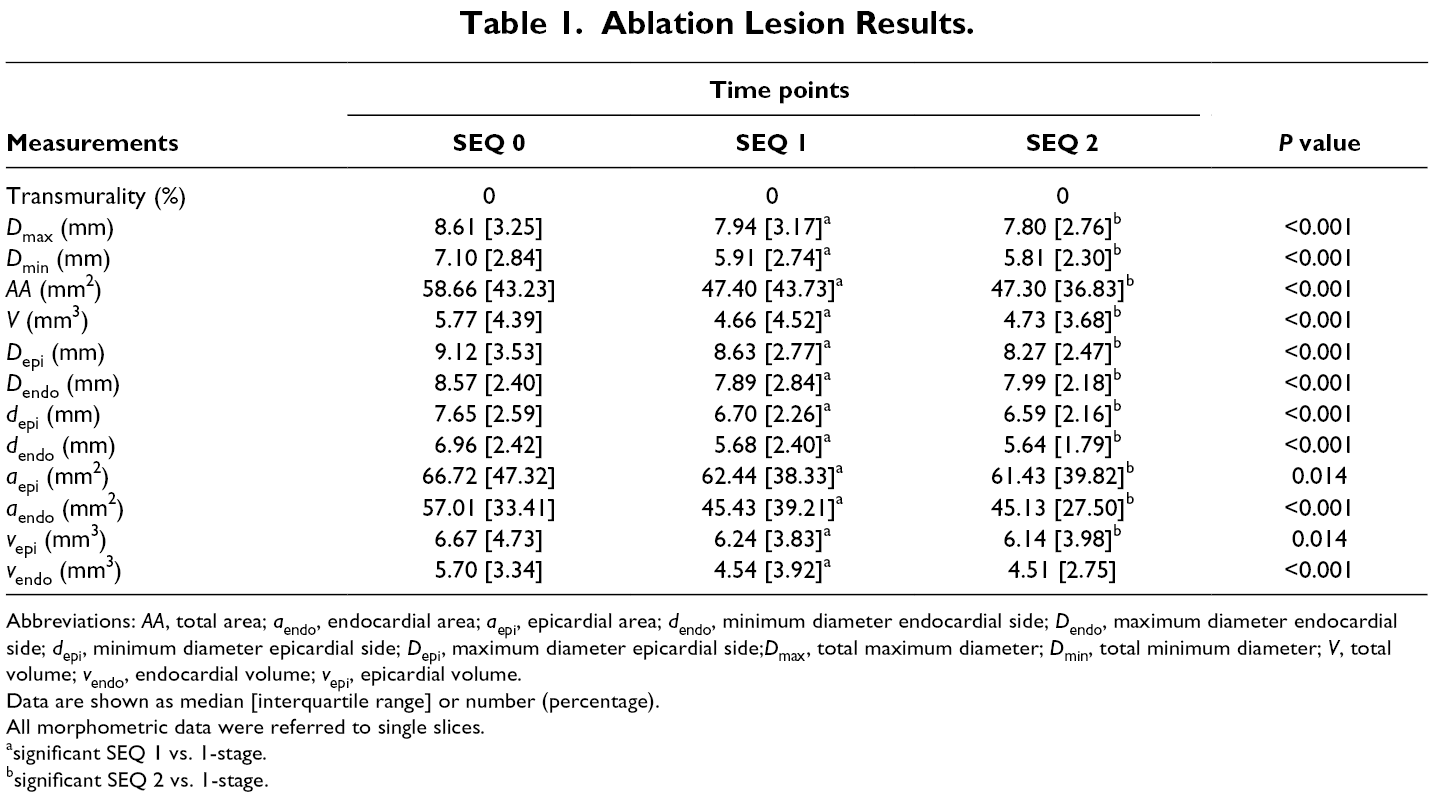
Abbreviations: AA, total area; a endo, endocardial area; a epi, epicardial area; d endo, minimum diameter endocardial side; D endo, maximum diameter endocardial side; d epi, minimum diameter epicardial side; D epi, maximum diameter epicardial side;D max, total maximum diameter; D min, total minimum diameter; V, total volume; v endo, endocardial volume; v epi, epicardial volume.
Data are shown as median [interquartile range] or number (percentage).
All morphometric data were referred to single slices.
asignificant SEQ 1 vs. 1-stage.
bsignificant SEQ 2 vs. 1-stage.
Lesion Size
D max was comparable between SEQ 1 and SEQ 2 (P = 0.149), but it was broader in SEQ 0 (P < 0.001). Also, D min was larger in simultaneous ablated tissue compared to both SEQ 1 and SEQ 2 (both, P < 0.001), which did not show any difference between them (P = 0.118). Moreover, AA was different between 1-stage and SEQ 1 (P < 0.001) and SEQ 2 (P < 0.001) that were comparable (P = 0.706).
Additionally, D epi did not differ in SEQ 1 and SEQ 2 (P = 0.121), while both resulted smaller than in the 1-stage (both, P < 0.001). Furthermore, D endo was similar in SEQ 1 and SEQ 2 (P = 0.842), but lesions in both were smaller than in the 1-stage (P = 0.018 and P < 0.001, respectively). Besides, d epi and d endo were wider in 1-stage (both, P < 0.001), but they were comparable in SEQ 1 and SEQ 2 (P = 0.780 and P = 0.161, respectively).
Likewise, a epi and a endo were larger in 1-stage (a epi, P = 0.031 vs. SEQ1 and P = 0.045 vs. SEQ2; a endo, P < 0.001 vs. SEQ 1 and P < 0.001 vs. SEQ 2, respectively) but they did not differ between the 2-stage procedures (P = 0.965 and P = 0.295, respectively).
Lesion Depth
Likewise, V was similar in SEQ 1 and SEQ 2 (P = 0.997) but it was larger in 1-stage (P < 0.001 vs. both). Finally, v epi and v endo were comparable between SEQ 1 and SEQ 2 (P = 0.965 and P = 0.295, respectively), while it was bigger in 1-stage (v epi, P = 0.031 vs. SEQ 1, P = 0.045 vs. SEQ 2; v endo, P < 0.001 vs. SEQ 1, P < 0.001 vs. SEQ 2).
Discussion
Hybrid AF ablation is a treatment approach that combines thoracoscopic epicardial ablation with endocardial transcatheter ablation. 2,5,7 The epicardial step is performed first because it allows long-lasting isolation of the pulmonary veins and the creation of additional lines in the left atrium, resulting in a so-called box lesion. 4 The second part, carried out by the electrophysiologist, consists of mapping-guided demonstration of entry/exit block and offers the possibility of additional tailored substrate modification.
Although hybrid AF ablation is well defined as a concept, the time frame between both steps is not so clearly specified yet, and varies from a few hours up to 6 months. 8,10,15 For example, Mahaptra et al. performed the endocardial procedure 4 days after the surgical ablation, 16,17 and the other clinicians 30 to 45 days after the surgical procedure. 15,18 Bulava et al. completed the second step 6 to 8 weeks after surgery, 10 and Gaita and co-workers carried out the catheter ablation after a blanking period of 3 months. 19 Other clinicians performed either 1-stage or 2-stage procedures. 20 -22 Supporters of the delayed approach claim that a catheter ablation performed in a second stage significantly increases the likelihood of identifying conduction gaps in the surgical lesions, which potentially translates in a higher mid-term success rate. 15,18 Advocators of a contemporary approach argue that it strengthens the cooperation and mutual understanding between the surgeon and the electrophysiologist. Nonetheless, results were comparable in series using both approaches. 22
However, the superiority of one technique over the others is still debated. 23 As far as we know, no study has specifically addressed this issue. Furthermore, no evidence exists on simultaneous ablation, performed in 1 step with the atrial tissue interposed between the catheters.
In this study, we tested whether simultaneous ablation results in deeper transmural lesions compared to 1-stage ablation (SEQ 1, after 60 minutes) or sequential ablation (SEQ 2, after 240 minutes). The tests were performed on the ABLA-BOX that allows to control contact force, blood temperature, and flow to mimic physiological conditions. 14
In our observations, we checked diameter maximum, diameter minimum, ablated area, ablated volume, as well as maximum and minimum diameters and volumes measured at epicardial and endocardial sides.
We observed smaller ablation diameters on the endocardial surface compared to the epicardial one (P < 0.001). This finding is not surprising because of the different sizes and shapes and differences in the applied force of the 2 catheters. Nonetheless, noteworthy is that all the lesions measured were larger in the 1-stage samples.
D max and D min, and consequently AA and V, were significantly larger in the 1-stage group. First, this might be the result of a deeper penetration during simultaneous ablation, as a result of higher forces, and thus better contact. Nonetheless, we failed to find transmurality in the samples. We might postulate that the pressure applied from both sides reduces the extracellular space of tissue, increasing its impedance, 24 leading to lateral irradiation of the radiofrequency. This lateral irradiation could lead to energy loss that may be responsible for the lack of transmurality. Importantly, we could expect that the tissue compression negatively affects the penetration of RF, but, in contrast, we found that the penetration into the tissue, expressed as volume v endo and v epi, was higher in the 1-stage group (P < 0.001). In contrast, we failed to find any difference between the 60- and 240-minute delay (P = 0.29). This difference could be attributed to the higher contact force of the epicardial catheter. Nonetheless, despite the contact, the force applied was the same in the 3 groups, and v epi and v endo were higher in the 1-stage samples, so this is more likely related to RF physics properties. Indeed, it is known that approximately 90% of the RF energy delivered is adsorbed in the first 1 mm of tissue, and the remainder of tissue heating occurs for indirect conduction. The heating transmission inside the tissue is slower than the RF, and, for this reason, the heating continues rising for several seconds after RF delivery interruption (thermal latency phenomenon). 25
In our previous experience, 14 we obtained transmural lesions using a different setup. In the experiments reported here, we tried to mimic clinical settings. For instance, the endocardial contact force was set up to 300 gr and the endocardial force at 30 gr and endocardial impedance close to ideal 120 Ω. 26 Therefore, we might postulate that the lower impedance, employed in our previous experiments, as well as the difference in tip temperature, could explain the different outcomes.
However, it can be concluded from our findings that the lack of transmurality was not related to different time frames between the endocardial and the epicardial step, and this is a call for specifically improved catheters with optimized design for hybrid AF ablation.
To address the lateral dispersion and enhance more penetration by the RF energy, after some preliminary tests, we are developing a specific “true-bipolar” catheter with the atrial tissue compressed between the 2 ablation poles. This prototype will be the object of an upcoming study. The aim is to obtain fully linear transmural ablations in 1 step and to get a complete, effective left atrium isolation, which is a key feature of AF ablation. 27,28
Also, the lack of postablation edema that, in vivo, may reduce the lesion effectiveness 10,23 prevents us from drawing conclusions. Hence, further in vivo studies to compare 1-step, sequential, and delayed approaches, including electromapping and pathophysiologic changes, are needed.
Finally, the findings from the study add to the voluminous experimental and clinical evidence that unipolar RF does not reliably produce linear, transmural lesions.
However, our findings must be read with caution. Indeed, the real application of simultaneous epi–endocardial ablation is far from being applied to the hybrid clinical arena. Further research is necessary to explore the reliability and feasibility of this technique and whether the challenges entailed in the implementation of this technique can be overcome.
Limitations
Our study suffers from some limitations that should be highlighted. First, we did not make histological evaluations to demonstrate the necrosis induced by the RF. The “TUNEL” (Terminal deoxynucleotidyl transferase [TdT] dUTP Nick-End Labeling) assay might have been the best method for indicating the necrosis but, in our setup, it was impossible due to the number of layers from each sample. This technique is mainly in use, and it has been designed to detect apoptotic cells undergoing extensive DNA degradation during the late stages of apoptosis. It is based on the ability of TdT to mark the blunt ends of double-stranded DNA breaks following apoptosis as well as other forms of cellular necrosis . 29 -31 Second, we employed tools that were not designed to be used simultaneously. Therefore, the lack of transmurality may be due to the in vitro setup and the characteristics of the catheters used, and this must be taken into account when reading our results.
Third, dead tissue was used, thus excluding all pathophysiologic mechanisms occurring in vivo. In addition, the absence of microvascularization that performs as a heat sink decreases the penetration of RF in alive tissue. 25 Fourth, we did not test other devices. This was beyond the aims of our paper, but it will be the object of further research.
Fifth, the delay from the first ablation not being longer than 240 minutes related to tissue conservation issues. Furthermore, we did not perform multiple ablations. This issue might have led to improved depth of penetration. Sixth, the tissue employed was not viable, and as a consequence, either microcirculation or intrinsic electrophysiological properties were absent, thus excluding all pathophysiologic mechanisms occurring in vivo in response to heating stress. In addition, the absence of microvascularization that performs as a heat sink decreases the penetration of RF in alive tissue. 25 These aspects may limit the applicability to the clinical settings, and thus data have to be confirmed in further studies with a different setup.
Finally, porcine tissue came from young, healthy, 7-month- to 1-year-old animals bred for human consumption. It is likely that the heart tissue did not show any pathologic event typical of AF, like fibrosis development.
Conclusions
In this ex vivo evaluation of different hybrid procedure techniques, 1-stage, sequential, and delayed, transmurality was not reached by any procedure. Nonetheless, the 1-stage approach showed deeper and broader lesions. Our findings need to be confirmed by further research.
Supplemental Material
Visual Abstract - Supplemental material for One-Stage Versus Sequential Hybrid Radiofrequency Ablation: An In Vitro Evaluation
Supplemental material, Visual Abstract, for One-Stage Versus Sequential Hybrid Radiofrequency Ablation: An In Vitro Evaluation by Francesco Matteucci, Bart Maesen, Kevin Vernooy, Carlo De Asmundis, Jos G. Maessen, Mark La Meir and Sandro Gelsomino in Innovations: Technology and Techniques in Cardiothoracic and Vascular Surgery
Footnotes
Authors’ Contributions
All authors actively contributed to the paper in the following domains: conception or design of the work, data collection, data analysis, and interpretation, drafting the article, critical revision of the article, final approval of the version to be published.
The last two authors equally contributed to the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
