Abstract
The widespread and persistent underrepresentation of groups experiencing health disparities in research involving biospecimens is a barrier to scientific knowledge and advances in health equity. To ensure that all groups have the opportunity to participate in research and feel welcome and safe doing so, we must understand how research studies may be shaped to promote inclusion. In this study, we explored the decision to participate in hypothetical research scenarios among African American adults (n = 169) that varied on the basis of four attributes (form of consent, reason for research, institutional affiliation and race of the researcher). Findings indicate that participants were largely willing to contribute to biobanks but significantly preferred opportunities where they had control over the use of their biological samples through tiered or study-specific forms of consent. Broad consent procedures, although common and perhaps preferred by participants with high trust in researchers, may amount to an exclusionary practice.
Keywords
Introduction
Diversity in research participation is critical to the achievement of health equity, yet studies rarely have adequate participation from the very populations that suffer most from health inequities (Knepper & McLeod, 2018; Oh et al., 2015). This lack of diversity can also potentially result in the exacerbation of health inequities. For example, the promise of precision medicine depends on mapping the human genome, but this information is only as good as participant samples and the Genomic Wide Association Studies (GWAS) Diversity Monitor reports that 95% of total GWAS participants from a total of 6263 studies are of European descent. Other descent groups including Asian (3%), African (.2%), African American or Afro-Caribbean (.5%), Hispanic or Latin American (.3%) and other/mixed descent (.7%) are underrepresented both in term of proportion of the world's population and the distribution of poor health outcomes (Mills & Rahal, 2020). The cause of the problem of representation in health research is complex and has implications well beyond genomics research. Indeed, a lack of diversity pervades the research enterprise including when the condition under study is a known health disparity (Boden-Albala et al., 2021; Knepper & McLeod, 2018). Causes include distrust of researchers and health professionals as a consequence of historical abuse and current experiences of discrimination in healthcare settings (Corbie-Smith et al., 2002; Passmore et al., 2020b; Quinn et al., 2013), the presence of bias and systemic racism in science and research (Gilmore-Bykovskyi et al., 2021; Griffith et al., 2020), as well as, a lack of rigor and attention to the study of research inclusion (Gilmore-Bykovskyi et al., 2021; Passmore et al., 2020b).
Our focus on the lack of diversity in research participation to date has been largely trained on issues of distrust (Corbie-Smith et al., 2002; Scharff et al., 2010). While accurate and important, this focus on the problem as located in underrepresented communities has not substantially moved us toward solutions and may have, in fact, compounded the problem somewhat (Passmore et al., 2021). There is evidence that investigators feel that distrust amounts to an insurmountable barrier to diversity in research and, ironically, a justification for not pursuing inclusive practices (Niranjan et al., 2019; Passmore et al., 2021). Recent discussions have accurately reframed the issue as a crisis of untrustworthiness in research and call for a more rigorous explorations (Gilmore-Bykovskyi et al., 2021; Griffith et al., 2020; Wilkins, 2018) and a rethinking of how we can minimize the deleterious effects of “exclusionary research practices” (Gilmore-Bykovskyi et al., 2021).
The difference between a focus on
Authors have argued, more recently, that researchers must overtly re-shape practices to increase accessibility for marginalized potential participants to build trustworthiness (Gilmore-Bykovskyi et al., 2021; Griffith et al., 2020; Wilkins, 2018). In short, they point out that, among other contributing factors, the lack of diversity in all research including those collecting biological specimens, is exacerbated by practices that are convenient for researchers and privilege certain groups of participants over others (Niranjan et al., 2019; Wilkins, 2018). Participants representing historically marginalized groups, in contrast, may require more and different approaches to feel welcome, safe or able to participate in research. Such participants may need materials in a range of languages, more opportunities to ask questions, and greater assurances that they will have access to the benefits of research (Lemke et al., 2012). As health equity requires the participation of more diverse groups of people in research, our job as researchers is to discover the diverse needs, goals and values of the people we hope to study so that we may accommodate them. Indeed, distrust in researchers and science is not so much a barrier as a call to innovation and action.
In this study, our team explored factors that contribute to the decision to participate in genomics research among African Americans adults living in the urban areas of Madison and Milwaukee, Wisconsin. A secondary goal was to test the feasibility of using a new methodology, called the research StoryDeck, to determine the acceptability of study attributes to be used to practically promote diversity in research participation. We asked our participants to assess the acceptability of hypothetical genomic studies that varied on four factors: race of the investigative team, research goal, sponsoring institution and form of consent. Each of these factors is strongly connected to research participation in the literature (Davis et al., 2013; Echeverri et al., 2018; Frierson et al., 2019; Fryer et al., 2015; Guillemin et al., 2018; Levkoff & Sanchez, 2003; Moulton et al., 2020). Our choice of “form of consent” was additionally motivated by previous studies that have indicated that the use of, specifically,
Methods
In order to determine how our factors (race of the investigative team, institution, form of consent, research goal) figured into participant decision making, we used the research StoryDeck, a card sort elicitation technique matched with a factorial survey design used to create research scenarios for participant review (Passmore et al., 2020a; Passmore et al., 2020b). This method produces both quantitative and qualitative data simultaneously to determine patterns in the acceptability of research scenarios as well as the process and meaning driving choices about acceptability. The StoryDeck also provides insight on the interaction of factors revealing the relative “weight” of barriers and facilitators to determine, among other things, how barriers may become acceptable in combination with facilitating attributes. To accommodate remote data collection, a virtual StoryDeck survey was developed and distributed to most interested participants to complete on their own (n = 131). An additional subset (n = 38) was created by offering an administrated version of the survey with a “think aloud” interview (Padilla & Leighton, 2017; Priede & Farrall, 2011) to every fourth interested participant. Adults (18 years of age and older) who self-identified as African Americans living in areas around Madison and Milwaukee were eligible to participate.
Following from Green, a sample size of
The University of Wisconsin – Madison Survey Center developed the infrastructure for a mobile based administration of the survey using “virtual” cards representing a range of research study attributes (form of consent, diversity of the research team, research goal, and institutional affiliation) that was optimized for mobile and other devices. As noted, participants were presented with eight research “opportunities” or scenarios. Each scenario involved a genomics research study (uses DNA to explore genetic connections to disease) that varied on a set of attributes (presented in Table 1). Each participant saw eight scenarios with random assignment of the 4 factors x 3 characteristics. As such, all participants saw individually generated scenarios varying on the constellation of characteristics. However, no participants were presented with the same constellation of characteristics more than once.
Attribute Categories and Characteristics for Research Scenarios.

Scenarios were presented using virtual cards, randomly selected in each category, which could be “flipped” by the participant with a click. Once all the study characteristics of a particular scenario were revealed, participants were able to respond to the item “How likely are you to participate in this study?” (See Figure 1). Participants were also asked to respond to demographic questions including race, ethnicity, income, education, age and gender. Race and ethnicity questions followed the format used in the United States 2020 census (Marks & Jones, 2020).
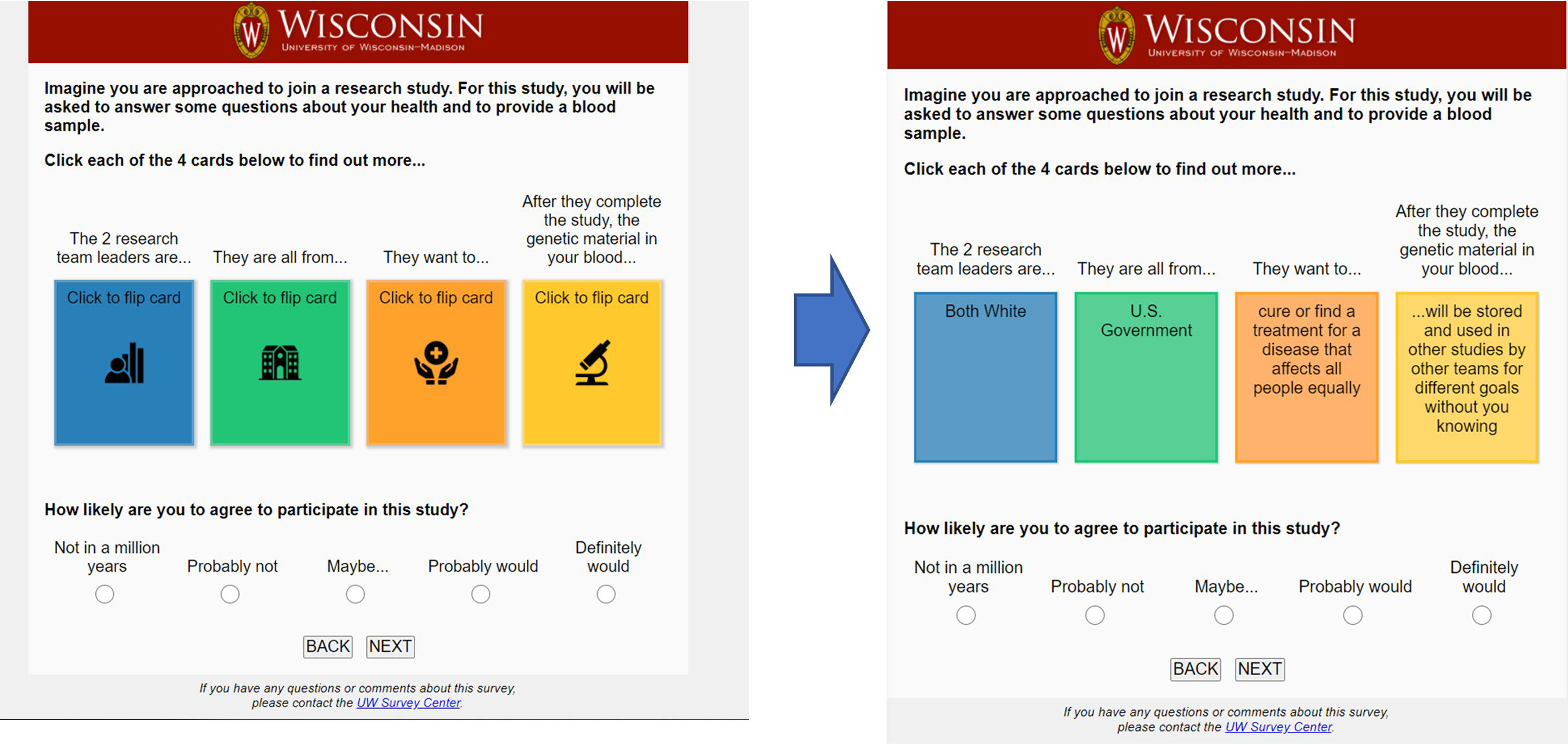
Screen shot of online story deck instrument.
Two groups (n = 18) of African American adults living in Madison and Milwaukee, identified with the help of community partners, reviewed the instrument prior to data collection. Reviewing participants made suggestions on language and format, which were adopted in the final instrument. Pilot testing with these groups revealed an average completion time of seven minutes of the survey alone.
Given public health restrictions at the time, participants were recruited through community outreach using existing social networks, distribution of electronic flyers by community partners, and advertisement in local publications. Those interested contacted the study team who provided individualized links to the online survey or an invitation for every fourth person to participate in a “think aloud” interview via Zoom (Padilla & Leighton, 2017; Priede & Farrall, 2011). In interviews, a team member led participants through the scenarios and asked them to explain their reasoning for their chosen level of likelihood to participate with relevance to each scenario characteristic. Specific questions varied based on the participant's decision and the relevant scenario. For example, if a participant chose that they “definitely would” participate in the relevant scenario, the interviewer first inquired about what led them to that decision. They followed that with probes regarding each factor in the scenario (i.e., “How did the use of study-specific consent influence your decision to participate?”). After participants fully explained their decision relative to each characteristic, they were probed about characteristic interactions (i.e., “Did it matter that that the researchers were White? Would it have been different if they had been African American?”). This process was repeated for each of the eight scenarios. The purpose of the interviews was to reveal what meaning participants ascribed to each characteristic as well as the interaction of characteristics in scenarios. While the survey data revealed discernable patterns in decision making, interviews afforded a view into the significance of characteristics and processes of decision making.
Participants received a $20 incentive for participation in the survey alone and $40 for participating in the “think aloud” interview. Interviews took approximately 20–30 min to complete. Surveys taken alone took an average of 10 min to complete.
Analysis
Quantitative Data
For each of the eight scenarios separately the previously mentioned CART analyses were used within SPSS 28. CART methods recursively partition the entire sample (the parent node) into smaller subgroups (child nodes) based on the outcome variable. At each partition this method identifies the attribute that best separates data into subgroups related to the outcome, repeating the process until the sample is divided into homogenous groups and resulting in a classification tree. The benefit of this strategy, relative to other analytical techniques, is that it automatically yields combinations of attributes that statistically significantly separate the data based on the outcome variable, in this case willingness to participate in the research under given scenarios. For these analyses the minimum parent size was
Qualitative Data
Interviews were audio recorded and professionally transcribed. We took a constructivist approach to the analysis with a focus on the meaning participants ascribed to the various factors in research scenarios as well as their meaningfulness in the decision to participate (or not). Our process was similar to the six phase analytical process of thematic analysis suggested by Braun and Clark (Braun & Clarke, 2021, 2022; Byrne, 2022). Coding was an iterative process following a period of data immersion. Codes and themes were data-driven, although the first pass of coding was done to label participant quotes by study attribute (form of consent, reason for research etc). From there, we used open coding to develop codes that were interpreted directly from participant statements. Two team members played an active role in analysis with one as primary coder. Throughout the coding process, the team discussed interpretation and emergent directions. Following coding, initial themes were interpreted from codes and used to develop a thematic map which was, in turn, used to continue to refine themes that cut across scenario factors. Initial themes were collapsed into larger conceptual categories. For example, concepts such as “shadiness/sketchiness” were collapsed with direct comments about ulterior motives (ex: use of DNA in the criminal justice system) to create the final theme of “hidden agendas.” Similarly, “sharing personal values with the goal of the research” and “supporting the black community” were both covered by the theme of “shared values.” Final interpretations of qualitative themes were accomplished with input from the entire team.
Informed consent was obtained from all subjects participating in this study. All methods were carried out in accordance with relevant guidelines and regulations and study procedures were reviewed and approved by the University of Wisconsin-Madison IRB (2019-1531).
Results
169 individuals participated in the online survey. Of these, 131 completed the survey on their own either on a computer, tablet, or cell phone. The remaining 38 participated in the “think aloud” interviews in which the survey was administered by a research team member. The sample had reasonable gender distribution and a mean age of 38 (range: 18–70). Surprisingly, as all participants identified as African American in the eligibility questionnaire (they would not have gained access to the survey if they had not), only 97% of participants self-identified as African American on the race/ethnicity items. Regarding race, 6 participants selected: Other – “Me” (1); White (1), American Indian (1), and no response (3). While recognizing the limitations of such definitions, we rely on the categories of ethnicity and race as used in the United Census in which Hispanic/Latino self-identification is recognized as an “ethnicity” and is, therefore, separated from “racial” categories (Marks & Jones, 2020). Characteristics of participants in the survey and the think aloud interviews (subset) are presented in Table 2. Participants in interviews were like those in the larger survey sample in most ways but did have somewhat higher incomes and education levels. It is possible that these participants were more likely to accept the invitation to participate in interviews than others.
Sample Characteristics (n = 169).
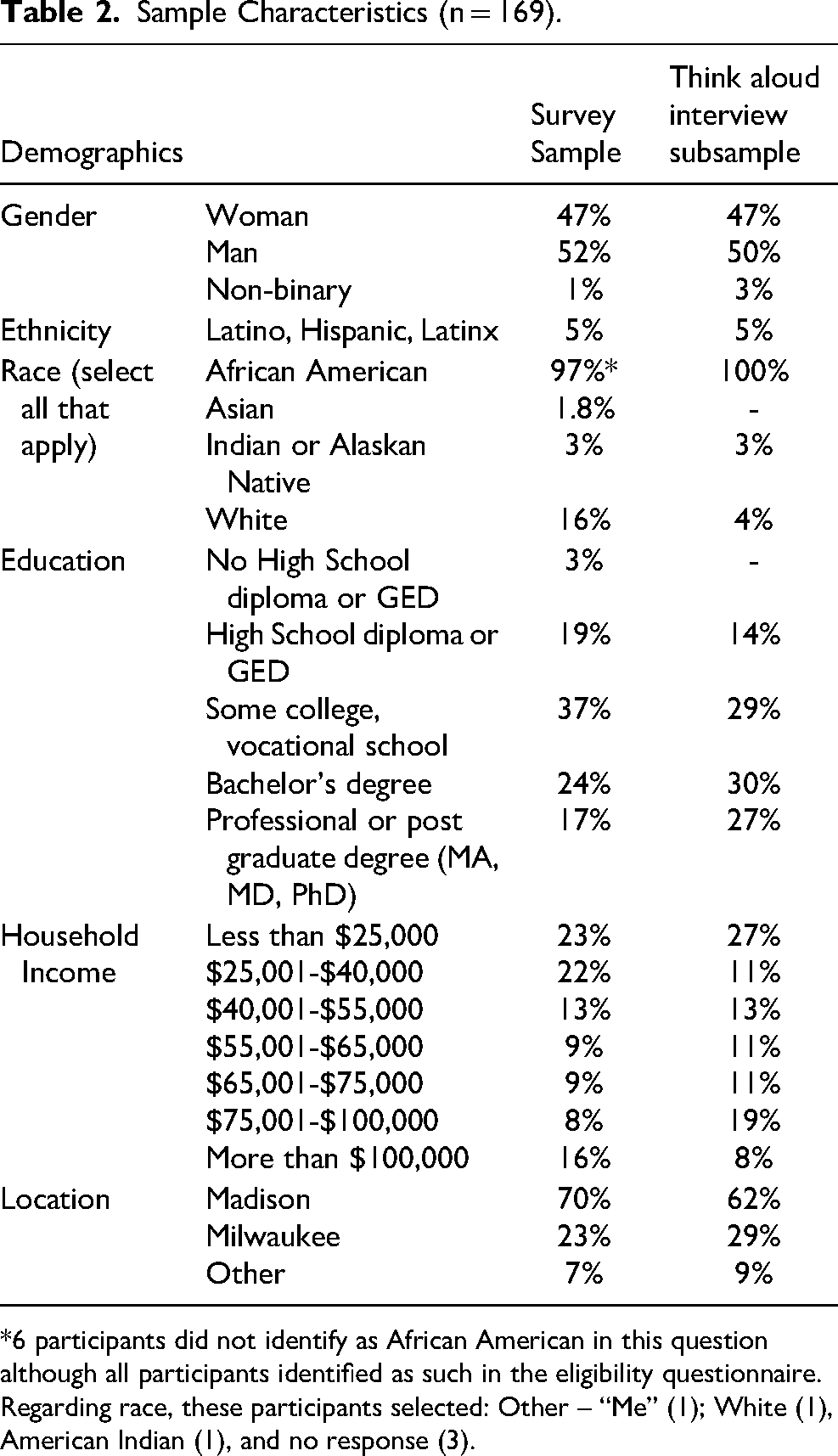
6 participants did not identify as African American in this question although all participants identified as such in the eligibility questionnaire. Regarding race, these participants selected: Other – “Me” (1); White (1), American Indian (1), and no response (3).
Quantitative Results
A slim majority (56%) of scenarios were found to be acceptable to participants (participants reported that they “probably would” or “definitely would” participate). Figure 2 presents a simple summary of quantitative data, percentage of scenarios which participants found acceptable (“probably would or “definitely would” participate) for each characteristic. The most dramatic differences are found regarding form of consent where a tiered form of consent (10.3%) was represented in acceptable scenarios about twice as much as broad consent (5.0%). There were less dramatic preferences for scenarios sponsored by universities rather than the government and those led solely by researchers who identified as African American. CART analysis confirmed that only form of consent was determined to drive decision making.
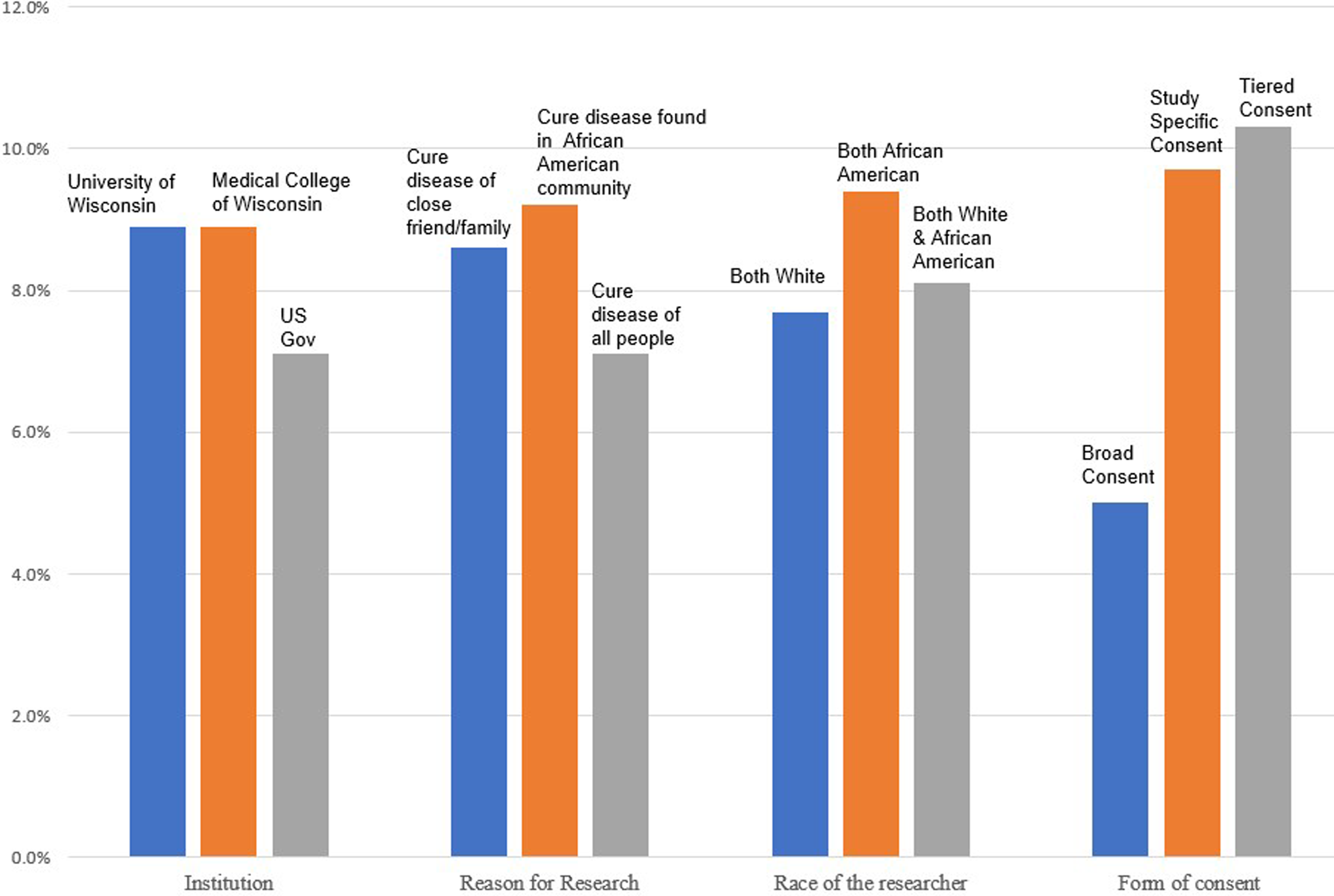
Percentage of acceptable scenarios in which each characteristic appeared (n = 169).
Without presenting the details of each of the eight CART analyses, we may consider the relative importance of each predictor variable summatively across all analyses. In seven of the eight analyses the most predictive variable was form of consent (it was the second most important predictor in the other analysis), making it far and away the most important predictor. Indeed, using a normed importance measure, d = form of consent averaged nearly three times the importance of the other measures (data use consent, 91.7; agency, 36.2; cure, 33.7; researcher race, 32.5). The bifurcation was between a specimen being used without consent vs. being destroyed or used with consent, typically creating subgroups more than a full standard deviation apart. The next most salient predictor in each analysis was evenly divided among the remaining three scenario characteristics. All analyses were repeated including demographic variables and previous research participation as eligible predictors, none of which altered the clear dominance of the consent variable in predicting participation.
Qualitative Results
Participants explained their decisions with reference to six themes we have termed,
Form of Consent: Regarding the different forms of consent, the themes of But the main factor is my blood, you know, my biology being out there, and who knows what's happening with it? Not knowing where my blood is going, where any of my genetic markers are going, that's a little spooky. I would read the small print because, you know, that's my DNA. I mean, is that tracing back to the police department or FBI? I mean, who's overseeing these studies? … Like are you sharing it with third parties? But if I don’t know what else they’re doing with my blood… I don’t want them to just use it, sacrificing my blood to animals or some weird stuff like that. I'd want to know everything that my blood is going to be used for. I don't want to be kept in the dark. That just sounds shady. So then I have And then I don't mind them being used for other studies, as long as somebody is just, you know, gives me a heads up. Hey, we're going to send it over here or over there, because that makes me more Because it says my blood will be destroyed. So once that test is over with, and the research is done, my blood is going to be, you know, destroyed, so it's not going to go nowhere else. So I have more, you know, But, yeah, I got a little more open when I heard it was to cure a disease that affects everybody, and then the samples that I then provide will be destroyed. So that made me feel, like, a bit more secure and stuff.
Institutional Affiliation: Participant preferences for either Medical College of Wisconsin (in Milwaukee) or the University of Wisconsin-Madison were influenced by their level of familiarity with the institution in question. In general, participants in Madison viewed Medical College of Wisconsin as less desirable and less well known. For example, Like, I feel like I know the school more and like trust the school more, yeah. I think that did feel better initially. I like the University of Wisconsin-Madison. I'm more familiar with that than the other places… I don’t trust government, the American government anyway . . . You know, just the past history of doing a lot of, you know, shady, just sort of . . . things. A lot of the policies they’ve passed have affected how black people view medicine research today, so it's just like, and they really haven’t done a lot to change that view. So I just wouldn’t care to be involved in the study. I definitely want something that's going to help African Americans…I don't necessarily believe that white folks, like, that's their priority. … if the results turn out to be something pretty impactful, it would be a good thing to have that credit go to some black scientists, black researchers because, do you know what I mean? So that's why I would definitely go for that one, yeah. But to see somebody of color, you know, I guess I'm culturally competent when it comes to people of color, or black people, so I can relate to them better. I just don't trust all white folks, and I’m just not sure, you know, what their agenda is sometimes. So it makes me feel better knowing that it's two black people. …so it's not just a white person that is on the research team. It's also African American, and they can relate to, you know, the struggles we have as African Americans when it comes to these types of research. And this also brings me back to the current vaccine. Like they came with this vaccine, and they promoted, oh, there's a black scientist who helped create it …But also, are they using that person as a prop? I’m going to say definitely would, and that's because it is very important to cure the diseases of people around me, you know. They do matter more to me... Well, if it's going to like help cure disease that affects African Americans, that's like, you know, impacts my family potentially, be beneficial to people I’m very close to. So that definitely is probably the biggest draw for me, the permission one too again. I like the fact that it's equally for all because I feel like that study might promote future studies to be done that way, to create for the greater good. Yeah, for that personally, I also have, like, a health background, so when I think of things, like, being affected people equally, I almost feel, like, that's just not realistic. Like, I think people are affected different ways. So I think, for me, that was, like, the first card that I was, like, oh, I don’t know. I have to learn a little bit more about this. I honestly don’t trust the U.S. Government. They’ve lied so many times about so many different studies and results…and then with them both being white, that's another, with me being black, that's another red flag for me. And then the other, I would honestly say, is the fact that they claim it would be destroyed. I can’t give you 100% consent that I trust that both of the white U.S. Government would actually destroy my blood after they say they will. So if it's somebody close to me, I definitely will want to participate most likely. So then without me knowing wouldn't be as impactful, using my blood without me knowing, because as long as it's being used for somebody that I know that's close to me, it's not as big a deal, you know.
Discussion
Qualitatively, our participants preferred research opportunities where they had control over the use of their DNA (tiered/dynamic consent; study specific consent), when there was an African American researcher (who might better understand their needs as well share their values), when the research goal was similar to their own goals, and when the affiliated institution had a positive reputation that was familiar to them. Each of these involved concepts that have been described elsewhere as components of trust and trustworthiness (Jamison et al., 2019; Passmore et al., 2020b; Twyman et al., 2008) and have been associated with greater willingness to participate in research in previous studies (Green-Harris et al., 2017; Passmore et al., 2016; Quinn et al., 2013).
Our findings uniquely add quantitative findings that clearly indicate that these attributes were not equally weighted in decision making. While participants ascribed meaning, both positive and negative, to all of the study attributes we offered in scenarios (form of consent, reason for research, institutional affiliation and race of the researcher), only form of consent was shown quantitatively to drive decision making about willingness to participate in research scenarios. In other words, the presence of broad consent in scenarios overrode all other factors in participant decision-making. Further, participants ascribed different meaning to the use of broad consent relative to study-specific or tiered consent. In particular, participants expressed significant concerns about losing control of their DNA which is not surprising given the history of research abuse and marginalization. In contrast, when participants maintained hypothetical control over their genetic material through study specific or tiered consent, participants felt control and, perhaps relatedly, a sense of comfort. Each of these contributed to a strong enough sense of trustworthiness for participants to consider participation in research. The close relationship between tiered and study consent is also evident in Figure 2. It is important to note that overall participants were willing to have their genetic material available for use in other, future studies (i.e., use of a biobank), as long as they were able to make decisions to govern the specific uses.
Currently, broad consent is the more common practice for various reasons. Certainly, it is the most convenient way to maximize the use of data. Other forms of consent are complicated and pose unique challenges. For example, tiered and dynamic forms of consent have been said to be potential threats to participant confidentiality (Petrini, 2010), may lead participants to accept the therapeutic misconception (Steinsbekk et al., 2013), and may place undue pressure on participants by offering too much choice (Ram, 2007). Moreover, there is some evidence that participants prefer the use of broad consent to other forms of consent. However, these assessments seem to be most relevant for those groups already well represented in research (N. A. Garrison et al., 2016; Simon et al., 2011).
Our finding indicate a need for a shift in consent patterns to account for issues of distrust or mistrust found among marginalized communities (Bogart et al., 2020; Corbie-Smith et al., 2002; Rajakumar et al., 2009; Scharff et al., 2010). It is clear that history, culture, politics and other contexts potentially create differences in participant views of science and research. Similar situations have been described with reference to participants recruited into biospecimen research from low- and middle-income countries (Nembaware et al., 2019; Tiffin, 2018; Tindana & de Vries, 2016). In Africa, for example, broad consent may not feasible due to the context of historical research abuse and the continued vulnerabilities (Tiffin, 2018). Moreover, such histories can shift the perception of what it means to participate in research. For example, Barsdorf and Wassenaar have shown that the meaning of “voluntariness” in research participation has been shaped in the specific context of South Africa (Barsdorf & Wassenaar, 2005).
Most importantly, our findings indicate that while “trustworthiness” may be driven by similar components (Jamison et al., 2019; Passmore et al., 2020b; Twyman et al., 2008), their meaning is uniquely connected to historical and current social contexts which must be supported in inclusive research. We cannot conclude on the acceptability of any approach, policy or procedure based on what works with any one segment of the population nor can we determine directions using the rubric of what best “benefits science.” As a community, we should all be sharply aware of the grievous ethical missteps using such a rubric has caused. Rather, our actions should reflect a recognition that science itself is shaped by a historical and current context (Buchanan et al., 2021; Henderson, 2022; Walsdorf et al., 2020) which should cause us to critically consider alternate approaches.
Conclusion
As trust in research and researchers is not randomly distributed in the population, practices such as the use of broad consent may equate to the exclusion of populations already bearing a disproportionate burden of illness in the form of health disparities. Exclusion which, in turn, contradicts our ethical obligation of justice to fairly distribute the benefits and burden of research. Our task to improve the inclusivity of research is to create studies that are welcoming to all (Gilmore-Bykovskyi et al., 2021; Griffith et al., 2020; Wilkins, 2018). Our success depends on our willingness to explore the range of participant perspectives and to build our studies accordingly. Increasing the trustworthiness of research and researchers is an important ultimate goal, albeit one that will require considerable cultural and structural shifts (Griffith et al., 2020; Wilkins, 2018). In the meantime, participant control of data, through limited consent processes, is an important asset in the absence of trust/ trustworthiness of researchers. Protocols that maximize participant choice may ensure that research involving biological samples are created in ways to support, not block, diverse participation and reflect the shift toward a focus on the trustworthiness of science and scientists urged recently across medical science(Gilmore-Bykovskyi et al., 2021; Griffith et al., 2020; Wilkins, 2018).
Study Limitations
While there are several benefits of the use of the StoryDeck to access factors contributing to decision making, there are also drawbacks. Among these is that the exploration is deductively framed by our choice of hypothetical research attributes which may have resulted in the loss of important insights. The reader may be interested in previous uses of the StoryDeck regarding additional factors (Passmore et al., 2023; Passmore et al., 2020b). Overall, we have found that including more factors can quickly led to an unacceptable participant burden. In addition, there also were choices, such as the use of Zoom interviews, necessary due to public health restrictions at the time, but that may have created limitations for this study. It is possible that the use of zoom may have been discouraging to participants unfamiliar with the platform. This could explain why participants in interviews had higher education and income levels than the general survey population.
Best Practices
Those interested in the engagement of study participants from a range of backgrounds should be aware that this goal may require a shift from the traditional ways of doing research. Investigators should be open to the perspectives of all potential participants, especially those underrepresented in research, to ensure that there are no undue barriers placed to participation. Researchers should also be aware that potential participants assign meaning to aspects of studies such as the diversity to be found on research teams and their institutional affiliations. Participants may feel more welcome in studies in familiar settings that are oriented to the needs their needs. Participants also may be more motivated to participate in studies in which they feel that they have a stake in the outcome. Certainly, researchers should strongly consider the use of tiered or dynamic consent in studies using biobanks.
Research Agenda
This study points to interesting questions about how forms of consent may present barriers for research participants. This could be explored further with other populations who are also underrepresented in research. Additionally, “deeper dives” into several for the findings in this study are warranted. For example, through the current study, we have only glimpsed the meaning participants ascribe to the diversity and institutional affiliations research teams and our findings indicate considerable nuance.
Educational Implications
Many investigators are unsure about practical strategies to increase diversity in research participation in their own work. The findings from this study will be incorporated into trainings, workshops and other educational programs for investigators through our
Footnotes
Acknowledgements and Funding
The project was supported by a grant from the National Human Genome Research Institute (R21 MD015130). This work also received support from the University of Wisconsin, Institute for Clinical and Translational Research (NCATS: UL1 TR002373), the School of Nursing as well as the Office of the Vice Chancellor for Research and Graduate Education with funding from the Wisconsin Alumni Research Foundation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Human Genome Research Institute, National Center for Advancing Translational Sciences, (grant number R21HG011503, UL1TR002373).
