Abstract
Keywords
Introduction
Iatrogenic bile duct injury after laparoscopic cholecystectomy is a rare but serious complication with an incidence ranging from 0.08% to 1.5%.1-3 Bile duct injuries are mostly the result of misidentification of the common bile duct (CBD) or the common hepatic duct (CHD) for the cystic duct (CD). Transection of the CBD or CHD leads to severe complications, long-term mortality rates up to 21%, and significant costs. 4 Important contributing factors are the learning curve associated with laparoscopic surgery and the presence of (misinterpreted) aberrant bile duct anatomy. Because laparoscopic cholecystectomy is currently one of the most performed surgeries worldwide, safer approaches are desirable. Use of the critical view of safety—a method of intraoperative identification of the CD and cystic artery—has been developed to minimize the risk of bile duct injury. 5 Although this strategy is now implemented worldwide, bile duct injuries still occur.
Identification of bile ducts during laparoscopic cholecystectomies can be enhanced by intraoperative cholangiography (IOC). This imaging technique requires injection of radio-opaque contrast into the CD, whereupon a series of X-rays are taken. IOC can accurately depict biliary anatomy and some advocate that it should be performed routinely.6,7 However, this technique is expensive, time-consuming, and exposes patients and health care personnel to radiation. Moreover, bile duct cannulation may cause bile duct injury itself and images can be difficult to interpret.8,9
An alternative intraoperative imaging technique that is less invasive, does not require radiation, and is not time consuming is near-infrared (NIR) fluorescence cholangiography. Feasibility of real-time NIR fluorescence imaging of bile ducts using indocyanine green (ICG) was first described in 2008. 10 After intravenous administration, ICG is cleared via the liver, enabling visualization of liver tumors and bile duct anatomy.11-13 ICG has a favorable safety profile; side effects are reported in less than 1 out of 40 000 patients. 14
Several studies have described fluorescence cholangiography during open, laparoscopic, and robotic cholecystectomy.15-19 However, only few tried to optimize the dose and timing of administration.15,20 This is important, because if fluorescence imaging is performed directly after administration of ICG, the liver will be highly fluorescent while bile ducts do not yet contain ICG. Optimal would be a high fluorescence signal in bile ducts, while liver tissue in the background stains dark. Excretion of ICG into the bile reaches a peak approximately 2 hours after dosing and remains detectable long after injection. 21 Yet most studies inject ICG within the first hour prior to imaging. It is already shown that discrimination between bile ducts and liver is enhanced when the time interval between ICG injection and fluorescence imaging is prolonged to 24 hours instead of 30 minutes. 15 Additionally, a recent study showed that a dose of 0.25 mg/kg, administered at least 45 minutes prior to fluorescence imaging, facilitates accurate bile duct identification. 20 Although several ICG doses (up to 0.25 mg/kg) were tested in this study, fluorescence cholangiography was performed only up to 180 minutes after ICG administration. The optimal dose for laparoscopic fluorescence cholangiography after an extended interval between dosing and imaging as determined by Verbeek et al, 15 i.e. 10 mg ICG, was not studied. Moreover, dosing times between 3 and 6 hours prior to surgery have never been studied, while these are clinically relevant.
In the current study, we first performed a systematic literature research to identify all used strategies for fluorescence cholangiography in clinical studies. Second, we optimized the dose of ICG and dosing time, ranging from 0.5 to 24 hours prior to surgery, to distinguish bile ducts from adjacent liver tissue.
Materials and Methods
Systematic Review of the Literature
We systematically searched PubMed for clinical trials about fluorescence cholangiography performed during open, laparoscopic, or robotic surgery since 2008. Only articles written in English and describing 3 or more patients were included. Selection of relevant titles was performed independently by 2 reviewers. Subsequently, all included articles were scanned on the content of the abstract, again by 2 independent reviewers.
Clinical Trial
This study was approved by the Medical Ethics Committee of the Leiden University Medical Center and performed in accordance with the ethical standards of the Helsinki Declaration of 1975 (Netherlands Trial Registry: NTR5623). Twenty-eight patients suffering from cholecystolithiasis and/or cholecystitis, and planned for laparoscopic cholecystectomy, were included. Exclusion criteria were based on contraindications for ICG: renal impairment (estimated glomerular filtration rate <55); allergy for iodine, shellfish, or ICG; hyperthyroidism and pregnancy. All patients provided informed consent.
Patients were first assigned into 2 dose groups receiving either 5 mg or 10 mg ICG. Patients assigned to the 5 mg group were planned to receive their ICG dose 30 minutes, 2, 4, or 6 hours prior to surgery (4 patients per time interval). Patients assigned to the 10 mg group were planned to receive their ICG dose 4, 6, or 24 hours prior to surgery. Patients were distributed over the different groups based on their order in the surgical program. Patients scheduled early in the morning were allocated in the brief interval groups, while patients late in the afternoon were allocated into the longer interval groups. Patients were selected for the 24hour interval group if their procedure was scheduled on Tuesday until Friday. Standard-of-care laparoscopic cholecystectomy was performed. When the surgeon considered it to be necessary, IOC was performed. Fluorescence imaging of bile ducts and adjacent structures was performed at least 3 times before the critical view of safety was assessed.
Intraoperative Near-Infrared Fluorescence Imaging System
Laparoscopic imaging was performed using a high-definition (HD) fluorescence laparoscope (Karl Storz Endoscopes, Germany) through a standard 12 mm subumbilical trocar port. The system was equipped with a visible and 760 nm (ICG modus) light source. No overlay of visible and ICG images was possible, but anatomical orientation could be maintained due to easy and fast switching between both channels using a foot pedal.
Preparation and Administration of Indocyanine Green
Twenty-five mg ICG (Pulsion Medical Systems, Munich, Germany) was resuspended in 10 cc sterile water to yield a concentration of 2.5 mg/mL. To obtain doses of 5 mg and 10 mg ICG, respectively, 2 and 4 mL of the solution was intravenously administered.
Postoperative Questionnaire for Surgeons
After surgery, the surgeon completed a questionnaire to score the following statements about fluorescence imaging: (a) it could decrease the number of bile duct complications; (b) it led to better detection of bile ducts; (c) the technique was of added value for this procedure; (d) it needs to be standard-of-care during laparoscopic cholecystectomy; and (e) it delays surgery. Surgeons scored these statements in the following range: totally disagree, somewhat disagree, neutral, somewhat agree, and totally agree.
Data Analysis
Data were analyzed using GraphPad Prism Software (Version 5.01, La Jolla, CA). Bile duct-to-liver ratios (BLR) were calculated by dividing the fluorescence signal of the CD by the fluorescence signal of the liver. Intraoperative fluorescence images of 3 different time points were analyzed using Image J (version 1.49b, National Institutes of Health, Bethesda, MD).
Results
Systematic Review
A total of 242 articles were identified (Figure 1). After reviewing all titles and subsequently, content of the abstracts, 27 eligible clinical studies were reviewed (Table 1).10,15-20,22-42 Used ICG doses ranged from 0.025 mg in total to 0.25 mg/kg. Most studies (17/27) used 2.5 mg administered within 1 hour prior to fluorescence imaging. Timing of ICG administration differed from 0 (intraoperatively) up to 24 hours. No clinical trial studied fluorescence cholangiography 3 to 24 hours after ICG administration. The majority of studies (24/27) performed ICG injection 30 to 60 minutes prior to surgery. Successful identification of the CD by fluorescence imaging was achieved in 98% (range: 48% to 100%) of all included patients (n = 1057). Two studies investigated fluorescence cholangiography using different doses of ICG and time of administration.15,20 They studied the doses 5 or 10 mg and 0.025 or 0.25 mg/kg, and time points of 10 to 180 minutes and 30 minutes or 24 hours prior to surgery.
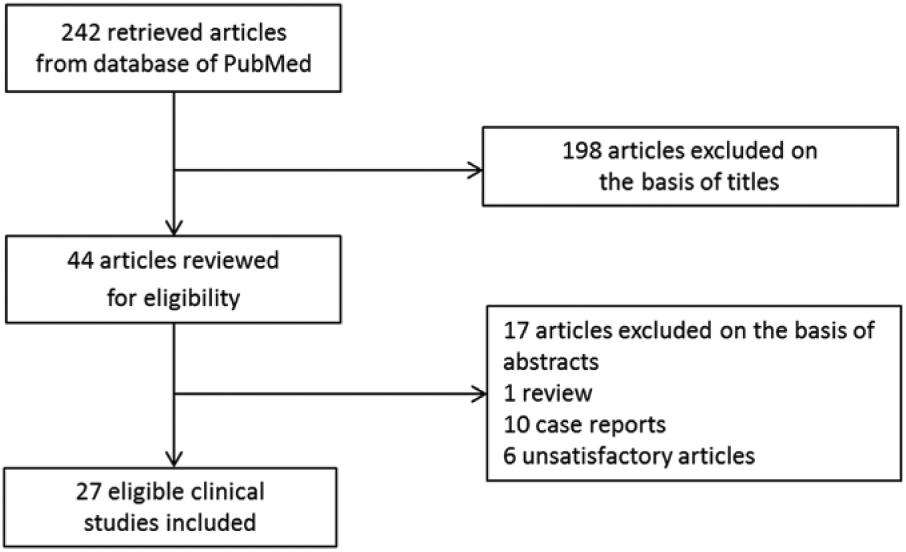
Flowchart of the literature search.
Relevant Articles About Fluorescence Cholangiography.

Abbreviations: ICG, indocyanine green; OC, open cholecystectomy; LC, laparoscopic cholecystectomy; SILC, single-incision laparoscopic cholecystectomy; SIRC, single-incision robotic cholecystectomy; LLR, laparoscopic liver resection; IR, identification rate; CD, cystic duct.
Open cholecystectomy performed during pancreaticoduodenectomy.
An additional ICG injection during surgery was given to evaluate fluorescence angiograph.
Five fluorescence laparoscopic imaging systems include (1) prototype and (2) improved version of the Hamamatsu Photonics laparoscope, the fluorescence imaging system of Olympus Medical Systems (3), the Karl Storz HD fluorescence laparoscope (4), and the fluorescence imaging system of Novadaq (5).
Patient Characteristics
A total of 28 patients were included. No adverse events related to ICG occurred. Sixteen patients received 5 mg ICG and 12 patients 10 mg ICG. Patient characteristics are summarized in Table 2. Three patients were excluded: one patient due to failed ICG administration, one due to technical issues during saving of images, and one patient with a history of Roux-en-Y gastric bypass who had a percutaneous drain in situ. In 3 patients, an IOC was performed. One patient, with a history of Roux-en-Y gastric bypass, underwent a transgastric endoscopic retrograde cholangiopancreaticography (ERCP) because of choledocholithiasis. Fluorescence cholangiography, performed 30 minutes after injection of 5 mg ICG, did not provide an overview of the bile duct anatomy (BLR 0.3). Another IOC was performed in a patient whose surgery was postponed. Fluorescence imaging 10 hours post dosing of 5 mg ICG was not optimal (BLR 0.4). The third IOC was performed in a patient who received 10 mg ICG 24 hours preoperatively. Fluorescence imaging showed the CBD (BLR 1.1), which was confirmed by IOC. No aberrant bile ducts were detected in the current study, but 2 patients showed aberrant arterial structures in close proximity to the gallbladder. Due to the lack of fluorescence, these structures could be identified as vasculature instead of bile ducts.
Patients’ Characteristics.
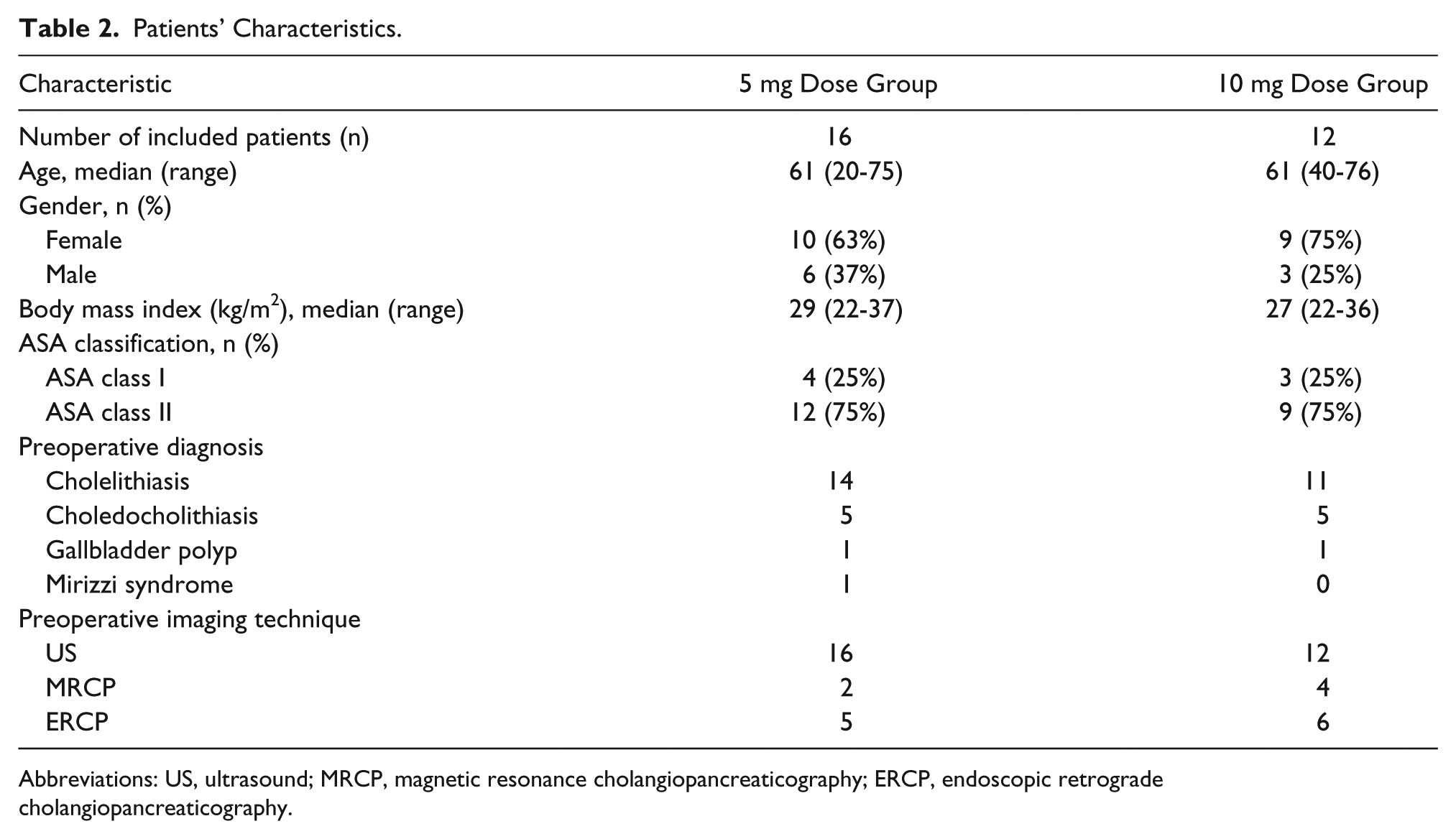
Abbreviations: US, ultrasound; MRCP, magnetic resonance cholangiopancreaticography; ERCP, endoscopic retrograde cholangiopancreaticography.
Optimization of Dose and Time Interval
Identification of the CD by fluorescence imaging was successful in 24 out of 25 patients (96%). The patient with unsuccessful bile duct identification was a patient who suffered from cirrhosis and Mirizzi syndrome and previously underwent stent placement and ERCP. During the laparoscopic surgery, strong adhesions between stomach, duodenum, and gallbladder were found. No clear overview of bile duct anatomy could be obtained, neither by normal inspection nor using fluorescence imaging. After conversion, the gallbladder appeared completely fused with the proximal duodenum and a fistula between gallbladder and duodenum, and a cyst originating from the CBD was found.
Of the remaining 24 patients, 14 patients received 5 mg ICG and 10 patients 10 mg ICG. Patients were intended to be divided in different groups based on time of fluorescence imaging after ICG injection, but it turned out to be difficult to time the exact moment between ICG administration and surgery due to the unpredictable course of the surgical program. In Figure 2, the BLR is plotted for every individual patient. In the 5 mg dose group, all BLRs between 3 to 7 hours post dosing were >1, with a mean ratio of 1.8 ± 0.5 (Video 1, available online at http://journals.sagepub.com/doi/suppl/10.1177/1553350617702311). Up to 3 hours after dosing, all measurements showed equally or more fluorescent liver tissue compared to the CD (BLR 0.8 ± 0.2), which was significantly lower (P < 0.01; Figure 3). In the 10 mg dose group, all BLRs between 5 and 25 hours were >1, with mean ratios of 1.4 ± 0.3. Up to 5 hours after dosing, the liver showed to be equally or more fluorescent than the CD (BLR 0.9 ± 0.2, respectively), which was again significant (P = 0.02; Figure 4). BLRs 3 to 7 hours post dosing of 5 mg and 5 to 25 hours post dosing of 10 mg were not statistically different (P = 0.12)
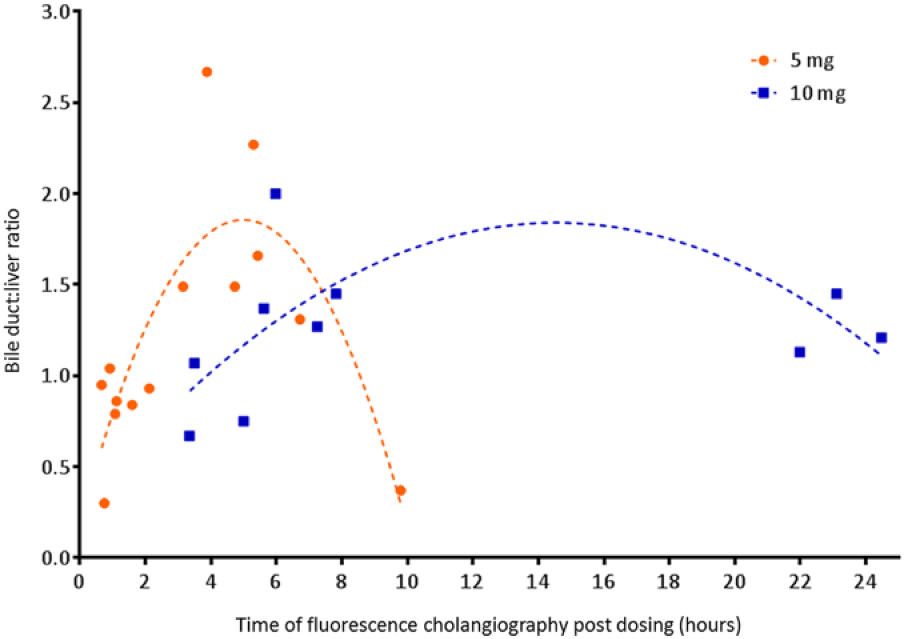
Fluorescence intensity of bile duct compared to adjacent liver tissue for each individual patient. The parabolic lines show our expectation of the mean BLRs over time.
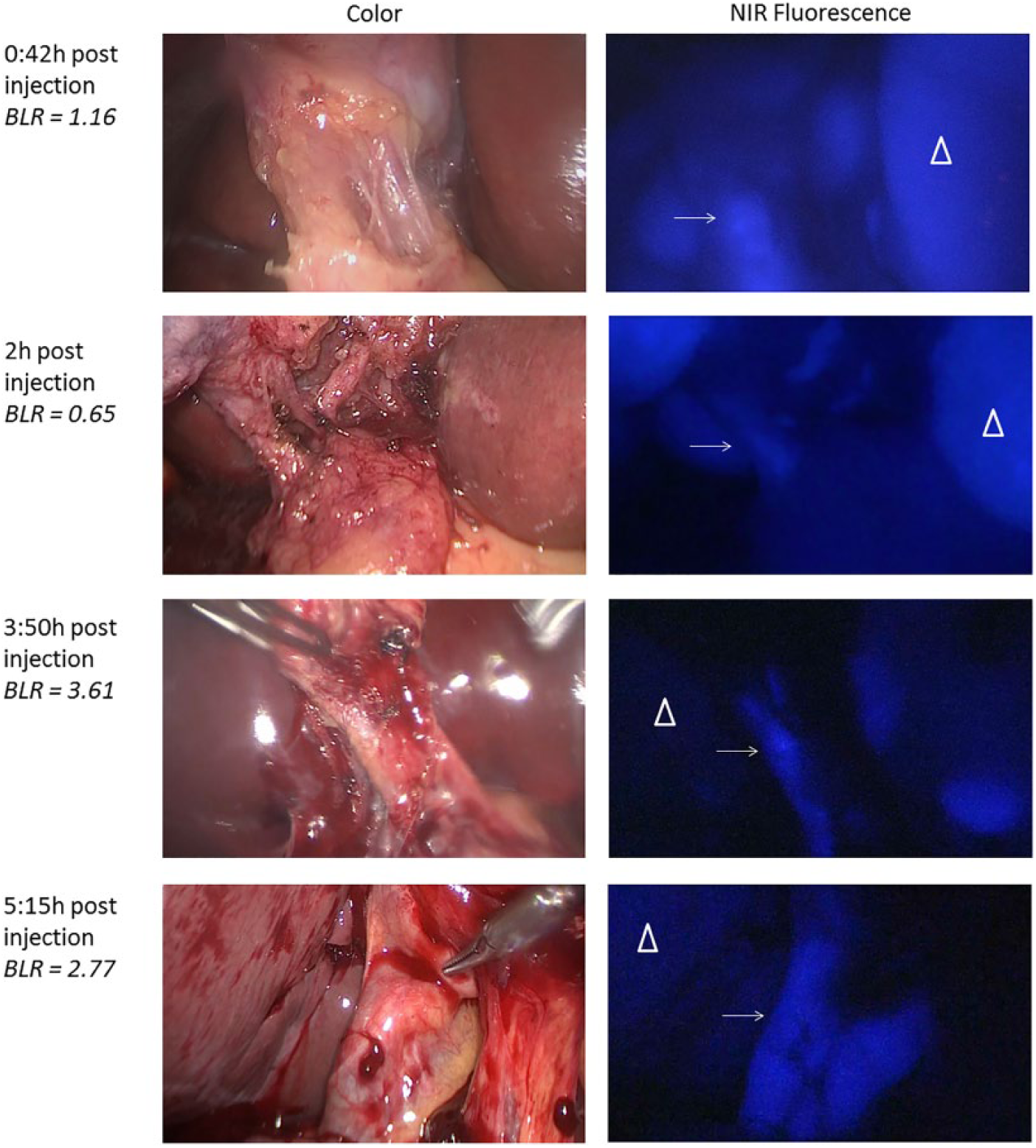
Fluorescence cholangiography over time after administration of 5 mg ICG. Shown are images of fluorescence detection of the cystic duct (arrow) and adjacent liver tissue (pyramid) over time. Bile duct-to-liver ratios were sufficient (ie, >1) in patients who received 5 mg ICG 3 to 7 hours prior to surgery. BLR was ≤1 when fluorescence imaging was performed less than 3 hours after administration.
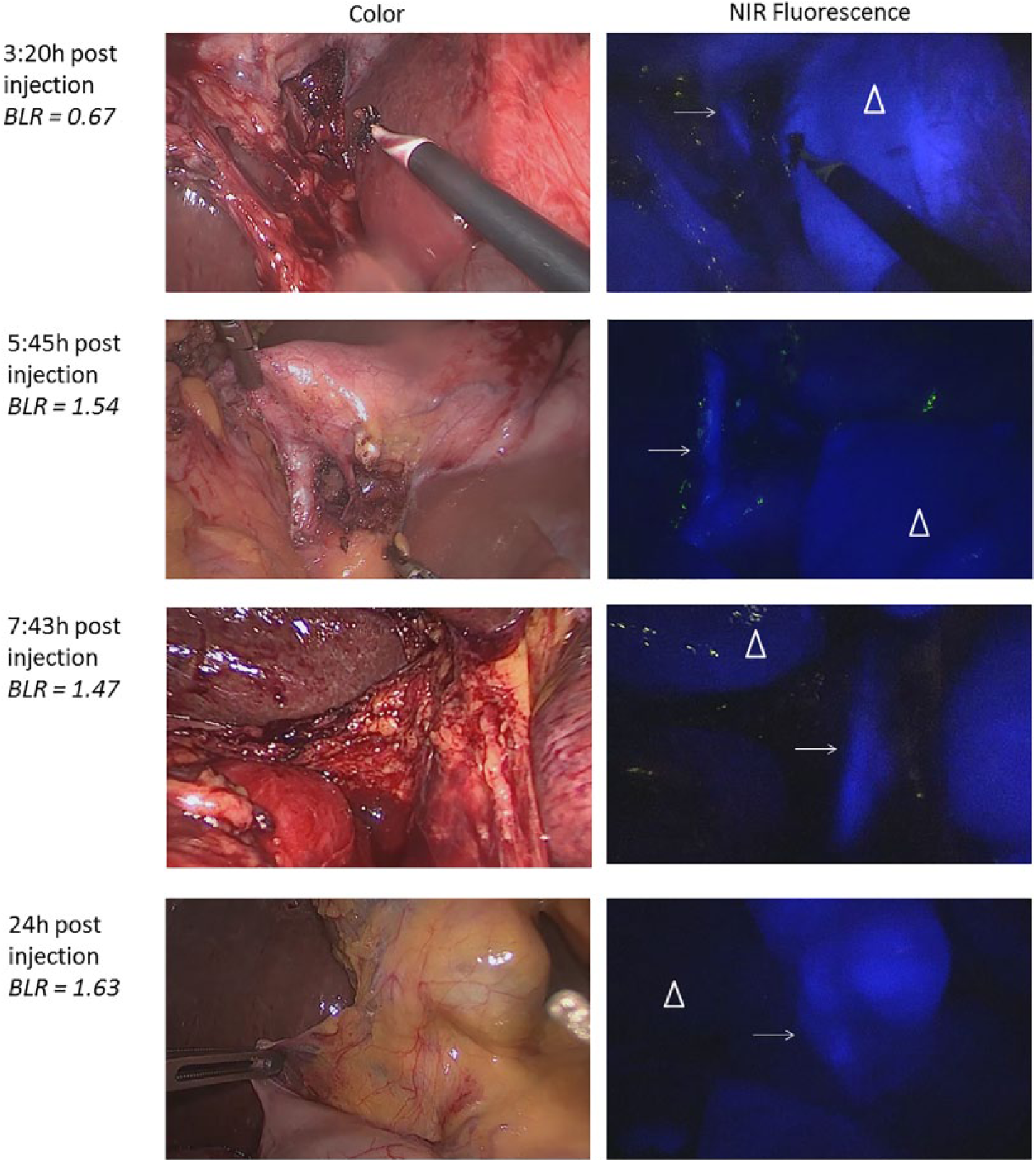
Fluorescence cholangiography over time after administration of 10 mg ICG. Shown are images of fluorescence detection of cystic duct (arrow) and adjacent liver tissue (pyramid) over time. Bile duct-to-liver ratios were sufficient (ie, >1) between 5 to 25 hours post dosing of 10 mg ICG.
Additional Finding
A patient planned to undergo a laparoscopic cholecystectomy and defenestration of a liver cyst received 10 mg ICG. Fluorescence cholangiography was performed at 3.5 hours post dosing. Although bile duct detection was hampered by the intense fluorescence of the liver (BLR 0.7), the liver cyst could be clearly demarcated by the lack of fluorescence inside the cyst (Video 2, available online at http://journals.sagepub.com/doi/suppl/10.1177/1553350617702311).
Evaluation of Fluorescence Cholangiography per Surgeon
Results of the questionnaire among all operating surgeons are displayed in Figure 5.
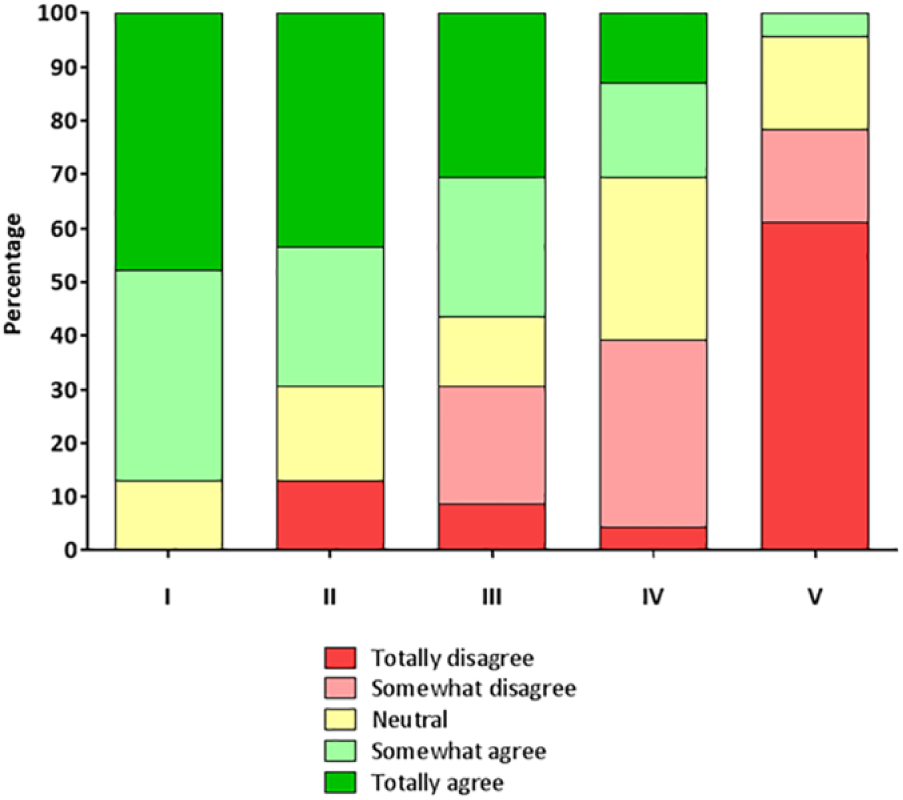
Results of questionnaire. Shown are all the scores that were given by surgeons after they performed a laparoscopic cholecystectomy with use of fluorescence cholangiography. The statements that were scored were respectively (I) it could decrease the number of bile duct complications; (II) it led to better detection of bile ducts; (III) the technique was of added value for this procedure; (IV) it needs to be standard-of-care during laparoscopic cholecystectomy; (V) it delays surgery.
Discussion
Fluorescence cholangiography can improve orientation of bile duct anatomy during laparoscopic cholecystectomies and potentially lead to a reduced risk of bile duct injury. This study is the first showing that the interval between administration of ICG and intraoperative fluorescence imaging should be prolonged to 3 to 6 hours post dosing of 5 mg, or 5 to 23 hours post dosing of 10 mg. By doing so, bile ducts are more fluorescent than liver tissue, resulting in improved discrimination of bile duct anatomy. Consequently, also unexpected aberrant bile ducts can be detected. 43 This may make laparoscopic cholecystectomy safer. Large studies focusing on patient outcome should demonstrate if fluorescence imaging reduces bile duct injuries.
In our systematic review, none of the identified articles studied fluorescence imaging at the optimal time points. Only 2 studies focused on optimizing the dose of ICG and timing of fluorescence imaging. Zarrinpar et al 20 performed a study in patients undergoing laparoscopic hepatic or biliary operations and injected doses ranging between 0.02 and 0.25 mg/kg, 10 to 180 minutes prior to fluorescence imaging. In this study, the authors also concluded that a prolonged interval appeared optimal for fluorescence cholangiography, but they did not study longer time intervals. Our group previously studied fluorescence cholangiography during open and laparoscopic surgery 0.5 and 24 hours after ICG administration. 15 The latter resulted in much better discrimination between bile ducts and liver tissue. However, administrating ICG 24 hours prior to surgery is less feasible in everyday clinical practice; most patients arrive in the morning prior to surgery at the hospital. According to our study results, we can advise a dose of 5 mg ICG, administered 3 to 7 hours prior to surgery, which is much more practical in everyday clinical practice. Although this study is performed with a relatively small group of patients, we believe these results should form the basis for larger studies that focus on the added value of fluorescence cholangiography. Currently, 3 large, multicenter, randomized clinical studies are ongoing to assess the potential added value of fluorescence cholangiography (Table 3). Unfortunately, probably due to logistical reasons, researchers choose to inject ICG directly prior to surgery. Prevention of bile duct injuries is a very difficult endpoint that requires huge amount of patients, but other useful endpoints could be reduced operating room time, cost-effectiveness, less use of radiation for IOC, and so on.
Recruiting randomized controlled trials using fluorescence cholangiography.
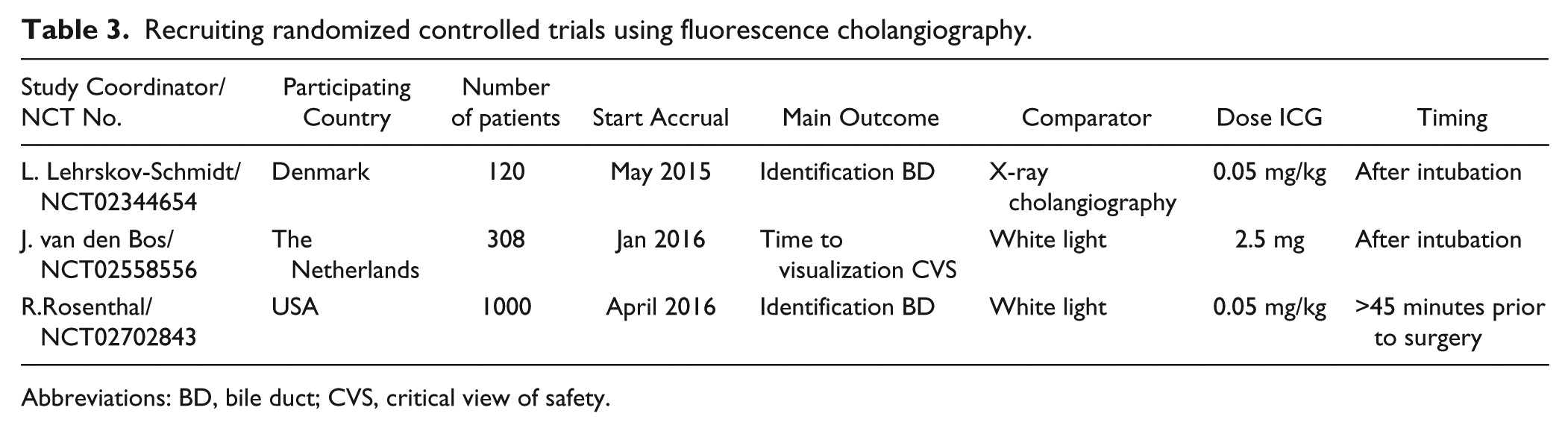
Abbreviations: BD, bile duct; CVS, critical view of safety.
A commonly mentioned drawback of fluorescence-guided surgery is the lack of standardization of fluorescence imaging systems. 44 Standardization is important for the determination of objective assessments during oncological fluorescence-guided surgery trials, 45 but differences in fluorescence imaging systems also hinder comparison between fluorescence cholangiography studies. Different lasers, LEDs, filters, image sensors, and image processing all make a fair comparison difficult. A recent study compared 5 different fluorescence laparoscopic imaging systems for fluorescence cholangiography: a prototype and an improved version of the Hamamatsu Photonics laparoscope, the fluorescence imaging system of Olympus Medical Systems, the Karl Storz HD fluorescence laparoscope, and the fluorescence imaging system of Novadaq. 19 An important conclusion from this study was that the contrast of ICG was significantly different among all the used laparoscopic imaging systems. Outcomes of studies (e.g. BLR) are therefore difficult to compare. The system used in the present study, the Karl Storz HD fluorescence laparoscope, showed not to be optimal for ICG detection, and therefore, ratios obtained in the current study are expected to be even higher when other imaging systems are used.
The intensity of the fluorescence signal depends on several factors, including the distance from the tip of the laparoscope to its target and the amount of covering tissue. For this reason, we chose in the current study to calculate a ratio between the fluorescence signal in the CD and liver, instead of measuring solely fluorescence intensity in the bile duct or liver. Moreover, to make the results more accurate, we analyzed the BLR at 3 different time points during surgery.
Intraoperative assessment of biliary anatomy can be improved using several techniques. 46 IOC, still the most frequent applied technique for bile duct clarification, is investigated in large cohort studies. A forest plot of the 6 largest population-based studies showed an odds ratio for bile duct injury of 0.60 (95% confidence interval = 0.52-0.70) when using IOC. 46 No randomized trial studying the effect of IOC exists and is almost impossible to conduct. Still the association between reduced risk of bile duct injury and use of IOC has been shown convincingly. 47 An important disadvantage of IOC is however that obtained radiographic images can be difficult to interpret and IOC is a technically challenging procedure. 48 Moreover, costs of IOC are high, caused by prolonged surgery time of 10 to 23 minutes, requirement of an additional medical specialist, and use of equipment. 49 On the other hand, treating patients with bile duct injuries is a financial burden for the health care system. Some physicians state therefore that routine use of IOC is cost-effective. 47 As indicated by our surgeons, fluorescence imaging does not prolong operating room time. Furthermore, it does not require another medical specialist or the use of radiation, and it certainly cannot cause bile duct injury. 35 We therefore advocate that fluorescence imaging should be an important part of laparoscopic cholecystectomy.
A limiting factor of fluorescence cholangiography compared to IOC is the inability to detect CBD stones. Moreover, the CBD is not always identified, due to the limited penetration depth of approximately 1 cm of light in the NIR spectrum. 12 Structures beneath a layer of (periductal) fat, especially in obese patients, are therefore more difficult to identify using NIR fluorescence imaging.12,42 Fluorescence cholangiography can however easily be combined with IOC, as shown in the current study. An advantage of fluorescence cholangiography is that it can be performed at all time during surgery without significant delay.
Intravenous ICG administration can also be valuable to assess intraoperative identification of (bile duct) vasculature.28,31 Because ICG binds to plasma proteins, it remains intravascular. 50 This in combination with its short half-life of approximately 3 minutes makes it a very useful contrast agent for perfusion angiography. When doubt exists about aberrant vasculature or identification of the cystic artery, an additional injection of ICG can be given. One should however bear in mind that intravenous ICG administration during surgery will result in intense fluorescence of the liver and, thus, should only be performed when bile duct anatomy is already clarified.
In conclusion, this study shows that a prolonged interval (at least 3 hours) between ICG injection and intraoperative fluorescence cholangiography results in much better contrast between bile ducts and liver tissue. Although this study was performed in a small group of patients, we believe that when the optimal dose and timing are used, fluorescence cholangiography can be of added value for both patients and surgeons.
Footnotes
Author Contributions
Study concept and design: Leonora S. F. Boogerd, Henricus J. M. Handgraaf, Wendeline J. van der Made, Cornelis J. H. van de Velde, Alexander L. Vahrmeijer
Acquisition of data: Leonora S. F. Boogerd, Henricus J. M. Handgraaf, Volkert A. L. Huurman, Hwai-Ding Lam, J. Sven D. Mieog, Wendeline J. van der Made
Analysis and interpretation: Leonora S. F. Boogerd, Cornelis J. H. van de Velde, Alexander L. Vahrmeijer
Study supervision: Volkert A. L. Huurman, J. Sven D. Mieog, Wendeline J. van der Made, Cornelis J. H. van de Velde, Alexander L. Vahrmeijer
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Dutch Cancer Society Grant UL2010-4732.
