Abstract
Objective
It is unknown whether epicardial and endocardial validation of bidirectional block after thoracoscopic surgical ablation for atrial fibrillation is comparable. Epicardial validation may lead to false-positive results due to epicardial tissue edema, and thus could leave gaps with subsequent arrhythmia recurrence. It is the aim of the present study to answer this question in patients who underwent hybrid atrial fibrillation ablation (combined thoracoscopic epicardial and endocardial catheter ablation).
Methods
After epicardial ablation of the pulmonary veins (PVs) and connecting inferior and roof lines (box lesion), exit and entrance block were epicardially and endocardially evaluated using an endocardial His Bundle catheter and electrophysiological workstation. If incomplete lesions were found, endocardial touch-up ablation was performed. Validation results were also compared to predictions about conduction block based on tissue conductance measurements of the epicardial ablation device.
Results
Twenty-five patients were included. Epicardial validation results were 100% equal to the endocardial results for the left superior, left inferior, and right inferior PVs and box lesion. For the right superior PV, 85% similarity was found. Based on tissue conductance measurements, 139 lesions were expected to be complete; however, in 5 (3.6%) a gap was present.
Conclusions
Epicardial bidirectional conduction block in the PVs and the box lesion corresponded well with endocardial bidirectional conduction block. Conduction block predictions by changes in tissue conductance failed in few cases compared to block confirmation. This emphasizes that tissue conduction measurements can provide a rough indication of lesion effectiveness but needs endpoint confirmation by either epicardial or endocardial block testing.
Central Message
For validation of ablation lines, epicardial and endocardial block testing correlate well and are therefore equally reliable as an electrophysiological endpoint. Tissue conduction measurement provides an indication of lesion effectiveness but needs confirmation by testing for bidirectional block.
Introduction
After thoracoscopic surgical ablation for atrial fibrillation (AF), bidirectional conduction block of the epicardial ablation lesions is usually checked epicardially with a sensing and pacing tool of a surgical ablation device. 1 During hybrid procedures, consisting of a combination of thoracoscopic epicardial and endocardial catheter ablation, epicardial ablation lesions are always endocardially checked for bidirectional conduction block by an electrophysiologist using an electrophysiological (EP) workstation. 2,3
To date, no studies have compared epicardial and endocardial validation tests for bidirectional block. Endocardial validation could possibly be more accurate since the epicardial tissue could be edematous after the ablation, giving false-positive outcomes for conduction block and thus leaving gaps in the ablation lesions. It is the aim of the present study to compare epicardial and endocardial validation of bidirectional block in a hybrid AF ablation setting. The validation results are also compared to predictions about conduction block based on the epicardial ablation device measuring tissue conductance.
Methods
Study Population
In 25 patients undergoing a hybrid AF ablation at the Maastricht University Medical Center, the Netherlands, epicardial and endocardial validation tests for bidirectional block across the epicardial lesions were conducted. Patients were referred for a hybrid procedure in case of failure of at least 1 antiarrhythmic drug Vaughan-Williams class I or III, left atrium volume index ≥29 mL/m2, persistent or long-standing persistent AF, one or more failed catheter ablations, or based on patient preference. The Institutional Review Board of Maastricht gave approval to the study.
Hybrid Ablation Technique
All ablation and validation procedures were carried out in the same hybrid operating room by 2 surgeons and 2 EPs. At the beginning of the procedure a transesophageal echocardiogram was made to confirm absence of intracardiac thrombi. The procedure was performed under general anesthesia, with double-lumen endotracheal intubation, using a video-assisted thoracoscopic surgical approach on the beating heart. One 5-mm camera port (midaxillary line) was inserted in the left fifth intercostal space and two 5-mm workings ports (anterior axillary line) were inserted in the left third and seventh intercostal space. The pericardium was opened posterior to the phrenic nerve. Via the oblique and the transverse sinus, the pericardial reflections around both caval veins were developed using blunt dissection. Antral isolation of the left pulmonary veins (PVs), medial to the ligament of Marshall, was performed using a biparietal bipolar radiofrequency clamp (Synergy System, AtriCure Inc., Cincinnati, OH, USA). The same location was ablated at least 2 times before the clamp was repositioned to perform the remaining ablations at the PVs. The total number of ablations depended on the conductance measurements shown by the ablation device. Connecting lines between both superior PVs (roof line) and inferior PVs (inferior line) were made epicardially using a unilateral bipolar radiofrequency rail device (Coolrail, AtriCure Inc, Cincinnati, OH, USA), creating a box lesion (Fig. 1). Thereafter, the right PVs were encircled with a rubber tape from the oblique sinus toward the transverse sinus using a light dissector (Lumitip, AtriCure Inc., Cincinnati, OH, USA). Guided by the rubber tape, the bipolar radiofrequency clamp was introduced in the oblique sinus and positioned around the right PVs. 4 In case it was not possible to target the right PVs from the left side, right PV isolation was performed through 3 right thoracic ports. For all epicardial lesions, as many applications as needed to reach an adequate drop in tissue conductance were performed. The left atrial appendage was closed using an epicardial clip (AtriClip, AtriCure Inc., Cincinnati, OH, USA).
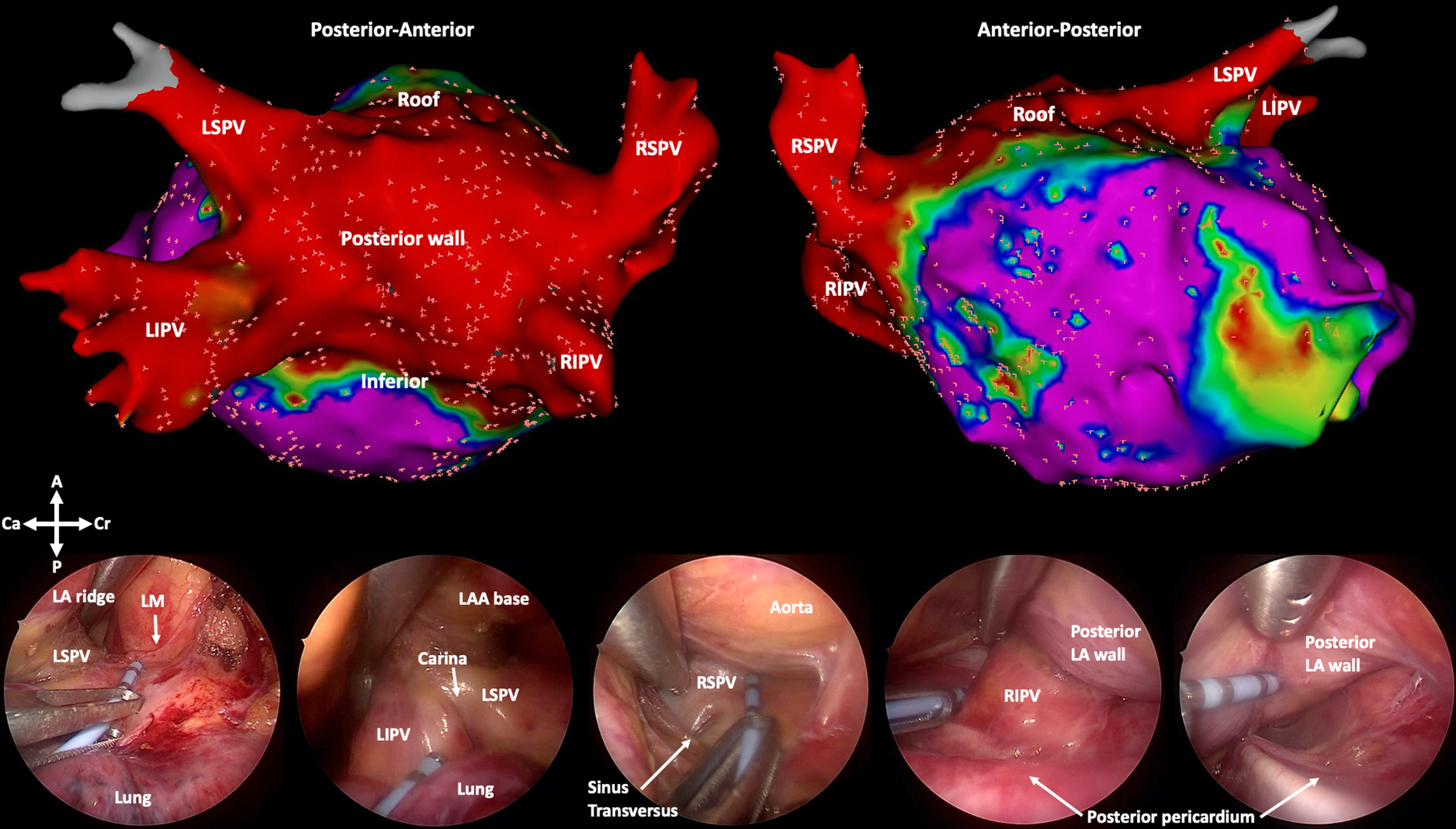
Electrophysiological validation. Upper part: Endocardial Carto voltage maps (Biosense Webster, cutoffs 0.5 to 1.5 mV) of a posterior and anterior view of the left atrium, showing a low-voltage area in red reflecting the box lesion. The little pink spots represent locations where electrophysiological signals were checked. This voltage map is only shown to present the locations of testing, and voltage maps were not further used for drawing conclusions in the current study. Lower part: Left-sided thoracoscopic views of each location during epicardial testing for bidirectional conduction block using an endocardial His bundle catheter. A, anterior; Ca, caudal; Cr, cranial; LA(A), left atrium (appendage); LIPV, left inferior pulmonary vein; LM, ligament of Marshall; LSPV, left superior pulmonary vein; P, posterior; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein.
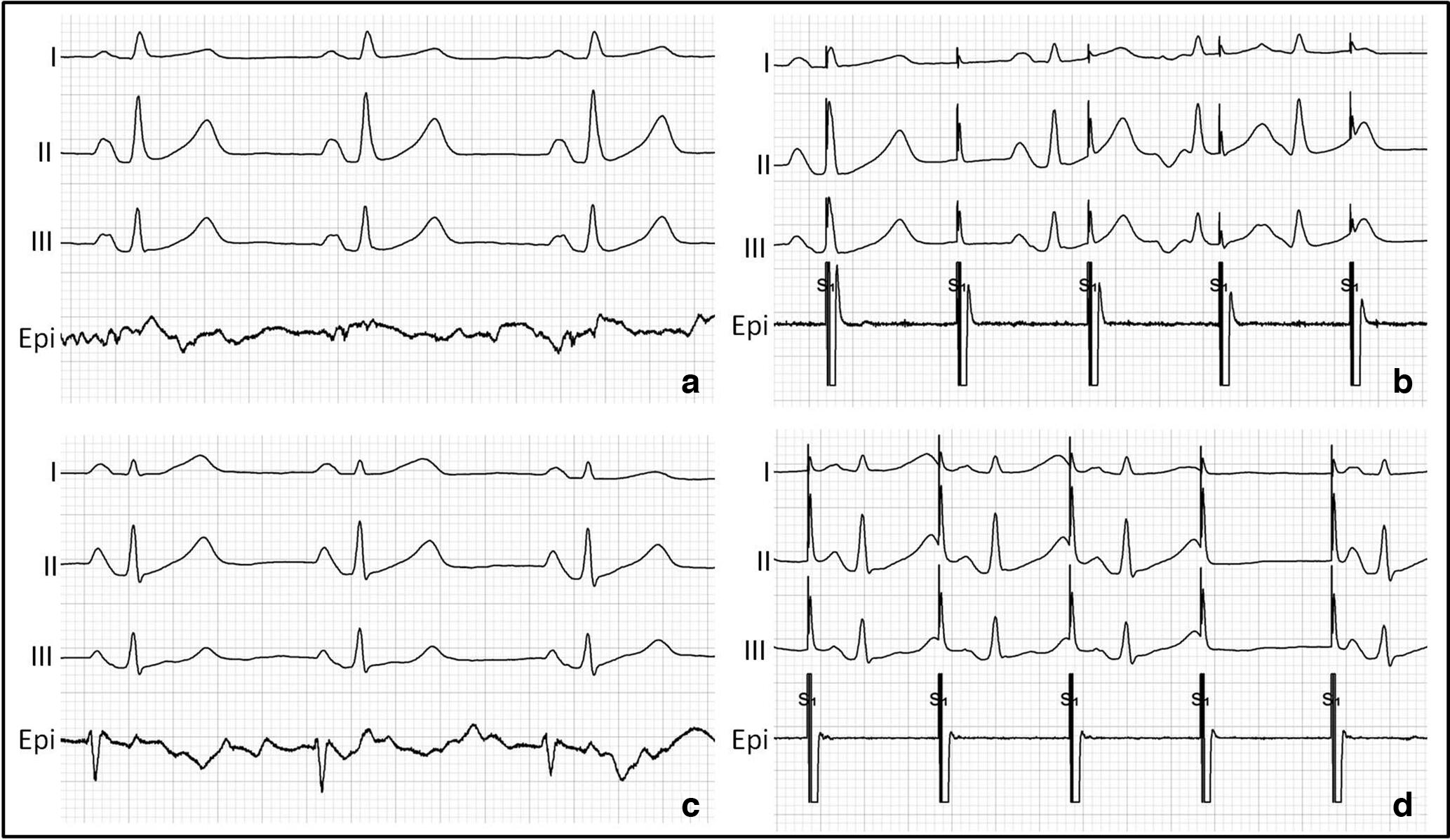
Electrophysiological recordings. Paper speed: 25 mm/s. (a) Entrance block: absence of potentials on the epicardially placed catheter. (b) Exit block: absence of conduction from pacing signals. If A and B are confirmed, the endpoint of bidirectional block is met. (c) Absence of entrance block: potentials recorded on the epicardially placed catheter. (d) Absence of exit block: conduction from pacing signals. If C or D is observed, the endpoint of bidirectional block is not met. Epi, epicardial testing using a His bundle catheter.
The epicardial ablation was immediately followed by an endocardial procedure. Via a 2-sided femoral venous approach, a bipolar His bundle catheter (St. Jude Medical, St. Paul, MN, USA), a coronary sinus catheter (Medtronic, Minneapolis, MN, USA) and a mapping catheter (Lasso or Pentaray, Biosense Webster, Diamond Bar, CA, USA) were placed under fluoroscopy. After transseptal puncture, an activated clotting time >300 seconds was maintained. If incomplete lesions were found, endocardial touch-up ablation was performed with a 3.5-mm cooled-tip radiofrequency catheter (SmartTouch, Biosense Webster). The endpoint for the ablation procedure was bidirectional block in each PV and in the box. If clinically necessary, additional right and/or left atrial lesions were made.
Electrophysiological Validation
Patients who were in AF after the end of the epicardial ablation were electrically cardioverted to restore sinus rhythm. The His bundle catheter and an EP workstation (Labsystem Pro, Boston Scientific, St. Paul, MN, USA) were used to epicardially validate the PVs and box for entrance (i.e., loss of conduction from the left atrium into each PV and the box, by sensing) and exit block (i.e., loss of conduction from each PV and the box into the left atrium, by pacing; Fig. 1, Fig. 2). The surgeon has direct vision on the target structures and the ablation line can easily be identified. This allows the surgeon to put the catheter just lateral from the ablation line but more toward the antrum of the atrium. Also, tissue contact can be evaluated in a direct manner by both tactile and visual feedback, ensuring good contact between the catheter and tissue. After correct placement of the catheter on the epicardial tissue, the EP workstation was checked for potentials. By absence of potentials, entrance block was confirmed. To check for exit block, pacing at the tip of the His bundle catheter using an output of 20 mA was performed with a speed faster than the heart rate of the patient. By absence of conduction, exit block was confirmed. A complete box lesion was defined as bidirectional conduction block in all PVs and in the box. Residual conduction was defined as persistent conduction from the left atrium in one or more PVs or in the box, revealed by sensing and/or pacing. Reverse capture was not tested.
After epicardial validation and placement of the endocardial catheters, the left atrium was mapped and, using a mapping catheter, bidirectional block of the PVs and box lesion were checked endocardially (Fig. 1). All signals on the Lasso or Pentaray catheter were evaluated for validation of the PVs and the box. After that, a voltage map was created. Further, exit block was tested at a location inside the PV with an output of 10 mA and a broad margin from the antral epicardial PV isolation, to be sure that the scar was not paced. The time between epicardial and endocardial validation was 20 to 30 minutes.
We also recorded predictions about conduction block based on a consistent drop of tissue conductance within 5 seconds for the PVs and within 20 seconds for the linear lesions, given by the epicardial radiofrequency ablation device (Synergy System, AtriCure Inc., Cincinnati, OH, USA and Coolrail, AtriCure Inc, Cincinnati, OH, USA, respectively). By absence of a consistent drop, even after multiple repeated application at the same location, absence of conduction block was predicted. This was compared to the actual findings of endocardial validation of bidirectional block.
Statistical Analysis
Data were prospectively entered into a database. Data were analyzed using SPSS Statistics, Version 25.0 (IBM Corp., Armonk, NY, USA). Continuous variables with normal distribution are presented as mean ± standard deviation, non-normally distributed variables as median and interquartile range (IR), and categorical variables as frequencies with percentages.
Results
Patient Characteristics
Baseline characteristics are summarized in Table 1. One patient was suffering from long-standing persistent AF; therefore, persistent and long-standing persistent AF patients were defined as nonparoxysmal. All paroxysmal AF patients underwent at least 1 catheter ablation in their history. Five patients (20%) were also known with atrial flutter, and 2 patients (8%) with atrial flutter and atrial tachycardia.
Baseline Characteristics.
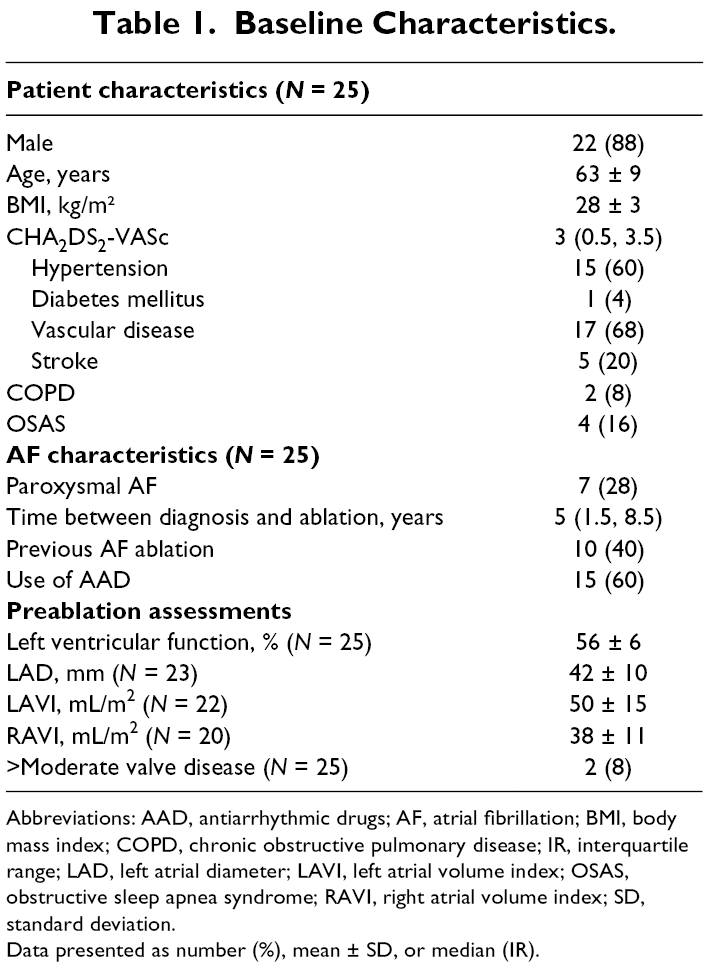
Abbreviations: AAD, antiarrhythmic drugs; AF, atrial fibrillation; BMI, body mass index; COPD, chronic obstructive pulmonary disease; IR, interquartile range; LAD, left atrial diameter; LAVI, left atrial volume index; OSAS, obstructive sleep apnea syndrome; RAVI, right atrial volume index; SD, standard deviation.
Data presented as number (%), mean ± SD, or median (IR).
Procedure Characteristics
Information on the procedure is summarized in Table 2. The median length of the procedure was 246 minutes (IR 222 to 299). Results of the conduction block validation tests are presented in Figure 3 and Table 3. Overall, presence of endocardial bidirectional block was always associated with epicardial bidirectional block, while epicardial bidirectional block could not be confirmed endocardially in 2/103 positive epicardial tests (1.9%).
Procedure Characteristics.
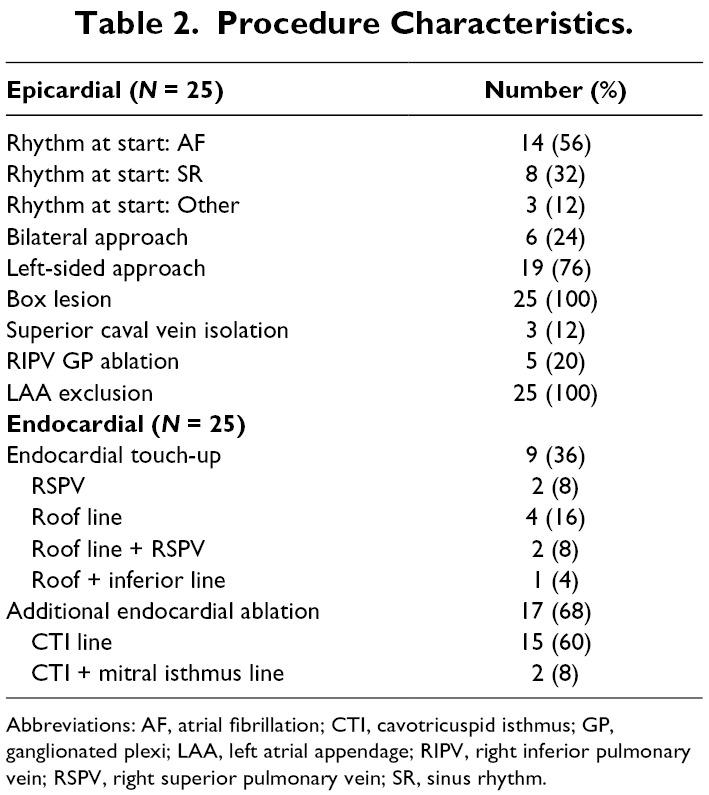
Abbreviations: AF, atrial fibrillation; CTI, cavotricuspid isthmus; GP, ganglionated plexi; LAA, left atrial appendage; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein; SR, sinus rhythm.
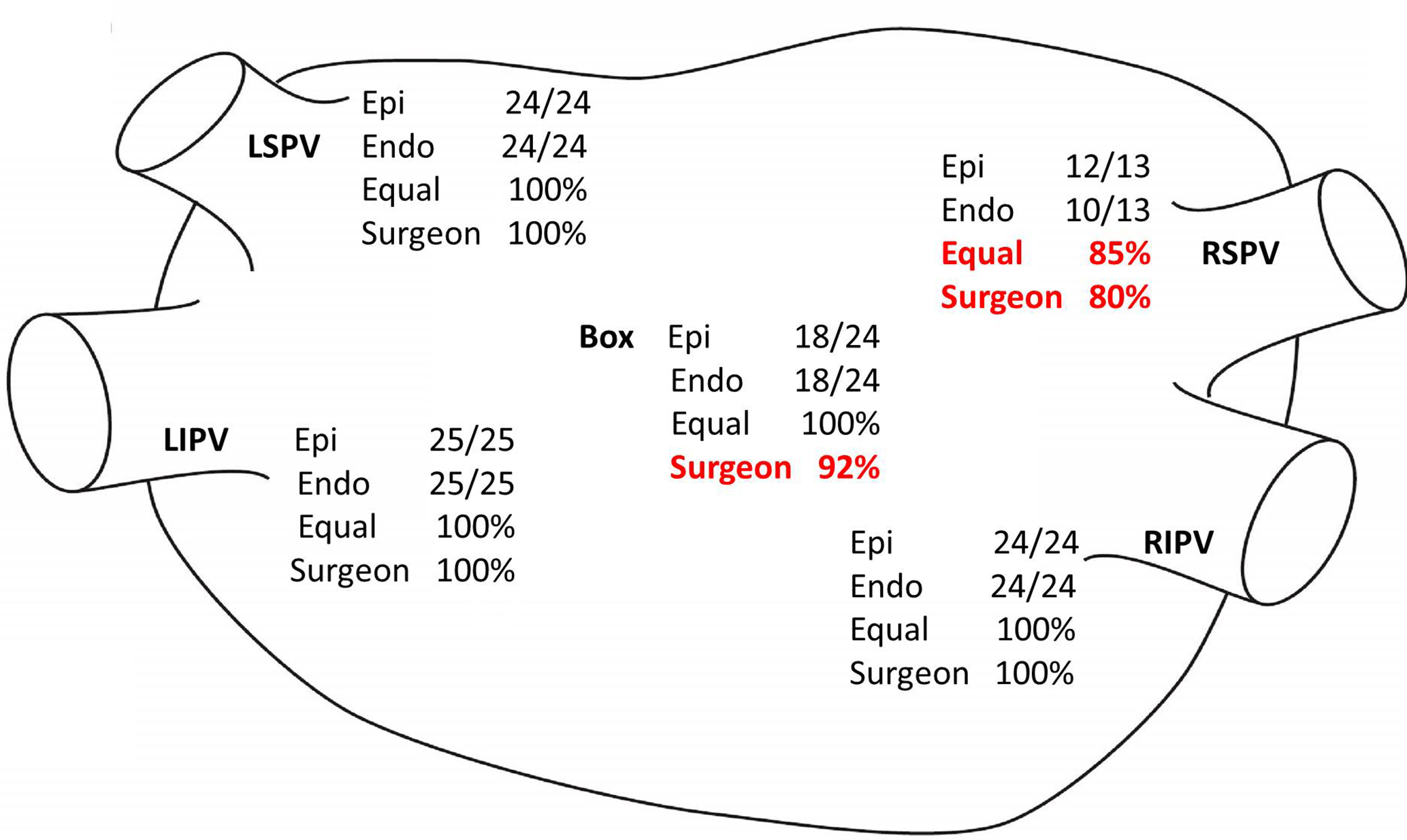
Results of testing for bidirectional conduction block. This posterior view of the left atrium shows the number of confirmed bidirectional conduction block in relation to the number of tested areas. Also, the percentage of similarity between epicardial and endocardial testing, and between endocardial testing and the correct estimation of the surgeon is shown. Endo, endocardial bidirectional block; Epi, epicardial bidirectional block; Equal, corresponding epicardial and endocardial conduction block; LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein; Surgeon, correct estimation of the surgeon if bidirectional block is present or not, based on a conductance graph given by the epicardial ablation device.
Conduction Block Comparison.
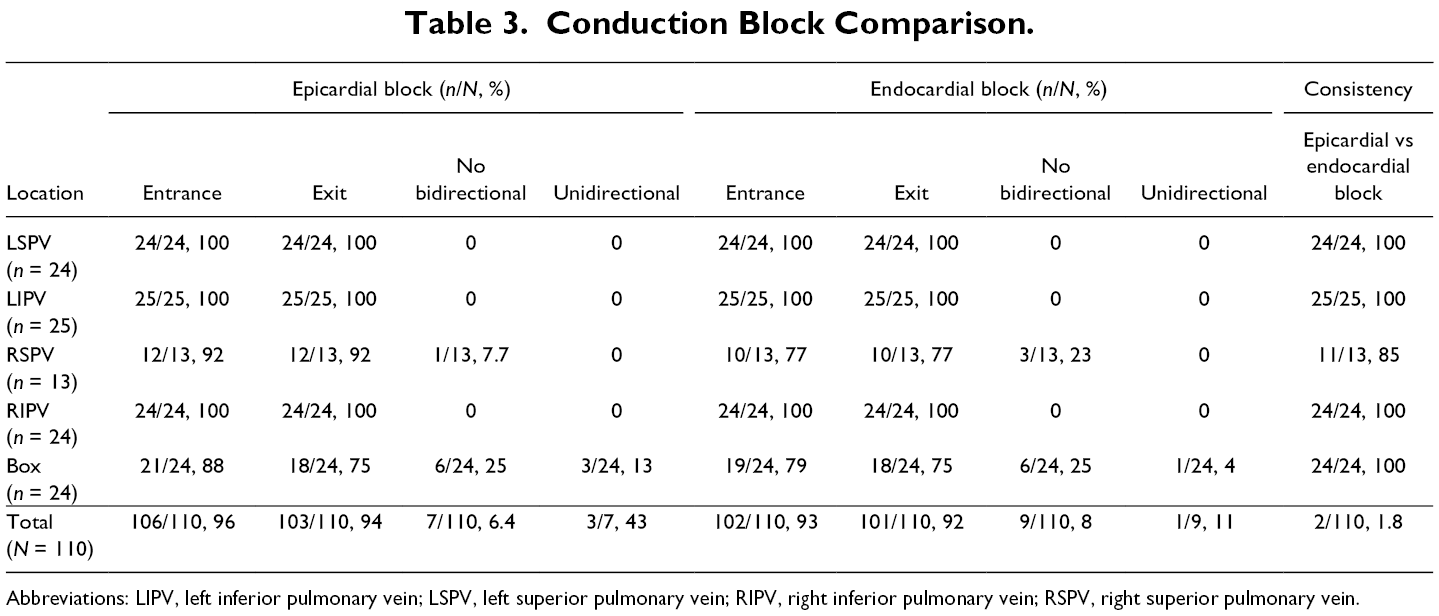
Abbreviations: LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein.
Right Superior Pulmonary Vein
The epicardial validation test results of the LSPV, LIPV, RIPV, and box lesion were completely consistent with the endocardial results. In 12 cases it was not possible to reach the RSPV for validation from the left side; therefore, the RSPV could only be validated epicardially in 13 cases. In 1/13 cases (7.7%) bidirectional block was epicardially absent. This was confirmed endocardially, leading to a 100% consistency for proving the absence of bidirectional block. In 12/13 cases (92.3%) bidirectional block was epicardially present. In 2 of these cases this could not be confirmed endocardially, resulting in a mismatch in 2/13 cases (15%). In both cases, the gap was located at the anterior side from the RSPV. Of the 12 cases that were only validated endocardially, a gap was found in 1 case.
Unidirectional Block
In 3/7 epicardial cases (43%) and in 1/9 endocardial cases (11%), a so-called unidirectional block was seen: an entrance block was apparent, while an exit block was absent. In all the cases this mismatch between entrance and exit block was found in the box (Table 3).
Touch-Up Ablation
In 9/25 patients (36%) endocardial touch-up ablation was performed for gaps found at 12/125 lesion locations (9.6%). In 1 patient with a gap in the roof and inferior line, the box could not be validated epicardially, and in 1 patient with a gap in the roof line and the RSPV, the RSPV could not be validated epicardially. Of the 9 remaining locations (in 8 patients) that could be validated epicardially, a gap was epicardially found at 7/9 locations (78%), so 2/9 gaps (22%) would have been missed and left untreated if no endocardial validation would have been performed.
Predictions
Based on how the conductance graph of the epicardial ablation device changed during the ablation, the surgeon expected gaps at 11 locations and a conduction block at the other 139 locations (roof and inferior line were predicted separately). These predictions were different from the endocardial validation results in 20% for the RSPV (5/25), and in 8% for the linear lesions of the box (4/50). Seven of the 11 expected gaps were indeed apparent, but 4 showed conduction blocks, resulting in a false-negative expectation of 36%. In one case the surgeon expected gaps at 3 different locations (roof, inferior, and RSPV), and in another case an incomplete RSPV, but epicardially and endocardially a bidirectional block, was found in all. Of the 139 expected complete lesions, only 134 showed conduction block, resulting in a false-positive expectation of 3.6%. In 2 of these 5 cases, only a gap at the RSPV was found. In 3 cases in which a gap already was expected, 1 additional gap was found in each (in 2 cases at the roof and in 1 case at the RSPV).
Discussion
In this study we explored the difference between epicardial and endocardial validation of bidirectional conduction block, and predictions about conduction block based on the epicardial ablation device measuring tissue conductance. Epicardial and endocardial validation tests corresponded very well, but not completely. Based on tissue conductance, completeness was misjudged in 20% of the RSPV isolations and in 8% of the linear lesions, of which 5 cases (3.6%) concerned a false-positive expectation. This emphasizes the importance of confirming valid EP endpoints at the end of the procedure.
In patients in whom catheter ablation is expected to be insufficient for achieving satisfying results, a thoracoscopic epicardial or a hybrid ablation can be an alternative. 5 -9 During hybrid ablation, conduction block is checked endocardially using a mapping catheter and EP workstation, but also for the success of a thoracoscopic procedure it is critical to make sure that the established endpoint of bidirectional conduction block is reached. 6,10 In current surgical practice non-EP guided conduction block validation is most commonly performed using an epicardial pen without the presence of an EP or an EP workstation. 1 In some centers EP guided validation is performed, which may contribute to the success rate. 11,12 While those studies used sophisticated custom-made epicardial mapping electrodes showing reliable testing results, we could reveal similar correlation with commercially available mapping catheters. 13 In our center, bidirectional block after thoracoscopic ablation procedures is always validated by an EP, using an endocardial His bundle catheter and an EP workstation. The reason for using an endocardial catheter is the possibility to connect it to an EP workstation, which allows to obtain more detailed signals and, if necessary, additional epicardial ablation can be guided more accurately.
In the current study we have shown that all validation tests, except for 15% of the RSPV, showed similar consistency in the presence or absence of conduction block. When performing a unilateral left-sided thoracoscopy, it is challenging to reach the RSPV. Therefore, validation of the RSPV could only be performed in 13 cases. In 2/13 (15%) cases, an epicardially found bidirectional block could not be confirmed endocardially. This could be a consequence of the catheter not picking up epicardial signals, due to edema or the anterior location of the gaps, or misplacement of the catheter in the unilateral approach due to limited visualization, since in the 6 bilateral approached cases no discrepancies were seen. This shows that in a left-sided approach it is important to make sure that the RSPV is reached to prevent false-positive results. Further, we found incomplete epicardial lesions in 9.6% of the performed lesions and in 36% of the patients. This is in line with previous studies of our group reporting on larger patient groups. 2,3,14 This is related to the fact that in a hybrid setting no epicardial reablation is performed if a gap is detected by epicardial testing, because it can be targeted more precisely from the endocardium after endocardial mapping.
Except testing for bidirectional block, predictions about conduction block based on tissue conductance graphs can also be of value. The ablation system measures tissue conductance, which is inversely proportional to tissue impedance, to control the amount of energy delivered to the tissue required to create a transmural lesion. As tissue impedance rises during ablation, the amount of energy delivered to the tissue decreases accordingly. Energy is delivered until conductance between the electrodes decreases and reaches a steady state. 15 The time necessary to create a transmural lesion depends on tissue thickness and tissue composition (i.e., the amount of epicardial fat). During the ablation, the ablation system shows a graph with conductance on the y-axis and time in seconds on the x-axis. If the conductance does not drop, it indicates unsatisfying tissue damage and thus a decreased chance of transmurality. Based on this algorithm, in the current study, a gap was predicted in 9 cases. However, 3 were false negative and 2 false positive predicted. This highlights the importance of measuring EP endpoints during the procedure to confirm bidirectional block.
Further, we confirmed that it is important to validate both exit and entrance block, as recommended by the current guidelines. 6 Prior reports also demonstrated the absence of exit block, while entrance block was present. 10,16 -18 It was even shown that a present entrance block recovered within 30 minutes in cases with absent exit block. 17 However, Duytschaever et al. state that entry block implies exit block, since they only found <1% unidirectional block. 19 The difficulty when testing exit block using pacing maneuvers is the possibility to have far-field capture of the left atrial appendage or superior vena cava, which might result in misinterpretation of apparent exit block. 20 In the current study we found entrance block but no exit block, in 11% of the cases with an endocardial absent bidirectional block, and in 43% of the cases with an epicardially absent bidirectional block. In one of the epicardial cases we found the same pattern endocardially, in the other 2 we found absence of entrance and exit block endocardially. In all 3 cases touch-up ablation was performed. This shows that, irrespective of possible far-field capture, it is important to also test for exit block, especially in epicardial ablation.
Limitations
It cannot be excluded that previous catheter ablation influenced the results in terms of bidirectional conduction block. Although this should not have an impact on the epicardial and endocardial validation test results, it could be that the percentage of bidirectional block was favorably influenced. The current results and its impact on rhythm outcome should, therefore be evaluated in a larger trial with longer follow-up.
Further, the value of a tissue conduction drop within 5 seconds as indicated by the generator as endpoint for predicted conduction block was not based on a dose–response investigation but from our earlier clinical experience resulting from EP testing of epicardial ablation lines. However, the clinical impact of false-negative prediction influenced by picking this value as cutoff endpoint appears to be minor in terms of efficacious ablation lines.
Conclusions
Epicardial bidirectional conduction block in the PVs and the box lesion corresponded well with bidirectional conduction block validated endocardially. Conflicting results could only be obtained for the RSPV in a few cases, which could be due to the chosen left-sided-only approach. As a result, it appears to be equally reliable whether endpoint testing is performed endocardially or epicardially. If the use of an endocardial catheter for validation of epicardial lesions is superior to validation with current surgical sensing and pacing devices, and thus leads to differences in outcome, should be explored in future studies. Further, this study emphasizes the importance to check for entrance and exit block, and to not only rely on predictions about conduction block based on tissue conductance graphs from the epicardial ablation system.
Supplemental Material
Presentation S1 - Supplemental material for Epicardial and Endocardial Validation of Conduction Block After Thoracoscopic Epicardial Ablation of Atrial Fibrillation
Supplemental material, Presentation S1, for Epicardial and Endocardial Validation of Conduction Block After Thoracoscopic Epicardial Ablation of Atrial Fibrillation by Mindy Vroomen, Bart Maesen, Justin L. Luermans, Jos G. Maessen, Harry J. Crijns, Mark La Meir and Laurent Pison in Innovations: Technology and Techniques in Cardiothoracic and Vascular Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mark la Meir is a consultant for AtriCure.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
