Abstract
Introduction
Endometriosis is a chronic, progressive gynaecological disease defined by the presence of endometrial-like tissue outside of the uterus. It is most commonly associated with debilitating pelvic pain and infertility but is also known to have a more widespread impact presenting through symptoms such as dyspareunia, dysuria, dyschezia and fatigue. In addition to the impact these physical symptoms can have on functional capacity, the disease diminishes quality of life through its negative consequences on intimate relationships, social activities, income and mental health. 1 This burden is further compounded by a delay in diagnosis and effective management. 2
Endometriosis can be divided into three subtypes based on the location and characteristics of the lesion. These are: superficial peritoneal endometriosis (SPE), ovarian endometrioma (OMA) and deep infiltrating endometriosis (DIE). SPE lesions can be further divided into red, black and white lesions which represent visual appearances corresponding to the increasing age of the lesions. Red lesions are thought to be more vascular compared to the older black or fibrosed white lesions. OMAs are brown fluid-filled cysts of the ovaries with endometrial lining. DIE is characterised by the invasion of the endometriosis into surrounding structures, including pelvic organs, the abdominal wall and retroperitoneal structures. In addition to these subtypes, endometriosis is also often classified intraoperatively by revised American Society for Reproductive Medicine (ASRM) stages. These stages are defined based on the number of lesions present and the depth of their infiltration, and range from stage I indicating minimal disease to stage IV indicating severe disease. 3
Currently, the gold standard for diagnosing and managing endometriosis is explorative laparoscopic surgery, with confirmation of the disease on histology required for diagnosis. In routine surgical practice, white light (WL) imaging is used to visualise endometriotic lesions on the peritoneal surface for excision. However, despite its central role in treatment, post-surgical symptom recurrence rates are concerning: about 21.5% within two years and between 40-50%, by the fifth year. 4 Incomplete excision of disease during the initial surgery may contribute to this recurrence. Challenges in visually identifying lesions during surgery, due to factors like their inaccessibility and varied appearances, might contribute to this incomplete excision and hence recurrence rates. 5 Furthermore because of these difficulties to the surgery, the experience of the surgeon may also influence outcomes. Consequently, there’s a push towards innovating surgical techniques, particularly in the realm of imaging, to enhance the detection of endometriotic lesions.
Moreover, there have been increasing developments of intra-operative imaging including the use of 3D cameras. Previous reviews in this area have also suggested that the use of contrast-enhanced imaging for the detection of endometriosis can improve the visualisation of endometriosis intra-operatively. 6 One promising method is the combination of indocyanine green (ICG) and near-infrared imaging (NIR). Originally developed in the 1950s for medical imaging, ICG when paired with NIR, offers real-time visualisation capabilities. ICG can be injected intravenously, into bile or into tissue, where it binds rapidly to proteins – in the plasma if injected intravenously - and becomes fluorescent when excited by a near-infrared light. 7 While NIR-ICG has proven effective in applications like retinal angiography, hepatic clearance, and cardiac output monitoring, 8 its potential in endometriosis surgery is still under exploration. 9 Given that angiogenesis (formation of new blood vessels) is a crucial process in the growth of endometriotic lesions, 10 ICG’s ability to highlight these hyper-vascular areas is seen as particularly promising.
Yet, a clear consensus on ICG’s overall utility as a contrast agent during endometriosis surgery remains elusive. There is a more unified body of evidence which validates the use of ICG within laparoscopic surgery for endometriosis in roles such as the evaluation of bowel anastomosis and the assessment of ureter perfusion.11-13 However, there is not a consensus about the capacity for ICG as the contrast-agent for the detection of endometriosis. This review seeks to consolidate research on NIR-ICG in detecting endometriosis, highlighting discrepancies that might account for the varied findings. Our goal is to provide a more definitive, evidence-based perspective on ICG’s role for detection of endometriosis lesions in laparoscopic endometriosis surgery.
Methods
Search Method
Search Strategies.

Eligibility Criteria
The key eligibility criteria are listed below:
Inclusion Criteria
- Primary or original research - Articles published in English - Year of publication: any - Intervention: NIR-ICG for intraoperative detection of endometriosis - Diagnosis of endometriosis confirmed on histology
Exclusion Criteria
- Case studies or video demonstrations - Letters to editor - ICG use in laparoscopic surgery for endometriosis in non-detection purposes
This narrative review included randomised clinical trials, pilot studies, case-control studies, prospective single-arm studies, case series and retrospective data analyses. Case studies and video demonstrations were not considered due to their lower reliability. For data that was only available within a conference abstract, the study was included if the inclusion criteria could be met from the abstract alone. If this were not the case, the corresponding authors were contacted for further data, and their studies were excluded if there was no response.
The participant population consisted of any person with suspected or previously confirmed endometriosis undergoing laparoscopic surgery for the investigation and management of the disease. The specific intervention selected for this review was the use of indocyanine green with near-infrared imaging (NIR-ICG) during laparoscopic surgery for the detection of endometriosis. The use of ICG for purposes other than detection – for instance bowel anastomosis assessment - was not considered. The primary outcome measure for the relevant studies was the intraoperative identification of endometriosis, confirmed by histopathological analysis of excised disease.
Screening and Data Extraction
Titles and abstracts of the articles produced by the search were screened independently by 2 reviewers according to the criteria outlined above. The data from the relevant articles was then extracted and summarised into an Excel spreadsheet by the primary reviewer. It was collated according to the sub-categories: - Title - Author - Year and Country of Publication - Study Type - Methodology details (sample size, ICG dose, timing of imaging) - Reported statistics about detection of endometriosis (quantity, PPV, NPV, sensitivity, specificity, false positive, false negative) - Reported sub-group analysis about different types of endometriosis
Results
The search strategy, described in Table 1 yielded 251 studies. These were uploaded to COVIDENCE and 70 duplicates were removed. Of the remaining 181 studies, 165 were excluded through screening titles and abstracts, and a further 7 were excluded on full-text review. This resulted in 9 studies that were included in this review (Supplemental Material). The screening process is summarised in a PRISMA diagram in Figure 1. PRISMA diagram.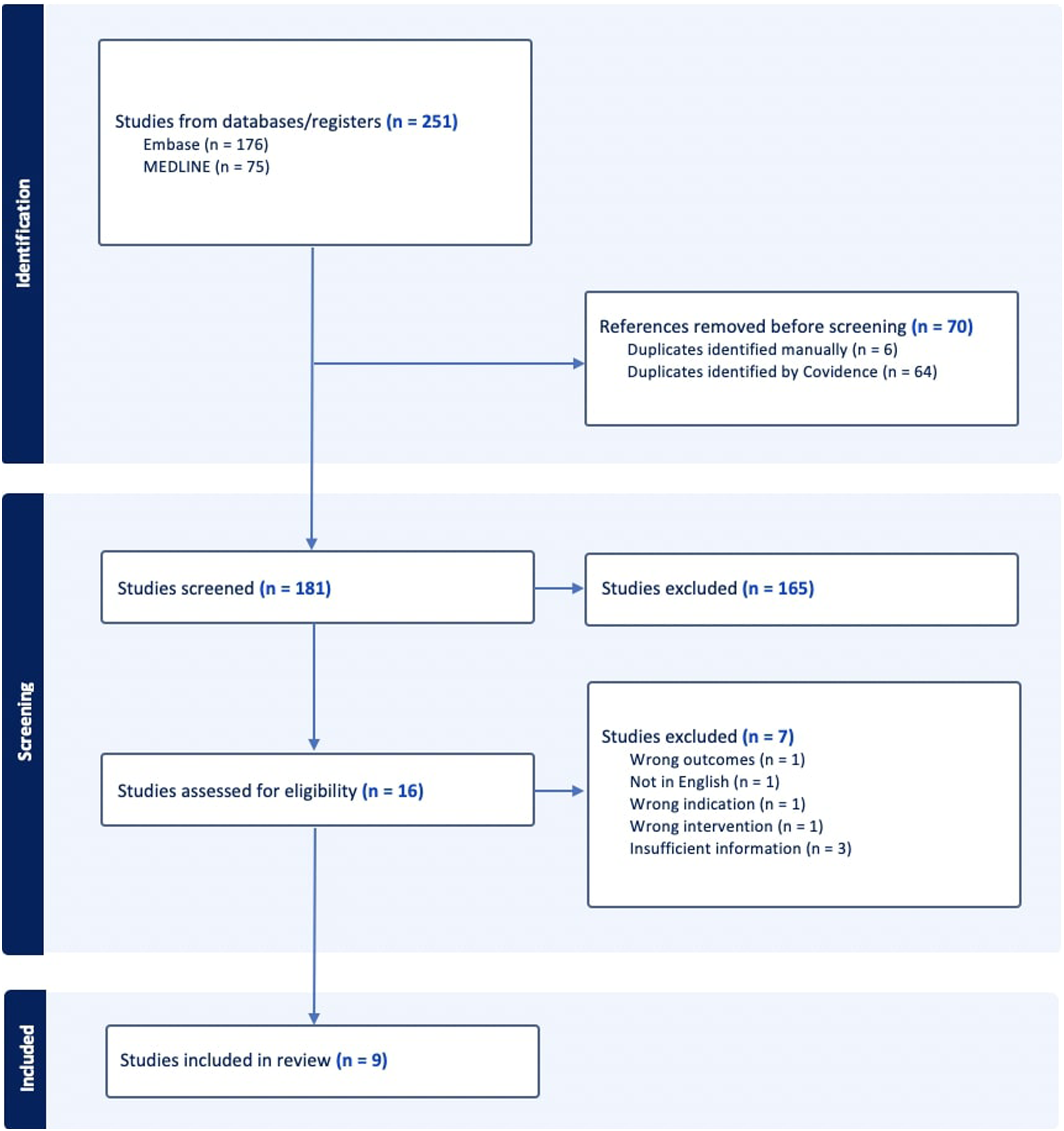
Study Characteristics.
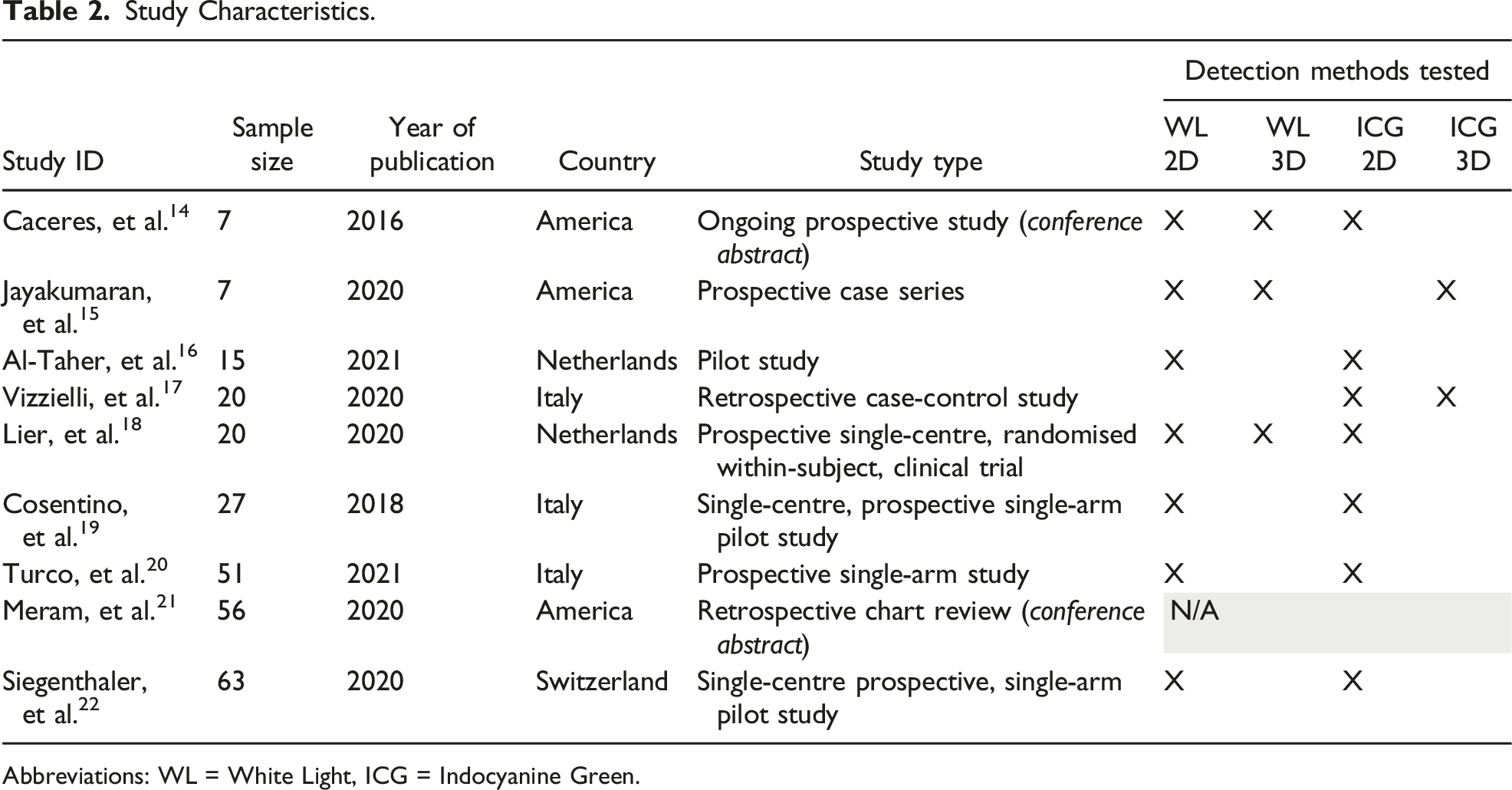
Abbreviations: WL = White Light, ICG = Indocyanine Green.
Summary of Statistics.
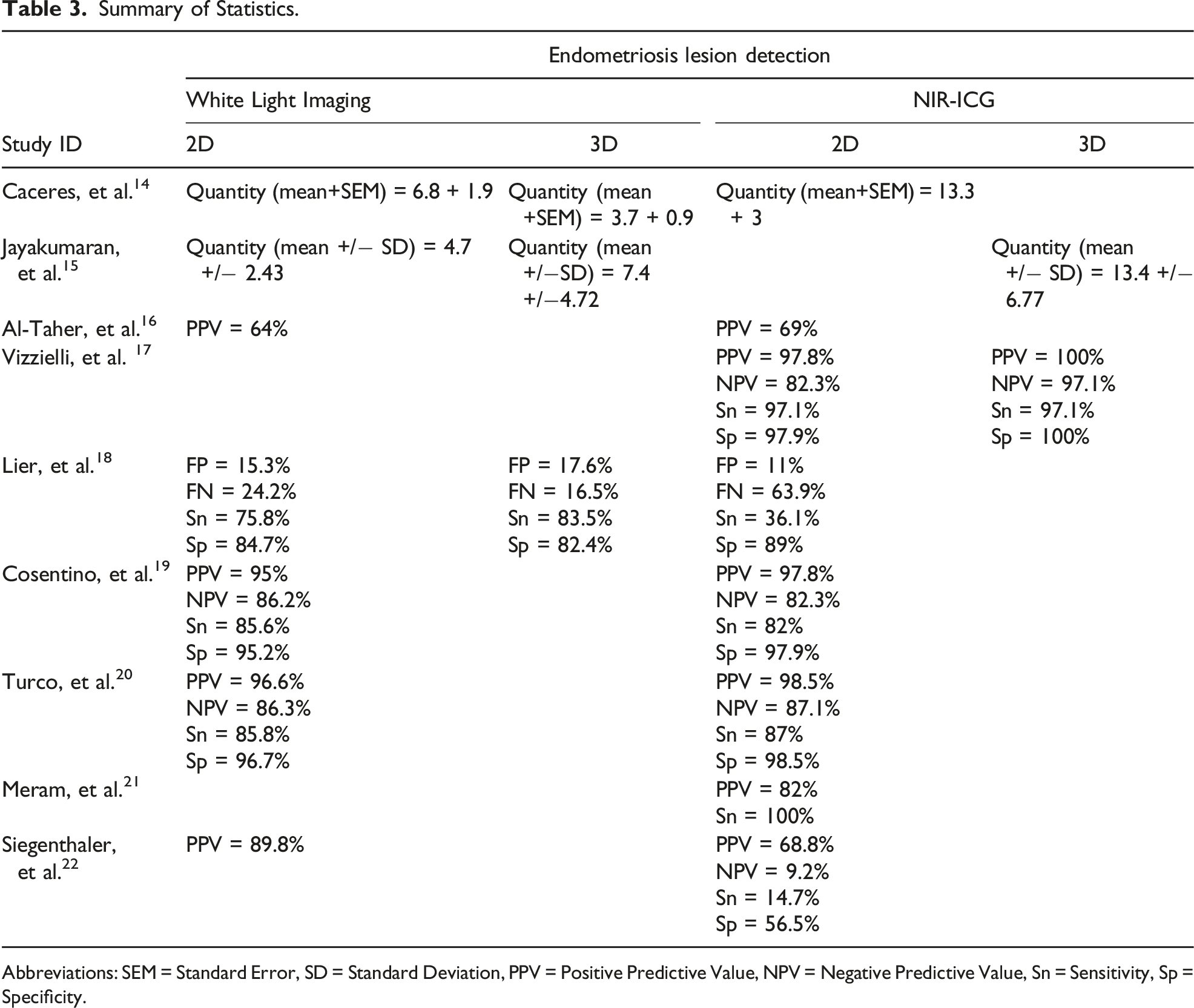
Abbreviations: SEM = Standard Error, SD = Standard Deviation, PPV = Positive Predictive Value, NPV = Negative Predictive Value, Sn = Sensitivity, Sp = Specificity.
Overall, six out of the nine studies described a promising role of NIR-ICG in the detection of endometriosis.14,15,17,19-21 Four of these studies were those comparing NIR-ICG to WL imaging and described improved detection of endometriosis with the addition of the fluorescence imaging modality.14,15,19,20 With NIR-ICG added, the number of average lesions detected per patient was reported to increase from 6.8 to 13.3, 14 and 7.4 to 13.4. 15 Similarly, Cosentino, et al. 19 and Turco, et al. 20 reported improved PPVs and specificity. While Vizzielli, et al. 17 and Meram, et al. 21 do not make the same comparisons between imaging modalities, they still conclude that NIR-ICG is an effective screening and diagnostic tool, with PPVs of 97.8% and 82% respectively.
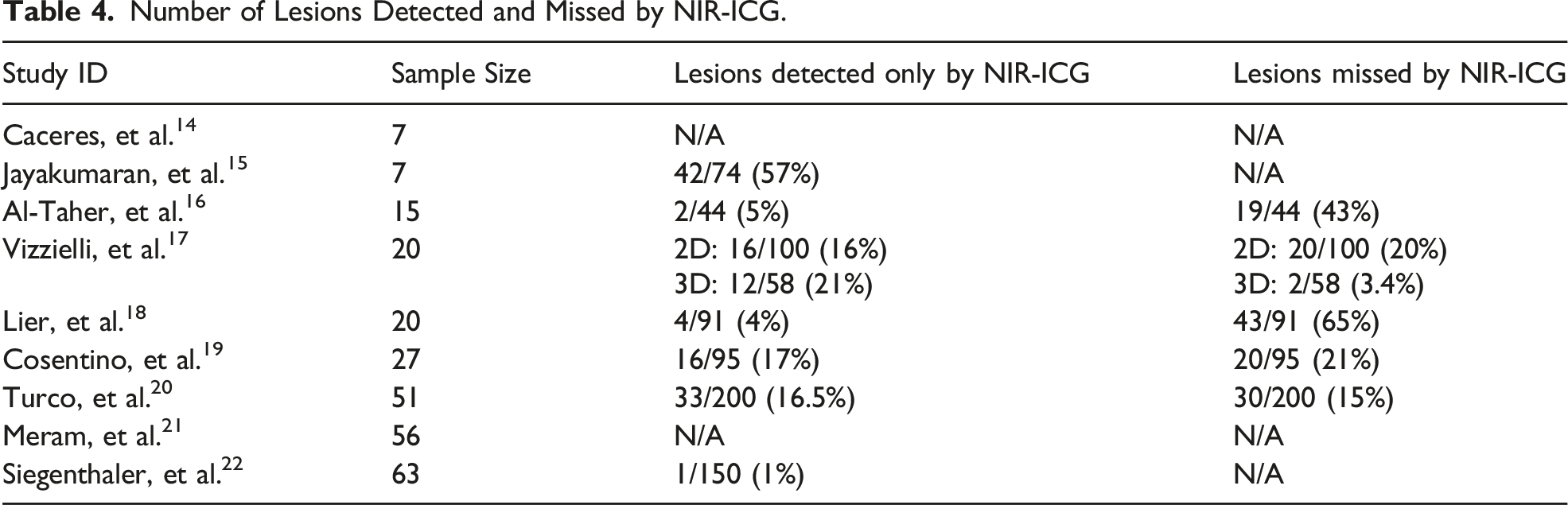
Number of Lesions Detected and Missed by NIR-ICG.
Study Methodologies.

Through subgroup analysis, Siegenthaler, et al. 22 demonstrated that longer time intervals between the dye administration and imaging led to significantly enhanced visualisation of lesions. Increasing the duration of exposure to the dye from 2-5 min to at least 20 min increased the detection rate from 4.7% to 44%.
Discussion
While the use of enhanced imaging techniques for the intraoperative detection of endometriosis has been presented as a promising tool for the future of endometriosis visualisation in laparoscopic surgeries, 6 there is a limited amount of evidence about the specific use of NIR-ICG as one of these tools, with this review encompassing just nine articles. A common thread among these studies is their limited samples sizes and varied methodologies, underscoring the pressing need for more in-depth research in this domain.
Dosage, Timing and Safety Data on NIR-ICG
Before considering its possible roles in laparoscopic surgery, it is important to discuss the safety and method of NIR-ICG. While there have been some rare reported cases of allergic reactions to ICG, 23 there were no reported adverse reactions to ICG within the scope of this review. Importantly, a known ICG allergy was considered an exclusion criterion for the majority of the included studies. Additionally, a comparison of the various methodologies utilised with this technique highlighted the importance of ICG dye dosage and imaging timing. The conclusion demonstrated by Siegenthaler, et al. 22 that longer time intervals between the dye administration and imaging enhanced visualisation of lesions, was reinforced by the fact that all three of the studies which did not support the overall efficacy of ICG used exposure times as low as two and five min within their protocols (Table 5). This difference is thought to be due to the time it takes for the dye to wash-out from the surrounding healthy tissue as the fluorescence remains in the neo-vascularised endometriotic lesion. 16 With regards to dosages, the studies within this review with the most efficacious results utilised dosages of 0.25-0.30 mg/kg (Table 5). Notably, the use of NIR-ICG within other specialties also utilising the injection of peripheral intravenous ICG is diverse in protocol. The consensus for the evaluation of anastomoses in colorectal surgery is a total ICG dose of 5-10 mg, 24 however biliary tract visualisation is performed with doses ranging from less than 0.05 mg/kg to 0.25-0.30 mg/kg.25,26 While the total maximum recommended ICG dosage reported in the literature is 2 mg/kg, 25 the highest dose at which it has an established role is 0.50 mg/kg for purposes such as liver tumour visualisation. 26 The reasoning for remaining within this lower range of ICG dosage is unclear, however it is possibly for the sake of cost and patient safety. In future research as well as clinical use, the ideal methodology for the initial assessment using NIR-ICG would thus involve a fixed amount of dye proportional to patient weight prior to surgery (0.25-0.30 mg/kg) followed by a longer waiting time before imaging (10-30 min). This extension of operating time was reported as negligible. 22 The use of additional boluses of ICG provided intra-operatively for repeated assessments throughout the surgery was shown to be a safe technique,16,18 however its impact on visualisation was not adequately explored within the included studies.
Effectiveness of NIR-ICG in Intraoperative Detection of Endometriosis
The goals of laparoscopic surgery for endometriosis can be summarised into two main aims; to identify the disease, and to safely remove it. This review focuses on the former, and particularly how the intra-operative use of NIR-ICG can improve the gaps of current surgical techniques. As a purely diagnostic tool, NIR-ICG was not found to provide additional value to conventional WL imaging. That is, it was not shown to identify the disease in patients where the disease was unable to be visualised with WL imaging. However, this finding may be limited by the fact that the majority of included participants were of later ASRM disease stage, where there is a larger number of lesions which are easier to identify. Still, its utility is more likely to be in how it can enhance the visualisation provided with WL imaging. It was a common theme across these papers that despite improved detection rates, positive lesions identified through WL imaging were missed by NIR-ICG with a 2D camera to extents ranging from 15% 20 to 65% 18 (Table 4). Thus, the possible clinical utility of NIR-ICG was suggested to only be in combination with WL imaging as either a confirmatory diagnostic tool, or to aid with more complete excision of the disease.
One previously identified barrier to obtaining complete excision of disease is the concept of occult endometriosis, which is histologically positive endometriosis in the peritoneum that is not normally visible on laparoscopy. Occult endometriosis was first reported in 1986 27 and has been subsequently confirmed over several studies including by Khan, et al. 28 who reported endometriosis detected in biopsies taken from normal appearing peritoneum, at depths between 10-80 micrometres from the surface and more commonly in women with visually identifiable endometriosis. Furthermore, they reported that these lesions were active and capable of growth. The combination of these conclusions suggests that identifying occult endometriosis for excision is particularly important to prevent symptomatic recurrence for those patients undergoing elective surgical management of their disease. In seven out of the nine included studies in this review, endometriotic lesions were identified through NIR-ICG which were not visualised through WL imaging. The range of additional occult lesions identified across the studies ranged from one 22 to forty-two. 15 The proportions which these additional lesions represented are summarised in Table 4, describing an inconsistency in the capacity for NIR-ICG to identify occult endometriosis. It is important to note that the significant variability in the methodologies of the included studies could contribute to this inconsistency. As detailed, the dosage and timing utilised had significant impacts on the techniques efficacy and furthermore there was some variety in the histological definition of a positive lesion. Turco, et al. 20 included both the presence of endometriosis foci and sclerosing inflammatory infiltrate in their definition of positive histology, and yielded a PPV of 98.5%, compared to a PPV of 68.8% by Siegenthaler, et al. 22 who only considered endometrial histology. The significance of these results is dependent on the clinical weight of a single unidentified lesion and on removing occult endometriosis, both of which are concepts not explicitly defined in the literature and reliant upon future research looking at long-term outcomes.
NIR-ICG in Detection of Deep Infiltrating Endometriosis
One of the factors that can make endometriosis difficult to detect during surgery is the heterogeneity of lesions in their location and appearances. Four of the included studies sub-categorised the identified endometriotic lesions as DIE or SPE.17,19,20,22 Some of the locations where DIE was described to be able to be visualised with NIR-ICG include the: periureteral region, ovarian fossa, uterosacral ligament, rectum, rectosigmoid colon, retrocervical region and urinary bladder. The reduced accessibility of some of these locations makes DIE more difficult to visualise on laparoscopy, 29 and provides a surgical challenge of resection without functional damage to the underlying structures.
Notably, NIR-ICG was found to be more effective in the identification of DIE, than for the identification of endometriosis as a whole. Cosentino, et al. 19 describes an increase in sensitivity from 82% to 89% when considering DIE alone. Furthermore, it was described to be more effective for the detection of lesions in the periureteral region, ovarian fossa and colorectal nodules, and less so for lesions of the bladder. Turco, et al. 20 reported an increase in sensitivity with NIR-ICG from 64.5% to 93.5% for periureteral/ovarian fossa nodules and 86.7% to 90.0% for colorectal nodules, but a decrease from 76.2% to 71.4% for bladder nodules.
The reasons for the differences in detection rates could be due in part to several factors related to the mechanism of ICG. As previously described, NIR-ICG allows real-time visualisation of the dye within the vasculature allowing for visualisation of structures that are not at the surface. Thus, the effectiveness of the fluorescence is dependent on the desired lesion being more vascular than the surrounding tissue. Jayakumaran, et al. 15 describes that the vascular endometriotic lesions when visualised with NIR-ICG present as a fluorescent island with a surrounding white, fibrotic ring. It has been hypothesised that the pathophysiology of DIE differs from the other subtypes of endometriosis. For instance, DIE has been reported to have a greater degree of vascularity than peritoneal lesions. 30 Furthermore, DIE of the rectum specifically has been reported to have a higher expression of vascular endothelial growth factor (VEGF) and thus vascularity than endometriosis of the bladder or ovaries, 31 which could explain the deficiency in the identification of bladder lesions. However, it is important to consider that a diversity of vascular patterns has been reported even within the category of rectosigmoid endometriosis (RSE). In a prospective pilot study utilising NIR-ICG, Raimondo, et al. 32 describes rectosigmoid lesions with both abundant and absent fluorescence indicating their respective hyper- and hypo-vascularity. In fact, they show a majority of RSE nodules (60%) presenting with a hypovascular pattern and an association of these hypovascular lesions with larger nodule size. Notably this study did report observing fluorescence of the RSE nodules up to 3 min. The hypo-vascularity could be explained by the possible role of fibrosis in the pathophysiology of endometriosis. That is, that chronic inflammation leads to the activation of signalling pathways for fibrosis which as it accumulates can both increase nodule size and reduce vascular density. 32 Moreover, several of the included studies excluded participants with ovarian or fallopian tube endometriosis because of the hypo- and hyper-vascularity of these structures respectively.19,20 Understanding the different degrees of vascularity of lesions is important to establish the limitations of this technique.
Additional Roles of NIR-ICG in Endometriosis Surgery
While this review focuses on the role of NIR-ICG specifically for detection, it is worthwhile mentioning the alternative roles in which this fluorescent technique can be used in laparoscopic surgery for endometriosis, to understand how it can be integrated into practice. Siegenthaler, et al. 22 describe two of these alternative roles. The first is to assist in identifying the margins of resection of DIE, and the second is to allow visualisation of the ureters when excising ureteric endometriosis. Both these serve to maximise the protection of healthy underlying tissue and hence the function of these pelvic organs, preventing infertility and urological dysfunction. In a systematic review of the wider role of ICG in endometriosis surgery, Ianieri, et al. 11 have previously concluded on the same utility for the evaluation of ureter perfusion assessment as well as intestinal anastomoses after segmental resection. Their conclusions about ureteral perfusion assessment are made based on a prospective case series by Raimondo, et al. 12 which showcased the success of the imaging technique in confirming ureteral ischaemia suspected with white light imaging, prompting ureteral stent placement. There is a larger body of growing evidence supporting the use of NIR-ICG for surgery of RSE. The technique was first reported in 2018 in a video description of a bowel resection for the management of a large rectal endometriotic nodule. In this case, ICG was used to select the margins for rectal resection, and assess rectal vascularisation after the formation of the anastomosis. 33 The feasibility of the technique was then tested in a larger preliminary study of thirty-two patients, which showed that NIR-ICG could be used in a safe and timely manner to identify sufficient perfusion at the anastomotic line. In this study, NIR-ICG prompted reinforcement of the anastomosis in one case and was followed by an anastomotic leak in another single case. 34 While the majority of the literature surrounding NIR-ICG focuses on the qualitative role of the fluorescence technique, a cohort study utilising the angiography technique after full-thickness bowel resection for RSE by Raimondo, et al. 13 went further as to consider quantitative evaluation. Rather than relying only on an individual’s identification of fluorescence patterns, their quantitative analysis used video software to consider fluorescence as well as changes to the intensity of this fluorescence over time. Both qualitative and quantitative methods were established as effective for anastomotic perfusion assessment. However, quantitative compared to qualitative analysis also showed an increased specificity from 71 to 93% in predicting post-operative bowel fistula, suggesting a promising role for the detection of major post-operative complications.
Limitations
There are several limitations to this review. As mentioned earlier, there are very few included studies with limited sample sizes and varied methodologies. Additionally, due to the nature of the intervention, there was no blinding of the surgeons to the NIR-ICG leading to a risk of bias in identification outcomes. The identification of lesions is a highly qualitative, and hence difficult task. The methodologies of the included studies stressed the importance of having surgeons experienced in minimally invasive gynaecological surgery for endometriosis at the centre of these trials for this reason. However, this could also be considered a limitation in the produced evidence as it does not allow a representation of how the technique may have utility for surgeons with less experience. Furthermore, most of the studies only included patients who were in the later stages of the disease (ASRM III-IV), in which endometriosis is often easier to visualise on laparoscopy. Not only does this potentially influence results, it does not accurately represent the clinical need for improved detection tools for people with endometriosis in earlier stages. Lier, et al. 18 propose that the sensitivity and specificity of imaging techniques on detection could also have different degrees of importance at different stages of the disease. For instance, in earlier stages, specificity may be more essential to confirm the presence or absence of disease whereas in later stages sensitivity is required for more thorough excision. These nuances could not be established with the study populations represented in this review. Finally, many of the statistics reported are purely theoretical, as you cannot truly report on the sensitivity of NIR-ICG as the actual total known number of lesions is impossible to determine without a total peritonectomy. The specificity also depends on whether negative controls were taken, which was not always the case in the included studies.
Conclusion
In conclusion, NIR-ICG has been shown to be a possibly promising tool for the enhanced intra-operative detection of endometriosis when used with conventional WL imaging. With adequate dosage and timing, it is capable of identifying occult endometriosis, in part through improving the visualisation of DIE. However, the current evidence is still limited in size and homogeneity. Based on the results of this review, we propose that future studies should consider a larger sample size and utilise an ICG protocol with a fixed amount of dye proportional to patient weight prior to surgery (0.25-0.3 mg/kg) followed by a longer waiting time before imaging (10-30 min). The studies can also consider repeat injection of ICG in their protocol. Future studies should also include long-term follow-up outcomes assessing the impact of the additional excised lesions using NIR-ICG on symptomatic relief and surgical re-intervention. These studies should also make sure to include patients from earlier as well as the later ASRM stages of the disease, surgeons with a diverse range of experience and focus at how NIR-ICG can specifically enhance DIE detection.
Supplemental Material
Supplemental Material - The Role of Indocyanine Green With Near-Infrared Imaging for the Intraoperative Detection and Enhancement of Endometriosis Lesions: A Narrative Review
Supplemental Material for The Role of Indocyanine Green With Near-Infrared Imaging for the Intraoperative Detection and Enhancement of Endometriosis Lesions: A Narrative Review by Minoli Rajasinghe, Tarana Lucky, and Shamitha Kathurusinghe in Surgical Innovation.
TL: literature screening, manuscript editor.
SK: literature screening, manuscript editor.
All authors reviewed final version of manuscript and agreed to submit it to SRI for publication.
Footnotes
Acknowledgments
Dr Catarina Ang, Dr Vanessa Ross and the University of Melbourne for their support.
Author Contributions
MR: literature screening, data extraction, manuscript author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
