Abstract
This scoping review explores current practices for obtaining consent in research on risky behaviours among adolescents in Canada. The JBI methodology for scoping reviews was used. The database search was conducted in August 2021 and updated in November 2022. Papers published in 2010 or later were included. Extracted data included study characteristics, sample characteristics, and consent procedures. The review included 83 reports covering 57 studies. Nearly 60% of studies relied on adolescent self-consent for participation. Adolescent self-consent was more common than parental/guardian consent for studies using in-person research methods, older adolescent groups, and particularly vulnerable populations. Parental/guardian consent was more common for studies using younger age groups and general population samples. Adolescent self-consent was more common than parental/guardian consent for most risky behaviours covered by this review. These results provide insight into current consent practices in this area and offer guidance to researchers and institutional review boards in Canada.
Keywords
Background
Adolescence is a developmental period that extends from around 10 years of age to 19 years of age (Patton et al., 2018; Sawyer et al., 2018). It is characterized by important physical and cognitive changes and often involves an increase in experimentation with risky behaviours. Risky behaviours are purposive behaviours that have a marked potential for negative consequences for the individual or for others (Boyer & Byrnes, 2018; Leather, 2009; Patton et al., 2018; Sanci et al., 2018). Two common examples are sexual activity and substance use (Hester, 2004; Leather, 2009; Patton et al., 2018; Sanci et al., 2018). Risky behaviours and their associated harms have been the topic of considerable research, particularly with respect to adolescents. Typically, the aim of such research is to identify factors associated with risky behaviours to inform prevention and intervention strategies (Patton et al., 2018; Sanci et al., 2018). Therefore, it is often prudent to recruit adolescents themselves to participate in this research.
Individuals who conduct research with adolescents often seek informed consent for participation from the adolescents’ parents or guardians. Information about the study is provided to the parents/guardians, who then give either active or passive consent if they are willing to let their adolescents participate (Liu et al., 2017; Scott, 2013). With active consent, parents/guardians sign a consent form explicitly permitting their dependents to participate. With passive consent, the study information is accompanied by a statement that consent will be implied if parents/guardians do not contact the researcher to exclude their dependents from the study (Liu et al., 2017; Scott, 2013). In addition to parental/guardian consent, adolescents must provide either consent or assent to participate in the research (Liu et al., 2017; Scott, 2013). Assent denotes an agreement to participate in the research but is considered less formal than consent. It is typically used in contexts where the individual might not have the capacity to fully understand the research or its implications (Liu et al., 2017; Scott, 2013).
Individuals conducting research with adolescents may aim to get informed consent from the adolescents without seeking consent from their parents/guardians (Balen et al., 2006; Coyne, 2010; Pickles, 2020). This practice, which is referred to as adolescent self-consent, is supported by evidence suggesting that adolescents have the capacity to make informed decisions about research participation by the age of 14 or 15 (Coyne, 2010; Hester, 2004; Ruiz-Canela et al., 2013; Scott, 2013). Obtaining consent directly from the adolescents has potential advantages from a research perspective. Researchers have noted that some adolescents may be excluded from research participation if their parents/guardians refuse to consent or if they do not feel comfortable asking their parents/guardians to consent, particularly for studies examining risky behaviours and sensitive topics (Hester, 2004; Liu et al., 2017; Pickles, 2020; Ruiz-Canela et al., 2013; Scott, 2013). By obtaining consent from adolescents themselves, researchers can increase the likelihood that their sample is representative of the target population and less likely to be biased.
In Canada, guidelines surrounding adolescent consent are inconsistent or unclear. The Tri-Council Policy Statement on Ethical Conduct for Research Involving Humans (TCPS 2; Canadian Institutes of Health Research et al., 2018) considers individuals under the age of majority (i.e., 18 or 19; Government of Canada, 2013) to be a vulnerable participant group that may or may not require a substitute decision maker to consent to research on their behalf. The TCPS 2 also states that informed consent procedures for individuals under the age of majority should be based on decision-making capacity, rather than age (Canadian Institutes of Health Research et al., 2018). Elsewhere, Health Canada and the Public Health Agency of Canada suggest that youth can consent to minimal risk studies at 16 years of age (Government of Canada, 2023). However, they also note that decisions concerning adolescent consent are often made on a case-by-case basis and are the responsibility of individual institutional review boards (Flicker & Guta, 2008; Lind et al., 2003). Although procuring a waiver of parental/guardian consent through institutional review boards is possible, the process is often onerous and can present substantial barriers for researchers (Coyne, 2010; Hester, 2004). It is currently unclear how often these waivers occur.
It would be useful to know more about how researchers are obtaining consent for research on risky behaviours among adolescents in Canada. Such information would provide insight into commonly accepted procedures for obtaining consent in this research area and may help to establish guidelines for best practices. A preliminary search of PROSPERO, the Cochrane Database of Systematic Reviews, and JBI Evidence Synthesis was conducted in January 2021 and no current or ongoing systematic reviews on this specific topic were identified. Two reviews of limited relevance were uncovered. One review considered consent and assent in research involving children and adolescents, but without a focus on risky behaviours specifically (Huang et al., 2016). The second review compared the influence of active and passive parental consent on outcomes such as response rates and sampling bias in risky behaviours research, although it provided little information on the consent procedures themselves (Liu et al., 2017). More generally, neither review focused on the Canadian context, and both reviews included older studies with practices that may be out of date. The present review contributes to the literature in this area by mapping current procedures for obtaining consent for research on risky behaviours among adolescents, with a specific focus on adolescents in Canada.
Review Question
What are the current procedures for obtaining informed consent in research on risky behaviours among adolescents in Canada?
Inclusion Criteria
Participants
This review focused on research studies that involve the participation of adolescents aged 14 to 19 years in Canada. The lower age of 14 was selected as research suggests that adolescents develop the capacity to make informed consent decisions by the age of 14 or 15 years (Coyne, 2010; Hester, 2004; Ruiz-Canela et al., 2013; Scott, 2013). The upper age of 19 was selected as the age of majority (i.e., legally considered an adult) in Canada is 18 or 19, depending on the province or territory (Government of Canada, 2013). It should be noted that this review included studies with participants younger than 14 and older than 19, provided the studies also captured the targeted age group.
Concept
This review addressed informed consent practices in research on risky behaviours among adolescents. We defined informed consent as the process through which a researcher gains permission for an individual to take part in a research study (Canadian Institutes of Health Research et al., 2018). We considered both active and passive parental/guardian consent, as well as adolescent assent and consent. Risky behaviours were defined as intentional behaviours that involve significant potential for negative consequences for the individual or for others (Boyer & Byrnes, 2018; Leather, 2009). Examples of risky behaviours include sexual activity, alcohol use, drug use, smoking, and self-harm (Killianova, 2020).
Context
This scoping review included research conducted within Canada, in community settings, primary care settings, and virtual environments. We excluded research studies that recruited participants from within primary or secondary school systems, as school boards commonly have well-established criteria and procedures for obtaining informed consent that differ from other institutions. For example, most Canadian school boards always require active parental/guardian consent for research involving any students under the age of majority (e.g., Government of Nova Scotia, 2019; Ottawa-Carleton Research and Evaluation Advisory Committee, 2023; Toronto District School Board, 2023; Vancouver School Board, 2023).We also excluded studies that recruited youth who were enrolled or enrolling in a treatment program (e.g., rehabilitative care), as their consent for research may have been tied to, or otherwise affected by, the criteria surrounding their consent for the medical intervention. However, we included studies that recruited youth from a clinical setting during routine care (e.g., primary care appointments).
Types of Sources
This review included peer-reviewed published research papers using any type of study design, including qualitative studies, quantitative studies, and mixed methods studies. This review excluded conference papers and proceedings, as well as review papers.
Methods
This review was conducted in accordance with the JBI methodology for scoping reviews (Peters et al., 2020). The protocol for the review was registered with Open Science Framework after the start of the review process, but before the data analysis was performed (MacNeill & MacNeill, 2023).
Search Strategy
The aim of the search strategy was to locate published research articles assessing risky behaviours among adolescents in Canada. An initial limited search was conducted in MEDLINE (Ovid) to identify articles on this topic. The text words contained in the titles and abstracts of these articles, as well as the index terms used to describe the articles, were used to develop a full search strategy. The search strategy was externally peer reviewed by a second librarian using the Peer Review of Electronic Search Strategies (PRESS) guidelines (McGowan et al., 2016). The search strategy, including all identified text words and index terms, was adapted for each database. Note that a search filter was used as part of the search to retrieve studies related to Canada, Canadian provinces, and the 100 largest Canadian centres (Campbell, 2020). Key terms relating to informed consent procedures (e.g., consent, assent, permission, ethics, research) were not included in the search strategy, as a preliminary search showed that many articles qualifying for this review would not be captured with these terms included. The main database search was undertaken on August 4, 2021, and an updated search was run on November 9, 2022. The full search strategy is provided in Appendix I.
The databases searched included Medline (Ovid), EMBASE (Elsevier), CINAHL with Full-Text (EBSCO), and PsycINFO (EBSCO). In addition to these databases, Google Scholar was searched using the key search terms: (adolescent OR teen OR youth) AND (“risky behaviour” OR “substance use” OR smoking OR vaping OR cannabis OR alcohol OR drinking OR sex OR pregnancy OR “STI” OR “HIV”) AND (Canada OR Canadian). Google Scholar orders listings by their relevance to the search topic; therefore, only the first 200 results were screened for this review, which is in line with other recent reviews (Bramer et al., 2017; Haddaway et al., 2015). Citation searching was performed by hand-searching the reference lists of studies included in the review.
Only full-text articles published in French and English, the official languages of Canada, were included for review. The main database search was limited to papers published in or after 2010, whereas the updated search was limited to papers published since the main database search. The review only considered papers published in or after the year 2010 because this year was when the TCPS 2 was first published in Canada. Papers published since that time reflect current ethical practices for research involving minors. Longitudinal studies that began before 2010 were excluded, unless there was evidence that ethics approval was reobtained after 2010.
Source of Evidence Selection
Following the database searches, all identified records were uploaded into EndNote v.X9 (Clarivate Analytics, PA, USA) and duplicate records were removed. The remaining records were imported into Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia). Duplicate records missed by EndNote were removed. Two independent reviewers screened each title and abstract against the established inclusion criteria. Full texts were retrieved for any studies that were included at this stage, and two independent reviewers screened each full text against the inclusion criteria. Full texts were retrieved for any studies identified during citation searching and were screened according to the same process. Disagreements that arose between the reviewers at any stage of the selection process were resolved through discussion or with a third reviewer.
Data Extraction
Two independent reviewers extracted data from the included studies using a data extraction tool developed for this review (see Appendix II). Extracted data included: (1) study characteristics, including author name(s), year of publication, study design, mode(s) of recruitment, mode(s) of data collection, and risky behaviour(s) assessed; (2) sample characteristics, including age, gender, and study population; and (3) informed consent procedures, including whether consent was obtained from a parent/guardian (yes/no), type of informed consent from the parent/guardian (active/passive); mode of informed consent from the parent/guardian (written/verbal), whether assent was obtained from the adolescent participant (yes/no), mode of assent from the adolescent participant (written/verbal), whether informed consent was obtained from the adolescent participant (yes/no), and mode of informed consent from the adolescent participant (written/verbal). Note that electronic consent (e.g., clicking a consent button in an online survey) was considered a written form of consent for the purposes of the review. Any data extraction disagreements that arose between the reviewers were resolved through discussion or with a third reviewer. Where required, attempts were made to contact study authors to acquire missing or additional data.
Data Analysis and Presentation
Data on study characteristics, sample characteristics, and informed consent procedures were summarized using descriptive statistics (e.g., frequency counts). The results are presented in a narrative summary that aligns with the objective of the review. The narrative summary is supplemented by summary tables in the text, as well as longer tables containing the full results in Appendix III.
Results
Search Results
Database searches produced 14,941 records, 3,591 of which were removed as duplicates. Titles and abstracts for 11,350 records were screened for eligibility and 9,954 of those were excluded. Full text reports of 1,396 records from the database searches were sought for retrieval. In addition, full text reports were sought for 49 records identified from citation searching, two of which could not be retrieved. Altogether, 1,443 full text reports were assessed for inclusion across database and citation searching, 1,360 of which were excluded. Reasons for exclusion are detailed in Appendix IV. The most common reasons for exclusion were that the research was conducted outside of Canada (n = 453) and that consent and data were collected within a primary or secondary school system (n = 319). A total of 83 reports were included for data extraction. Screening results are presented in a Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) flow diagram (Page et al., 2021; see Figure 1).
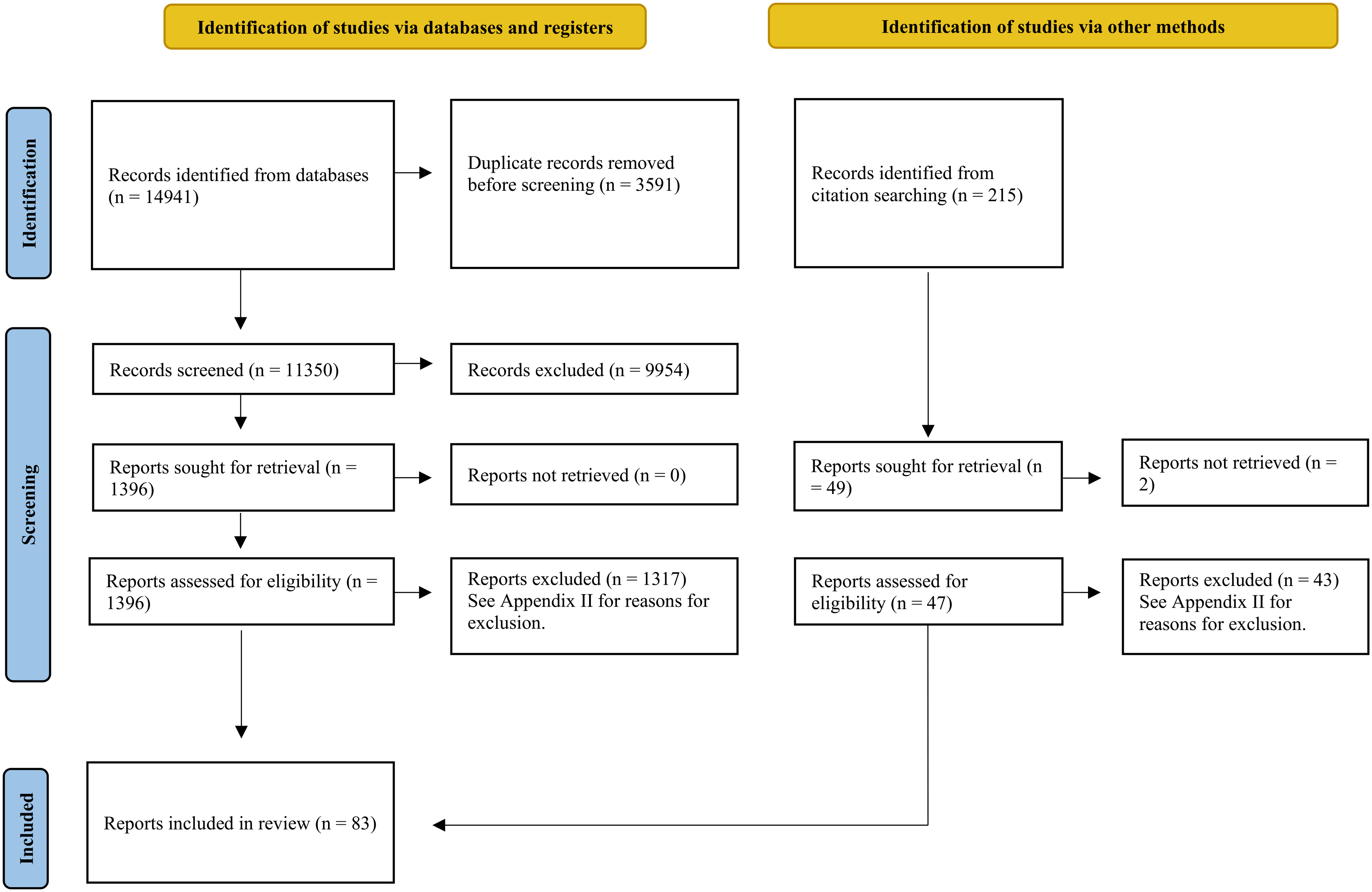
Search results and screening process (Page et al., 2021).
Review Findings
Characteristics of included studies
This scoping review included 83 reports published between 2011 and 2021. See Appendix V for a reference list of the included reports. All reports were peer-reviewed journal articles. The 83 reports corresponded to 57 distinct studies (there were instances where multiple articles described the same study). Most of these studies were observational studies (n = 52) and the remaining studies were interventional studies (n = 5). Some studies recruited participants from across Canada (n = 12), whereas others recruited specifically from Ontario (n = 23), British Columbia (n = 9), Quebec (n = 7), Alberta (n = 4), New Brunswick (n = 4), Manitoba (n = 2), Nova Scotia (n = 2), the Northwest Territories (n = 1), Prince Edward Island (n = 1), and Saskatchewan (n = 1). The most common forms of recruitment were advertisements in the community (n = 12), targeted emails (n = 12), advertising through community organizations (n = 11), respondent driven sampling/snowball sampling (n = 10), and online consumer panels (n = 10). The most common mode of data collection was remote online surveys (n = 21), followed by in-person interviews (n = 12) and in-person paper surveys (n = 10). The risky behaviours most commonly assessed were alcohol use (n = 22), cannabis use (n = 19), and sexual activity (n = 16). It should be noted that several studies used multiple modes of recruitment and data collection and measured more than one risky behaviour. See Appendix III for more details on the study characteristics.
Characteristics of study samples
The lower age limit of participants varied across studies. Twelve studies included participants under the age of 14, four studies focused on participants aged 14 years and older, five studies focused on participants 15 years and older, 26 studies focused on participants 16 years and older, and three studies focused on participants 17 years and older. Most studies recruited participants of any gender, although four studies recruited only women and three studies recruited only men. Nineteen studies recruited participants from the general population, whereas 12 studies recruited from ethnic and sexual minority groups, 11 studies recruited substance users, eight studies recruited street-involved or homeless individuals, two studies recruited participants with specific medical conditions, two studies recruited participants who were pregnant, one study recruited females from a rural setting, one study recruited twins, and one study recruited undergraduate students. See Appendix III for more details on the sample characteristics.
Informed consent procedures
Of the 57 studies 1–83 covered by this review, 34 studies 2–7, 9,10–25,27,29–31,33, 35,36,38,39,41,43,44–50,52–56, 60,62,65,67,70,72− 76,78–80,83 used adolescent self-consent for participation. By comparison, 22 studies 1,8,26,28,32,34,37,40,43,51,57–59, 63,64, 66,68−,71,77,81,82 required parental/guardian consent. Among the studies that required parental/guardian consent, 18 studies 1,26,28,32,37,42,58,63,64,66,68–71,77,81,82 used active consent procedures, one study 8 used passive consent procedures, and three studies 34,40,57,59 did not specify whether consent was active or passive. Nine studies requiring parental/guardian consent also obtained adolescent consent 1,8,42,51,57–59,71,77,82 , five studies obtained adolescent assent 26, 32,37,63,70 , six studies obtained either adolescent assent or consent (depending on age) 28,64,66,68,69,81 , and two studies did not specify whether assent or consent was obtained from adolescents 34,61 . Written consent was the most common mode of consent for parental/guardian consent 1,26,28,37,51,63,64,66,68,70,81 , adolescent assent 26,28,37,63,64,66,68–70,81 , and adolescent consent 1–4,6,8–16–19,21–23,25,27,28,30,31,35,36,38,39,41,47,48,50,51,53–55,60,62,64–70,72,73–76,80,81,83 . See Appendix III for more details on informed consent procedures.
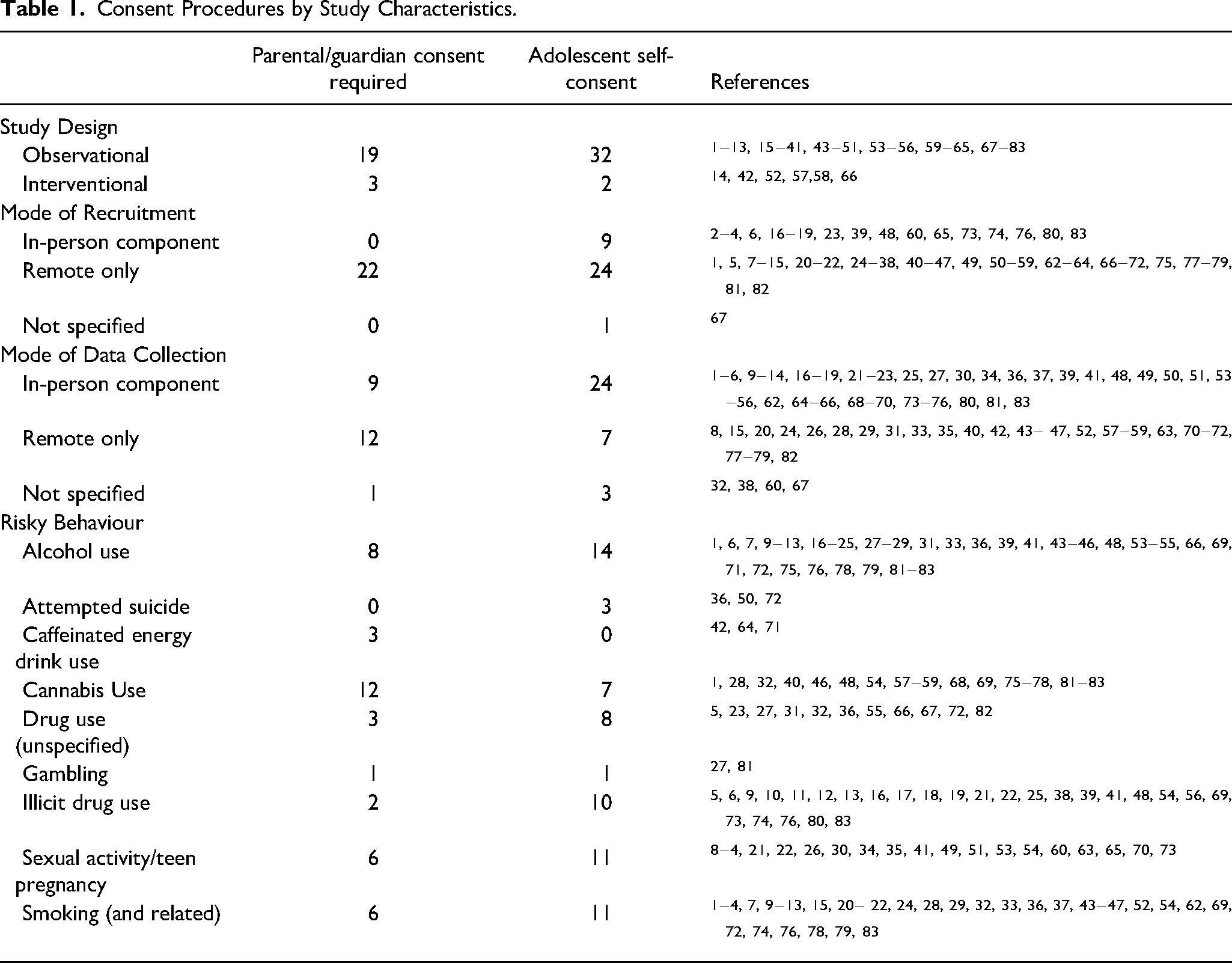
Results indicated that consent procedures varied by study characteristics (Table 1). For instance, adolescent self-consent was more common than parental/guardian consent for observational studies, whereas the few interventional studies were more likely to use parental/guardian consent than adolescent self-consent. For mode of recruitment and mode of data collection, we distinguished between studies that contained an in-person component and studies that were remote only, based on the notion that studies with in-person components could present more perceived privacy and confidentiality risks to participants and therefore may have consent procedures that favor participant discretion. Consistent with this idea, adolescent self-consent was more common than parental/guardian consent for studies that used in-person recruitment or data collection, whereas consent procedures were more mixed among studies that only used remote recruitment or data collection. Finally, consent practices varied by risky behaviour. Adolescent self-consent was more common than parental/guardian consent for most of the risky behaviours covered by this review, but there were a few exceptions (e.g., caffeinated energy drink use, cannabis use).
Consent Procedures by Study Characteristics.
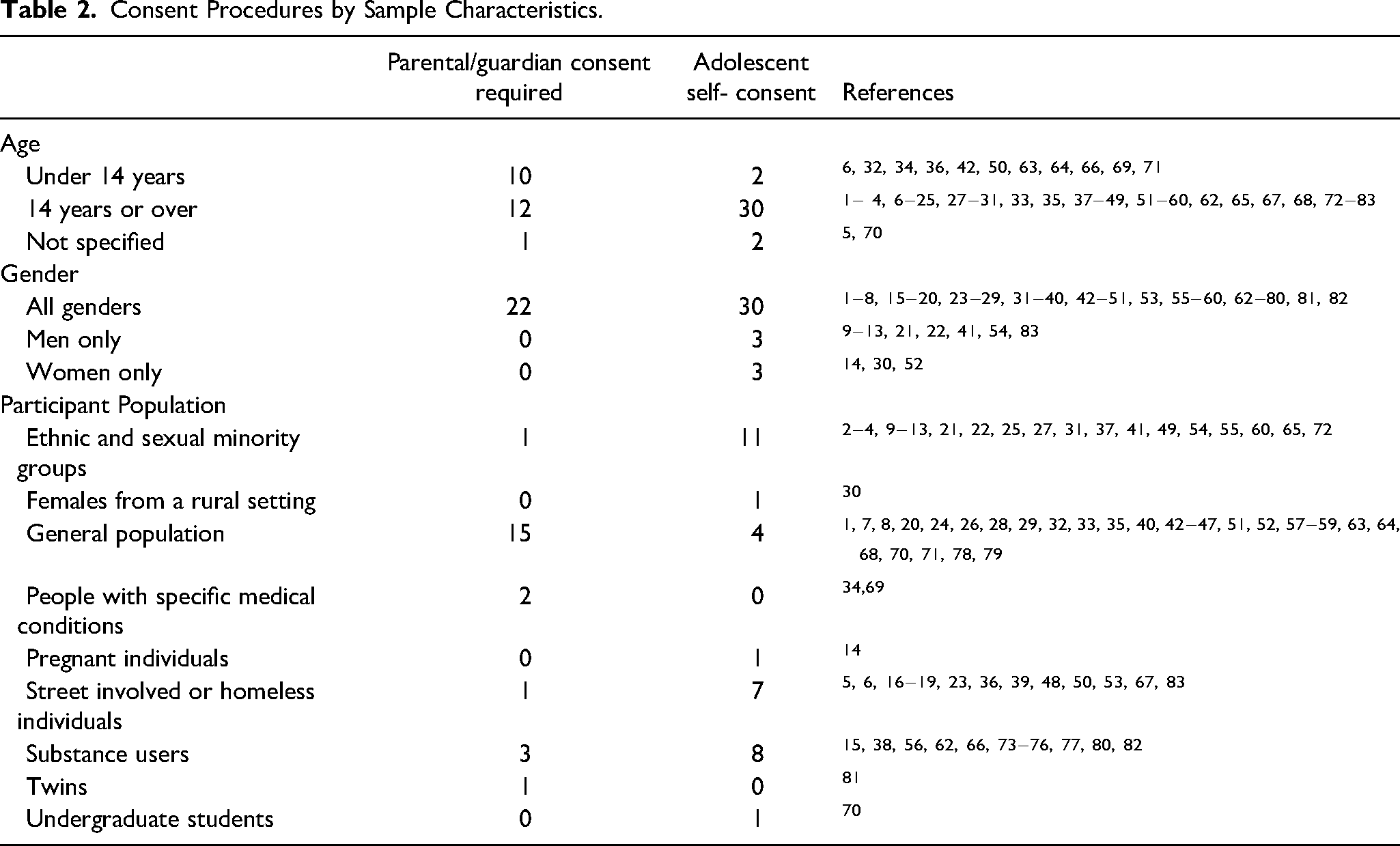
Results also indicated that consent procedures varied by sample characteristics (Table 2). In terms of age, we distinguished between studies that only included individuals aged 14 and older from those that also included individuals under 14, based on the idea that the latter studies would have more stringent parental/guardian requirements. Indeed, studies that only included individuals aged 14 and older were more likely to use adolescent self-consent than parental/guardian consent, whereas parental/guardian consent was more common if the study also included individuals under 14. In terms of gender, there did not appear to be any clear consent patterns based on studies’ gender inclusion criteria. There did seem to be some differences with participant populations, however. Studies that recruited from the general population were more likely to use parental/guardian consent than adolescent self-consent. By comparison, the reverse pattern was seen for studies that recruited from populations with an elevated risk of vulnerability, such as ethnic and sexual minority groups, street-involved or homeless individuals, and substance users.
Consent Procedures by Sample Characteristics.
It should be noted that, of the 57 studies covered by this review, we had to reach out to the corresponding authors for 45 of the studies in an attempt to obtain more information. This finding indicates that, in general, consent procedures are not well reported in the literature.
Discussion
The aim of this scoping review was to map current procedures for obtaining informed consent in research on risky behaviours among adolescents in Canada. This review included 83 reports covering 57 studies that assessed a variety of risky behaviours across a range of settings. Nearly 60% of studies relied on adolescent self-consent for participation, suggesting that many researchers studying risky behaviours with adolescents consider their participants to be capable of providing consent for research participation (Hildebrand et al., 2015; Liu et al., 2017; Sims & Nolen, 2021). The use of adolescent consent was also common in studies requiring parental/guardian consent, although sometimes it was paired with the use of assent for younger adolescents. Researchers favored active versus passive consent practices when acquiring consent from parents or guardians; in other words, they ensured that they secured explicit permission from parents or guardians before proceeding with the study. In terms of mode of consent, written consent was used almost exclusively among the studies included in the review.
The consent procedures described in the included studies varied based on certain study characteristics. For instance, studies that included in-person recruitment and data collection were less likely to require parental/guardian consent than studies using remote-only methods. There could be a few reasons for this finding. It is possible that in-person research methods are simply more common in research areas where parental/guardian consent is not possible (e.g., studies of homeless youth). There could also be situations where parental/guardian consent is possible but not ideal for privacy or confidentiality reasons. If an in-person research study has a parental/guardian consent requirement, then the parent or guardian would likely be in close proximity to their dependent during recruitment or data collection. This proximity could deter adolescents from participating due to concerns about privacy and confidentiality (Fourie et al., 2020; Hildebrand et al., 2015; Liu et al., 2017; Ruiz-Canela et al., 2013; Scott, 2013; Sims & Nolen, 2021). Researchers may seek adolescent self-consent in these situations so that adolescents have some assurance that their parents/guardians will not learn about their participation and any risky behaviours that they might have engaged in.
Concerns over privacy and confidentiality may account for other findings as well. For instance, adolescent self-consent was more common than parental/guardian consent for most of the risky behaviours covered in this review, including alcohol use, drug use, and sexual activity. Many of these behaviours are illegal for minors in Canada, although specific laws vary by jurisdiction. Adolescents who are concerned that parental/guardian consent requirements will disclose their involvement in risky and potentially illegal behaviours may decline to participate in this type of research (Fourie et al., 2020), which would lead to lower response rates and systematic bias in study results (Hildebrand et al., 2015; Liu et al., 2017; Ruiz-Canela et al., 2013; Scott, 2013; Sims & Nolen, 2021). Researchers may be seeking adolescent self-consent to avoid these issues. Notably, parental/guardian consent was more common in studies assessing caffeinated energy drink consumption and cannabis use. Caffeinated energy drinks are widely accessible to children and young adolescents (Goldman, 2013; Ruiz & Scherr, 2018), and researchers studying this topic may want to include these younger age groups in their studies; the inclusion of these particularly young age groups would necessitate parental/guardian consent for the study. It is less clear why research on cannabis use often seeks consent from a parent or guardian. Many of the cannabis studies included in this review used remote research methods, which were more likely to involve parental/guardian consent than in-person research methods. Perhaps the frequency of parental/guardian consent in cannabis studies reflects methodological factors more than the risky behaviour itself.
The consent procedures described in the included studies also seemed to vary based on certain sample characteristics. For instance, adolescent self-consent was more common in studies involving older adolescents. In the past, some researchers have expressed the view that youth have the capacity for making informed decisions about research participation by age 14 or 15 (Hester, 2004; Nelson et al., 2016; Scott, 2013). Furthermore, some research indicates that by age 15 adolescents are capable of understanding the purpose of a research study, how research procedures will affect them, and how to balance risks and benefits of research participation (Nelson et al., 2016). The fact that the studies included in this review tended to adopt the age of 14 to 16 as a cutoff for acquiring adolescent self-consent suggests that many researchers ascribe to this perspective. When researchers include participants below this age cutoff, they are more likely to apply a broader or more general requirement for parental/guardian consent or have different consent procedures for different age groups. For example, some of the studies using parental/guardian consent in this review sought consent from older adolescents and assent from younger participants.
Consent procedures also varied according to the recruited populations. Studies involving adolescents from the general population commonly required parental/guardian consent, whereas those recruiting from more vulnerable adolescent populations often omitted this requirement. It may seem counterintuitive that self-consent was more common among studies that recruited from particularly vulnerable adolescent populations; however, this form of consent can be a useful and effective method for engaging these populations, which are otherwise challenging to access (Pickles, 2020; Sims & Nolen, 2021). For instance, researchers have argued that parental/guardian consent requirements present potential harm for LGBTQ+ adolescents when their parents or guardians hold prejudicial attitudes, such that these adolescents may decline to participate in research with these consent requirements (Pickles, 2020; Sims & Nolen, 2021). Adolescent self-consent offers a safe and confidential means by which these individuals can participate in a research study. Parental/guardian consent requirements also present unique barriers for recruiting street-involved or homeless youth, as these youth often do not have direct contact with their parents or guardians (Scott, 2013). In these studies, adolescent self-consent is the only practical method for acquiring consent.
Guidelines for Consent Procedures
Although adolescent self-consent appears to be common in research on risky behaviours in adolescents, ethics policies surrounding adolescent consent practices remain somewhat unclear. For example, the TCPS 2 states that consent procedures should be based on decision making capacity rather than a specific age requirement; however, no definition or subjective measure of decision-making capacity is provided (Canadian Institutes of Health Research et al., 2018). Given these ambiguous guidelines, decisions concerning adolescent consent are often made on a case-by-case basis and the onus usually falls on individual institutional review boards to make these decisions (Government of Canada, 2023). This situation can give rise to challenges, as some researchers are reluctant to involve adolescents in their studies due to the often-challenging process of securing institutional review board approval for such research (Hester, 2004; Lind et al., 2003).
To address these issues, research policy-making entities, such as the Tri-Council funding agencies in Canada, should develop comprehensive standards and guidelines for adolescent consent practices. These standards and guidelines should clearly define adolescent capacity to consent to research and indicate situations that are suitable for adolescent self-consent. In addition, institutional review boards should develop more explicit and consistent policies surrounding consent requirements for the involvement of adolescents in research (Scott, 2013). For example, in the absence of an age-based framework, institutional ethics policies might present standard measures of decision-making capacity. At the same time, it is important to make sure safeguards are in place when conducting research with adolescents (Scott, 2013; Sims & Nolen, 2021). These safeguards could include recruitment and data collection approaches that reduce stress and mitigate power imbalances as much as possible, as well as consent procedures that protect adolescents from coercion or exploitation while respecting their autonomy (Scott, 2013; Sims & Nolen, 2021). By establishing clear and standardized guidelines for obtaining adolescent consent, institutions and organizations can promote consistency in research practices and enhance the integrity of research findings.
Limitations
The findings from this review should be considered in light of the following limitations. It is somewhat difficult to determine how informed consent is acquired in research on risky behaviours among adolescents because many papers do not provide details regarding the consent process. Consider a recent review that examined consent practices in research on risky behaviours in American adolescents. The results of the review showed that around 90% of published studies did not present details on consent procedures (Liu et al., 2017). In the current scoping review, 77 articles were excluded because they did not mention the consent process. Moreover, many of the papers included in the review did not present full details surrounding consent procedures. Although attempts were made to fill these gaps, it was not possible to reach all of the authors.
Another limitation with this review is that we only included published journal articles, which resulted in a somewhat narrow view of the literature. We excluded systematic reviews, scoping reviews, and meta-analyses from our search as these reviews typically aggregate studies from various locations or focus on non-Canadian settings. Given that our emphasis was solely on research conducted within Canada, these sources would have been of limited use to us. However, it is possible that we may have missed articles by excluding these sources. Grey literature (e.g., reports, government documents, conference presentations) was also excluded, as it often lacks the methodological detail necessary for this scoping review. Nonetheless, it is worth noting that grey literature could have directed us to other sources that were eligible for inclusion.
Conclusion
This scoping review provides information on current procedures for obtaining consent for research on risky behaviours among adolescents in Canada. The majority of the studies included in this review relied on adolescent self-consent for participation, suggesting that many researchers in this area see this practice as a reasonable alternative to parental/guardian consent. Adolescent self-consent was particularly common in certain types of studies, such as those using in-person research methods and those focusing on older adolescent age groups. Taken together, the current results provide insight and guidance to researchers who conduct research on risky behaviours, as well as the institutional review boards that evaluate this research against current standards.
Research Agenda
The results of this review suggest that adolescent self-consent is a common and accepted procedure in risky behaviours research, especially for studies involving in-person research methods, older adolescent groups, and particularly vulnerable populations. Future research should evaluate what safeguards are in place to protect adolescents from potential exploitation within this research.
Educational Implications
While it remains crucial for institutional review boards to use the TCPS 2 as a foundational resource for evaluating research proposals, it would be prudent for these boards to establish clear and consistent guidelines concerning parental/guardian consent requirements in research involving adolescents. Guidelines should be clearly communicated to researchers and incorporated into learning settings.
Best Practices
Researchers should make a concerted effort to include detailed consent information when describing their methodology in research communications. This information will provide other researchers with a better understanding of how the research community is acquiring consent in research on adolescent risky behaviours.
Supplemental Material
sj-docx-1-jre-10.1177_15562646241253953 - Supplemental material for Obtaining Consent for Research on Risky Behaviours Among Adolescents in Canada: A Scoping Review
Supplemental material, sj-docx-1-jre-10.1177_15562646241253953 for Obtaining Consent for Research on Risky Behaviours Among Adolescents in Canada: A Scoping Review by Lillian MacNeill, A. Luke MacNeill, Shelley Doucet, Alison Luke and Alex Goudreau in Journal of Empirical Research on Human Research Ethics
Footnotes
Acknowledgments
We would like to thank Richelle Witherspoon, Librarian with the University of New Brunswick Libraries, for peer reviewing the search strategy for this scoping review. We would like to thank Julia Besner and Kendra Hebert for their assistance with title and abstract screening and full-text screening.
Declaration of Conflicting Interests
The authors declared no actual or potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
