Abstract
Nano-sized extracellular vesicles enclosed by a lipid bilayer and secreted by various cell types including mesenchymal stem cells, exosomes act as natural transporters, carrying bioactive molecules such as proteins, lipids, and nucleic acids that mediate intercellular communication. Exosomes influence a range of cellular processes, including immune modulation, tissue repair, and disease progression. Compared to whole-cell therapies, exosomes provide anti-inflammatory, immunosuppressive, and regenerative effects with reduced risks linked to cellular components, such as infusion toxicity, immunogenicity, and tumorigenic phenomena. This article reviews isolation, modification, characterization, and storage techniques, challenges in clinical translation, and innovative engineering strategies to enhance targeting and efficacy. It also examines preliminary evidence suggesting that exosomes may have potential in managing degenerative disorders such as osteoarthritis, intervertebral disc degeneration, osteoporosis, osteonecrosis, and tendinopathy, as well as non-degenerative disorders such as sciatic nerve injury, fractures, and soft tissue trauma.
Introduction
Stem cell-based therapies, especially those using mesenchymal stem cells (MSCs), have emerged as a promising modality for regenerative medicine, particularly for the treatment of orthopedic and musculoskeletal pathology. The clinical role of MSCs has evolved since they were first described by Arnold Caplan in 1991 as the basis for the emergence of a new therapeutic technology of self-cell repair. 4 The term “mesenchymal stem cells” was fitting, as these cells were shown to possess the potential to proliferate into various musculoskeletal tissues of the mesodermal lineage, including cartilage, bone, adipose, and tendon. 44 More recently, Caplan suggested that MSCs may be more accurately described as “medicinal signaling cells” that have no direct morphogenic effects through differentiation but rather secrete bioactive factors that are immunomodulatory and regenerative. 3 Regardless, MSCs have been shown to possess anti-inflammatory and tissue repair properties, with evidence suggesting possible clinical application in managing degenerative diseases such as osteoarthritis (OA), as well as promoting bone and soft tissue healing. 50
Stem cell-based therapies in orthopedics have important limitations, including infusion toxicity, immunogenicity, tumorigenic potential, and ethical issues—many that originate from the necessity of whole-cell transplantation. 69 To address these challenges, researchers have investigated the underlying mechanisms through which MSCs promote tissue regeneration. Initial research focused on growth factors, cytokines, and chemokines; however, no single factor could account for all therapeutic effects. Studies have shown that many of the mechanistic benefits of MSCs involve paracrine signaling via the release of extracellular vesicles (EVs).23,58 This research has revealed more defined and precise active subcellular “organelles,” structures that may be responsible for advancing morphogenic and paracrine functional pathways. It is mainly through the secretion of EVs that MSCs exert a therapeutic effect. 56
EVs are membrane-bound vesicles with distinct functions classified into 3 types: exosomes, microvesicles, and apoptotic bodies. These vesicles vary in size, morphology, density, cargo, biogenesis, and biological activity, all of which distinguish them from one another. 2 The content and biodistribution of EVs are highly heterogeneous and dependent on their cell of origin, surface molecules, microenvironment, isolation methodologies, and the active signals within a cell. 65 Given their role in cellular regulation, EVs have gained interest in clinical research for possible use in vaccines and immunosuppressants, as well as in healing, repair, and differentiation processes following tissue injury. 45
Exosomes are the smallest and most extensively studied of EV particle types. (EV biogenesis and release are summarized in Figure 1.) Exosomes originate as intraluminal vesicles (ILVs) formed by the inward budding of the endosomal membrane. During this process, bioactive substances are captured and packaged into ILVs, which are contained within multivesicular endosomes (MVEs). The exosomes are subsequently released into the extracellular space when the MVE fuses with the plasma membrane. 55 Exosomes are nano-sized spherical vesicles, 30 to 150 nm in diameter and are composed of a lipid bilayer. Secreted by a variety of cell types, they act as intercellular messengers that facilitate the transfer of bioactive molecules such as proteins, lipids, and nucleic acids. 72 They also act as transport pods, carrying molecular “cargo” or signaling proteins that mediate cell-to-cell communication and promote regulation of cellular processes. 42 Exosomes interact with target cells through several mechanisms, including internalization, direct membrane fusion, and receptor-ligand interactions. Upon internalization by the target recipient cell, exosomes release their molecular contents, which can influence various cellular functions such as immune modulation, tissue repair, and disease progression, depending on the exosome origin and cargo and the recipient cell’s metabolic state. 22
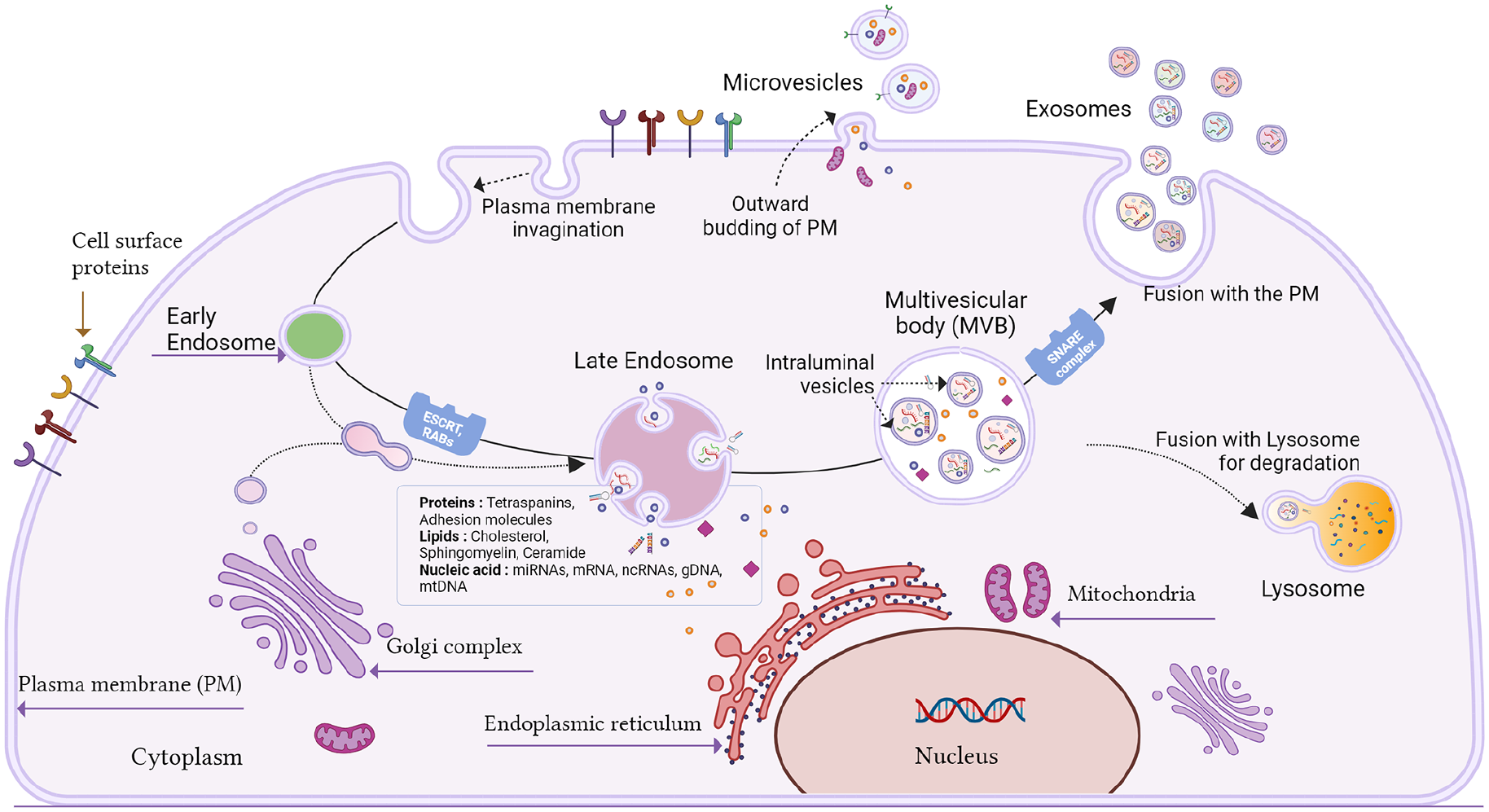
Overview of EV biogenesis and release. EVs, such as exosomes and microvesicles, are released from cells through distinct pathways. Early endosomes are formed by the invagination of the plasma membrane or the trans-Golgi network. These mature into late endosomes where inward budding of the endosomal limiting membrane forms ILVs, containing cytosolic cargo such as proteins, lipids, and nucleic acids. These endosomes containing ILVs are multivesicular bodies that can either fuse with the plasma membrane, releasing their ILVs as exosomes, or fuse with lysosomes for degradation. Microvesicles are formed by direct outward budding of the plasma membrane and may encapsulate cytosolic components as they form. 55 Created with BioRender.com.
Exosomes may offer an alternative to aggregate cellular therapies due to their smaller size, modifiable surface and cargo, stable long-term storage, and efficient transport properties. Furthermore, they pose a significantly lower risk of immunogenicity and tumorigenesis, making them an attractive clinical treatment alternative. 54 Interest is increasing in the use of exosomes for regenerative medicine.
This review examines exosomes’ origins and the techniques used in their isolation, modification, characterization, and storage. It also describes several clinical applications and the strategies used to enhance their targeting and efficacy.
Exosomes: The Importance of Cellular Sources
The cellular source of exosomes plays a fundamental role in determining their usability and applications. Common sources include stem cells, macrophages, platelets, synoviocytes, adipocytes, dendritic cells, and tumor cells. Among these, MSCs have been identified as a particularly viable source due to their prolific production of EVs. 68 Although bone marrow MSC-derived exosomes (BM-MSC-Exos) and adipose tissue MSC-derived exosomes (AD-MSC-Exos) are frequently cited sources, studies have also found benefits in reducing articular cartilage damage and joint inflammation through exosomes from the synovium, the infrapatellar fat pad, and platelet-rich plasma (PRP). 33 Paired with the ability to proliferate and differentiate into musculoskeletal tissues, MSCs are considered ideal candidates for exosome-based clinical trials, particularly in musculoskeletal therapies.
Exosomes contain cell-specific biomarkers that link them to their parent cells. 54 This suggests that MSC-derived exosomes inherit their therapeutic properties, which include being anti-inflammatory, immunosuppressive, and promoting tissue regeneration without the disadvantages of infusion toxicity, immunogenicity, and tumorigenic phenomena. 46 MSCs are highly heterogeneous, and this variability extends to the EVs they produce. Beyond differences in vesicle size, there is also variability among exosome subpopulations. 30 Several factors influence exosome production, including the MSC source, passage number, growth media, ex vivo atmospheric conditions, culture substrate, and collection methods. 2
Isolation and Purification
Techniques for exosome isolation and purification are based on their size, density, surface charge, immunoaffinity, and solubility. 27 Because exosomes are heterogeneous in size, content, surface markers, and source, it is difficult to standardize their isolation. Ultracentrifugation is the most widely used isolation method, but it is costly, time-intensive, and not easily scalable. In addition, repeated centrifugation damages exosomes, reducing their quality. 66 To overcome these limitations, studies have investigated alternative exosome isolation techniques such as immunoaffinity chromatography, ultrafiltration, and size-exclusion chromatography. 61 Immunoaffinity chromatography is a separation technique that relies on the binding between antibodies or ligands and target molecules on the surface of exosomes. This is a rapid method that offers high purity, specificity, and yield. However, the antigen/protein coupling needs to be present on the surface of the exosomes. 13 On the other hand, size-based isolation techniques such as ultrafiltration and size-exclusion chromatography are associated with more expeditious processing and are well-suited for large-scale applications. However, these methods can lead to pore clogging, exosome loss, and lower purity, which has hindered widespread use. 26 Typically, the faster the exosome isolation method, the more likely it is to produce structural damage and low yield. 74 Consensus has shifted toward “gentle” isolation methods that optimize exosome purity and preserve structural integrity; however, each technique possesses its own disadvantages. 33 Given the limitations of each technique, combining methods—such as immunoaffinity chromatography with size-based techniques or adding precipitation-based and microfluidics-based methods—may provide a more effective solution, enabling researchers to meet the various demands for exosome isolation and purification. 54
Modification
Exosomes can be biochemically modified to enhance, alter, or expand their therapeutic potential. These modifications fall into 2 main categories: internal modifications (which alter the exosomal cargo) and external modifications (which alter the surface lipid membrane of the exosome). 54 Internal modifications are further classified into pre-secretion and post-secretion loading techniques; this depends on whether MSCs are treated before exosome secretion, or if the secreted exosomes are the direct target of the treatment. Pre-secretion methods include incubation, transfection, and electroporation, while post-secretion methods include incubation, freeze-thaw cycles, sonication, extrusion, and hypotonic dialysis.27,72 While incubation is the least disruptive internal modification, it is only effective for small, hydrophobic molecules that can diffuse easily. 29 For larger or hydrophilic cargo, such as protein, microRNA (miRNA), small interfering RNA, and messenger RNA, more invasive techniques such as extrusion, electroporation, and sonication are required. External modifications involve various strategies such as the genetic engineering of exosomal membranes or origin cells, chemical conjugation of targeting ligands, electrostatic interactions, and magnetic nanoparticle technologies. 72 The primary goal of surface modification is to enhance the targeted delivery of exosomes to specific cell types, thereby increasing therapeutic effectiveness.
Characterization
Methods for exosome characterization focus on verifying its size, morphology, and cargo profile. 34 Size-based analysis techniques include nanoparticle tracking analysis (NTA), dynamic light scattering, and tunable resistive pulse sensing. Morphology is typically assessed using scanning electron microscopy and transmission electron microscopy (TEM). 13 Cargo analysis is further subdivided into proteomic, lipidomic, and genomic profiling, with techniques such as western blot, enzyme-linked immunosorbent assay, flow cytometry, mass spectroscopy, and polymerase chain reaction. 28 Each method has inherent strengths and limitations, and so it is common practice to use multiple characterization techniques for a thorough analysis. One common combination of techniques is TEM for structural visualization, NTA for size distribution and concentration analysis, and western blotting for vesicle protein marker verification. 54 This multifaceted approach enhances the accuracy and reliability of exosome characterization.
Storage
The primary methods for long-term exosome preservation include cryopreservation, lyophilization (freeze-drying), and spray-drying. 72 Cryopreservation is the most frequently used, with temperature and cryoprecipitates being critical for maintaining exosome integrity. Cryoprecipitates, such as trehalose, are added to storage buffers to protect exosomes during freezing and thawing. The literature suggests that storing exosomes at −80 °C in a phosphate buffer solution supplemented with trehalose cryoprotectants offers optimal protection for both short- and long-term preservation. 21 However, despite its efficacy, this approach can be costly and time-intensive, making it impractical for large-scale transportation from laboratories to hospitals or clinical settings. 74 Lyophilization is another effective preservation method, particularly for thermolabile materials like exosomes. This process allows for cheap, easy storage and simple reconstitution by adding water. As with cryopreservation, adding cryoprotectants can maintain exosomal protein and RNA activity for up to 4 weeks, even at room temperature. 5 Spray-drying, in contrast to freeze-drying, is a single-step process that eliminates the need for costly equipment and extensive multistep processing. However, factors such as the exosome feeding rate, atomization pressure, and outlet temperature must be carefully controlled, as they can impact exosome stability and cargo integrity. 31 While both lyophilization and spray-drying may be alternatives to cryopreservation, comparative studies evaluating their efficacy remain limited. Further research is needed to establish standardized, cost-effective protocols for scalable exosome storage and transportation.
Improved Retention
Researchers currently lack sufficient evidence on the optimal dose and frequency of exosome administration for therapeutic applications. In many animal models, therapeutic effects require higher dosing regimens, leading to substantial exosome production demands that limit preclinical research. This poses a barrier to clinical translation, as the larger anatomical structure and increased disease complexity in humans likely require even higher doses and prolonged treatment durations.33,51 To address this, researchers have developed several methods to increase exosome production. These include biochemical approaches (eg, lipopolysaccharide, bone morphogenetic protein-2, hypoxia-inducible factor 1-alpha, and cytokines such as interferon-gamma, and tumor necrosis factor-alpha [TNF-α]), physical methods (eg, hypoxia, thermal stress, and starvation), mechanical techniques (eg, shear stress and 3D cell culture), and instrumental approaches (eg, hollow-fiber bioreactors and stirred tank bioreactors). 1 Despite advances, efficient, large-scale exosome production remains a major challenge.
Improving the retention of exosomes following delivery is another method of enhancing therapeutic efficacy while reducing the need for increased dosage and treatment frequency. One strategy involves loading exosomes onto biocompatible hydrogels or scaffolds. 48 These have been shown to maintain the local concentration of exosomes by enabling controlled release, extending exosomal half-life, and preventing clearance.7,8,70 Chen et al created 3D-printed scaffolds using bioink composed of BM-MSC-Exos, decellularized cartilage extracellular matrix (ECM), and gelatin methacrylate hydrogel; this facilitated cartilage regeneration better than exosome treatment alone in a rabbit osteochondral defect model. 8 The outcome may be attributed to higher exosome retention and the controlled release of both exosomes and their cargo in the exosome-loaded scaffold treatment group. 3D-printed scaffolds can effectively control exosome administration, providing a 1-step operation system, which is a more convenient way to maintain therapeutic exosome concentration than conventional methods of repetitive intra-articular injections.8,48 Therefore, the use of hydrogels and scaffolds to enhance exosome retention remains an area for further study.
Clinical Applications
Animal and in vitro studies have suggested the potential use of exosomes in treating musculoskeletal and neurological conditions: degenerative disorders such as OA, intervertebral disc degeneration (IDD), osteoporosis, osteonecrosis, and tendinopathy and non-degenerative disorders such as sciatic nerve injury, fractures, and soft tissue trauma. These conditions are characterized by tissue degradation, collagen breakdown, inflammation, and impaired regenerative capacity, making exosomes an attractive therapeutic option due to their ability to modulate cellular processes and promote tissue repair (Figure 2).
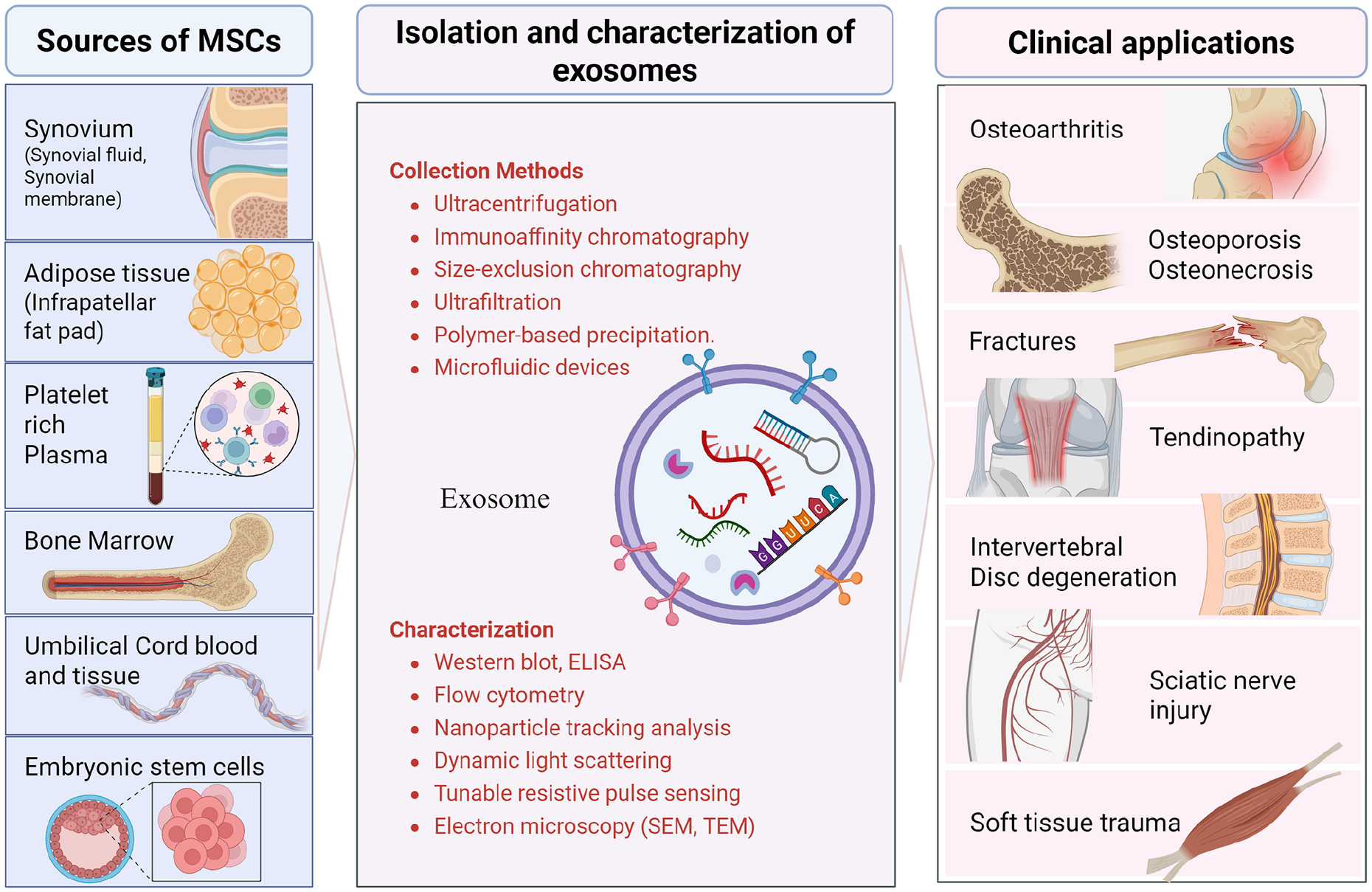
Sources of MSCs for exosome derivation, methods for exosome isolation and characterization, and potential therapeutic targets. Created with BioRender.com.
Degenerative Disorders
OA, the most common joint disease, is characterized by cartilage degradation, synovial inflammation, and disordered subchondral bone remodeling, leading to chronic pain and functional impairment. 19 Exosomes have shown potential in treating OA by targeting key catabolic and anabolic pathways using miRNA cargo; specifically, these include BM-MSC-Exos, AD-MSC-Exos, umbilical cord-derived MSC exosomes (UC-MSC-Exos), synovium-derived MSC exosomes, and infrapatellar fatpad-derived MSC exosomes.11,24,25,36,40,63 Exosomes promote chondroprotection and ECM synthesis by upregulating anabolic genes such as the ACAN (aggrecan) gene and alpha-1 chain of collagen type II, while downregulating catabolic enzymes such as matrix metalloproteinase 13 and A disintegrin and metalloproteinase with thrombospondin motifs 5, as demonstrated in rodent studies.11,36 Additionally, exosomes alleviate pro-inflammatory cytokines such as interleukin-1β (IL-1β), IL-6, and TNF-α to suppress inflammation.25,36 Human MSC studies show exosomes that target Wnt5a, a key mediator of cartilage degradation, inhibiting its expression through Wnt signaling and nuclear factor-κB (NF-κB) pathways, thereby reducing catabolic activity.24,40 Notably, exosomes derived from PRP were shown to be a superior treatment than PRP alone in a rabbit model, with a possible role in reversing the decrease in collagen II and runt-related transcription factor-2 (RUNX2) protein expression, promoting cartilage repair, and inhibiting OA progression through modulation of Wnt/β-catenin signaling. 39 In vivo rat studies have shown that the chondroprotective effects of intra-articular injected EVs are dose dependent. 36
Osteoporosis is a systemic bone disorder marked by reduced bone mass, mineral density, and microarchitectural deterioration, leading to an increased risk of fractures. It results from an imbalance between osteoclastic bone resorption and osteoblastic bone formation. 10 BM-MSC-Exos, AD-MSC-Exos, and UC-MSC-Exos have shown potential in restoring bone homeostasis by promoting osteoblast activity and inhibiting resorptive osteoclasts in rabbit and in vitro studies. These exosomes can enhance osteoblast proliferation and differentiation by delivering miRNAs and growth factors that activate osteogenic pathways.17,49 They also inhibit osteoclast activity through the OPG/RANKL/RANK and Wnt/β-catenin pathways.35,47
IDD is a leading cause of chronic back pain and disability, characterized by ultrastructural deterioration of the nucleus pulposus and annulus fibrosus. It has a similar pathogenesis to OA through ECM degeneration, inflammation, oxidative stress, apoptosis, as well as senescence and reduced autophagy. 16 BM-MSC-Exos have demonstrated therapeutic potential in mitigating IDD by reducing senescence and apoptosis in nucleus pulposus cells and promoting ECM synthesis in in vitro studies.12,53 BM-MSC-Exos were shown to alleviate oxidative stress and inflammation by targeting endoplasmic reticulum stress and mitochondrial dysfunction, thereby restoring ECM homeostasis in rat and rabbit studies.37,64
Osteonecrosis, particularly of the femoral head (ONFH), is a progressive and functionally debilitating condition with causes including trauma, corticosteroid use, and alcohol intake. 18 Glucocorticoid (GC)-induced osteonecrosis is one of the most common etiologies of ONFH, and if progressive, can require the need for total hip arthroplasty. 43 Exosomes derived from human-induced pluripotent stem cell-derived MSCs have been shown to promote endothelial cell proliferation and migration via the PI3K/AKT pathway, improving blood supply to necrotic bone in cases of GC-induced ONFH. 38 In addition, BM-MSC-Exos upregulate osteogenic genes such as BMP2 and SOX9, activating TGF-β/BMP pathways to enhance bone formation. 15
Tendinopathy involves tendon injury, breakdown, degeneration, and ECM disruption, often leading to tendinosis, tendon collagen fibril pathology, and possible rupture. Exosomes derived from tendon-derived MSCs have shown potential in promoting tendon repair by upregulating ECM components such as COL1A1 and suppressing catabolic enzymes like matrix metalloproteinase 3. 60 AD-MSC-Exos also exert anti-inflammatory effects by inhibiting NF-κB signaling and reducing pro-inflammatory cytokines, thereby alleviating tendon inflammation and many of the common soft tissue disorders. 52
Non-Degenerative Disorders
Soft Tissue Trauma: Untreated muscle, tendon, and ligamentous trauma, resulting from acute injury or chronic overuse, can lead to functional impairment and long-term disability. 57 BM-MSC-Exos and AD-MSC-Exos have shown potential in promoting tendon healing by stimulating collagen synthesis and tenocyte proliferation and migration, thereby improving tendon-muscle unit strength and elasticity in murine and rabbit studies.9,41 For muscle tears, AD-MSC-Exos have been shown to reduce atrophy, inflammation, and fatty infiltration while promoting biomechanical properties in a rabbit study. 59 The most common soft tissue injuries are tendon-bone enthesopathic insertional injuries, as well as anterior cruciate ligament (ACL) injury and rotator cuff tears. BM-MSC-Exos have demonstrated an ability to promote tendon-bone healing by stimulating M1 to M2 macrophage polarization, increasing angiogenesis, and enhancing osteogenesis through upregulation of transcription factors SOX-9, RUNX2, and osterix in rodent studies. 75 For purely ligamentous injury, such as ACL tears, exosomes from human embryonic stem cell-derived MSCs demonstrated enhanced healing through fibroblast proliferation, migration, and collagen synthesis, while simultaneously restoring ECM homeostasis 62 in a rabbit study.
Fractures are a common traumatic orthopedic condition that can result in functional morbidity if not properly treated. Fracture healing is characterized by an anabolic phase of increasing skeletal tissue volume, followed by a catabolic phase in which the new tissue is remodeled. 14 Exosomes have shown potential in accelerating a more organized bone healing process by enhancing angiogenesis and osteogenesis. Most cells responsible for fracture healing, such as osteoblasts and vascular endothelial cells, can take up exosomes, allowing exosomes to modify gene expression in those cells. 67 BM-MSC-Exos and UC-MSC-Exos were shown to accelerate the proliferation of osteoblasts and endothelial cells, further promoting osteogenesis (via the BMP-2/Smad1/RUNX2 pathway) and angiogenesis (through the HIF-1a-VEGF pathway) in rat studies.71,73 Disordered fracture healing is a clinical challenge that may be improved by more advanced exosome-derived therapies.
Sciatic nerve injury can result in pain and functional impairment. Sciatic nerve regeneration is regulated by the interaction between neurons and Schwann cells, with additional complexity due to inflammatory cell infiltration. 20 AD-MSC-Exos have shown potential in promoting nerve regeneration by enhancing Schwann cell proliferation, reducing inflammation, and stimulating axonal growth in a rat study. 6 LPS-preconditioned BM-MSC-Exos could accelerate peripheral nerve regeneration and target inflammatory cells by enhancing M2 macrophage polarization in a rat study. 32
Future Directions
The future of exosome-based therapies lies in addressing current limitations in standardization, production, and clinical translation. Standardizing protocols for isolation, purification, characterization, and storage—which are costly, time-consuming, and prone to compromising exosome integrity—will be critical to ensuring consistency and reproducibility across studies, as will developing scalable and cost-effective production methods. Research efforts should focus on optimizing therapeutic strategies, such as cargo loading, surface engineering, and incorporating exosomes in biocompatible materials, such as hydrogels or scaffolds, to enhance targeted delivery and therapeutic efficacy through controlled release. Identifying optimal doses, exosome sources, and administration frequencies, along with rigorous preclinical and clinical studies, is necessary to validate safety, efficacy, and long-term outcomes; these studies are key to achieving therapeutic effects in humans. Finally, collaboration among researchers, clinicians, and regulatory bodies will be essential in navigating regulatory hurdles and accelerating the translation of exosome therapies from bench to bedside.
Supplemental Material
sj-docx-1-hss-10.1177_15563316251362179 – Supplemental material for Clinical Status of Exosomes: A Review
Supplemental material, sj-docx-1-hss-10.1177_15563316251362179 for Clinical Status of Exosomes: A Review by Jonathan Sgaglione, Eric V. Neufeld, Pooja Swami and Daniel A. Grande in HSS Journal®
Supplemental Material
sj-docx-2-hss-10.1177_15563316251362179 – Supplemental material for Clinical Status of Exosomes: A Review
Supplemental material, sj-docx-2-hss-10.1177_15563316251362179 for Clinical Status of Exosomes: A Review by Jonathan Sgaglione, Eric V. Neufeld, Pooja Swami and Daniel A. Grande in HSS Journal®
Supplemental Material
sj-docx-3-hss-10.1177_15563316251362179 – Supplemental material for Clinical Status of Exosomes: A Review
Supplemental material, sj-docx-3-hss-10.1177_15563316251362179 for Clinical Status of Exosomes: A Review by Jonathan Sgaglione, Eric V. Neufeld, Pooja Swami and Daniel A. Grande in HSS Journal®
Supplemental Material
sj-docx-4-hss-10.1177_15563316251362179 – Supplemental material for Clinical Status of Exosomes: A Review
Supplemental material, sj-docx-4-hss-10.1177_15563316251362179 for Clinical Status of Exosomes: A Review by Jonathan Sgaglione, Eric V. Neufeld, Pooja Swami and Daniel A. Grande in HSS Journal®
Footnotes
Informed Consent
Informed consent was not required for this review article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Daniel A. Grande, PhD, declared relationships with ICRS and Cresilon. The other authors declare no potential conflicts of interest.
Human/Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article as supplemental material.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
