Abstract
Electroencephalographic (EEG) alterations have been demonstrated in acute, chronic, and experimentally induced musculoskeletal (MSK) pain conditions. However, there is no cumulative evidence on the associated EEG characteristics differentiating acute, chronic, and experimentally induced musculoskeletal pain states, especially compared to healthy controls. The present systematic review was performed according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses guidelines (PRISMA) to review and summarize available evidence for cortical brain activity and connectivity alterations in acute, chronic, and experimentally induced MSK pain states. Five electronic databases were systematically searched from their inception to 2022. A total of 3471 articles were screened, and 26 full articles (five studies on chronic pain and 21 studies on experimentally induced pain) were included for the final synthesis. Using the Downs and Black risk of assessment tool, 92% of the studies were assessed as low to moderate quality. The review identified a ‘very low’ level of evidence for the changes in EEG and subjective outcome measures for both chronic and experimentally induced MSK pain based on the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) criteria. Overall, the findings of this review indicate a trend toward decreased alpha and beta EEG power in evoked chronic clinical pain conditions and increased theta and alpha power in resting-state EEG recorded from chronic MSK pain conditions. EEG characteristics are unclear under experimentally induced pain conditions.
Introduction
Spinal and supraspinal structures play an essential role in pain processing and modulation.1–3 Neuroplastic changes in various brain regions involving pain processing and modulation occur in persistent pain and could influence pain perception independent of peripheral neural activation.2,4–6 Such alterations have been proposed to maintain pathological pain states in musculoskeletal (MSK) conditions.4,7–15 Mounting evidence suggests that the symptomatology associated with pain is determined by the complex dynamic neuronal interaction between multiple brain regions and is susceptible to alterations with nociceptive signals.15–22
Electroencephalography (EEG) is a non-invasive tool for evaluating and recording brain function.23,24 EEG can record and process functional brain changes at a high temporal resolution, in milliseconds, and analyze the recorded oscillation phase.25–27 Brain activity and functional connectivity changes are extensively studied in various pain conditions as well as healthy populations. In addition, numerous studies have investigated the EEG responses to experimentally induced pain. Thus far, however, there is limited evidence available on the associated EEG characteristics differentiating acute, chronic, and experimentally induced musculoskeletal pain states.
Moreover, these studies have used various methodological paradigms and experimental designs. The diverse nature of these studies produces heterogeneity in reported results and warrants a systematic synthesis of the existing literature to identify EEG changes associated with experimentally induced pain contexts. A current meta-analysis has provided evidence on the noxious stimulation and associated brain changes in the chronic pain population. However, the review has included only studies that utilized functional magnetic resonance imaging (fMRI) and was limited to the chronic pain population. 28
MSK pain is no different from experimentally induced pain context, considering the associated brain changes. Key alterations in the EEG-based oscillatory and synchrony patterns in the cognitive, affective, and somatosensory cortices have been identified in individuals with MSK pain.8,17,29,30 Such EEG characteristics can also explain the dynamics of the brain regions linked to the pain experience. 31 Previous systematic reviews confirmed EEG-quantified brain activity changes in lower back pain (LBP) and fibromyalgia in lower and higher EEG frequency bands.8,32,33 However, no clear overview exists of evidence-based EEG studies describing if and how brain response might be altered in people with MSK pain when compared with healthy controls. Moreover, there is limited evidence on whether there is a difference in EEG response associated with acute, chronic, and experimentally induced MSK pain conditions. This systematic review aims to address the existing limitations and bridge the aforementioned research gap by answering the research question – What is the available evidence for cortical brain activity and connectivity? (Outcome; O) abnormalities using resting-state electroencephalography (Exposure; E) in people with MSK pain (Population; P) compared to healthy controls (Comparison; C)?. 34
Methods
Protocol Registration
This systematic review followed the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).35,36 The protocol registration can be found under the PROSPERO ID CRD42021270187.
Search Strategy and Information Sources
A structured and comprehensive search was performed using a well-developed search strategy to identify the relevant studies in the following electronic bibliographic databases 37 ; Ovid-Allied and Complementary Medicine (AMED), PsycINFO, PubMed, Scopus, and EMBASE from their inception to August 2021. The search strategy and terms were developed with the assistance of an experienced medical librarian from the University. The identified search terms were combined using appropriate BOOLEAN operators to perform the global search in the aforementioned databases. 38 A preliminary search combination was generated using PubMed advanced search strategy, and the search output was; ((eeg) OR (electroencephalogr*)) AND (pain). Periodic alerts from all the relevant databases were activated using the saved search strategy, and the search was updated on 31st January 2022. The details of the search terms are provided in the supplementary file (Supplementary file). We limited the search only to human studies and studies published in English, although no limits regarding period of publication were used.
Study Selection
A single reviewer (JM) collated the list of all possible studies for inclusion and exported them to a team-shared folder in EndNote, where duplicates were removed. The articles were then exported to Rayyan software39,40 by JM, and two independent reviewers (JM and TP) conducted a multilevel, blinded screening of the articles (titles and abstracts and full-text articles for eligibility). Any uncertainty regarding the eligibility of the study was resolved through discussion and where needed, by consulting the third reviewer (RM). Study exclusions were documented with reasons.
Eligibility Criteria
Definition and Participants
The following international definitions were used to define the participant population to include relevant articles for the systematic review.
According to the International Association for the Study of Pain (IASP) classification of chronic pain for ICD-11 defines chronic MSK pain as ‘chronic pain in bones, joints, and tendons arising from an underlying disease classified elsewhere’ lasting for more than three months. 41 Acute pain is awareness of noxious signaling from recently damaged tissue (< three months), complicated by sensitization in the periphery and within the central nervous system (CNS). Experimental pain refers to the experimental induction of pain through the application of electrical or thermal, or chemical stimuli in healthy individuals.42,43 In evoked chronic clinical pain, the examiner and/or participant elicit factors that initiate or exacerbate the pain.44,45 This review considered studies that included participants aged 18 years or older with acute or chronic MSK pain conditions or who underwent experimental pain stimulation.
Types of Study and Outcome Measures
This review included studies that compared the resting state EEG (rs-EEG) characteristics in people with acute or chronic MSK pain with healthy participants. Studies with evoked chronic clinical pain must have a control group as a comparator. Studies with experimental pain induction in healthy individuals must have baseline EEG data as a comparator to be included in the review. Studies that investigated resting-state EEG (rs-EEG) characteristics of pain associated with neurological conditions, migraine/headaches, fibromyalgia, and studies without a control group /condition were excluded from the review. Study designs such as cross-sectional, longitudinal, and case-control studies were considered.
Data Extraction
Data extraction was carried out using a customized Microsoft Excel (2020) sheet by JM. The following data were extracted from the included studies; study characteristics (author and year, study design), participant characteristics (sample size, age, and gender distribution, study population/diagnosis), type of pain, EEG characteristics (type of measure, EEG parameters, frequency bands of interest, analysis methods), clinical outcome measures, missing data, and key results. If data are incomplete or clarification is required, corresponding authors were contacted via e-mail. Two attempts were made to contact the authors, and we waited for two weeks to receive a response.
Assessment of Methodological Quality
The methodological quality of the studies was assessed by two independent reviewers (JM and TP) using the Downs and Black quality index originally developed to assess the methodological quality of both randomized and non-randomized studies using a 27-item checklist. 46 For the purpose of this review, we used a modified version of the tool, including 18 items in five domains (reporting: 8 items, external validity: 2 items, internal validity-bias: 4 items, internal validity-confounding: 3 items, and power: 1 item). We omitted items 4, 8, 13, 14, 17, 19, 23, 24, and 26 as they assessed variables related to interventional studies and hence were not relevant to this review. Item 9 was applicable only for longitudinal studies. All the items were scored as ‘yes’/’no’/UD (unable to determine)/’ NA’ (not applicable). Not applicable items were not included in the final percentage calculation. The final quality index was determined as follows; high quality: scored ‘yes’ for >75% of the total criteria; moderate-quality: 50-74% and low quality: <50%.47,48 Inter-rater reliability between the reviewers was calculated using Cohen's Kappa, 49 and a consensus was reached by both reviewers. Any disagreement between reviewers was resolved by consulting the third reviewer (RM).
Synthesis of the Level of Evidence (LoE) – GRADEpro GDT Analysis
Overall study quality was assessed for the LoE and strength of recommendation based on the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) criteria 50 using the GRADEpro GDT software accessed online from https://gradepro.org/. 51 GRADE has six components (study design, risk of bias, inconsistency, indirectness, imprecision, other considerations-publication bias, large effect, plausible confounding, dose-response gradient) assessing the certainty of the study. Quality was allocated as high (there is a lot of confidence that the true effect lies close to that of the estimated effect), moderate (the true effect is probably close to the estimated effect), low (the true effect might be markedly different from the estimated effect.), or very low (the true effect is likely to be substantially different from the estimated effect). 52 The GRADE scoring was performed based on the overall reported EEG changes if the methods of EEG acquisition, filter parameters, frequencies of interest, and computational units were varied.
Results
Study Selection
We extracted 9481 published articles from various electronic databases till August 2021 (AMED: 37; EMBASE: 1103; PubMed: 3046; Scopus: 3903 and PsycINFO: 1378), while 14 articles (Scopus) were manually added during the review process retrieved from periodical alerts from September 2021 to January 2022. After removing duplicates, we screened 3471 articles based on titles and abstracts, and 34 full-text articles were assessed for eligibility. Finally, 26 articles were included in the review. Figure 1 shows further detailed information on the inclusion and exclusion of study articles in the review.
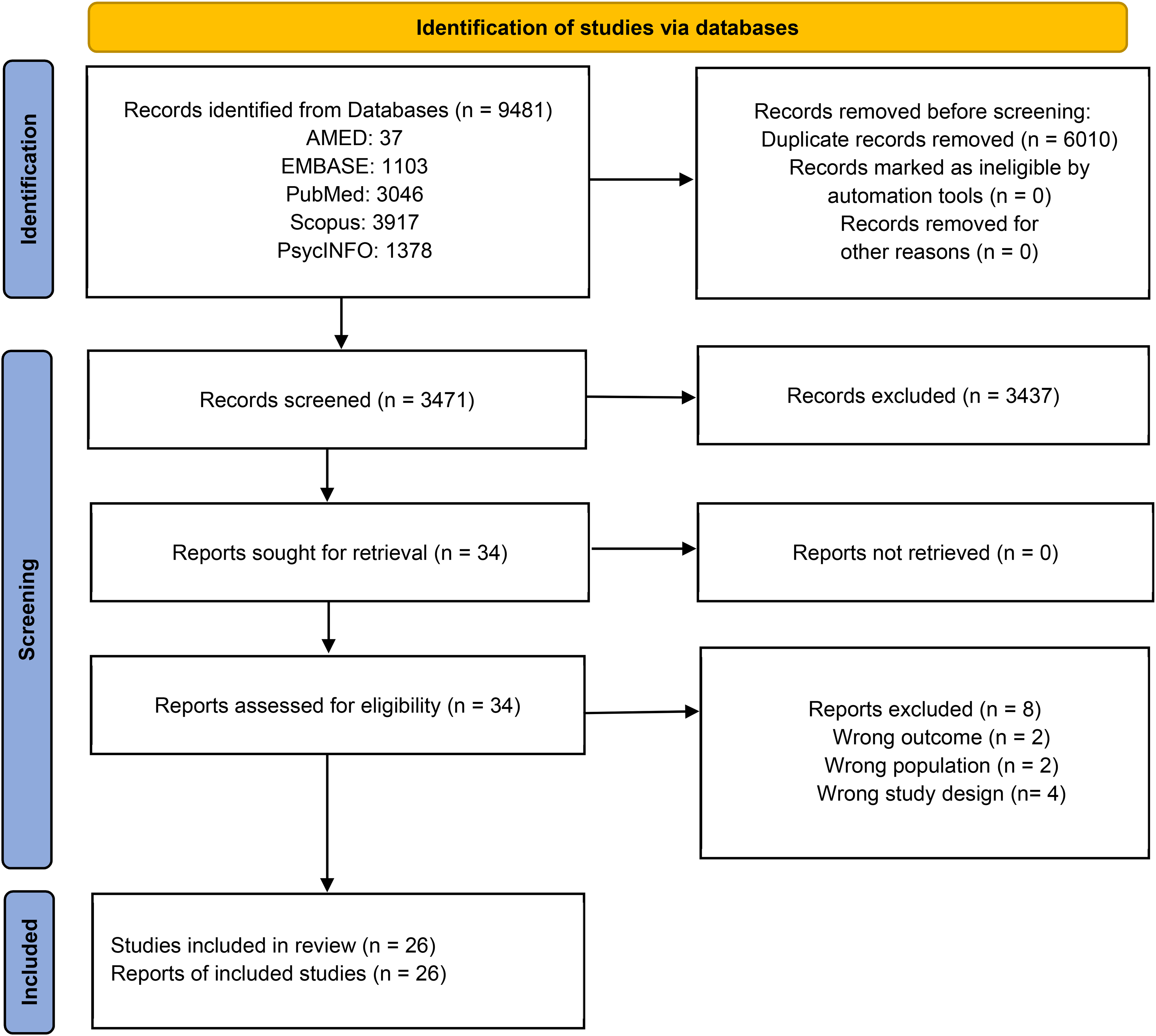
PRISMA flow diagram of the present review. Adopted from PRISMA 2020 statement.
Study Characteristics
The inclusion process yielded 26 published articles,19,29,53–76 including five studies on chronic pain53–57 and 21 studies on experimentally induced pain. No studies on acute musculoskeletal pain were identified. Overall, there were 23 cross-sectional and three longitudinal study designs.19,29,58 Studies were published between 1998 and 2022. The key characteristics of the included studies are presented in Tables 1 and 2.
Chronic Pain Study Characteristics.

Abbreviations: CS, cross-sectional; LD, longitudinal design; NR, not reported; N, numbers; SD, standard deviation; PN, pain group; HC, healthy control; MSK, musculoskeletal; CLBP, chronic low back pain; RS-EEG, resting-state electroencephalogram; CoG, center of gravity; EO, eyes open; EC, eyes closed; dB, Decibel; ↑, increase; ↓, decrease.
All five studies used activity-based analysis. * log-transformed sum of all power spectral density (PSD) values.
Experimental Induction of Pain Study Characteristics.
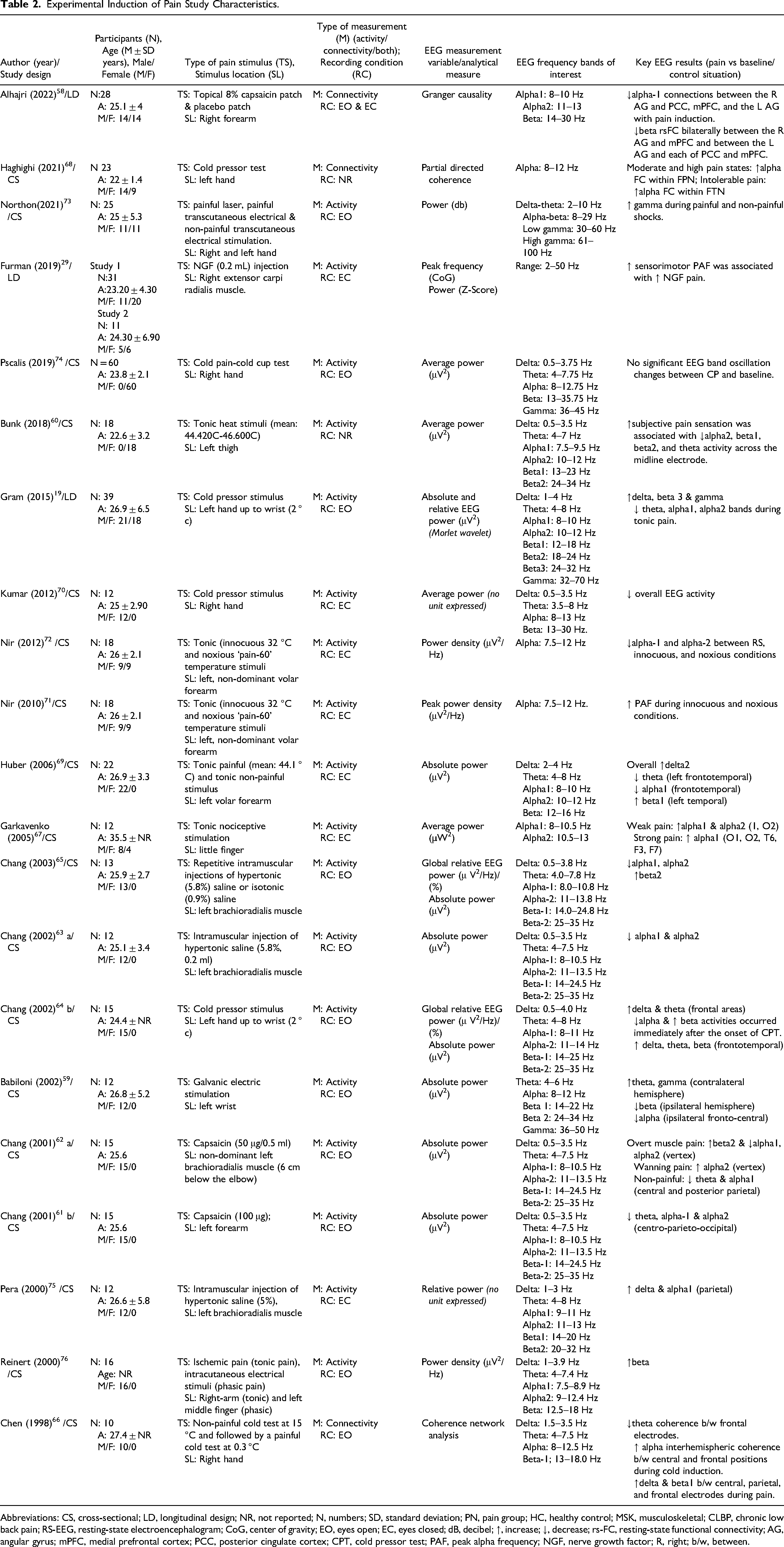
Abbreviations: CS, cross-sectional; LD, longitudinal design; NR, not reported; N, numbers; SD, standard deviation; PN, pain group; HC, healthy control; MSK, musculoskeletal; CLBP, chronic low back pain; RS-EEG, resting-state electroencephalogram; CoG, center of gravity; EO, eyes open; EC, eyes closed; dB, decibel; ↑, increase; ↓, decrease; rs-FC, resting-state functional connectivity; AG, angular gyrus; mPFC, medial prefrontal cortex; PCC, posterior cingulate cortex; CPT, cold pressor test; PAF, peak alpha frequency; NGF, nerve growth factor; R, right; b/w, between.
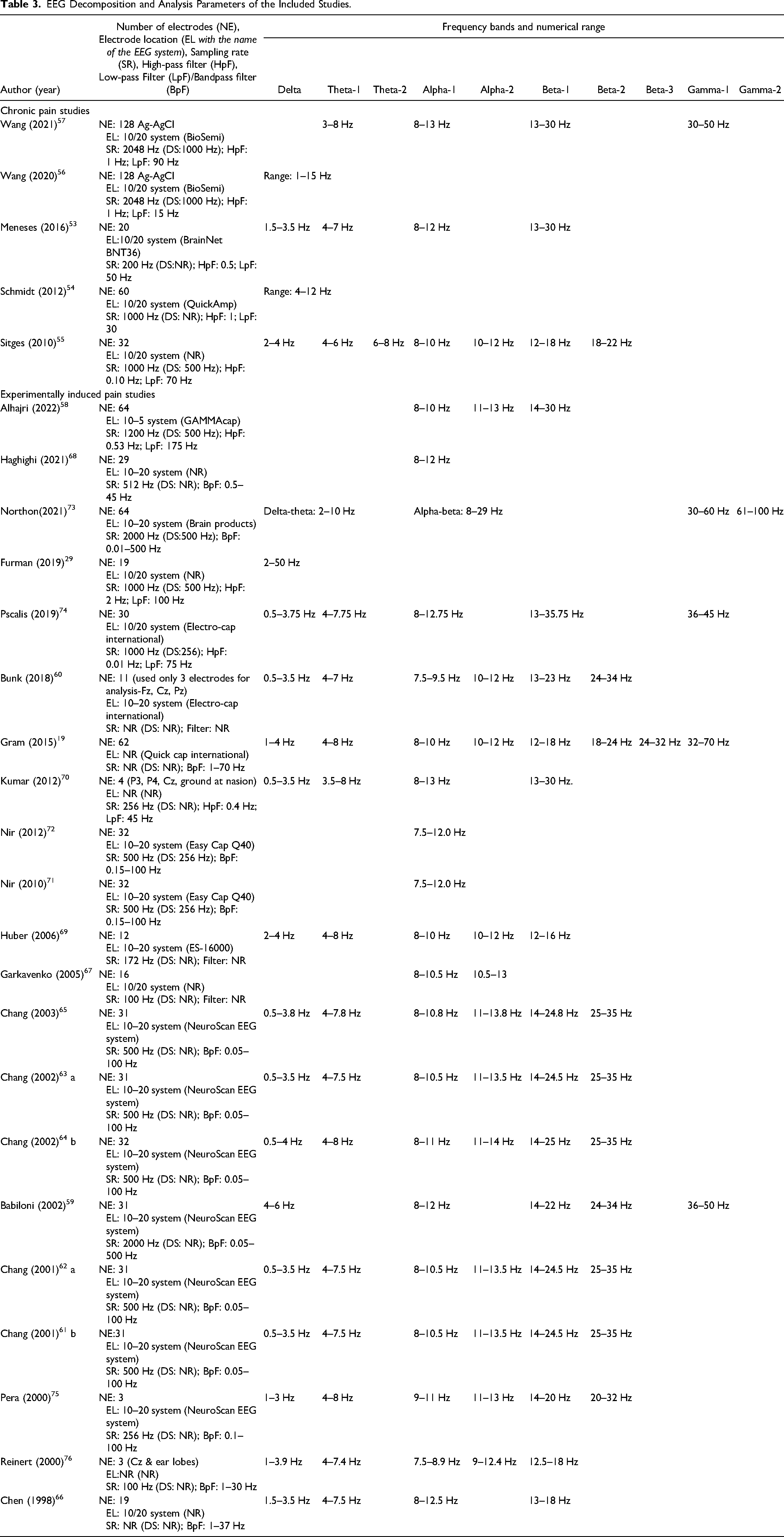
Due to the inconsistent division and overlapping of the frequency bands, we used semantic categories of frequencies (delta, theta, alpha, beta, and gamma) while describing and reporting the review results like previous reviews. 77 The most common inconsistency in frequency range description was observed with delta, theta, and alpha bands across the included studies. The EEG decomposition and analysis parameters of the included studies are summarized in Table 3.
EEG Decomposition and Analysis Parameters of the Included Studies.
Chronic Pain
A total of five studies of cross-sectional designs investigated EEG features in the chronic pain population against a healthy control group. Out of the five studies, two studies were in chronic low back pain (CLBP),54,57 one study in rheumatoid arthritis (RA), 53 one study in chronic jaw pain (CJP), 56 and one study examined a mixture of chronic MSK pain conditions 55 (Table 1). The sample size varied from 18 to 37 in the pain group and 16 to 37 in the healthy group. Studies involved both males and females with a mean age range from 32 to 65 years, with most of the studies including adults over 40 years of age. Two studies involved only females,53,55 and all other studies had a sample of 62–75% females. The mean pain duration of the participants included in the studies ranged from six to 25 years. Two studies investigated RS-EEG,53,54 and two studies recorded EEG during evoked chronic clinical pain conditions.56,57 One study captured EEG during non-painful somatosensory stimulation under pleasant and unpleasant affective conditions. 55 EEG data were recorded under different ocular conditions. Of the five chronic pain studies, three acquired EEG data during EO conditions, one during EC conditions, and one during EO and EC conditions.
All studies performed a sensor-level cortical level activity analysis, with most of the studies analyzing theta, alpha, and beta frequency bands of interest. Gamma band was measured in one study evaluating CLBP, 57 while delta was measured in two studies.53,55 Studies have measured pain intensity using various subjective outcome measures, including the Visual analogue scale-VAS (60% of studies) and Mcgill pain questionnaire-MPQ (40%). Psychological function (80%), physical activity (40%), physical function (20%), pain self-efficacy (20%), and quality of life (20%) was also reported in the studies.
In general, decreased alpha and beta EEG power was demonstrated over parieto-occipital regions by the two movement evoked pain studies in CLBP and CJP.56,57 However, out of the two rs-EEG studies, one study concluded increased theta and alpha power in the RA population compared to the healthy controls but did not find any correlation between the associated EEG changes and clinical pain outcomes. 53 The other study in CLBP 54 did not find any significant difference in EEG bands between the pain and healthy groups but did indicate a correlation between EEG power and higher pain ratings at various time points (refer to Table 2). In contrast, a non-painful somatosensory stimulation study in chronic MSK pain conditions demonstrated decreased theta and beta power over sensorimotor and temporal cortices. 55 However, this study included different MSK pain conditions, including conditions with neurological involvement, and the confidence in reporting the observed results may not be specific to the MSK pain according to the reviews definitions.
Experimentally Induced Pain
The review yielded 21 full articles investigating EEG changes associated with experimentally induced pain conditions in the healthy population. Three studies were longitudinal designs, while 18 studies were cross-sectional in nature (Tables 1 and 2). Overall, the sample size varied from 10 to 60 participants with 8 studies included both males and females,19,29,58,67,68,71–73 11 studies included only males59,61–66,69,70,75,76 and 2 studies with only females.60,74 Studies have used various testing stimuli to induce experimental pain, with three studies using capsaicin injection,58,61,62 three studies used hypertonic saline injection,63,65,75 nerve growth factor (NGF) injection in one study, 29 six studies performed cold-induced pain,19,64,66,68,70,74 three studies used electrical stimulation59,73,76 and five studies with tonic heat60,69,71,72 or pressure pain. 67
Studies have used various EEG recording methods with electrode numbers varying from 3 to 64 and 10–20 or 10–5 system of electrode placements. Most of the studies have decomposed EEG recording to theta, alpha, beta, and gamma frequency bands, while five studies investigated only the alpha EEG power67,68,72 or peak alpha frequency29,71 in the studied population. Three studies have performed connectivity analysis, out of which two studies explored sensor-based coherence network analysis.66,68 A single study used source-based analysis between the bilateral angular gyrus (AG) and posterior cingulate cortex (PCC), and medial prefrontal cortex (mPFC) at alpha and beta oscillations under EC and EO states using Granger causality as a measure for effective, ie, directional connectivity. 58 Furthermore, 11 studies performed EEG acquisition during EO conditions, seven during EC conditions, one during both EO & EC conditions, and one study did not report the ocular conditions during the EEG recording. All the studies have measured pain intensity using either VAS (33% of the studies), Visual rating scale-VRS (14%), Numerical rating scale-NRS (24%), MPQ (14%), or Numerical pain scale-NPS (24%). Pain catastrophizing thoughts were measured in two studies,29,57 and sleep quality, pain vigilance, and depression constructs were measured in one study. 58
Overall, studies that used capsaicin injection demonstrated reduced alpha-1 and alpha-2 EEG power over various electrodes and regions, especially in the centero-parieto-occipital regions.58,61,62 Similarly, two studies from the same authors63,65 demonstrated decreased alpha-1 and alpha-2 activity during muscle pain induced by 5.8% intramuscular hypertonic injection and were correlated with muscle pain. However, another study that used the same testing methods couldn't replicate the similar findings, instead demonstrated increased delta and alpha-1 power with tonic muscle pain from 5% hypertonic saline injection. 75 The two studies that examined PAF demonstrated consistent results indicating a strong association between increased PAF and muscle pain despite using different testing stimuli and locations.29,71 Overall, four studies that utilized cold-induced pain testing demonstrated increased beta and delta and decreased alpha EEG activity.19,64,66,74 Further, all the studies utilizing painful electrical stimulation demonstrated increased gamma oscillation in the studied population. Two studies60,69 from tonic heat pain have demonstrated decreased alpha and theta activity, while three studies67,71,72 showed increased alpha power with tonic heat pain induction.
Assessment of Methodological Quality
Two studies were assessed as high quality, five studies were low quality, and 19 were moderate quality (Table 4). The interrater level of agreement between the two reviewers (JM and TP) was ‘substantial agreement’ (Kappa = 0.74). 78 The most common bias observed across studies was the lack of sample size estimation, except for one study. 54 Additionally, only 31% of the included studies measured and performed adequate adjustments for confounding variables in the analysis.
Methodological Quality Assessment of the Included Studies Using Downs and Black Risk of Assessment Tool.
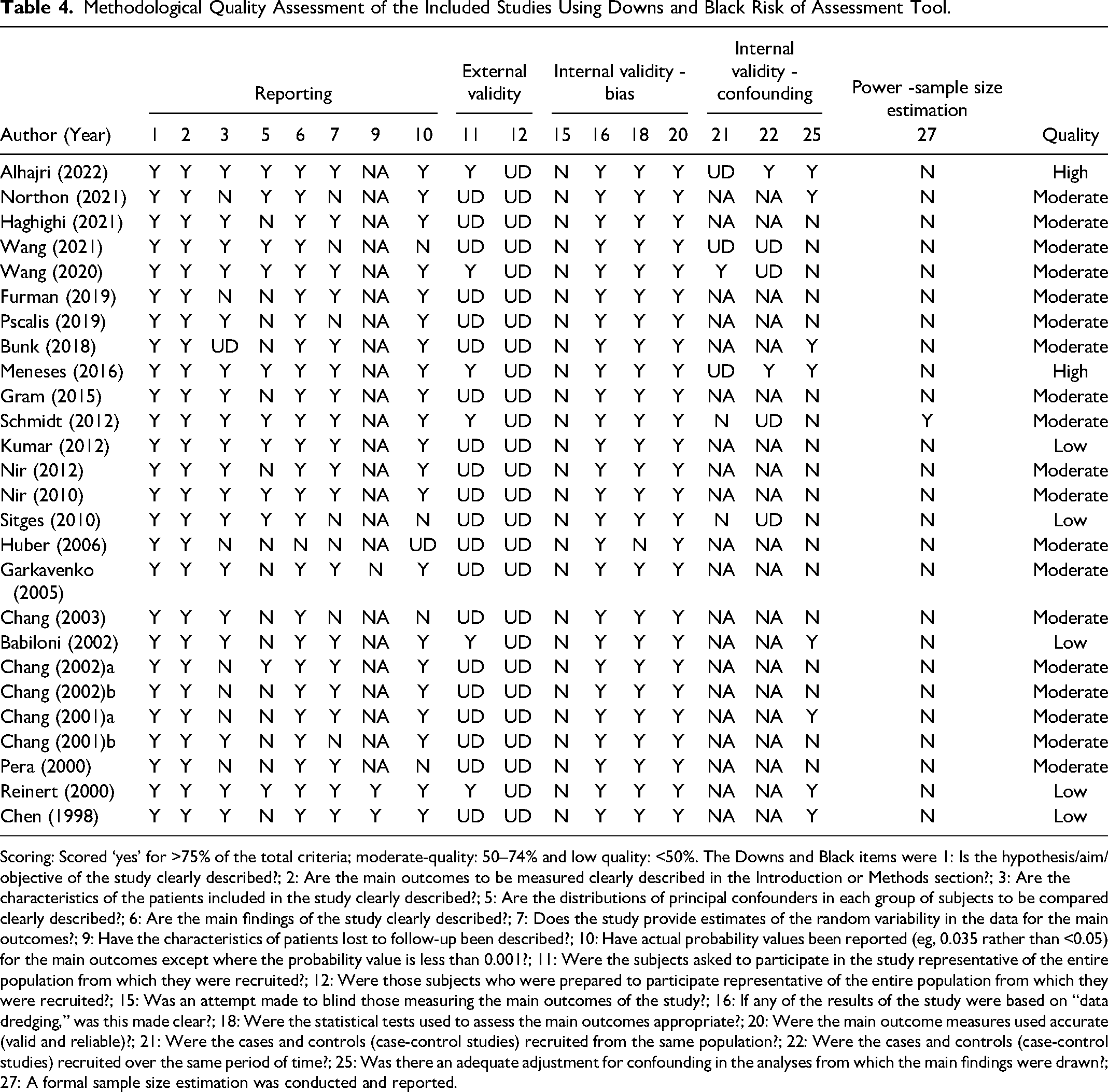
Scoring: Scored ‘yes’ for >75% of the total criteria; moderate-quality: 50–74% and low quality: <50%. The Downs and Black items were 1: Is the hypothesis/aim/objective of the study clearly described?; 2: Are the main outcomes to be measured clearly described in the Introduction or Methods section?; 3: Are the characteristics of the patients included in the study clearly described?; 5: Are the distributions of principal confounders in each group of subjects to be compared clearly described?; 6: Are the main findings of the study clearly described?; 7: Does the study provide estimates of the random variability in the data for the main outcomes?; 9: Have the characteristics of patients lost to follow-up been described?; 10: Have actual probability values been reported (eg, 0.035 rather than <0.05) for the main outcomes except where the probability value is less than 0.001?; 11: Were the subjects asked to participate in the study representative of the entire population from which they were recruited?; 12: Were those subjects who were prepared to participate representative of the entire population from which they were recruited?; 15: Was an attempt made to blind those measuring the main outcomes of the study?; 16: If any of the results of the study were based on “data dredging,” was this made clear?; 18: Were the statistical tests used to assess the main outcomes appropriate?; 20: Were the main outcome measures used accurate (valid and reliable)?; 21: Were the cases and controls (case-control studies) recruited from the same population?; 22: Were the cases and controls (case-control studies) recruited over the same period of time?; 25: Was there an adequate adjustment for confounding in the analyses from which the main findings were drawn?; 27: A formal sample size estimation was conducted and reported.
Synthesis of the Level of Evidence (LoE) – GRADEpro GDT Analysis
There is a ‘very low’ LoE for the changes in EEG and subjective outcome measures for both chronic and experimentally induced MSK pain. A detailed LoE assessment using GRADEpro GDT rating is reported in Tables 5 and 6.
Level of Evidence for Chronic Musculoskeletal Pain Compared to Healthy Controls for EEG Changes.

Level of Evidence for Experimentally Induced Pain Compared to Baseline/Control for EEG Changes.

Discussion
The purpose of this systematic review was to summarize the available evidence regarding the EEG characteristics in people with acute, chronic, and experimentally induced MSK pain contexts. Although there is very low confidence in the level of evidence, overall, the findings of this review indicate a trend toward decreased alpha and beta EEG power in evoked chronic clinical pain conditions and increased theta and alpha power in rs-EEG recorded from chronic MSK pain conditions. On the other hand, the review failed to identify a consistent difference in EEG characteristics in response to experimentally induced pain in healthy individuals using various testing stimuli.
Chronic MSK Pain
In general, a reduction in alpha and beta EEG activity was demonstrated in all four chronic pain studies over various cortical regions, primarily over the premotor and supplementary motor areas. Similar findings were reported by the two movement evoked pain studies in CLBP and CJP.56,57 Previous studies have also reported a reduction in beta power with movement evoked pain in hip osteoarthritis and CLBP.45,57 The shift in alpha and beta bands over the somatosensory cortices evidences the remodeling and somatotrophic reorganization with persistent MSK pain.79–82 This would explain and support the basis for pain development and maintenance beyond physiological tissue healing time.83–85 Moreover, this is imperative knowledge indicating that the neuronal alterations in the alpha and beta bands could be an important EEG correlate reflecting brain change associated with chronic MSK pain state.4,7–14 The findings are also of value while selecting regions of interest and frequency bands for designing neuromodulatory interventions for the management of pain experience in MSK conditions. Furthermore, three studies have investigated the relationship between the observed EEG changes with their pain experience.53,54,57 Notably, the higher pain ratings were associated with increased EEG power indices, 54 and movement evoked pain was associated with an attenuated reduction in beta power in the premotor and supplementary motor cortex. 57 One study did not find any significant association between the EEG changes and pain measures. 53 This could be merely due to comorbidities in people with pain when compared to controls. Co-morbidities associated with chronic pain, especially the psychosocial variable can influence the resting state EEG indices.86–88 Future studies may also consider investigating the association between the EEG and subjective and objective pain measures. This will increase the confidence in implying that the EEG changes were related to the pain not influenced by other co-morbidities.
Experimentally Induced Pain
Despite the inconsistencies and heterogeneous results of individual studies, it is of value to discuss some consistent findings observed across the studies. All the studies utilizing painful electrical stimulation found consistent results showing an increased gamma oscillation in the studied population. Evidence from electrocorticography (ECoG) and EEG studies has demonstrated high gamma activity as a reliable biomarker for perceiving electrical stimulation and as an early brain response correlated with localized nociceptive stimuli.89–93
In summary, 52% of experimentally induced studies have demonstrated decreased alpha power, while 28% of studies reported an increase in alpha power. Additionally, these changes were associated with subjective pain sensation. This suggests and supports the existing evidence8,33,94,95 that the changes in alpha power can be an important EEG marker in chronic and experimentally induced pain states. However, our findings suggest a lack of evidence for the direction of alpha power changes associated with chronic and experimentally induced pain conditions. Studies have also reported increased delta (19%), beta (19%), gamma (9%), and decreased theta (9%) with various testing stimulus paradigms used for experimental induction of pain. This suggests that the brain response can vary according to the type of noxious stimuli. This could be due to the complex adaptive nature of the brain in response to translating the noxious stimuli into different dimensions of pain involving the activation of deeper subcortical structures.4,96–98 Previous systematic reviews have also reported similar findings with noxious stimulation in chronic pain conditions. 28
Strengths, Limitations, and Future Recommendations
This is the first comprehensive systematic review conducted to identify and compare the level of evidence in EEG changes associated with acute, chronic, and experimentally induced MSK pain against healthy controls. It is evident from this review that there is a paucity of literature investigating EEG changes in chronic MSK pain conditions against age and gender-matched healthy controls. Future EEG studies must consider incorporating a matched control group to compare the EEG changes associated with pain. This will increase the confidence in drawing a definitive conclusion regarding the associated EEG changes and is of clinical value.
We initially planned to perform a meta-analysis of the included studies if there was adequate homogenous data. However, the results from the data extraction indicated a large variation in EEG decomposition, filter parameters, definitions and division of each frequency range, and computational unit expression across the included studies. For example, if one study reported absolute EEG power, while another study reported overall EEG power (computed as the log-transformed sum of all PSD values within the empirically defined regions of interests), synthesis was not possible since the measures are distinct calculatory expressions of EEG power concerning volume conduction and field spread (refer to Tables 1–3).99,100 A similar issue can be observed in studies that conducted connectivity analysis. Also, most of the studies have not provided estimates of the variability in the EEG data. Most of the results were presented in topographical earth images, causing graphical data extraction difficult using data visualizing software. Additionally, studies have reported the EEG data either based on the scalp sensors or region-specific (lobes). This increases the uncertainty with data synthesis and performing a meta-analysis. Future studies might benefit from employing more measures for reporting the results of spectral analyses, with a special focus on potential differences in EEG spectral features among different pain conditions. Also, studies might consider reporting the estimates of the random variability of the EEG data, such as standard deviations and confidence intervals for each frequency band. This will help future systematic reviews in data synthesis and analysis.
However, we contacted the authors of the included studies. Unfortunately, most authors could not locate the raw data, due to various reasons, and others didn't respond to the e-mail requests. Moreover, future EEG decompositions could consider dividing the frequency bands according to the available guidelines and recommendations. 101 This will aid future reviews in conducting a meta-analysis based on the reported frequencies and computational units. However, the existing guidelines do not provide a consensus on the frequency bands <1 Hz. In such cases, future studies can refer to existing divisions used for neuromodulatory approaches utilizing slow and infraslow oscillations for various conditions.102–105 Surprisingly, an important observation is that none of the included studies has evaluated a full band EEG in the studied population. A growing body of literature is available indicating the important role of frequency bands below 1 Hz (slow and infraslow bands) in pain processing and modulation.4,106,107 Future studies in EEG might therefore benefit from considering recording full-band EEG and analyzing frequency bands below 1 Hz.
We observed a wide variability with studies reporting the pre-processing methods used for removing artifacts from the EEG signals. The studies have used different ocular conditions for EEG acquisition based on the purpose and study objectives. However, there is a greater risk of artifacts from eye blinking and laterality movements affecting the EEG data with EO conditions. This can affect the overall quality of the EEG data if appropriate artifact rejection methods are not used. Only a few studies have recorded electro-occulogram during the EO EEG recording conditions and reported the artifact rejection from the EEG data during pre-processing. Future studies should consider using robust methods for artifact rejection (eg, Independent component analysis, blind source separation), and the quality of the manuscript might improve by reporting this process in the manuscript. Furthermore, future studies could consider including a whole-brain mapping analysis without any ‘a priori’ assumptions to understand the whole brain dynamic activity changes observed in the studied population. This will help the researcher to include brain regions that are significantly different in comparison with a control group to do a further activity or connectivity analysis along with the ‘a priori’ regions. Finally, recent evidence supports the idea that chronic pain can be understood not only as altered brain activity but also as a consequence of the aberrations in the functional connectivity within and between various pain mediating networks.15,17,21 Future studies could therefore consider including more explorative connectivity and network-based analysis to understand the involvement of brain networks in chronic pain. This will also aid the development of neuromodulation approaches targeting cortical networks for the management of chronic pain experiences.
This review included studies that compared the rs-EEG characteristics in people with acute or chronic MSK pain compared with healthy/control participants. We have excluded the studies that investigated event-related potential (ERP). However, we have included studies that have investigated both resting state and ERP but the systematic review considered only the data corresponding to the resting state EEG variables. Stimuli-induced ERP variables could also be a potential marker in acute and chronic pain conditions. This needs to be considered in future systematic reviews/meta-analysis.
Deviation from Protocol
The review was initially planned to use Newcastle-Ottawa Scale (NOS) for assessing cross-sectional studies. However, the studies have exhibited a large variation with EEG acquisition and reporting methods upon the initial pilot search. To capture all the methodological considerations, the NOS tool requires an enormous level of modification to adapt for the purpose of this review. To avoid the plausible selection and information (misclassification) (https://online.stat.psu.edu/stat507/lesson/3/3.5) 108 bias associated with this modification, the authors decided to use a modified Downs and Black scale for non-randomized studies/case-control studies.
Conclusions
Alpha (and beta) band alterations are evident in people with chronic musculoskeletal pain. However, there is uncertainty in the direction of the observed differences. EEG characteristics are unclear under experimentally induced pain conditions, possibly due to the varying nature of pain induction procedures. Overall, the included studies were of moderate to low methodological quality. Future studies might therefore benefit from following the appropriate existing guidelines for reporting methods and results of the study. Also, future studies in EEG could consider recording full-band EEG and analyzing frequency bands below 1/0.5 Hz.
Supplemental Material
sj-docx-1-eeg-10.1177_15500594221138292 - Supplemental material for Is There a Difference in EEG Characteristics in Acute, Chronic, and Experimentally Induced Musculoskeletal Pain States? a Systematic Review
Supplemental material, sj-docx-1-eeg-10.1177_15500594221138292 for Is There a Difference in EEG Characteristics in Acute, Chronic, and Experimentally Induced Musculoskeletal Pain States? a Systematic Review by Jerin Mathew, Tyson Michael Perez, Divya Bharatkumar Adhia, Dirk De Ridder and Ramakrishnan Mani in Clinical EEG and Neuroscience
Footnotes
Availability of Data and Other Materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
