Abstract
This study aimed to evaluate children's capacity for informed consent. We translated into Azerbaijani language and adapted the University of California, San Diego Brief Assessment of Capacity to Consent (UBACC). We enrolled four healthy groups: children aged 11, 12, and 13 years and adults. We provided the participants with information about the simulated research proposal and a related informed consent form. Subsequently, they were administered the UBACC. The mean total UBACC scores were 11.9 (11-year-olds), 12.7 (12-year-olds), 14.0 (13-year-olds), and 16.0 (adults). The gradual increase in the mean UBACC scores with age suggests the continuous maturation of the capacity to comprehend the informed consent process. There was no specific cutoff age to decide whether the children were competent enough to provide informed consent.
Keywords
Introduction
The ethical conduct of research involving human subjects depends on an individual's capacity to provide informed consent based on the information provided to the subject or representative. In reviewing research involving children in the United States, the Department of Health & Human Services 45 Code of Federal Regulations (CFR) Part 46 (The Common Rule), Subpart A obligates the relevant Institutional Review Board (IRB) to consider the potential benefits, risks, and discomforts of the research specific to children and assess the justification for their inclusion. Children are considered unable to provide informed consent. The 45 CFR subpart D on additional protections for children involved as subjects in research 46.402(c) defines permission as the agreement of the biological or adoptive parent(s), or guardian authorized under the applicable state or local law, to provide consent for the child's participation in the research. In Azerbaijan, the guidelines for implementing research involving human subjects, including children, require adherence to the Good Clinical Research Practice guidelines of the World Health Organization (WHO) administered by the Board of the Ministry of Health (2020).
In general, informed consent involves four components: the subject's understanding of the relevant information, appreciation of how the information applies to one's own condition, reasoning with the information provided, and evidence of choice regarding participation in the research (Appelbaum & Grisso, 2001; Gupta, 2013; Undurraga et al., 2023). Therefore, the capacity to consent represents an individual's ability to understand the information, reason, and reflect to make decisions. An individual's competence, however, is a legal concept that refers to a person's right to provide an opinion when making an autonomous decision (Michaud et al., 2015; Baltag et al., 2022). Competence is task and context-specific. Competent decision-making is required for valid informed consent and the voluntariness of the participatory process (Grisso & Appelbaum, 1995).
An objective assessment of decision-making capacity among vulnerable and disadvantaged populations such as children and adolescents (aged 10 to 19 years) is required (WHO, 2021). The lack of appropriate assessment may result in the involvement of such subjects, which contradicts the basic ethical principles of voluntariness and comprehension of informed consent. The absence of objective measures of the decision capacity of children and adolescents in transitional ages remains an important barrier to promoting a greater degree of research inclusion, which may also lead to better quality information that can improve their well-being, especially as they stand to benefit directly from the research at hand (Yan & Munir, 2004).
A pediatric research subject's chronological age is currently considered the best parameter for assessing their competence in providing informed consent. Little is known about the maturational capacity of pediatric subjects in the transition to adolescence to independently participate in the informed consent process (Hein et al., 2015a). The age cutoff is often decided arbitrarily, with limited knowledge of children's developmental abilities. Therefore, designing a strict chronological age threshold for informed consent comes at the price of underestimating the inclusion of children below the cutoff who may have already developed this capacity. Arguably, this approach neglects the principle of respect for the autonomy of children capable of providing informed consent (Wendler , 2006). However, asking for assent from children who lack the capacity for informed consent may upset them and may undermine the study outcomes if they are included (Whittle et al., 2004). Indeed, assent entails a child's affirmative agreement (instead of a mere failure to object) to participate in the research. In this regard, the IRB is charged with considering the child's maturity rather than chronological age, with an implicit effort to enable the child's understanding to the extent capable. Arguably, a positive aspect of requesting assent is to make children feel more independent and allow them to learn from participating in research.
Significant variability exists in the mandatory age limits for obtaining informed consent among patients in pediatric clinical trials (PCTs). In the United States, the highest age below which a parent or authorized guardian must provide informed consent on behalf of a child or adolescent is 18 years in 11 states, the lowest being 16 years in 31 states along with the District of Columbia, and 17 years in the rest of the country. The age of consent for PCTs in Europe ranges from 18 years in most countries to 16 years in the United Kingdom, with some countries requiring consent from both parents (excluding those countries that require consent from one parent if the other is not listed on the child's birth certificate, has died, or is younger than 18 years of age). PCTs show considerable variation in the interpretation of the age for assent (Lepola et al., 2016), with several countries not specifying any age. Cayouette et al. (2022) confirm that the most frequently reported criterion used to determine a child's ability to provide assent is chronological age. The reported range for assent varied considerably, from 5 to 13 years, with a minority of studies (5.1%) specifying the cognitive ability of pediatric subjects as a criterion for obtaining assent. Indeed, Whittle et al. (2004) demonstrated that only half of the IRBs have a specified method to determine assent; most of them rely on age cutoffs, and the remainder rely predominantly on investigator judgment. Therefore, the IRB guidelines have great variability that investigators are required to follow in order to obtain assent (Kon, 2006).
When choosing chronological age as a criterion for informed consent from pediatric subjects, the two following types of mistakes should be avoided. The first one involves subjects placed above the age cutoff who are deemed capable of consent but lack capacity, as shown by poor performance in objective measures. Imposing complex decisions on individuals who are unable to make independent choices is highly undesirable. The second one includes subjects placed below the cutoff who are otherwise perfectly capable of making informed choices and desire to participate in decision-making (de Vries et al., 2010). Setting an age limit in such cases indicates that all pediatric subjects younger than the cutoff age are incapable of making a voluntary consensual choice, and their participation is contingent upon the substitute judgment of parents or authorized guardians.
Few empirical studies have investigated children's ability to comprehend the informed consent process. Hein et al. (2015b) studied children's ability to make treatment decisions using the MacArthur Competence Assessment Tool for Treatment (MacCAT-T), a structured instrument that addresses all four relevant criteria. The research findings showed that the mean age of children judged as being non-competent was 9.3 years, with a mean age of 12.1 years deemed competent to provide consent. Children younger than 10.0 years were judged as non-competent with 90% sensitivity, and those older than 11.8 years were judged as competent, with 90% specificity (Hein et al., 2015b). The MacCAT-T is a 21-item structured interview with 4 subscales assessing the main dimensions of decision-making: understanding (13 questions), appreciation (3 questions), reasoning (4 questions), and expressing a choice (1 question). Despite its good psychometric characteristics, this tool requires 15–20 min to be administered and substantial training and experience for valid interpretation, limiting its routine use in clinical research. The University of California, San Diego Brief Assessment of Capacity to Consent (UBACC) is a 10-item and easy-to-use tool that takes 5 min to administer. It was found to have good psychometric properties (internal consistency, interrater reliability, concurrent validity, high sensitivity, and acceptable specificity) among middle-aged and older outpatients with schizophrenia and healthy control subjects participating in research on informed consent (Jeste et al., 2007). These two tools, when applied to the same population, agreed in categorizations as capable/incapable in most cases. When discrepancies were observed, 97 percent of these involved instances in which individuals were categorized as capable by the MacCAT version for Clinical Research (MacCAT-CR) and not categorized by the UBACC as having inadequate responses.
Beyond chronological age, intellectual quotient (IQ) has been noted as the second most important contributing factor explaining variance in children's capacity to consent (Hein et al., 2015a). However, IQ testing is not operable, as it does not reflect a subject's understanding, which is task and context-dependent (Grisso & Appelbaum, 1995). According to Hein et al. (2014), factors such as socioeconomic status, ethnicity, gender, and disease experience do not significantly add to the explained variance in competence judgment, suggesting the generalizability of competence studies in diverse populations. A fundamental question exists regarding the dimensional (continuous) model of competence assessment versus the dichotomous capacity determination. According to Buchanan and Brock (1989), decision-making capacity is a matter of minor differences (gradual model) rather than an all-or-none competence assessment (threshold model). The salience of the dimensional (continuous) model for children and adolescents in transition ages is not surprising, given the continuous growth and emergence of differences inherent with increasing age.
We administered the UBACC to children and adolescents of transition age to evaluate their capacity for informed consent and to learn if there was an age at which a significant change in capacity for informed consent could be observed. The overarching aim of this study was to enhance the principle of inclusive research participation among children transitioning to adolescence. Currently, more than 20,000 children and adolescents in Azerbaijan are not under parental care, and as they age, it is important to enhance their autonomous research participation with potential improvements in their status and welfare. This situation is also applicable to other regions of the world. We used the UBACC as a standardized tool designed to operationalize and screen participants who may lack informed consent capacity and compared scores among children and adolescents at different transitional ages, as well as comparing them with adults. We hypothesized a distinct increase in the UBACC mean scores among children aged 12 years and older compared to 11-year-olds, based on findings in previous research (Hein et al., 2015b). Finally, we studied the performance of individual UBACC items among children and adolescents of different ages and compared them with those of adult subjects.
Methods
Study Instrument
The UBACC is an open-source structured questionnaire consisting of 10 items covering 3 domains of informed consent (Table 1). The tool is designed to be administered by a bachelor's degree–level research assistant. A research assistant can attempt to repeat or rephrase the question in a language appropriate to the individual if the participant seems to be having difficulty understanding the wording of a specific question. Each UBACC item is scored on a scale of 0 to 2 points, with 0 and 2 indicating incapable and capable responses, respectively. An intermediate score of 1 may be used for partially appropriate responses or uncertainty even after additional probing. The total score can range from 0 to 20.
University of California, San Diego Brief Assessment of Capacity to Consent (UBACC) Domains.
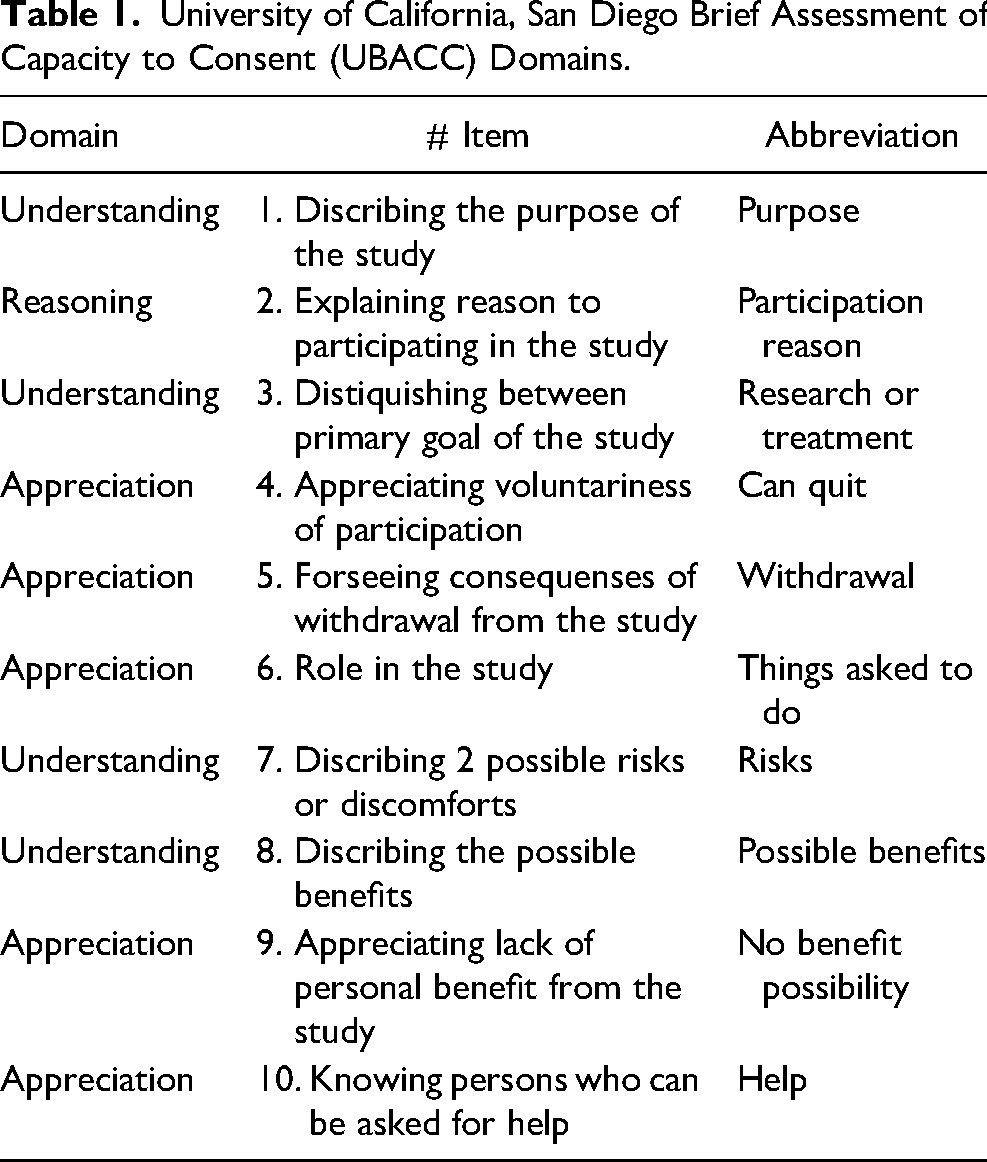
The UBACC was translated into Azerbaijani by a medical professional at the Azerbaijan Medical University with experience in the translation and adaptation of similar tools. Only forward translation was conducted because the items reflected specific situational descriptions. The UBACC does not include medical terms that can lead to ambiguous interpretations. Nevertheless, prior to study initiation, we modified two items because the original items were not related to the proposed simulated research template in the current study and might have caused distress in young participants. Namely, in item #5 we changed “If you withdraw from this study, will you still be able to receive regular treatment?” to “If you withdraw from this study, will it cause any problems?”; subsequently, in item #10, we changed “Who will pay for your medical care if you are injured as a direct result of participating in this study?” to “Who is the person you will ask for help if you have any problem as a direct result of participating in this study?”
Participants
We enrolled 4 groups of healthy subjects: 11-year-olds (n = 31), 12-year-olds (n = 26), and 13-year-olds (n = 23) in transition ages and adults (ages 18–42, mean 24 ± 6 years) (n = 27). We recruited children and adolescents from randomly selected public schools in Baku: 30% from suburban neighborhoods (two schools) and 70% from the city (six schools). Although the study involved a non-random selection of children, the participants came from a broad range of public schools, reflecting the characteristics of children attending public schools. No youth was recruited from private schools (fewer than 5% of the children in Baku attend private schools). We approached the school staff to announce the invitation to participate in the study and requested permission to contact parents willing to agree to participate. We further explained the research and obtained consent from the parents to enroll the children. All children who participated in the study assented to do so.
The prospective adult subjects were consecutively and similarly approached in city venues; of them, 78% were blue-collar workers (sales, restaurant service, and food delivery services), 15% were unemployed, and 7% were white-collar workers (business owners and administrators).
Procedure
Each participant was given a copy of the simulated research proposal and an accompanying consent document prepared by the Office for Human Research Protection, Federal Policy for the Protection of Human Subjects 45 CFR Part 46 (Department of Health and Human Services, 2021) standard informed consent template, which included a description of the following components: i) the purpose of the study, that is, to help understand the relationship between mobile phone usage in schoolchildren and their physical well-being, ii) the voluntariness of their decision to participate, iii) the importance of confidentiality and anonymity of individual responses, iv) anticipated duration and any inconvenience or mild discomfort in completing the questionnaire, and v) potential benefit in helping understand children's participation in studies. We ensured that the readability level of the simulated research proposal and consent form did not exceed the reading skills of eighth-grade students. The participants could access the simulated research proposal and consent form throughout the process; therefore, they did not have to rely solely on their ability to memorize any details.
Each participant was, then, administered the UBACC with adequate time provided to complete the questionnaire, which took an average of 15 min to complete. When a participant seemed to have difficulty understanding the simulated research proposal, informed consent form, or wording of a UBACC question, the research assistant explained that part, repeating or rephrasing the question as necessary to facilitate their understanding in a semi-structured format.
Statistical Analysis
Statistical analyses were performed using SPSS (version 19.0, Chicago, IL, USA). The mean UBACC scores for each group were calculated and compared. We used an independent samples t-test to compare the mean UBACC scores between genders. We performed individual item analysis using a one-way analysis of variance (ANOVA). A cutoff score of 14.5 was proposed as acceptable, and participants who scored higher were considered capable of consent (Jeste et al., 2007). We applied this cutoff score for group comparison.
Results
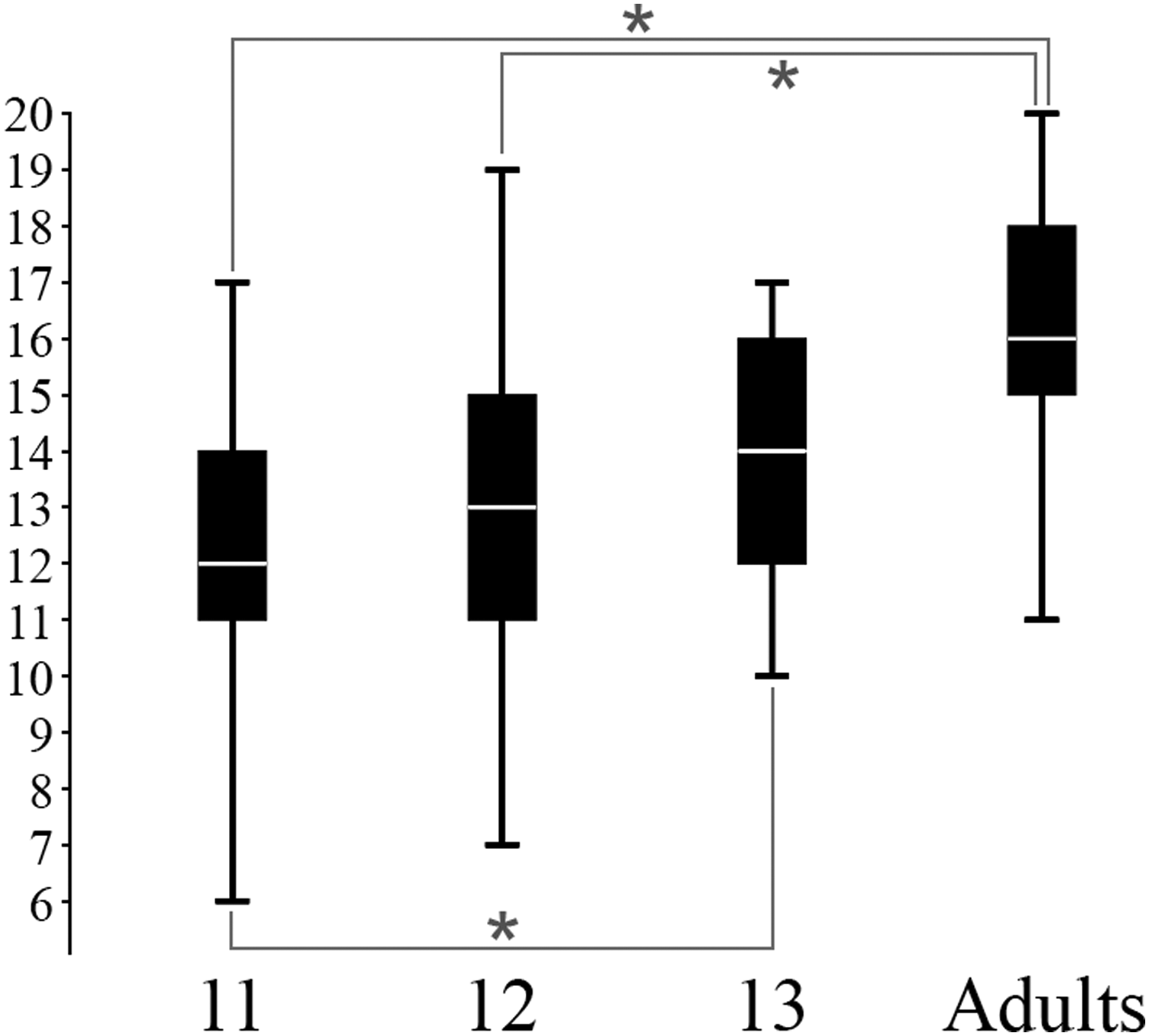
Of all participants, 52 percent were male. The gender difference in the UBACC scores was insignificant. The mean UBACC scores were 11.9, 12.7, 14.0, and 16.0 in 11-year-olds, 12-year-olds, 13-year-olds, and adults, respectively. (Figure 1).
Mean UBACC scores.
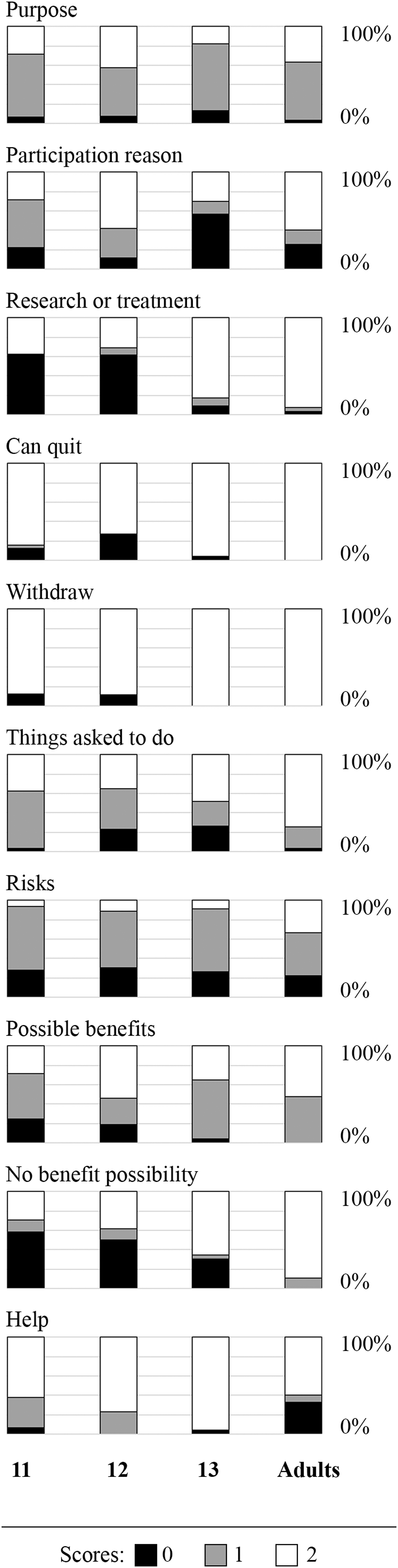
There was a gradual increase in the proportion of participants scoring higher than the cutoff of 14.5 with age: 12.9% of 11-year-olds (4 out of 31), 34.6% of 12-year-olds (9 out of 26), 47.8% of 13-year-olds (11 out of 23), and 74.0% of adults (20 out of 27). Figure 2 shows the scores of the individual UBACC items across the groups.
Individual items across the groups.
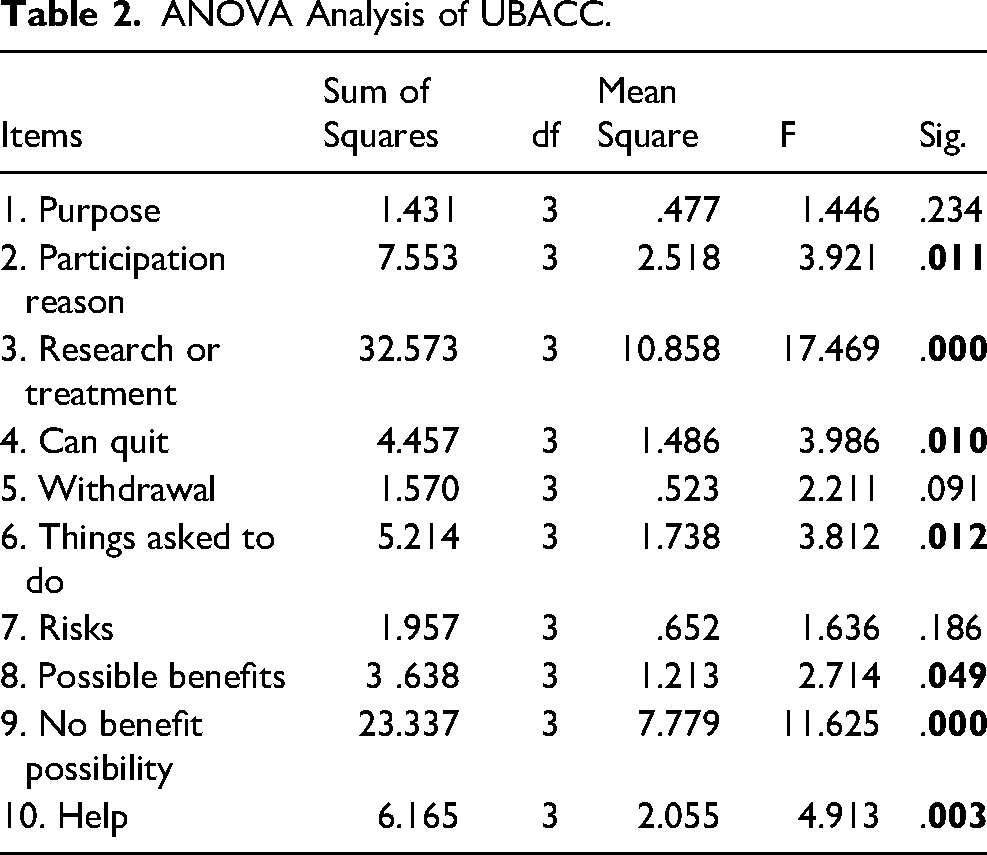
The mean UBACC score of adults was significantly higher than those of the 11- and 12-year-old children (p = 0.000). The mean UBACC score of 13-year-old children was significantly higher than that of 11-year-olds (p = < 0.05). Table 2 shows the one-way ANOVA of the individual UBACC items between groups.
ANOVA Analysis of UBACC.
Discussion
This study provides empirical data on developing the decisional capacity to consent among children transitioning to adolescence in Azerbaijan, one of the most densely populated low- and middle-income countries in the Caucasus region. This study shows that chronological age can be used to differentiate competence in terms of voluntary consensual participation in research; this is an arbitrarily defined threshold for children below the legal age for consent to the treatments or procedures involved. Although the protection of pediatric subjects remains a worthy goal, the threshold rule may be insufficient to do so; however, it is also relevant to ensure investigator and institutional compliance. Nevertheless, this practice may restrict the self-expressive choice of maturing subjects in transition ages to participate in research more meaningfully.
The study showed a gradual increase in the mean UBACC scores with chronological age, making it difficult to assign a specific age threshold to define children's capacity to consent. These findings support the notion that children's capacities gradually develop with age. These findings are consistent with prior research indicating a gradual increase in children's capacity to consent without evidence to support a clear cutoff age. In a study by Hein et al. (2014), competence for informed consent was more probable among children older than age 11.2 years as measured by the MacCAT-T and unlikely among children younger than 9.6 years. Transition-age children 11.8 years and older and those 10.0 years and younger were very likely and unlikely to be considered competent to consent, respectively.
The study findings also support the view that children's ages vary in the continuity of understanding the different foundational domains of informed consent, as reflected in the UBACC scores in the present study (Figure 2). These variations are likely influenced by the different levels of complexity inherent in research protocols, which further makes it difficult to apply an all-or-none phasic rule in determining children's capacity to consent. Individuals in all pediatric age groups, as well as in the adult group, underperformed on the UBACC, suggesting that these subjects may need further capacity assessment, irrespective of chronological age. Interestingly, even some of the youngest participants in the present study performed well, necessitating the respectful acknowledgment of their opinions, although they had not attained the legal age to consent. Nevertheless, in defining children, 45 CFR 46.402(a) does not specify the age of consent, leaving it to state and local laws to pronounce the legal age of adulthood. This gray area requires further research to establish developmentally appropriate pediatric tools and approaches for objectively measuring children's consent capacities.
In this light, the study findings also underscore that not all adults were equally competent, with only 74% scoring higher than the cutoff compared to 47.8% of the 13-year-old children. It is likely that, for these older adolescents, the capacity maturation process has approached adult competency levels. Consistent with this finding, Fisher et al. (2021) showed that 16–17-year-old adolescents were capable of understanding and appreciating consent information at the same level as 18–19-year-olds, with the majority (83%) of 14–15-year-olds demonstrating similar competencies. However, Kreniske et al. (2023) showed that 10–14-year-olds scored significantly lower than 15–17-year-old and 18–19-year-old adolescents on the MacCAT-CR and suggested that adolescents older than 15 years could provide independent informed consent for sexual and reproductive health studies, whereas 10–14-year-olds may benefit from supported decision-making approaches. Interestingly, health literacy and family affluence (including socioeconomic status) were significant predictors of overall capacity, whereas age was no longer associated with capacity after controlling for these factors in the multivariate analyses (McGregor & Ott, 2019).
In the current study, all UBACC items differed significantly between groups (Table 2), except for three, namely Purpose (#1), Withdrawal (#5), and Risks (#7). In the original UBACC study by Jeste et al. (2007), item-wise analysis of adult participants categorized as capable showed that Purpose and Risks were the items missed most frequently. Interestingly, in the current study, the Purpose and Risks items failed to show a significant difference between the groups (Table 2). The understanding of Purpose seemed more difficult than other items because it asked participants to describe the purpose of the research project, which many failed to report correctly (Figure 2). Likewise, the Risks item asks participants to list two risk instances related to the research project, and most participants in all groups named either one risk or none. In contrast, the Withdrawal item was the easiest for all age groups, and only a few children in the younger groups were incorrect (Figure 2).
Most items that showed a clear tendency toward an increase in correct answers with age (Figure 2) may be considered foundational with respect to developing the capacity to consent. Two of these items, Research or treatment (#3) and Withdrawal (#5), showed a steep increase in the correct response rates between ages 12 and 13. In contrast, Things asked to do (#6), Possible benefits (#8), No benefit possibility (#9), and Help (#10) displayed a pattern of more gradual increase with age—with no specific age connoting a rapid phasic increase. Therefore, all these items reflect the gradually evolving maturation process of children's capacity to consent.
Prior research has also shown a high rate of disagreement among experts on competency ratings, as measured by the MacCAT-CR (Hein et al., 2015b). This also renders the assignment of a threshold-based model questionable, even among expert judgments in deciding competence. One can argue that the dimensional continuous model of competence also applies to the adult population. Arguably, difficulties may arise when defining a threshold for measuring any continuous process, such as brain development, physical growth, and development of skills, to fit into predefined categories. A similar trade-off between the sensitivity and specificity of a test occurs when the cutoff value of the test increases or decreases. In terms of research ethics, by increasing the threshold age for capacity, we increase the probability of undermining the principle of respect for the autonomy of those who wish to and can decide for themselves. However, lowering the threshold may complicate the informed consent process by causing undue distress in children.
The dimensional approach, instead of the dichotomous model, is more likely to incorporate the heterogeneous nature of developing children's skills and abilities to assess their competence to consent (Vellinga, 2006). The foundational components of the competence to consent, such as understanding, appreciation, reasoning, and evidence of choice (Applebaum & Grisso, 2001) also each have their own continuous and distinct maturational trajectories, with little empirical data on their interplay and unique growth with increasing age.
The interplay between legal and normative ethical considerations in the inclusion of pediatric subjects in the transition ages to adolescence should emphasized individual autonomy and choice. In the current study, we used the UBACC score to study the differences in performance between groups. According to Jeste et al. (2007), participants who scored at least 14.5 on the UBACC were considered eligible for participation. When we applied this cutoff score to our participants, a clear age-dependent increase was observed in the proportion of participants who scored above it, with a significant number of adults underperforming. Although normative adult subjects are legally considered competent in providing informed consent, questions remain regarding their ability to comprehend certain components of consent. However, in all groups of children, some participants scored higher than the cutoff. This highlights the importance of an individual's ability to comprehend consent, along with the legal consideration of using an age threshold for research participation. The current use of a cutoff age (as opposed to a cutoff score) to include children in research involving human subjects has an underlying legal basis, rather than reflecting their developmental understanding of the consent process.
The results of the current research, therefore, suggest that the knowledge of a child's chronological age is not sufficient as a threshold for consent. We propose a hybrid model that considers age with measures of individual capacity to provide consent within a more inclusive framework for pediatric transition-age groups. In parallel, the application of assent guidelines should be based on prior measures of individual capacity in particular research circumstances. Accordingly, the assent may be respected and waived in the case of children with and without the capacity, respectively, to accede or veto participation, regardless of parental consent. Children deemed capable should have the right to informed consent, as well as affirmative assent, as legally deemed appropriate. For very young children, who incidentally are also unlikely to have the capacity for valid assent (a topic that also requires empirical elucidation), the requirement for assent is developmentally not advisable. For adults, ages 18 and higher, whose consent is legally mandated, the general informed consent permissible guidelines apply with modifications for special categories.
The findings of the current study are relevant for both high-income and resource-poor countries in promoting a more inclusive pediatric research framework. Globally, most children and adolescents of transition age reside in low- and middle-income countries. This study raises several important questions. First, the children deemed incapable of giving consent can still be enrolled in a study if their parents favor participation with their affirmative assent. However, similarly aged children deemed capable of giving informed consent may refuse, raising questions about differential responses that may compromise scientific validity. Second, the issue is further complicated by the varying capacity of pediatric subjects in transitional ages to provide consent based on the complexity of research protocols, for example, in understanding such questions of randomization and double-blinding. The third query is regarding the emerging capacity of children to consent for studies conducted over several years, during which a pediatric subject reaches the legal age of consent. Although the Office for Human Research Protections notes that informed consent is an ongoing process throughout a research project, no specified guidance exists on applying such developmental expectations, and it is left to IRB discourse. A relevant question is, when should the children's emerging capacity for informed consent be re-evaluated if they initially fail to meet a consent “threshold”? Fourth, as the UBACC item-wise analysis suggests, incorrect answers to some items point to differing rates of understanding on some important aspects, such as voluntariness of participation or potential risks; in such situations, further probing of specific items is necessary before the participant can be enrolled. Finally, decisional capacity depends on context (Appelbaum & Roth, 1982). Many environmentally salient factors can influence the capacity to consent beyond the complexity of the research protocol, such as the quality of the consent form and consent procedures and ways of presenting information to participants. These latter factors are highly salient in research involving pediatric subjects. The simulated research proposal used in this study is relatively simple. However, in the case of more complex research protocols, the participants’ performance may be lower.
In summary, the capacity to provide consent has long been used to protect children and adolescents, which may invariably lead to their exclusion from research, especially in vulnerable circumstances (Yan & Munir, 2004). The present results show a growing capacity of pediatric transition-age subjects to understand informed consent, as evidenced by a gradual increase in the UBACC score across age groups. A categorical threshold age model is, therefore, unlikely to optimally differentiate competence for informed consent. The UBACC is a promising tool for screening children's capacity for consent. Future studies should examine the possibility of greater individualization of the informed consent process among pediatric participants in the transition years to adolescence, with greater attention paid to their developmental maturation and greater promotion of their autonomous choices in research participation.
Limitations
This study has several limitations. First, the children were not selected randomly but represented a population comprising those whose parents had consented to participate. This may represent a selection bias and imply a higher level of competence than expected by chance. Nevertheless, all children studied at public schools and did not reflect privileged or merit-based highly selective private school populations. Second, we did not assess outcome measures of competence, such as using an expert-level external review panel or measuring children's levels of intellectual and adaptive functioning. Future studies can benefit from a direct assessment of children's cognitive and adaptive functioning, which, in turn, can be useful for developing integrated measures to further elicit their maturational characteristics. The current study was limited to analyzing differences in children's capacity to understand the consent process based on the normative influence of chronological age.
Best Practices
A clear gradual increase was observed in the UBACC scores with the participants’ age. On the one hand, younger children scored high on the UBACC; on the other hand, some older children underperformed. Best practices regarding informed consent in children include an individual approach for each participant, evaluating their capacity to comprehend the informed consent process. The performance on the UBACC, as with other questionnaires, may be influenced by several circumstantial factors such as the hospital environment, illness-related circumstances, and complexities of proposed research protocols that can individually, or in different combinations, impact an individual's attention, change the motivational level, or increase the level of anxiety. Therefore, the initial low scores on the UBACC of a research study participant may not correctly reflect their full capacity and may be remediated later. Objective tools, such as the UBACC, are useful for evaluating participants’ comprehension levels. Although the age threshold for the assent requirement may be legally determined, it varies from one jurisdiction to another. Some children develop decision-making capacity at an earlier age, whereas others do not until later. Investigators and IRBs should be mindful of the significant proportion of younger children who can understand the informed consent process and express their attitudes toward a study. The current data agree with the recommendation of tailoring research practices to the abilities of specific children, instead of assuming a general threshold for all children (Wendler, 2006).
Research Agenda
Future research should investigate children's capacity to comprehend the informed consent process. Validation of tools for assessing the capacity to consent among children of different ages is paramount. The influence of different mental disorders, including intellectual developmental disorders, on children's comprehension of the informed consent process should also be studied. These steps will lead to the individualization of the informed consent process among children as well as their broader inclusion in research studies—a foundational goal in research ethics. Future studies would greatly benefit from addressing children's capacity from diverse cultural or linguistic backgrounds.
Educational Implications
The results demonstrate that children can comprehend the goals and benefits of the study, as well as the possible risks related to their participation. Researchers must explain the study to younger participants during the informed consent process and consider their attitudes with a broader compass. We would encourage researchers working with pediatric populations to assess the individual decision capacity of a child, especially during the transition ages to adolescence, instead of relying on a chronological cutoff age for informed consent.
Footnotes
Acknowledgments
This work, in part, was supported by Fogarty International Center / National Institutes of Health: International Bioethics Research Postdoctoral Training - C. Asia Network (Award Number D43TW011237, KM Munir, PD/PI) Boston Children's Hospital, Division of Developmental Medicine, Harvard Medical School, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fogarty International Center, (grant number D43TW011237).
